Abstract
To monitor vulnerability of countries to poliovirus (PV) outbreaks, serosurveillance of anti-PV neutralization antibody is conducted by conventional PV neutralization test (cPNT), which uses live PV strains. We previously developed a pseudovirus PV neutralization test (pPNT) as an alternative to cPNT, which uses PV pseudovirus that expresses luciferase as a reporter in the infection without producing infectious PV. In the present study, we established a high-throughput pPNT (HTpPNT) for a large-scale serosurveillance. The HTpPNT system was evaluated with 600 human serum samples obtained from a broad range of age groups of healthy volunteers (ages of 0–89 years). HTpPNT showed high correlation with cPNT (R2 for anti-type 1, 2, and 3 PV neutralization antibody titres are 0.90, 0.84, and 0.90, respectively). By using HTpPNT, we analyzed relative neutralizing antibody titre of the sera against a type 1 PV wild-type strain (Mahoney strain) to that against the type 1 Sabin strain. As a result, a correlation between the age (≥ 60 years) and the relative neutralizing antibody titre was observed (n = 15–16, P = 0.0000023–0.041), while the types of PV vaccine (i.e., oral PV vaccine and Sabin strain-based IPV) had no effect. HTpPNT would serve as a useful alternative to cPNT in a large-scale serosurveillance.
Similar content being viewed by others
Introduction
Poliovirus (PV) is a small non-enveloped virus with a positive-sense single-stranded RNA genome of approximately 7500 nt and belongs to the genus Enterovirus, the family Picornaviridae. PV is the causative agent of poliomyelitis, and is the target of the global eradication led by the World Health Organization (WHO) since 1988. Through the global vaccination program with live oral PV vaccine (OPV) and/or inactivated PV vaccine (IPV), as of 2022, only Pakistan and Afganistan remain as the endemic countries of wild PV. However, imported cases are still detected, such as the recent reports from Malawi and Mozambique. In Japan, Sabin strain-based IPV (sIPV) has been introduced in routine immunization since 2012 followed by the concomitant elimination of the vaccine-associated paralytic poliomyelitis cases1.
To monitor potential vulnerability to a poliomyelitis outbreak in Japan, annual national serosurveillance has been implemented in prefectural public health institutes since 1962, and approximately 1100–1800 individuals in 6–8 prefectures in a wide range of age groups (ages of 0–> 60) have been tested every year2. Until 1983, the serosurveillance was performed by using wild-type PV strains as the challenge viruses, which were then replaced with attenuated vaccine strains (Sabin strains). In 2015, the WHO published the third edition of the WHO Global Action Plan to minimize poliovirus facility-associated risk after type-specific eradication of wild polioviruses and sequential cessation of oral polio vaccine use (GAP III)3, which was recently revised as GAP IV4. The GAP III/ IV restricts the use of type 2 PV strains, including the Sabin 2 strain that causes the vast majority of circulating vaccine-derived PV (cVDPV) cases5, resulting in cessation of conventional PV neutralization test (cPNT) with the type 2 strains in the prefectural public health institutes in Japan; therefore, since 2017, the test has begun to be conducted only in the National Institute of Infectious Diseases. Subsequently, commercial services for the PV neutralization test in Japan have closed since 2019.
Under these circumstances, pseudovirus PV neutralization test (pPNT) has been established as an alternative to cPNT6,7,8,9,10. pPNT utilizes PV pseudovirus (PVpv) that possesses a luciferase-encoding PV replicon in the capsid and shows single cycle infection in susceptible cells (adsorption/uncoating/replication) without producing an infectious virus11. One of the major advantages of pPNT is the safety, which is free from infectious PV and allow the test to be performed in biosafety level (BSL) 2 laboratories. Neutralization tests with pseudotyped viruses have also been developed for other viruses, especially for those required to be handled in high BSL laboratories (e.g. Ebola virus, SARS-CoV, SARS-CoV-2, lyssaviruses, Nipah virus, highly pathogenic avian influenza A viruses)12,13,14,15,16,17,18.
Despite the feasibility of pPNT, a high-throughput system, which could be utilized in serosurveillance has yet to be established. In the present study, we automated most steps in pPNT and established the high-throughput system (HTpPNT); enable to test approximately 300 serum samples at a time by one to two persons. By using the HTpPNT, we analyzed the difference of neutralization antibody titres against a type 1 wild-type strain (Mahoney strain) and a vaccine strain (Sabin 1 strain) with human serum samples (a total of 731 samples) before and after the introduction of sIPV in routine immunization program.
Results and discussion
Automation of pPNT
In the previous study, we established pPNT by manual handling6,9. To enable a large-scale pPNT which can be used in serosurveillance, we attempted to automate pPNT in the steps of serum dilution, plate copying, and removal of supernatant from the plates; an automatic dispenser/diluter/plate-copying machine (EDR-384SX work station) and a plate centrifuge for removal of supernatant (GYROMINI) were introduced (Fig. 1). Other equipment (dispenser and luminometer) were already employed in the previous systems6,9. The most difficult step for the automation was diluting human serum on a 384-well plate system, because serum shows a high resistance to be mixed due to a high concentration of high-molecular weight solutes including albumin (total protein concentration of about 6.5–8.0%)19. Human serum samples were mixed with 1% FCS-DMEM (fourfold dilution of the serum) by manual pipetting 10 times, and then subjected to the automatic diluter with mixing rate of 40 μL/s. Condition of the removal of supernatant by the plate centrifuge was optimized to recapitulate that by the manual decanting (1000 rpm for 1 s). These automations allowed testing of approximately 300 serum samples at a time by one to two persons.
Evaluation of high-throughput pPNT (HTpPNT)
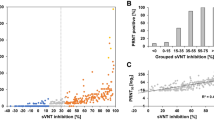
To evaluate the utility of HTpPNT in serosurveillance, we compared the results of HTpPNT and those of cPNT by testing 600 human serum samples collected in 2017, 2018 and 2019 (Fig. 2, Supplementary Data 1, 2, 3 and 4). The results obtained by these tests showed high correlations, as previously observed for manual-handling pPNT (coefficient of determination R2 was 0.82–0.91)9. The high correlations were observed in the all age groups (Fig. 3, Supplementary Data 5, 6, 7 and 8). Marked increase was observed in the titre against the type 3 among the young age group (aged 0–19 years) after 2017 compared to that in 2009 due to the switch from OPV to sIPV in routine immunization program in 2012. According to the National serosurveillance, anti-PV neutralizing antibody titre is maintained at least until school-entry age in Japan (https://www.niid.go.jp/niid/ja/y-graphs/9664-polio-yosoku-serum2020.html), as well as the population vaccinated with cIPV (reviewed in20). Meanwhile, waning of the immunity in children/adolescents (mostly cIPV vaccinees, aged 1–14 years) has been observed possibly due to intrinsic nature of IPV without booster21. Therefore, further monitoring of the immunity induced by sIPV is essential to confirm the effectiveness.
Comparison of results of cPNT and HTpPNT. Scatter plot of neutralizing antibody titres against PV by cPNT and HTpPNT. The size of the circles is proportional to the number of serum samples with the indicated titres. The coefficient of determination (R2), regression line (red), 95% confidence interval (green), and 95% prediction interval (blue) are shown.
Transition of anti-PV neutralization antibody titre before and after the switch from OPV to sIPV in Japan. Distributions of neutralizing antibody titres against PV obtained by cPNT and HTpPNT in different age groups before and after the switch from OPV to sIPV in 2012 in routine immunization in Japan are shown.
Analysis on difference of the neutralizing antibody titre against type 1 WT strain (Mahoney strain) and Sabin 1 strain
As of 2022, only type 1 WT PV strains are circulating; type 2 and 3 WT strains are considered to be eradicated (declared by WHO in 2015 and 2019, respectively. The last cases were in India, 1999, and in Nigeria, 2012, respectively)22,23, while the great majority of currently circulating PV is the type 2 vaccine-derived PVs (682 cases vs. 6 cases caused by type 1 WT in 2021)5. Type 1 PV strains show a varied antigenicity, and the WT strains show striking difference compare to the vaccine strain, Sabin 1; neutralizing antibodies induced by the Sabin 1 strain have lower titre against the parental Mahoney strain than that to the Sabin 1 strain by approximately to four to eightfold9,10 and to the WT strains circulated in Africa by approximately fourfold24, while the WT strains circulating in Afghanistan/Pakistan had similar antigenicity to the Sabin 1 strain25. As a viral factor that could cause this antigenic difference, a VP3-T60K mutation in the Sabin 1 strain has been identified26,27. However, the human factors that could affect on the difference remains to be clarified. The national serosurveillance in Japan has been conducted by using the Sabin strains since 1984, and currently no data is available in terms of the difference in the neutralizing antibody titres against the WT and the Sabin strains. To monitor the neutralizing antibody titre against the type 1 WT strain, we performed pPNT with PV1pv with capsid proteins of the type 1 wild-type (WT) Mahoney strain (PV1pv[Mahoney]), and compared the neutralization titre against PV1pv with capsid proteins of the Sabin 1 strain (PV1pv[Sabin 1], Fig. 4). In the young age group, the titre against PV1pv(Mahoney) is approximately fourfold lower on average compared to the PV1pv(Sabin 1). The difference was age-dependent, and the elderly group (age of ≥ 60 years) consistently showed relatively higher titre against PV1pv(Mahoney) than that to PV1pv(Sabin 1) (approximately one to fourfold higher titre) (n = 15–16, P = 0.0000023–0.041). A gradual shift of the high-titre age groups (age of ≥ 40 years as of 2009) was observed between the samples collected in 2009 and samples collected in 2017, 2018, and 2019, suggesting a fixation of WT-specific immunity in the elderly group. The last poliomyelitis case by an indigenous WT strain in Japan was observed in 198028. This might suggest a possibility that the elderly groups (age of ≥ 40 years as of 2009) had an asymptomatic infection by indigenous WT strains in their early childhood. Moreover, we analysed the effect of vaccine types (i.e., OPV or sIPV) in the young age group (aged 0–7 years), before and after the switch from OPV to sIPV in 2012 (Fig. 5). As a result, the vaccine types did not show significant effects on the relative titre (n = 13–45, P = 0.047–0.74).
Difference of neutralizing antibody titre against type 1 PV wild-type (Mahoney strain) and that against type 1 Sabin strain among age groups. Log2 ratio of the neutralizing antibody titre against PV1pv(Mahoney) to that against PV1pv(Sabin 1) is shown. Each box represents 25–75 percentile with median (a line in the box), between maximal and minimal values. Values of mean and standard deviation (in parenthesis) are shown.
Difference of neutralizing antibody titre against type 1 PV wild-type (Mahoney strain) and that against type 1 Sabin strain between OPV and sIPV-received children. Log2 ratio of neutralizing antibody titre against PV1pv(Mahoney) to that against PV1pv(Sabin 1) in OPV vaccinees (serum samples collected in 2009) and sIPV vaccinees (serum samples collected in 2017, 2018, and 2019) are shown. Each box represents 25–75 percentile with median (a line in the box), between maximal and minimal values. Values of mean and standard deviation (in parenthesis) are shown.
In summary, we established an HTpPNT that could serve as a useful alternative to cPNT. The HTpPNT would be suitable for laboratories that perform a large-scale serosurveillance with limited manpower. Under different circumstances of country/laboratory, the HTpPNT can be implemented in the serosurveillance in combination with other proposed methods, such as cPNT with hyper-attenuated PV strains (S19), an alternative of live PV strains29,30. Flexibility in the implementation of the systems would help laboratories to continue the activities in the global PV eradication.
Methods
Cells, viruses, and human sera
RD cells (human rhabdomyosarcoma cells) and HEK293 cells (human embryonic kidney cells) were cultured as monolayers in Dulbecco’s modified Eagle medium (DMEM) supplemented with 10% fetal calf serum (FCS). Vero cells (African green monkey kidney cells) were cultured as monolayers in Eagle’s Minimum Essential Medium (EMEM) supplemented with 0.11% bovine serum albumin (BSA) (fraction V, Sigma). The RD cells were used for pPNT titration and the Vero cells were used for cPNT. PV pseudoviruses (PVpvs), which composed of a luciferase-encoding PV replicon based on the Mahoney strain and capsid proteins derived from PV1(Mahoney), PV1(Sabin 1), PV2(Sabin 2), or PV3(Sabin 3), were used in this study9,11. In house standard anti-PV serum (monkey sera, 32 U per 50 μL for each serotype of Sabin strain determined by cPNT) was used as control in pPNT. Human sera were collected from healthy volunteers (ages of 0–89) who gave informed consent by themselves, their parents or legal guardians. Experiments performed here were approved by the Committee for Ethical Regulation of the National Institute of Infectious Diseases, Japan. All experiments were performed in accordance with relevant guidelines and regulations.
cPNT
cPNT was performed according to the standard procedure recommended by the WHO with modifications31 as previously described6,9. Briefly, a twofold dilution series of human sera was prepared with EMEM supplemented with 0.11% BSA resulting in 1/4–1/1024 dilutions. Then, 50 μL of the diluted sera or EMEM supplemented with 0.11% BSA was added to the wells of three 96-well plates (one plate for each serotype of PV with a total of three plates). Next, 50 μL of the type 1, 2, or 3 Sabin strains (100 50% cell culture infective dose (CCID50) ) was added to each well of the plates, and incubated at 37 °C for 3 h. After the incubation, 100 μL of Vero cell suspension in EMEM supplemented with 0.11% BSA (1.0–2.0 × 105 cells) was added to each well of the plates, and the plates were incubated at 37 °C for 7 days. Neutralizing antibody titre of the serum was determined as 50% endpoints suppressing the cytopathic effect of the cells.
HTpPNT
Basic principle of pPNT is described in previous reports6,9. DMEM supplemented with 1% FCS (1%FCS-DMEM) were dispensed into 384-well plates (Thermo Fischer Scientific, 264574) as follows; 45 μL/well for rows 1, 9 and 17, and 30 μL/well for other rows. Then, 15 μL of human serum samples, standard anti-PV serum, or 1% FCS-DMEM (for mock treatment) were added to the wells in rows 1, 9, and 17 (total volume of 60 μL/well) followed by 10 times manual pipetting. A twofold dilution series of the serum samples and the standard serum were prepared by using an automatic diluter/dispenser for 384-well plates by mixing eight times for the first row and then six times for other rows by the rate of 40 μL/s (EDR-384SX 12 stage work station, BIOTEC) (the final dilution of 1/4–1/512). Next, 5 μL of the diluted samples were dispensed into the wells of four 384-well plates (Greiner Bio-One, 781080) by using EDR-384SX 12 stage work station (one plate for each serotype of PVpv(Sabin) and PV1pv(Mahoney) with a total of three plates). Then, 5 μL of PV1pv(Mahoney), PV1pv(Sabin 1), PV2pv(Sabin 2), or PV3pv(Sabin 3) solution (400 IU) was dispensed into the wells of the each plate by using an automatic dispenser (WellMate, Matrix). The plates were centrifuged (200×g, 1 min) (5910, KUBOTA), and then incubated at 4 °C overnight. Subsequently, 20 μL of RD cell suspension (4.0 × 105 cells/mL) in 5% FCS-DMEM was dispensed into each well of the plates by using an automatic dispenser (WellMate, Matrix) (total 8000 cells/well). The plates were centrifuged (200×g, 1 min) (5910, KUBOTA), and then incubated at 37 °C for 7 h. After the incubation, the supernatant was removed by centrifugation (1000 rpm for 1 s) using a plate centrifuge (GYRO mini, micronix), and then 5 μL of Steady-Glo solution was added to each well of the plates by using an automatic dispenser (WellMate, Matrix). Luciferase signal was measured by 2030 ARVO X luminometer (PerkinElmer). PVpv infection was calculated from the percentage of luciferase signal of infected cells. The luciferase signal in mock-treated cells was taken as 100%. Neutralizing antibody titre was determined by a reciprocal number of the highest dilution of the serum that suppressed PVpv infection to less than 5% (for PV1pv[Mahoney], PV1pv[Sabin 1], and PV2pv[Sabin 2]) or 10% (for PV3pv[Sabin 3]). As measures of quality controls of HTpPNT, the titre of the standard serum (less than fourfold difference from the expected titre for each type, thus > 8 and < 128), coefficient of variation of luciferase signals in mock-treated PVpv-infected cells (less than 30%) were used.
Statistical analysis
Results of experiments are shown with means and standard deviations. P values < 0.05 by Kruskal–Wallis test and Steel–Dwass test were considered statistically significant and were indicated by asterisks (*P < 0.05; **P < 0.01; ***P < 0.001). Statistical analysis was performed by OriginPro software (regression analysis) or by R using an R commander EZR (Kruskal–Wallis test and Steel–Dwass test)32.
Data availability
Raw data sets not included in the manuscript or the supplementary information are available from the corresponding author upon request.
References
Shimizu, H. Development and introduction of inactivated poliovirus vaccines derived from Sabin strains in Japan. Vaccine 34, 1975–1985. https://doi.org/10.1016/j.vaccine.2014.11.015 (2016).
Iwai, M. et al. Evaluation of a two-dose administration of live oral poliovirus vaccine for wild and virulent vaccine-derived poliovirus type 1, 2, 3 strains in Japan. Scand. J. Infect. Dis. 40, 247–253. https://doi.org/10.1080/00365540701596003 (2008).
WHO. http://www.polioeradication.org/Portals/0/Document/Resources/PostEradication/GAPIII_2014.pdf.
WHO. https://polioeradication.org/polio-today/polio-now/this-week/circulating-vaccine-derived-poliovirus/.
Arita, M., Iwai, M., Wakita, T. & Shimizu, H. Development of a poliovirus neutralization test with poliovirus pseudovirus for measurement of neutralizing antibody titer in human serum. Clin. Vaccine Immunol. 18, 1889–1894. https://doi.org/10.1128/CVI.05225-11 (2011).
Liu, S. et al. A safe and reliable neutralization assay based on pseudovirus to measure neutralizing antibody titer against poliovirus. J. Med. Virol. 89, 2075–2083. https://doi.org/10.1002/jmv.24909 (2017).
Jiang, Z. et al. A simple and safe antibody neutralization assay based on polio pseudoviruses. Hum. Vaccines Immunother 15, 349–357. https://doi.org/10.1080/21645515.2018.1526553 (2019).
Arita, M. & Iwai-Itamochi, M. Evaluation of antigenic differences between wild and Sabin vaccine strains of poliovirus using the pseudovirus neutralization test. Sci. Rep. 9, 11970. https://doi.org/10.1038/s41598-019-48534-1 (2019).
Liu, S. et al. Comparison of the neutralizing activities of antibodies in clinical sera against both Sabin and wild-type polio pseudoviruses. J. Virol. Methods 300, 114376. https://doi.org/10.1016/j.jviromet.2021.114376 (2022).
Arita, M., Nagata, N., Sata, T., Miyamura, T. & Shimizu, H. Quantitative analysis of poliomyelitis-like paralysis in mice induced by a poliovirus replicon. J. Gen. Virol. 87, 3317–3327. https://doi.org/10.1099/vir.0.82172-0 (2006).
Rimoin, A. W. et al. Ebola virus neutralizing antibodies detectable in survivors of Theyambuku, Zaire outbreak 40 years after infection. J. Infect. Dis. 217, 223–231. https://doi.org/10.1093/infdis/jix584 (2018).
Fukushi, S. et al. Evaluation of a novel vesicular stomatitis virus pseudotype-based assay for detection of neutralizing antibody responses to SARS-CoV. J. Med. Virol. 78, 1509–1512. https://doi.org/10.1002/jmv.20732 (2006).
Han, D. P., Kim, H. G., Kim, Y. B., Poon, L. L. & Cho, M. W. Development of a safe neutralization assay for SARS-CoV and characterization of S-glycoprotein. Virology 326, 140–149. https://doi.org/10.1016/j.virol.2004.05.017 (2004).
Wright, E. et al. Investigating antibody neutralization of lyssaviruses using lentiviral pseudotypes: A cross-species comparison. J. Gen. Virol. 89, 2204–2213. https://doi.org/10.1099/vir.0.2008/000349-0 (2008).
Tamin, A. et al. Development of a neutralization assay for Nipah virus using pseudotype particles. J. Virol. Methods 160, 1–6. https://doi.org/10.1016/j.jviromet.2009.02.025 (2009).
Wang, W. et al. Establishment of retroviral pseudotypes with influenza hemagglutinins from H1, H3, and H5 subtypes for sensitive and specific detection of neutralizing antibodies. J. Virol. Methods 153, 111–119. https://doi.org/10.1016/j.jviromet.2008.07.015 (2008).
Zettl, F. et al. Rapid quantification of SARS-CoV-2-neutralizing antibodies using propagation-defective vesicular stomatitis virus pseudotypes. Vaccines (Basel) https://doi.org/10.3390/vaccines8030386 (2020).
Cussler, E. Difussion 3rd edn, 117–160 (Cambridge University Press, 2009).
Vidor, E. Poliovirus vaccine-inactivated. In Plotkin's Vaccines Edtsevebth edn (eds Plotkin, S. A et al.) 858 (Elsevier, 2018) doi:https://doi.org/10.1016/C2013-0-18914-3.
Reinheimer, C., Friedrichs, I., Rabenau, H. F. & Doerr, H. W. Deficiency of immunity to poliovirus type 3: A lurking danger?. BMC Infect. Dis. 12, 24. https://doi.org/10.1186/1471-2334-12-24 (2012).
WHO. https://polioeradication.org/news-post/global-eradication-of-wild-poliovirus-type-2-declared/. (2015).
WHO. https://www.who.int/news-room/feature-stories/detail/two-out-of-three-wild-poliovirus-strains-eradicated. (2019).
Drexler, J. F. et al. Robustness against serum neutralization of a poliovirus type 1 from a lethal epidemic of poliomyelitis in the Republic of Congo in 2010. Proc. Natl. Acad. Sci. USA 111, 12889–12894. https://doi.org/10.1073/pnas.1323502111 (2014).
Alam, M. M. et al. Antigenic structure of wild poliovirus type 1 strains endemic in Pakistan is highly conserved and completely neutralized by Sabin’s Oral Polio Vaccine. J. Infect. Dis. https://doi.org/10.1093/infdis/jiab555 (2021).
Diamond, D. C. et al. Antigenic variation and resistance to neutralization in poliovirus type 1. Science 229, 1090–1093 (1985).
Blondel, B. et al. Mutations conferring resistance to neutralization with monoclonal antibodies in type 1 poliovirus can be located outside or inside the antibody-binding site. J. Virol. 57, 81–90 (1986).
Hara, M., Hagiwara, A., Yoneyama, T., Saito, Y. & Shimojo, H. Antigenic and biochemical characterization of poliovirus type 1 isolates of non-vaccine origin. Microbiol. Immunol. 27, 1057–1065. https://doi.org/10.1111/j.1348-0421.1983.tb02939.x (1983).
Knowlson, S. et al. New strains intended for the production of inactivated polio vaccine at low-containment after eradication. PLoS Pathog. 11, e1005316. https://doi.org/10.1371/journal.ppat.1005316 (2015).
Farcet, M. R. et al. Continued use of poliovirus after eradication: hyper-attenuated strains as a safe alternative for release testing of human immunoglobulins. Transfusion 58(Suppl 3), 3084–3089. https://doi.org/10.1111/trf.15048 (2018).
World Health Organization. Manual of Laboratory Methods for Potency Testing of Vaccines Used in the WHO Expanded Programme on Immunization. WHO Publication No. WHO/BLG/95.1 1995 (World Health Organization, 1995).
Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 48, 452–458. https://doi.org/10.1038/bmt.2012.244 (2013).
Acknowledgements
We are grateful to Yuzuru Aoi for her excellent technical assistance, Minami Kikuchi-Ueno for proof reading the manuscript, and to Hiroyuki Shimizu and Masamichi Muramatsu for kind support. This study was supported in part by AMED under Grant Number 21fk0108085j0003, 22fk0108627j0001 and by the Health and Labor, Sciences Research Grant (grant number 22HA1003) to M.A.
Author information
Authors and Affiliations
Contributions
M.A. conceived the project. M.A. designed the study. M.I. prepared human serum samples and performed cPNT with Sabin strains. M.A. performed pPNT. M.A. wrote the manuscript. M.I. commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arita, M., Iwai-Itamochi, M. High-throughput analysis of anti-poliovirus neutralization antibody titre in human serum by the pseudovirus neutralization test. Sci Rep 12, 16074 (2022). https://doi.org/10.1038/s41598-022-20544-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-20544-6
- Springer Nature Limited