Abstract
Laser therapy is the most effective treatment considered for retinopathy of prematurity (ROP). We compared the foveal morphology of the retina in eyes with a history of ROP to that of full-term children. This cross-sectional comparative study included 74 patients with a history of ROP, aged 4–6 years. Among them, 41 underwent laser treatment for ROP. The clinical findings and retinal morphology in these patients were compared to that of 33 patients who had spontaneous ROP regression and 30 age-matched full-term controls. All the patients with ROP had 20/40 or better best-corrected visual acuity (BCVA). The foveal thickness was significantly thicker in laser-treated ROP eyes than in regressed ROP eyes and controls. The outer nuclear layer was significantly thicker, and the inner segment (IS) of the photoreceptors and the inner retinal layer were significantly thicker in the laser-treated ROP eyes than that in the control eyes. In the patients with ROP and controls, better BCVA was associated positively with deeper foveal depression, which was associated with a later gestational age. Our results suggest that prematurity and laser treatment affect the foveal morphology and BCVA.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Retinopathy of prematurity (ROP), which warrants treatment, has increased with the development of perinatal care1. ROP has a higher incidence of myopia, amblyopia, retinal detachment, strabismus, cataracts, glaucoma, and macular folds1,2,3,4,5. Angiogenesis in the retina begins from the posterior pole at 24–28 weeks of gestational age, and the peripheral retina is finally vascularized at 40 weeks6. Thus, retinal development, including the migration and redistribution of the photoreceptor cells, occurs ex utero in premature infants7.
Spectral-domain optical coherence tomography (SD-OCT) studies have demonstrated several microstructural abnormalities in the eyes of premature infants, such as retention of the inner retinal layers (IRL) at the fovea and an absence of foveal depression in individuals aged 2–18 years with a history of premature birth8,9,10,11,12.
Laser therapy is considered to be the most effective treatment for ROP2,4. However, laser therapy destroys the peripheral retina and retards abnormal growth of peripheral blood vessels2. There is no consensus regarding the effect of laser treatment on retinal immaturity; however, the signs of immaturity could be a predictor of visual function.
Therefore, this study aimed to determine whether laser photocoagulation affects retinal maturation. To accomplish this, we analyzed the foveal morphology in children treated with laser photocoagulation for ROP. The findings in these children were compared to those in children whose ROP spontaneously regressed and age-matched full-term children.
Results
Seventy-four children with a history of ROP were divided into two groups: the laser-treated ROP group and the spontaneously regressed ROP group. The laser-treated ROP group consisted of 41 patients, including 20 boys and 21girls, and the mean age was 5.0 ± 0.8 years (mean ± standard deviation). The spontaneously regressed ROP group consisted of 33 patients (18 boys and 15 girls) with a mean age of 4.9 ± 0.8 years. The control group consisted of 30 children (12 boys and 18 girls) with a mean age of 5.0 ± 1.4 years (Table1).
The age and sex distributions were not significantly different between the patients with ROP and the control group (analysis of variance, ANOVA).
The gestational age and birth weight were significantly lower in the laser-treated ROP group than that in the spontaneously regressed ROP and control groups (P = 0.0001, ANOVA).
The mean better best-corrected visual acuity (BCVA) at the initial examination was 0.04 ± 0.13, 0.06 ± 0.18, and − 0.04 ± 0.05 logarithm of the minimum angle of resolution (logMAR) units in the laser-treated, regressed ROP, and control groups, respectively. The BCVA was significantly better in the control group (P = 0.009, ANOVA). All the patients had a BCVA of 20/40 or better.
The mean refractive error was − 1.50 ± 3.00, + 1.00 ± 1.75, and + 0.73 ± 1.41 diopter (D) in the laser-treated, regressed, and D control eyes, respectively (P = 0.090, ANOVA). The mean axial length was 21.7 ± 1.3, 21.4 ± 0.6, and 22.2 ± 0.9 mm in the treated, regressed, and control eyes, respectively (P = 0.006, ANOVA; Table 1). In the laser-treated ROP group, ROP was treated at stage 3 in all the eyes. In the regressed ROP group, ROP was spontaneously regressed at stage 0, 1, 2, and 3 in 10, 9, 5, and 7 eyes, respectively.
Foveal thickness
The mean foveal thickness was 264.0 ± 26.0, 241.1 ± 22.9, and 214.4 ± 13.4 µm in the laser-treated, spontaneously regressed, and control eyes, respectively (P = 0.0001, ANOVA; Table 2). The fovea was significantly thicker in the laser-treated eyes compared to the regressed and control eyes. The inter-observer reproducibility was excellent (intraclass correlation coefficients = 0.91).
The mean foveal depression was 59.0 ± 31.0, 75.3 ± 34.7, and 124.5 ± 17.1 µm in the treated, regressed, and control eyes, respectively. The foveal depression was significantly shallower in the laser-treated eyes than that in the regressed and control eyes (P = 0.0001, ANOVA; Table 2). The mean thickness of the outer nuclear layer (ONL) was significantly thicker in the treated eyes than that in the regressed eyes and control eyes (P = 0.0001, ANOVA; Table 2). The length of the inner segment (IS) was significantly thicker in the treated and regressed ROP eyes than that in the control eyes (P = 0.0001, ANOVA; Table 2). IRL was significantly thicker in the laser-treated eyes than that in the regressed eyes and control eyes (P = 0.0001, ANOVA; Table 2). IRL was detected in 51.2% of the treated eyes and 12.0% of the regressed eyes; thus, IRL was significantly preserved in the laser-treated eyes, whereas it was not detected in the control eyes (P = 0.0001, χ2-test; Table 2). The resistance of IRL was attributed to a delay in foveal maturation9. In the analysis of covariance (ANCOVA) adjusted with the gestational age and ROP, the foveal, IS, and IRL thickness was thicker in the laser treated group than regressed ROP and control group (Table 3).
In the univariate analysis, better BCVA was positively associated with mature gestational age, larger birth weight, smaller refractive error, deeper foveal depression, and thinner IS thickness in ROP treated eyes (Pearson’s coefficient; Table 4). In the regressed and control eyes, BCVA was not associated with the foveal morphology (Pearson’s coefficient; Table 4). In the multiple linear regression analysis of ROP and controls, better BCVA was significantly associated with deeper foveal depression (Standardized β = − 0.278, P = 0.039; Table 5). In the multiple linear regression analysis of the ROP and control eyes, deeper foveal depression was significantly associated with mature gestational age (Standardized β = 0.733, P = 0.008; Table 6).
Representative ROP patient
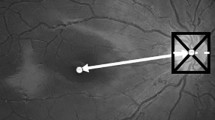
Case 1: A 6-year-old boy was born at 25 weeks gestation and weighed 690 g at birth. He was classified with Zone 1 and stage 3 ROP with plus disease. The patient was treated with laser photocoagulation at 33-weeks-of-age. His BCVA was 20/40 Snellen equivalent in both eyes. The foveal depression was shallow at 22.5 µm in the OCT images, and the IRL of the fovea was preserved (Fig. 1A). The fovea was 239 µm thick. The ONL was 133 µm, IS was 50 µm, Outer segment (OS) was 40 µm, and IRL was 33 µm.
(A) Optical coherence tomography (OCT) images of a representative patient with retinopathy of prematurity (ROP) who underwent laser treatment. The foveal depression is shallow, at 22.5 µm in the OCT images, and IRL of the fovea is preserved (Fig. 1A). The inner retinal layer (blue line) at the fovea is still present. The foveal thickness (inner retinal layer and outer retinal layer, i.e., blue and red line, respectively) is 239 µm, the IS and OS lengths are 50 and 40 µm, respectively. Yellow line: foveal depression; blue line: inner retinal layer; red line: outer retinal layer; green line: IS length. (B) OCT image of a representative eye from the spontaneously regressed ROP group. The foveal depression is 108.5 µm in the OCT images (Fig. 1B). IRL of the fovea is not detected and the foveal thickness (red line, outer retinal layer) is 237 µm. IS length is 44 µm and OS length is 43 µm. Yellow line: foveal depression, red line: outer retinal layer, green line: IS length.
Case 2: A 4-year-old girl was born at 24 weeks gestation and weighed 640 g at birth. She was classified as Zone 3 stage 3 ROP without plus disease. ROP spontaneously regressed after 44 weeks in this case. Her BCVA was 20/25 Snellen equivalent in both eyes. The foveal depression measured 108.5 µm in the OCT images (Fig. 1B). IRL of the fovea was not detected and the foveal thickness was 218 µm. The ONL was 92 µm, IS was 44 µm, OS was 43 µm, and IRL was not detected.
The foveal depression was deeper, IRL was thinner, and the visual acuity was better in the second case than that in the first treated case.
Discussion
We found that the patients with a history of ROP demonstrated macular morphological differences, such as, retention of IRL, shallower foveal depression, and thicker ONL and IS, compared to full-term control children. These findings suggest an alteration in the retinal developmental process in eyes with ROP.
OCT findings
We observed a thicker ONL and resistance of IRL in children with ROP, which is in agreement with the results of previous reports9,13,14. Preterm birth between 24 and 28 weeks of gestation has been proposed to be a critical period associated with failure of normal migration of IRL away from the fovea, resulting in increased foveal thickness and resistance of IRL13,14. The migration of IRL appears to be independent of the outer retinal development, as previously suggested15. This disparity in maturation could be associated with the variation in the vascular supply between the inner and outer retina. The vasculature of the outer retina develops during the late stage of gestation16. IRL matures completely at the time of full-term birth; however, maturity of the outer retina is not attained until a few years after birth13,14. The maturation of the outer retina includes the development of the IS and OS layers, which occurs after birth and continues up to 5 years of age7,17. These processes are disrupted by preterm birth and ROP. Thicker ONL and IS in children with ROP could be attributed to a delay in foveal maturation18.
Visual acuity.
BCVA of the treated and regressed ROP patients was worse than that of the control children, although all the patients with ROP had 20/40 or better BCVA. In previous reports, some studies found that a thicker fovea resulted in poorer visual acuity in childhood10,19. Another study found no relationship between foveal thickness and visual acuity20.There is no consensus regarding the relationship between visual acuity and infant prematurity. In our study, BCVA of the treated ROP patients was significantly associated with early preterm birth, light birth weight, large refractive error,, thick IS, and shallow foveal depression. Early preterm birth and light birth weight are signs of infant prematurity. Large refractive error in patients with ROP is affected by laser treatment10,19. Shallow foveal depression and thicker IS in patients with ROP were signs of immaturity of the foveal development18. In our study, thicker IS and foveal thickness was significantly associated with the laser treatment. Thus, BCVA is related to infant prematurity, foveal prematurity, and laser treatment.
The laser treatment of ROP
IS in the eyes of patients with ROP was thicker than that in the control eyes. Premature neonates had a thin photoreceptor layer at the fovea on the SD-OCT images7. These studies demonstrated the presence of significant foveal cone specialization in the absence of a foveal pit21. The migration of the cells in IRL was independent of the photoreceptor development in the outer retinal layer15. The results of a recent study showed that maturation of the outer retinal layer and photoreceptors is delayed in laser-treated ROP eyes22. Vogel et al. reported that infants treated with intravitreal injection of bevacizumab (IVB) for ROP had more rapid outer retinal thickening at the fovea, while those treated with laser therapy showed delayed development of the ellipsoid zone at the fovea22. However, the structure–function correlations in these regions remain poorly understood. We studied children aged 4–6 years when the retina is still developing16,23. In these ages, foveal development can last, and the outer retina of patients with ROP is still developing23. Celik G et al. reported that at 5 years of age, foveal thickness was significantly thicker in laser treated ROP eyes than in IVB treated ROP eyes24. In this report, foveal morphology was not discussed. Our results suggest that infant prematurity and laser treatment affect foveal morphology. Deeper foveal depression was associated with mature gestational age and laser treatment, which affected the foveal, IRL, and IS thickness.
Limitations
This study had certain limitations. The number of patients with ROP and controls was small. In addition, all the participants were Japanese. Considering previous studies, Hispanic neonates reportedly have higher rates of severe ROP compared with non-Hispanic neonates, while non-Hispanic Black neonates reportedly have lower rates of ROP than non-Hispanic White neonates25,26.
We manually measured the retinal layers owing to segmentation errors during the use of automated software27. The retinal thickness was measured using Spectralis OCT and Cirrus OCT. The differences between the two SD-OCT systems were owing to the differences in their built-in software algorithms28; we measured the retinal thickness using ImageJ software. We did not have an additional IVB group.
Conclusion
In conclusion, the morphology of the fovea in the eyes of children with laser-treated ROP was significantly different from that of spontaneously regressed ROP and control eyes. In the patients with ROP and controls, better BCVA was associated with deeper foveal depression, which was associated with mature gestational age. Our results suggest that infant prematurity and laser treatment affect foveal morphology and visual acuity.
Methods
This multicenter retrospective study included patients with a history of ROP who were diagnosed and followed up at the Nara Medical University Hospital, Tokushima University Hospital, Hyogo Medical University Hospital, Kobe University Hospital, Sapporo City General Hospital, Shiga Medical University Hospital, and Kurume University Hospital between April 2012 and December 2019.
The protocol for this study conformed to the tenets of the Declaration of Helsinki and was approved by the Internal Review Boards of the Nara Medical University, Tokushima University, Hyogo Medical University, Kobe University Hospital, Sapporo City General Hospital, Shiga Medical University Hospital, and Kurume University Hospital. For research involving human participants who were minors, informed consent was obtained from their parents for permitting performing measurements and reviewing their medical records.
All the patients had a history of ROP and were followed up for 4–6 years. Patients who were capable of cooperating with the OCT examinations were included. Patients with organic eye diseases, history of intraocular surgery, cataracts, glaucoma, or any retinal disorders were excluded. During acute phase ROP, ROP was examined at least at one visit per week. Laser-treated ROP patients were treated at the pre-threshold ROP or threshold ROP stage29. Laser treatment was performed in all the quadrants in the laser treated eyes. The patients with spontaneously regressed ROP presented with stage 3 or less, and none of them had received any treatment for ROP previously.
Age-matched, full-term children with normal ocular findings were recruited from the Nara Medical University Hospital and the nursery school of the Tokushima University Hospital for the control group. In control group, children with myopia greater than -1.00 D or with hyperopia greater than + 2.00 D were excluded.
All the patients and controls were examined using slit-lamp biomicroscopy, extraocular motility assessment, subjective cycloplegic refractions (1% cyclopentolate and 2.5% phenylephrine), dilated ophthalmoscopy, and SD-OCT imaging. The same examination procedures were used for both the eyes of the patients with ROP and the right eyes of the controls. One severe eye per patient was selected for the analysis.
The retinal thickness, axial length, and BCVA of 18 eyes of the control patients have been reported30,31,32. The visual acuity was measured using a standard Snellen chart at 5 months and was converted to the logMAR units for statistical analyses. The axial length was measured using IOL Master (Carl Zeiss Meditec, Jena, Germany) and AL-2000 (TOMEY, Nagoya, Japan). The refractive error (spherical equivalent), gestational age, birth weight, sex, and history of laser treatment were also evaluated.
Measurements of retinal layer thickness
The retinal layer thickness was determined using SD-OCT (Spectralis, Heidelberg Engineering, Heidelberg, Germany; Cirrus SD-OCT, Carl Zeiss Meditec, Jena, Germany and RS-3000, Nidek, Gamagori, Japan). The thicknesses of the fovea, IRL, ONL, and photoreceptor IS and OS layers were measured in the temporally scanned OCT images of the fovea. The thickness measurements at 1 mm nasal and temporal to the fovea were also measured as the parafoveal thickness. The foveal depression was calculated by subtracting the central foveal thickness from the mean parafoveal thickness.
The thickness of each retinal layer was determined by two experienced retinal specialists who were blinded to the diagnosis. They evaluated the retinal thickness independently and manually using an open-access software ImageJ (version 1.50a; NIH, Bethesda, Maryland, USA). The ONL thickness was measured as the distance from the outer border of the inner limiting membrane (ILM) to the external limiting membrane (ELM). The IS length was measured as the distance between the ELM and outer border of the ellipsoid zone (EZ). The OS length was measured as the distance between the outer border of the EZ and the inner border of the retinal pigment epithelium. The arithmetic means of the two examiners were used for the statistical analyses. The inter-observer reproducibility was evaluated using intraclass correlation coefficients.
Statistical analyses
The data are expressed as means ± standard deviation. The gestational birth date, age at the time of the examination, BCVA, axial length, refractive error (spherical equivalent), and foveal structures of the ROP and control eyes were compared using one-way ANOVA with post-hoc Tukey tests and ANCOVA adjusted with the gestational age. The presence of the IRL was compared using theχ2-test. Univariate and multivariate linear regression analyses were performed to determine the significance of the correlations between the BCVA and OCT findings of ROP and control eyes. The potential confounders (gestational age, birth weight, axial length, laser treatment, and ROP stage) were included in the multivariate linear regression analyses. The standardized coefficients (ß) were calculated for each independent variable. Statistical significance was set at p < 0.05. Statistical analyses were performed using licensed statistical software (SPSS version 22.0; SPSS Inc., Chicago, IL, USA).
Data availability
The data that support the findings of this study are available from the corresponding author, T.N., upon reasonable request.
References
Smith, B. T. & Tasman, W. S. Retinopathy of prematurity: late complications in the baby boomer generation (1946–1964). Trans. Am. Ophthalmol. Soc. 103, 225–34 (2005).
Wheeler, D. T. et al. Retinopathy of prematurity in infants weighing less than 500 grams at birth enrolled in the early treatment for retinopathy of prematurity study. Ophthalmology 118, 1145–1151. https://doi.org/10.1016/j.ophtha.2010.09.031 (2011).
Hartnett, M. E., Gilbert, M. M., Hirose, T., Richardson, T. M. & Katsumi, O. Glaucoma as a cause of poor vision in severe retinopathy of prematurity. Graefes Arch. Clin. Exp. Ophthalmol. 231, 433–438. https://doi.org/10.1007/BF02044227 (1993).
Salvin, J. H., Lehman, S. S., Jin, J. & Hendricks, D. H. Update on retinopathy of prematurity: Treatment options and outcomes. Curr. Opin. Ophthalmol. 21, 329–334. https://doi.org/10.1097/ICU.0b013e32833cd40b (2010).
Moskowitz, A., Hansen, R. M. & Fulton, A. B. Retinal, visual, and refractive development in retinopathy of prematurity. Eye Brain 8, 103–111. https://doi.org/10.2147/EB.S95021 (2016).
Provis, J. M. Development of the primate retinal vasculature. Prog. Retin. Eye Res. 20, 799–821. https://doi.org/10.1016/s1350-9462(01)00012-x (2001).
Maldonado, R. S. et al. Dynamics of human foveal development after premature birth. Ophthalmology 118, 2315–2325. https://doi.org/10.1016/j.ophtha.2011.05.028 (2011).
Villegas, V. M., Capó, H., Cavuoto, K., McKeown, C. A. & Berrocal, A. M. Foveal structure function correlation in children with history of retinopathy of prematurity. Am. J. Ophthalmol. 158, 508-512.e2. https://doi.org/10.1016/j.ajo.2014.05.017 (2014).
Bowl, W. et al. OCT-based macular structure-function correlation in dependence on birth weight and gestational age-the Giessen long-term ROP study. Invest. Ophthalmol. Vis. Sci. 57, 235–241. https://doi.org/10.1167/iovs.15-18843 (2016).
Stoica, F. et al. Clinical relevance of retinal structure in children with laser-treated retinopathy of prematurity versus controls - using optical coherence tomography. Acta Ophthalmol. 96, e222–e228. https://doi.org/10.1111/aos.13536 (2018).
McLoone, E., O’Keefe, M., McLoone, S. & Lanigan, B. Long term functional and structural outcomes of laser therapy for retinopathy of prematurity. Br. J. Ophthalmol. 90, 754–759. https://doi.org/10.1136/bjo.2005.068304 (2006).
Gunn, D. J., Cartwright, D. W. & Gole, G. A. Prevalence and outcomes of laser treatment of aggressive posterior retinopathy of prematurity. Clin. Exp. Ophthalmol. 42, 459–465. https://doi.org/10.1111/ceo.12280 (2014).
Yanni, S. E. et al. Foveal avascular zone and foveal pit formation after preterm birth. Br. J. Ophthalmol. 96, 961–966. https://doi.org/10.1136/bjophthalmol-2012-301612 (2012).
Park, K. A. & Oh, S. Y. Analysis of spectral-domain optical coherence tomography in preterm children: Retinal layer thickness and choroidal thickness profiles. Invest. Ophthalmol. Vis. Sci. 53, 7201–7207. https://doi.org/10.1167/iovs.12-10599 (2012).
Thomas, M. G. et al. Structural grading of foveal hypoplasia using spectral-domain optical coherence tomography a predictor of visual acuity?. Ophthalmology 118, 1653–1660. https://doi.org/10.1016/j.ophtha.2011.01.028 (2011).
Provis, J. M. & Hendrickson, A. E. The foveal avascular region of developing human retina. Arch. Ophthalmol. 126, 507–511. https://doi.org/10.1001/archopht.126.4.507 (2008).
Vajzovic, L. et al. Delay in photoreceptor development in very preterm compared to term infants. Invest. Ophthalmol. Vis. Sci. 56, 908–913. https://doi.org/10.1167/iovs.14-16021 (2015).
Bringmann, A. et al. Muller cells in the healthy and diseased retina. Prog. Retin. Eye Res. 25, 397–424. https://doi.org/10.1016/j.preteyeres.2006.05.003 (2006).
Balasubramanian, S. et al. Relationship between retinal thickness profiles and visual outcomes in young adults born extremely preterm: The EPICure@19 study. Ophthalmology 126, 107–112. https://doi.org/10.1016/j.ophtha.2018.07.030 (2019).
Akerblom, H., Larsson, E., Eriksson, U. & Holmström, G. Central macular thickness is correlated with gestational age at birth in prematurely born children. Br. J. Ophthalmol. 95, 799–803. https://doi.org/10.1136/bjo.2010.184747 (2011).
McAllister, J. T. et al. Arrested development: High-resolution imaging of foveal morphology in albinism. Vision Res. 50, 810–817. https://doi.org/10.1016/j.visres.2010.02.003 (2010).
Vogel, R. N. et al. Foveal development in infants treated with bevacizumab or laser photocoagulation for retinopathy of prematurity. Ophthalmology 125, 444–452. https://doi.org/10.1016/j.ophtha.2017.09.020 (2018).
Lee, Y. S. et al. Comparison of foveal thickness in preschool children with a history of retinopathy of prematurity and laser photocoagulation or anti-vascular endothelial growth factor treatment: A prospective, longitudinal study. Br. J. Ophthalmol. 106, 106–112. https://doi.org/10.1136/bjophthalmol-2020-317284 (2022).
Celik, G., Gunay, M. & Kizilay, O. Swept-source optical coherence tomography findings in premature children with a history of retinopathy of prematurity at 5 years of age. Arq. Bras. Oftalmol. 83, 490–496 (2020).
Ying, G. S., Bell, E. F., Donohue, P., Tomlinson, L. A. & Binenbaum, G. G-ROP research group. Perinatal risk factors for the retinopathy of prematurity in postnatal growth and Rop study. Ophthalmic Epidemiol. 26, 270–278 (2019).
Karmouta, R. et al. Association between social determinants of health and retinopathy of prematurity outcomes. JAMA Ophthalmol. 140, 496–502 (2022).
Chen, Y. H. et al. Outer retinal structural alternation and segmentation errors in optical coherence tomography imaging in patients With a history of retinopathy of prematurity. Am. J. Ophthalmol. 166, 169–180. https://doi.org/10.1016/j.ajo.2016.03.030 (2016).
Sander, B., Al-Abiji, H. A., Kofod, M. & Jørgensen, T. M. Do different spectral domain OCT hardwares measure the same? Comparison of retinal thickness using third-party software. Graefes Arch. Clin. Exp. Ophthalmol. 253, 1915–1921. https://doi.org/10.1007/s00417-015-3075-2 (2015).
Good, W. V. & Early Treatment for Retinopathy of Prematurity Cooperative Group. Final results of the early treatment for retinopathy of prematurity (ETROP) randomized trial. Trans. Am. Ophthalmol. Soc. 102, 233–248 (2004).
Nishi, T., Ueda, T., Hasegawa, T., Miyata, K. & Ogata, N. Choroidal thickness in children with hyperopic anisometropic amblyopia. Br. J. Ophthalmol. 98, 228–232. https://doi.org/10.1136/bjophthalmol-2013-303938 (2014).
Nishi, T. et al. Choroidal structure in children with Anisohypermetropic amblyopia determined by binarization of optical coherence tomographic images. PLoS ONE 11, e0164672. https://doi.org/10.1371/journal.pone.0164672 (2016).
Nishi, T., Ueda, T., Hasegawa, T., Miyata, K. & Ogata, N. Retinal thickness in children with anisohypermetropic amblyopia. Br. J. Ophthalmol. 99, 1060–1064. https://doi.org/10.1136/bjophthalmol-2014-305685 (2015).
Funding
There were no grants and funds received for this work from any of the following organizations: National Institutes of Health (NIH); Welcome Trust; Howard Hughes Medical Institute (HHMI); and other(s). None declared under financial, general, and institutional competing interests for this study.
Author information
Authors and Affiliations
Contributions
T.N., T.U., Y.M. and N.O. wrote the manuscript. T.N., K.S., N.K., A.M., T.K., S.O., and T.T. collected data. Y.M., F.G., M.N., M.O. and S.Y. revised the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nishi, T., Ueda, T., Mizusawa, Y. et al. Effect of foveal morphology on visual acuity in 4–6-year-old children with retinopathy of prematurity: a J-CREST study. Sci Rep 12, 16431 (2022). https://doi.org/10.1038/s41598-022-20956-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-20956-4
- Springer Nature Limited