Abstract
We studied metabolic activity of the symbiont-bearing large benthic foraminifer Heterostegina depressa under different light conditions. Besides the overall photosynthetic performance of the photosymbionts estimated by means of variable fluorescence, the isotope uptake (13C and 15N) of the specimens (= holobionts) was measured. Heterostegina depressa was either incubated in darkness over a period of 15 days or exposed to an 16:8 h light:dark cycle mimicking natural light conditions. We found photosynthetic performance to be highly related to light supply. The photosymbionts, however, survived prolonged darkness and could be reactivated after 15 days of darkness. The same pattern was found in the isotope uptake of the holobionts. Based on these results, we propose that 13C-carbonate and 15N-nitrate assimilation is mainly controlled by the photosymbionts, whereas 15N-ammonium and 13C-glucose utilization is regulated by both, the symbiont and the host cells.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
General introduction
Large benthic foraminifera (LBF) are essential components of shallow marine ecosystems like coral reefs and seagrass meadows. LBFs are sensitive to climate change and react almost immediately to changing physical parameters like temperature or salinity1. Recently, numerous studies have shown that foraminiferal communities are sensitive bioindicators for monitoring environmental parameters and their change: Schmidt et al.2 investigated the combined effects of warming and ocean acidification on LBFs, showing that elevated temperature had more negative effects on calcareous organisms than increased concentrations of dissolved CO2. This impact is, however, dependent on the examined taxa3,4,5. Especially those species which incorporate more Mg2+ into the carbonate tests are important for the chemical equilibrium in reefs as they have (post mortem) a high buffer capacity against daily pH fluctuations caused by community metabolism6. Generally, the assemblages of living LBFs are highly dependent on physical parameters like habitat depth, light supply and water motion7. Observing LBFs activity is suitable to detect chemical contaminants in seawater8. Thus, regular monitoring of LBFs can be used as an important tool to characterize the health state of coral reefs. This approach was first established by Hallock et al.9 as the FORAM index (Foraminifera in Reef Assessment and Monitoring) and is based on the changes in the foraminiferal assemblages associated with environmental changes. Following that, it is possible to classify the state of health of coral reefs by simply investigating the foraminiferal fauna composition9, 10.
Light as key factor for LBFs
LBFs contribute a significant amount to the carbonate sediments worldwide due to their high abundance11. They also play a fundamental role in global carbon cycling and in sediment production in reefs11. Generally, LBFs contain photosymbionts which play a pivotal role in the development of large sizes up to several mm12. Species hosting photosymbionts have an increased test size, a special chamber arrangement and ultrastructural modifications to optimize the light supply within the foraminiferal organisms13. Especially in environments with elevated light supply paired with depleted dissolved organic matter content, algal symbionts are advantageous14. Interestingly, although most LBFs are mixotrophic, they are not able to survive for longer periods without their endosymbionts15. Not only a high diversity of eukaryotic algal symbionts are known for LBFs, but also cyanobacteria and bacteria are assumed to contribute significantly to the LBFs metabolism16. The endosymbionts are controlled by several factors like food availability of the host, water temperature, light supply and salinity17.
In this study we use Heterostegina depressa as a model, representing large rotalids, which host obligatory symbionts (diatoms). The adaption to different light intensities from this symbiosis is reflected by their morphological plasticity. At this point it should be mentioned, that also some miliolids are part of the LBFs. In contrast to the adaptive strategy to different light conditions from rotalids, miliolids establishing symbioses with a variety of algal symbionts10.
Pecheux18 measured test sizes of LBFs collected from different water depths (20–130 m) and found that their size is directly related (negative) to light supply. The importance of irradiance for symbiont bearing foraminifera is obvious and was already observed by earlier studies19. However also other factors might be significant for the abundance of LBFs: Nobes et al.20 found that irradiance flux only explained a small proportion of foraminifera distribution (based on the observation of large rotalids). Contrarily, the distance from the coast turned out to be the most important factor for LBF occurrence, whereby potentially also the nutrient flux will play a role in the foraminiferal distribution, but this aspect was not clarified by Nobes et al. In laboratory experiments, the same authors also found that the growth of the LBF Heterostegina depressa increased significantly at reduced light supply under continuous irradiance supply; therefore this taxon is considered a low light species. High irradiance of ~ 1200 µmol photons m−2 s−1 leads to increased mortality (50%) within a few weeks, whereas low light supply (60 µmol photons m−2 s−1) turned out as the light optimum for H. depressa20. These results fit to the findings of Röttger21, who postulated highest growth rates of H. depressa at low light supply. H. depressa is a species which is obligatorily dependent on the metabolic by-products of their symbionts and therefore shows a mixotrophic life style (= host cells are heterotrophic but obtain metabolites from their autotrophic symbionts) like other LBFs22. Because of the direct dependency on irradiance supply, this species is used for paleo-reconstruction of past water depths by analysing the occurrence of fossil LBFs23.
Though some studies18, 20 have been conducted on the growth and size distribution of LBFs related to irradiance supply, no study has dealt with nutrient uptake of LBFs as dependent on light supply to our knowledge. We assume that the utilization of certain carbon- and nitrogen-related compounds is conducted by the symbionts or is enhanced by their presence under light. However, other compounds, like dissolved organic material, will also be taken up and assimilated by the foraminifera itself or by osmosis where the symbionts are not involved. For that purpose, we measured nutrient uptake (nitrate, ammonium, carbonate and glucose) during prolonged darkness and compared it with foraminifera grown at a diurnal light cycle. In addition, pulse amplified modulated fluorescence analyses were conducted with an imaging fluorescence instrument to study potential effects of irradiance supply and prolonged darkness on symbiont performance.
With this study we want to clarify several aspects. First, it should be observed whether foraminifera absorb dissolved components of carbon and nitrogen in complete darkness. Based on this observation, further experiments with a normal daylight rhythm will be carried out to investigate the proportion of the up taken amount of elements by the symbionts. Finally, since LBFs are often used as model organisms, a statement should be obtained about which isotopes are best suited for further laboratory cultivation experiments.
These results contribute to a better understanding of the host-endosymbiont relationship between foraminifera and diatoms and clarify which nutrients are more likely to be taken up by the diatoms and which by the foraminifera itself. In addition, these results can also be used for paleontological studies. Since foraminiferal assemblages are often used as proxies for the reconstruction of paleoenvironments, light-factor experiments in particular provide new data on the distribution patterns of certain species.
Material and methods
Main culture
We used individuals of a permanent culture of H. depressa, hosted at the Department of Palaeontology at the University of Vienna. All selected foraminifera had a diameter of approximately 1250 µm. The main culture is maintained in an aquarium at 25 °C and 30 µmol photons m−2 s−1 photosynthetically active radiation (PhAR).
Photosynthetic performance of the photobiont
Experiments were performed in six-well plates with placing a single individual in each well. The specimens were covered with 5 ml sterile filtered artificial seawater and were incubated at 25 °C. Six individuals were each incubated in total darkness or under a light:dark-cycle of 16:8 h at 30 µmol photons m−2 s−1, respectively (12 specimens in total). Photosynthetic performance of the photobiont symbionts of LBFs was measured several times during a period of 15 days using maximum variable chlorophyll fluorescence imaging of photosystem II (PSII; Imaging PAM Microscopy Version–Walz GmbH; excitation at 625 nm). Both, dark and light incubated foraminifera were measured at day 1, 3, 5 and 7 (Fig. 1). For this purpose, the same 12 individuals were measured every timepoint. The measured variable fluorescence as a proportion of maximum fluorescence yield (Fv/Fm) describes the difference between maximum fluorescence and minimum fluorescence (variable fluorescence), divided by maximum fluorescence, which is used as a measure of the maximum potential quantum efficiency of photosystem II24. Fv/Fm serves as a proxy for the integrity and physiological activity of the photosymbionts, ranging between 0.79 and 0.84, lower value indicating photobiont stress24. The PAM—images were evaluated using the software WinControl-3 (Walz GmbH); the photosynthetic area of each specimen was calculated with the software Image J (version 1.53 k, Java).
Isotopic uptake experiments
Foraminifera were incubated for 1, 3 and 7 days in crystallisation dishes filled with 280 ml sterile filtered artificial seawater. Six foraminifera were placed into each dish, supplied separately with either isotopically enriched Na15NO3, 15NH4Cl, NaH13CO3 or 13C-glucose to a final concentration of 0.2 mM each. One set of foraminifera was incubated at a light: dark-cycle of 16:8 h at 30 µmol photons m−2 s−1, a second one in continuous darkness. In total 6 × 4 × 2 (number of replicates × isotopically enriched compounds × light conditions) foraminifera were incubated for this experiment. After the respective incubation times, the foraminifera were collected from each irradiance treatment and nutrient addition. For each treatment, 6 replicates were analysed individually. After incubation, the foraminifera were rinsed with distilled water and transferred to pre-weighted Sn capsules. The specimens were then dried for 3 days at room temperature and weighed. Then the tests were dissolved in 12.5 µl 2 M HCl and the organic remains dried at 50 °C for 3 days and reweighed. Measurements of the isotope ratios of each sample were carried out in the Stable Isotope Laboratory for Environmental Research (SILVER) of the University of Vienna. For a detailed description of the measurement and the calculation of the incorporated amount of isotopes, see Lintner et al.25.
Statistics
The following hypothesis should be testes: different lighting conditions affect the activity of the symbionts (PAM experiments). Additionally, the hypothesis that the activity of the foraminifera is influenced by different chemical nitrogen or carbon sources components will also be investigated (isotopic uptake experiments). For statistical analysis, repeated measurement one-way ANOVA (level of significance = 95%) was performed for the PAM experiments over time to test if, prolonged darkness significantly altered the overall photosynthetic performance of the photobiont compared to natural irradiance supply. Two-way ANOVAs were used for the isotopic uptake to test if light supply and time affected the uptake of enriched 13C- and 15N-compounds. We used the software Past 4.03 and set the level of significance to 95%.
Results
Performance of the photosymbionts
The results of the PAM observations after experimental start and after day 1, 3, 7 and 15 are shown in Fig. 1 (values are provided in the supplementary file). During the whole experiment, Fv/Fm of all individuals was in the range between 0.6 and 0.8, which indicates a healthy state of the photosymbionts. Two-way ANOVA of the photosynthetic area over a period of 15 days, between the dark- and light-incubated foraminifera show a significantly difference between the light cycle (p = 0.027) and time (p < 0.001) and also their interaction (p < 0.001).
Within the dark incubated foraminifera, we observed no significant change in photosynthetic area over 7 days (rm-ANOVA, p = 0.110). Just a significant increase (rm-ANOVA, p < 0.001) of the photosynthetic area was observes from day 7 to 15.
Isotopic uptake experiments
The rate of isotope incorporation differed significantly (one-way ANOVA) depending on the type of offered carbon form (carbonate > glucose, p < 0.001) and nitrogen form (nitrate > ammonium, p < 0.001). Two-way ANOVA (cycle × time) was performed to see if there are differences in the uptake of isotopes during light exposure and over time. Natural light supply in contrast to complete darkness, highly significantly increased the uptake of carbonate, nitrate and ammonium (p < 0.001) and significantly for glucose (p = 0.048). The interaction between cycle and time was significant for glucose (p = 0.020), carbonate (p < 0.001) and nitrate (p < 0.001), but not for ammonium (p = 0.164). Tracer uptake increased with time (Table 1) for all compounds under light conditions, except ammonium. Under dark conditions tracer uptake only increased for carbonate and ammonium, but not for glucose (p = 0.087) and nitrate (p = 0.376) (Table 1).
For nitrate, ammonium and carbonate, the element uptake during darkness was negligible (Fig. 2). Nitrate and carbonate uptake were higher than that of ammonium and glucose under natural light conditions (16:8 h light:dark). The uptake of nitrate and carbonate in the light was approximately twice compared to ammonium and glucose, respectively. In prolonged darkness, a substantial uptake of tracer was only recorded for glucose.
Incorporated amount of isotopes during dark and light experiments over 7 days. The blue line represents the amount of up taken N or C compound at light exposure (16:8 h light:dark), the orange line the element uptake in complete darkness. The mean of 6 individuals was used for each data point. Bars indicating standard error.
Discussion
Heterostegina depressa is known as a low light species (oligophotic), i.e. well-adapted to grow under low light conditions20. We found that the photosynthetic area of the foraminiferal symbionts remained constant over 7 days of continuous darkness and show a slightly increase between 7 and 15 days (Fig. 1). This means that even after 15 days without light, the photosymbionts of H. depressa were alive and adapt to these conditions. Interestingly, there was no uptake and assimilation of carbonate and inorganic nitrogen during this time (Fig. 2).
Past experiments with dissolved carbonate show that LBFs can take it up by diffusion26. This uptake then follows a linear increase in the C concentration in the cytoplasm of the foraminifera as a function of time. However, we were only able to record a linear increase of the C concentration in the foraminifera during the experiments, which were carried out under light exposure. The dark incubated foraminifera show no uptake of carbon, which suggests that the C uptake does not take place by diffusion but by enzymatic activity as already suspected by Ter Kuile et al.26.
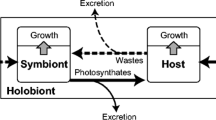
During prolonged darkness, foraminifera operate purely heterotrophic, as shown by the uptake of dissolved glucose, which was likely metabolized for energy generation in the absence of any transfer of photosynthates and other metabolites from the photosymbionts. Glucose uptake might also be promoted by the presence of bacteria, since some foraminifera also contain heterotrophic bacteria as symbionts, which can quickly digest glucose12. Another explanation could be an active uptake and digestion of enriched bacteria—its presence cannot be ruled out during an experiment for more than 3 days. Röttger et al.27 reported, that H. depressa can active feed on algae, but this food uptake just play a minor role in the energy budget of the foraminifera. It can therefore also be hypothesized that the uptake of glucose is caused indirectly by the phagocytosis of bacteria that have previously enriched themselves with 13C. The bacteria uptake and the so called “bacteria farming” is a widely known strategy of small benthic foraminifera28. At the moment, this feeding strategy was only observed from non-symbiont bearing foraminifera. However, it cannot be ruled out that bacteria settle on the surface of the foraminifera, which then metabolize glucose. The 13C-enriched metabolites of the bacteria can then be released into the culture water and absorbed through close contact by the foraminifera. In order to understand this more closely, further studies using TEM or NanoSIMS must be carried out. These studies would also clarify whether this species is able to uptake glucose via osmotrophy.
The isotope incorporation increased with time under natural light conditions (16:8 h light: dark). There was the same rising pattern for nitrate, ammonium, carbonate and glucose incorporation, which was fundamentally different from the pattern under continuous darkness. Although glucose uptake was similar, nitrate, ammonium, and carbonate uptake increased under irradiances supply substantially already after 7 days. We assume that glucose uptake is mostly driven by the heterotrophic foraminiferal host cell or by bacterial symbionts29 and used to generate energy and carbon skeletons for physiological processes. Under light supply, additional energy and organic compounds for foraminiferal growth are generated by the photobionts. Lintner et al.25 investigated the element uptake of the obligatory heterotrophic Cribroelphidium selseyense. During the first 7 days, they found only a marginal assimilation of carbonate and nitrate25, which however increased afterwards probably because of symbiotic bacterial activity. At the same time, C. selseyense showed a continuous uptake of ammonium during the whole experiment25. We conclude from our data, that assimilation of carbonate, nitrate and ammonium are light-dependent and triggered by the activity of the phototobionts, while glucose uptake is continued in darkness thus maintaining the holobionts metabolism. This helps the foraminifera to survive prolonged dark phases.
We studied nitrogen using the two inorganic compounds ammonium and nitrate and were able to prove a much higher nitrate uptake (Fig. 2). For both, nitrate and ammonium, there was no uptake in the dark, which implies that inorganic nitrogen assimilation was performed by only the photosymbionts, mediated under light supply. Interestingly, during the experiments, which were carried out completely in the dark, no uptake of nitrate was recorded (see Fig. 2). From studies with marine diatoms, however, it is known that diatoms accumulate nitrate in the cells during darkness30. This now suggests that the foraminifera itself is not active even in the dark, nor does it have osmotrophy during this period, allowing dissolved nitrate to be carried to the symbionts. Such behaviour could be compared to dormancy in foraminifera31. Dormancy can be caused by exogenous factors such as stressful environmental conditions (here lack of light during dark conditions) and leads to a strong reduction in metabolism. This hypothesis can now be reconciled very well with our results. It now appears that the here investigated foraminifera goes into a kind of dormancy during complete darkness and reduced the metabolism to an absolute minimum. However, since there is no uptake of any isotope during total darkness, this strategy applies not only to the foraminifera but also to their photosymbionts.
In general, both inorganic nitrogen forms (nitrate and ammonium) can be used by photoautotrophs (algae and higher plants) as nitrogen source32. For metabolic pathways (amino acids, proteins, nucleic acids and else), both inorganic nitrogen forms first need to be incorporated into amino acids, which in the case of nitrate requires additional reduction equivalents, energy and enzymatic reactions33. For many photoautotrophic organisms a mixture of both compounds led to the highest nitrogen uptake in plants30. Kronzucker et al.32 reported that nitrate uptake and assimilation is inhibited at high ammonium concentrations. This aspect can be excluded for our results since we incubated the foraminifera separately with nitrate and ammonium. Further, Dortch34 postulated that the preferred nitrogen source of phytoplankton is ammonium, which does not fit to our results. These differences can probably be explained by the positive effect of nitrate uptake on the cation–anion balance of phototrophic organisms (phytoplankton), allowing higher nitrogen uptake and growth rates with nitrate than with ammonium33.
In the past, some cultivation experiments were carried out with foraminifera, which had either light or temperature as a stress factor35. However, the temperature effect on LBFs is species specific and it has been shown that temperatures above 31 °C lead to a rapid death of the photosymbionts in H. depressa36. Since the light condition was constant in the experiments by Schmidt et al.36 and the temperature in our experiments, it cannot be stated which parameter has a stronger effect on the foraminifera. In order to examine this aspect more closely, cross-design experiments with 2 variables (temperature × light supply) must be carried out in the future.
It was shown that the availability of light is the essential factor for the distribution of foraminifera with depth35. Presumably not only the daylight but also the moonlight plays a role here. Observations showed that LBFs grown in the natural environment have oscillations in their chamber volume, which is probably caused by lunar and tidal cycles37. It is assumed that the lunar cycle influences the productivity of the photosymbionts in LBFs and thus has a positive effect on the activity of the symbionts at full moon night37. However, the light intensity of moonlight is much lower than that of sunlight, only about 0.0024 μmol m−2 s−138 which is around 12.5 k times lower than that in our experiment. It should be noted, that in sunlight all visible wavelengths are relatively equally present, whereas in moonlight the wavelengths are generally cantered around 400 nm38. If this wavelength-dependent irradiation affects the metabolism of H. depressa or not has not yet been investigated and could certainly shed more light on whether moonlight has an effect on the LBF symbionts.
Laboratory experiments have shown that H. depressa is a low light species21 and can therefore survive even in very poor light conditions. However, based on the results of our study, it can be clearly shown that in complete darkness the foraminifera do not absorb any essential nutrients. Recent studies have even shown that sequestered chloroplasts in foraminifera degrade within a few days when exposed to high light conditions and also have a photobleaching effect39. Even foraminifera, which have neither photosymbionts nor sequestered chloroplasts, can cope better with less light than with high light intensities40. All of these results and the data from this study suggest that high light intensities has a significant negative effect on their metabolism, but light is an essential factor for foraminifera with photosymbionts to survive.
Conclusion
The uptake of carbonate, nitrate, ammonium and glucose in H. depressa is highly dependent on the availability of light. Under dark conditions, the organisms take up mainly glucose to provide energy for maintaining the metabolic processes. If foraminifera are exposed to light, the photosymbionts are primarily responsible for uptake and assimilation of carbonate, nitrate and ammonium. Based on these results, in future uptake experiments with H. depressa it is recommended to enrich the culture water with carbonate and ammonium nitrate in order to offer best conditions to study the activity of the foraminifera and their symbionts with changing environmental conditions.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Change history
06 June 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41598-023-36353-4
References
Wernberg, T. et al. An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nat. Clim. Change 3(1), 78–82 (2013).
Schmidt, C., Kucera, M. & Uthicke, S. Combined effects of warming and ocean acidification on coral reef Foraminifera Marginopora vertebralis and Heterostegina depressa. Coral Reefs 33(3), 805–818 (2014).
Reymond, C. E., Lloyd, A., Kline, D. I., Dove, S. G. & Pandolfi, J. M. Decline in growth of foraminifer Marginopora rossi under eutrophication and ocean acidification scenarios. Glob. Change Biol. 19(1), 291–302 (2013).
Uthicke, S., Vogel, N., Doyle, J., Schmidt, C. & Humphrey, C. Interactive effects of climate change and eutrophication on the dinoflagellate-bearing benthic foraminifer Marginopora vertebralis. Coral Reefs 31(2), 401–414 (2012).
Hikami, M. et al. Contrasting calcification responses to ocean acidification between two reef foraminifers harboring different algal symbionts. Geophys. Res. Lett. 38(19), L19601 (2011).
Yamamoto, S. et al. Threshold of carbonate saturation state determined by CO2 control experiment. Biogeosciences 9(4), 1441–1450 (2012).
Baker, R. D., Hallock, P., Moses, E. F., Williams, D. E. & Ramirez, A. Larger foraminifers of the Florida reef tract, USA: Distribution patterns on reef-rubble habitats. J. Foraminifer. Res. 39(4), 267–277 (2009).
Lintner, M., Schagerl, M., Lintner, B., Nagy, M. & Heinz, P. Photosynthetic performance of symbiont-bearing foraminifera Heterostegina depressa affected by sunscreens. Sci. Rep. 12(1), 1–7 (2022).
Hallock, P., Lidz, B. H., Cockey-Burkhard, E. M. & Donnelly, K. B. Foraminifera as bioindicators in coral reef assessment and monitoring: The FORAM index. Environ. Monit. Assess. 81(1), 221–238 (2003).
Hallock, P. Symbiont-bearing foraminifera. In Modern foraminifera 123–139 (Springer, Dordrecht, 1999).
Langer, M. R. Assessing the contribution of foraminiferan protists to global ocean carbonate production 1. J. Eukaryot. Microbiol. 55(3), 163–169 (2008).
Prazeres, M. & Renema, W. Evolutionary significance of the microbial assemblages of large benthic Foraminifera. Biol. Rev. 94(3), 828–848 (2019).
Renema, W. Terrestrial influence as a key driver of spatial variability in large benthic foraminiferal assemblage composition in the Central Indo-Pacific. Earth Sci. Rev. 177, 514–544 (2018).
Hallock, P. Algal symbiosis: A mathematical analysis. Mar. Biol. 62(4), 249–255 (1981).
Lee, J. J. Algal symbiosis in larger foraminifera. Symbiosis (2006).
Bernhard, J. M., Edgcomb, V. P., Casciotti, K. L., McIlvin, M. R. & Beaudoin, D. J. Denitrification likely catalyzed by endobionts in an allogromiid foraminifer. ISME J. 6(5), 951–960 (2012).
Lee, J. J. & Anderson, O. R. Biology of Foraminifera (Academic Press, 1991).
Pecheux, M. J. F. Ecomorphology of a recent largeforaminifer, Operculina ammonoides. Geobios 28(5), 529–566 (1995).
Hallock, P. Light dependence in Amphistegina. J. Foramin. Res. 11(1), 40 (1981).
Nobes, K., Uthicke, S. & Henderson, R. Is light the limiting factor for the distribution of benthic symbiont bearing foraminifera on the Great Barrier Reef?. J. Exp. Mar. Biol. Ecol. 363(1–2), 48–57 (2008).
Röttger, R. Ecological observations Heterostegina depressa (Foraminifera, Nummulitidae) in the laboratory and its natural habitat. In International Symposium of Benthic Foraminifera of Continental Margins, vol. 1, 75–79 (1976).
Uthicke, S. & Altenrath, C. Water column nutrients control growth and C: N ratios of symbiont-bearing benthic foraminifera on the Great Barrier Reef, Australia. Limnol. Oceanogr. 55(4), 1681–1696 (2010).
Fujita, K., Omori, A., Yokoyama, Y., Sakai, S. & Iryu, Y. Sea-level rise during Termination II inferred from large benthic foraminifers: IODP Expedition 310, Tahiti Sea Level. Mar. Geol. 271(1–2), 149–155 (2010).
Parkhill, J., Mailett, G. & Cullen, J. Fluorescence-based maximal quantim yield fpr PSII as a diagnostic of nutrient stress. J. Phycol. 37, 517–529 (2001).
Lintner, M. et al. Assimilation of particular organic matter and dissolved organic or inorganic compounds by Cribroelphidium selseyense (Foraminifera). Front. Mar. Sci. 8, 1709 (2021).
Ter Kuile, B., Erez, J. & Padan, E. Mechanisms for the uptake of inorganic carbon by two species of symbiont-bearing foraminifera. Mar. Biol. 103(2), 241–251 (1989).
Röttger, R., Krüger, R. & de Rijk, S. Trimorphism in foraminifera (Protozoa)—Verification of an old hypothesis. Eur. J. Protistol. 25(3), 226–228 (1990).
Langer, M. R. & Gehring, C. A. Bacteria farming; a possible feeding strategy of some smaller motile Foraminifera. J. Foraminifer. Res. 23(1), 40–46 (1993).
Bernhard, J. M., Tsuchiya, M. & Nomaki, H. Ultrastructural observations on prokaryotic associates of benthic foraminifera: Food, mutualistic symbionts, or parasites?. Mar. Micropaleontol. 138, 33–45 (2018).
Raimbault, P. & Mingazzini, M. Diurnal variations of intracellular nitrate storage by marine diatoms: Effects of nutritional state. J. Exp. Mar. Biol. Ecol. 112(3), 217–232 (1987).
Ross, B. J. & Hallock, P. Dormancy in the foraminifera: A review. J. Foraminifer. Res. 46(4), 358–368 (2016).
Hachiya, T. & Sakakibara, H. Interactions between nitrate and ammonium in their uptake, allocation, assimilation, and signaling in plants. J. Exp. Bot. 68(10), 2501–2512 (2017).
Marschner, H. (ed.) Marschner’s Mineral Nutrition of Higher Plants (Academic press, 2011).
Dortch, Q. The interaction between ammonium and nitrate uptake in phytoplankton. Mar. Ecol. Prog. Ser. Oldendorf 61(1), 183–201 (1990).
Fujita, K., Kanda, Y. & Hosono, T. Light is an important limiting factor for the vertical distribution of the largest extant benthic foraminifer Cycloclypeus carpenteri. J. Earth Sci. 33(6), 1460–1468 (2022).
Schmidt, C., Heinz, P., Kucera, M. & Uthicke, S. Temperature-induced stress leads to bleaching in larger benthic foraminifera hosting endosymbiotic diatoms. Limnol. Oceanogr. 56(5), 1587–1602 (2011).
Eder, W., Briguglio, A. & Hohenegger, J. Growth of Heterostegina depressa under natural and laboratory conditions. Mar. Micropaleontol. 122, 27–43 (2016).
Breitler, J. C. et al. Full moonlight-induced circadian clock entrainment in Coffea arabica. BMC Plant Biol. 20(1), 1–11 (2020).
Lintner, M., Wildner, M., Lintner, B., Wanek, W. & Heinz, P. Spectroscopic analysis of sequestered chloroplasts in Elphidium williamsoni (Foraminifera). J. Photochem. Photobiol. B 238, 112623 (2023).
Lintner, M. et al. Effect of light on the metabolism of the foraminifera Cribroelphidium selseyense lacking photosymbionts and kleptoplasts. J. Photochem. Photobiol. 11, 100133 (2022).
Author information
Authors and Affiliations
Contributions
ML selected and cultivated the foraminifera and performed the experiments. BL supported the experimental effort and helped write the manuscript. MS organized the measurements using PAM and carefully read the manuscript. WW organized the isotope measurements and helped to evaluate this data and improved the manuscript. PH enabled the cultivation of the foraminifera in their laboratory and helped with both the manuscript and the organization of the experiments.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: In the original version of this Article, Michael Lintner was incorrectly listed as a corresponding author. The correct corresponding author for this Article is Petra Heinz. Correspondence and request for materials should be addressed to petra.heinz@univie.ac.at.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lintner, M., Lintner, B., Schagerl, M. et al. The change in metabolic activity of a large benthic foraminifera as a function of light supply. Sci Rep 13, 8240 (2023). https://doi.org/10.1038/s41598-023-35342-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-35342-x
- Springer Nature Limited