Abstract
Transthoracic echocardiography (TTE) and cine cardiac magnetic resonance imaging (CMR) are established imaging methods of the aortic root. We aimed to evaluate the comparability of measurements in TTE and standard cine CMR sequences of the aortic root. Our study included 741 subjects (mean age 63.5 ± 8 years, 43.7% female) from the Hamburg City Health Study (HCHS). Subjects underwent CMR and TTE. Aortic root measurements were performed at the level of the aortic annulus (AoAn), sinus of Valsalva (SoV), and sinotubular junction (STJ) by standard cine CMR in left ventricular long axis and left ventricular outflow tract view. Measurements were performed applying the leading-edge to leading-edge (LL) convention and inner-edge to inner-edge (II) convention in TTE and the II convention in CMR. Inter correlation coefficients (ICCs) demonstrated high inter- and intraobserver reproducibility for CMR and TTE measurements of SoV and STJ (ICCs 0.9–0.98) and moderate reproducibility for AoAn (ICCs 0.68–0.91). CMR measurements of SoV and STJ showed strong agreement with TTE: while correlations were comparable (r = 0.75–0.85) bias was lower with TTE II (bias − 0.1 to − 0.74) versus TTE LL measurements (mean bias − 1.49 to − 2.58 mm). The agreement for AoAn was fair (r = 0.51–0.57) with variable bias (mean bias 0.39–3.9). Standard cine CMR and TTE derived aortic root measurements are reproducible and comparable with higher agreement for TTE II instead of LL measurements. These results support an interchangeable application of TTE and standard CMR for screening of aortic root diseases thereby possibly reducing redundant multimodality imaging.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Dilatation of the aortic root is a frequent finding in clinical practice and is strongly associated with aortic regurgitation, an increased risk for aneurysm formation, and aortic dissection1,2. Timely diagnosis of aortic root dilatation is crucial because the abovementioned pathologies are associated with a high morbidity3 and mortality4.
Transthoracic echocardiography (TTE) is the most frequently used imaging method in the evaluation of the aortic root dimensions and most reference values for aortic diseases are derived from TTE studies5. However, cardiovascular magnetic resonance (CMR) has become increasingly available and allows for visualization and quantification of cardiac anatomy with accurate measurements of the aortic root6. It is performed in various clinical settings such as imaging of cardiomyopathies and in the setting of chest pain as it is part of major guideline recommendations7. CMR based aortic root diameter measurements overcome limitations of TTE such as a limited acoustic window and allow measurements perpendicular to the centreline of the aorta using multiplanar reconstructions as recommended in ESC guidelines5,8. Nevertheless, CMR is often used task-specific and not all possible sequences and angulations are performed. Previous studies report high agreement of specifically angulated CMR derived aortic root diameter measurements with TTE9. However, it is recommended to perform measurements of the aorta in a 3D dataset or sinus planes using a double-oblique orientation perpendicular to the aortic lumen6,10. Nevertheless, in clinical routine, specifically angulated planes or 3D sequences are only performed in patients with suspected aortic root disease. The cine CMR 3-chamber left ventricular (LV) view, which is part of the basic CMR sequences recommended by the Society for Cardiovascular Magnetic Resonance (SCMR) and the LV outflow tract (LVOT) view allow for a measurement of aortic root diameters as well11. However, there is a lack of data regarding the usefulness of these additional aortic root diameter measurements in the CMR 3-chamber LV and the LVOT cine-view in comparison to the established measurements performed by TTE.
Therefore, the purpose of this study was to provide data on the comparability of aortic root diameters obtained by cine-CMR and TTE in a large sample of the general population.
Materials and methods
Study population and study design
The Hamburg City Health Study (HCHS) was approved by the local ethics committee (PV5131, State of Hamburg Chamber of Medical Practitioners) and this study was approved by the review board (HCHS steering committee) of the HCHS. All participants gave written informed consent. This study and the HCHS were conducted in agreement with the Declaration of Helsinki.
The HCHS has been previously described12. In brief, the study prospectively includes a random sample of 45.000 participants between 45 and 74 years of age from the general population of Hamburg, Germany, investigating the interaction of socioeconomic risk factors, modern imaging techniques, physiological measurements, clinical variables, and targeted major diseases. At a baseline visit at the HCHS Epidemiological Study Center from the University Medical Center Hamburg all participants undergo a standardized interview, clinical examination, laboratory assessment, and TTE.
In this prospective study we analyzed a subgroup of the first 1000 HCHS participants who received a CMR13. Exclusion criteria were missing TTE data due to structural limitations at the beginning of the study (n = 209) or insufficient image quality in CMR and/or TTE (n = 57).
Demographics and clinical parameters
Demographics and clinical parameters were investigated by standardised interviews and questionnaires conducted by medical professionals following standard operating procedures12. At the baseline visit at the HCHS Epidemiological Study Center blood samples were withdrawn under fasting conditions. BP was measured twice at the right upper arm in sitting position after 5 min of rest, results were averaged. Arterial hypertension was defined as systolic blood pressure ≥ 140 mmHg and diastolic blood pressure ≥ 90 mmHg, or the use of one or more of the following antihypertensive drugs: ACE inhibitors, angiotensin II receptor blockers, beta blockers, calcium channel blockers, renin inhibitors, or loop diuretics. Diabetes mellitus was determined by fasting glucose levels of ≥ 126 mg/dl, or the use of antidiabetic drugs. Coronary artery disease was self-reported by questionnaire and defined as having had a history from one or more of the following conditions: myocardial infarction, percutaneous coronary intervention (PCI) or coronary bypass surgery.
Aortic root measurements
TTE
All TTE studies were evaluated and quantified at a single reading center blinded to the clinical information of the subjects using commercially available Siemens syngo SC2000 software (Siemens syngo SC 2000 Version 4.0, Siemens Healthineers, Erlangen, Germany). All TTE standard views were assessed in 2-dimensional echocardiography. The acquisition and analysis were performed according to the latest recommendations of the European Society of Cardiovascular Imaging (EACVI) and the American Society of Echocardiography (ASE)14.
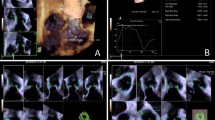
The aortic root was assessed in transthoracic parasternal long axis view as recommended by the American Society of Echocardiography (ASE) and the European Association of Cardiovascular Imaging (EACVI) and previously described by our group15,16. Systematic measurements were performed perpendicular to the proximal aorta axis in end-diastole (ED) including the following: (a) aortic annulus (AoAn), (b) sinus of Valsalva (SoV), (c) sinotubular junction (STJ). Both for TTE and CMR, end-diastole was defined visually with a maximal distended left ventricle right before aortic valve opening. The AoAn was measured as the largest diameter between the hinge points of the right- and non-coronary cusps of the aortic valve. The SoV was measured as the maximum diameter of the aortic bulb. The STJ was assessed at the demarcated transition between the SoV and the tubular portion of the ascending aorta. All parts of the aortic root were measured perpendicular to the long axis of the aorta by the inner-edge to inner-edge (II) convention as well as the leading-edge to leading-edge (LL) convention (Fig. 1).
Measurements of the aortic root in TTE and CMR. Measurements were performed in end-diastole at the level of the aortic annulus, sinus of Valsalva and sinotubular junction. In TTE measurements were performed both from inner-edge to inner-edge (II) and from leading-edge to leading-edge. In CMR measurements were performed using the II convention.
CMR
CMR was performed on 3 Tesla scanner (MAGNETOM Skyra, Siemens Healthineers, Erlangen, Germany). CMR protocols in the HCHS were described in detail in a previous publication13. balanced steady-state free precession imaging (BSSFP) cine-sequences were acquired in the following orientations: left ventricular (LV) short axis, 2,3,4-chamber LV long axis (LAX) view and left ventricular outflow tract (LVOT) view. Acquisition was performed standardized as recommended in the 2020 update for standardized cardiovascular magnetic resonance imaging (CMR) protocols published by the Society for Cardiovascular Magnetic Resonance (SCMR) Board of Trustees Task Force on Standardized Protocols11. BSSFP parameters were chosen as follows: voxel size 1.6 × 1.6 × 8 m3, FoV 340 mm2, TR 48 ms, TE 1.5 ms, FA 80°, parallel acquisition technique with a factor of 3.
All measurements were performed in the 3-chamber LV LAX cine-view and LVOT cine-view as they are part of standardized CMR protocols as recommended by the SCMR and comparable to the TTE PLAX17. Measurements were performed from the inner-edge to inner-edge at the predefined levels (Fig. 1): AoAn, SoV, and STJ. Diameter measurements were performed using a task specified plugin in ImageJ (v1.52, NIH, USA), which allowed for automatic export of diameter measurements.
Intra- and interobserver variability
For the calculation of intra- and interobserver agreement by interclass correlation coefficients (ICC [95%-CI]) a subset of 50 TTE and CMR exams was selected at random. For each imaging modality there were two individual readers resulting in a total of four different readers who exclusively measured CMR or TTE. Reader 1 performed two separate measurements (intraobserver variability) while reader 2 performed one measurement (interobserver variability) of all aortic root parts using the LL and II convention for TTE and II for CMR. For the prevention of recall bias there was a two-week interval between the reading sessions of reader 1.
Statistical analysis
Statistical analyses were performed using R version 4.0.3. Continuous data are presented as median and inter-quartile-range (IQR). Categorical data are presented as absolute and relative frequencies. Missing values were not included in the calculation of frequencies.
The relationship between TTE and CMR variable was investigated by the Spearman’s correlation coefficient and Bland–Altman analysis (mean difference [95%-confidence interval (CI)]). A significance in the correlation was assumed if p-values were < 0.05.
Declaration of Helsinki
The authors do hereby declare that their study complies with the Declaration of Helsinki.
Results
Study cohort
Among the first 1000 subjects with CMR enrolled into HCHS, 741 subjects were included in this analysis. The 741 subjects represented a middle-aged European population with 324 (43.7%) women, a median of 65 [IQR: 58–70] years, and a median body mass index (BMI) of 26.3 [IQR 23.8–29.4] kg/m2 (Fig. 2, Table 1). Hypertension was present in 516 (73.8%) subjects, 71 (10.4%) subjects had diabetes and 41 (8.3%) subjects coronary artery disease while atrial fibrillation and peripheral artery disease were present in 24 (3.3%) and 16 (2.3%) subjects, respectively. The median LDL-cholesterol level was 121 [IQR 98–143] mg/dl and the median TTE left ventricular ejection fraction 57.7 [IQR 54.9–61.5] %.
Study PRISMA. From the first 1000 HCHS subjects with CMR data, 798 subjects had undergone TTE examination, 57 were excluded due to insufficient echocardiographic or CMR image quality for measuring the aortic root. Inter and Intraobserver correlation coefficients were calculated for 50 at random selected subjects. CMR cardiac magnetic resonance imaging, ICC intra-/interclass correlation coefficient, TTE transthoracic echocardiography.
Median aortic root diameters in CMR LAX view were 21.3 [IQR 19.4, 23.5] mm for AoAn, 32.4 [29.8, 35.3] mm for SoV, and 26.6 [24.3, 29.4] mm for STJ, TTE and CMR LVOT diameters are presented in Table 2.
Intra- and interobserver agreement of CMR and TTE measurements
ICCs of ≥ 0.9 demonstrated high inter- and intraobserver reproducibility for all CMR and TTE measurements of the SoV and STJ (Table 3). Generally, CMR showed higher ICCs than TTE. CMR LAX measurements showed higher ICCs than CMR LVOT measurements. Reproducibility of TTE LL and TTE II measurements did not differ significantly. Measurements of the SoV were both in CMR and TTE the most reproducible (SoV interobserver: TTE II ICC = 0.97 [0.95; 0.98]; TTE LL ICC = 0.94 [0.9; 0.97]; LAX ICC = 0.97 [0.95; 0.98]; LVOT ICC = 0.94 [0.9; 0.97].
In contrast, measurements of the AoAn showed lower intra- and interobserver reproducibility, indicated by lower ICCs both in TTE and CMR (AoAn interobserver: TTE LL ICC = 0.68 [0.5; 0.8], TTE II ICC = 0.75 [0.6; 0.85]; CMR LAX ICC = 0.86 [0.76; 0.92], CMR LVOT ICC = 0.8 [0.68; 0.88]).
Comparison of CMR vs. TTE leading-edge aortic root measurements
CMR measurements of the SoV and STJ demonstrated strong correlations with TTE LL measurements (Table 4, Fig. 3, Supplemental Fig. 1). Concerning SoV diameters, the correlations with TTE LL varied depending on the CMR imaging plane: it was higher for the LAX view compared to the LVOT view (SoV TTE LL vs. CMR: LAX: r = 0.847, p < 0.001; LVOT: r = 0.746, p < 0.001). In contrast, correlations of the STJ with TTE LL diameters did not relevantly differ between LVOT or LAX view (STJ TTE LL vs. CMR: LAX: r = 0.804, p < 0.001; LVOT: r = 0.818, p < 0.001). CMR measurements generally resulted in smaller SoV and STJ diameters compared to TTE LL measurements (SoV TTE LL vs. CMR: LAX bias = − 1.49 ± 4.39 mm; LVOT bias = − 2.12 ± 5.79 mm; STJ TTE LL vs. CMR: LAX bias = − 2.54 ± 4.24 mm; LVOT bias = − 2.58 ± 4.01 mm).
Comparison of TTE and CMR measurements of the aortic root. The following scatter plots show the linear regression line for two values as well as the spearman correlation coefficient and p-value. The Bland–Altman plots show the mean bias between the two values (CMR measurements subtracted by the corresponding TTE measurement) and the 95% confidence interval. CMR cardiac magnetic resonance imaging, LAX long-axis view, TTE transthoracic echocardiography.
Regarding the AoAn, CMR and TTE LL measurements showed fair agreement, regardless of the CMR imaging plane (AoAn TTE LL vs. CMR: LAX: r = 0.573, p < 0.001; LVOT: r = 0.541, p < 0.001). CMR measurements significantly overestimated the AoAn diameter compared with TTE LL measurements with moderate limits of agreement and a higher bias for the CMR LVOT view than for the LAX view (AoAn TTE LL vs. CMR: LAX bias = 0.39 ± 4.66 mm; LVOT bias = 3.02 ± 5.23 mm).
Comparison of CMR vs. TTE inner-edge aortic root measurements
CMR measurements of the SoV and STJ, both in LAX and LVOT view, showed a high correlation with TTE II measurements (Table 4, Fig. 3, Supplemental Fig. 1). The mean difference of CMR vs. TTE II SoV and STJ measurements was lower compared to CMR vs. TTE LL measurements (SoV TTE II vs. CMR: LAX bias = − 0.1 ± 4.37; LVOT bias = − 0.71 ± 5.74 mm; STJ TTE II vs. CMR: LAX bias = − 0.7 ± 4.17; LVOT bias = − 0.74 ± 3.85 mm).
The correlation of AoAn CMR measurements with TTE II measurements was fair (AoAn TTE II vs. CMR: LAX: r = 0.559, p < 0.001; LVOT: r = 0.514, p < 0.001). CMR measurements overestimated AoAn dimensions compared to TTE II measurements. The overestimation was stronger with broader limits of agreement for the LVOT view (AoAn TTE II vs. CMR: LAX bias = 1.26 ± 4.72; LVOT bias = 3.9 ± 5.34).
Discussion
Aortic root measurements in standardized cine CMR sequences are comparable to state-of-the-art TTE measurements in a contemporary, prospectively enrolled, middle-aged sample from the general population. The SoV and STJ showed a high degree of agreement, whereas it was only fair for the AoAn. Comparability improved if CMR measurements were compared to II instead of LL TTE measurements.
Aortic root dimensions can be interchangeably measured by CMR or TTE
Few studies with only small cohorts compared aortic root measurements by TTE and CMR: In retrospective analyses with a maximum of 140 patients, diameters of the aortic root measured by CMR and TTE strongly correlated18,19. However, in those studies measurements were performed in specifically acquired sequences for measurements of the aortic root through the true cross sectional aortic valve plane18. To the best of our knowledge, there is only one retrospective study with a limited number of patients with suspected Marfan syndrome comparing standardized cine CMR measurements of the aortic root with TTE17. Hence our large prospective study in a general population is the first study to demonstrate that measurements assessed by LAX and LVOT cine CMR are comparable to TTE measurements. This is of clinical relevance as it supports not only the application of established TTE measurements but also of measurements derived from CMR orientations which are included in almost every CMR protocol for screening for aortic root diseases.
However, in CMR, the LAX view is part of routine clinical protocols whereas the LVOT view is not routinely included8. As LAX and LVOT view measurements for reliably assessing aortic root dimensions both demonstrated high correlations with TTE, the necessity for an additional LVOT view, at least regarding aortic root measurements, might be questionable.
Furthermore, the highest agreements were found for TTE according to LL convention with CMR rather than TTE II measurements and CMR9. This is in contrast to our results from our large cohort, which demonstrate comparable correlations, but a systematic overestimation of diameters measured by LL convention compared to CMR II measurements.
Diameters measured according to LL convention include the outer wall of one side of the aortic root. Therefore, an overestimation compared to diameters acquired by II convention, even though from different imaging modalities, seems comprehensible. Up to now, there is an ongoing discussion about which convention to use for echocardiographic measurements of the aortic root. While the ASE guidelines recommend to use the LL convention, the pediatric guidelines, supported by the 2010 American College of Cardiology and American Heart Association guidelines, support the II convention14,20,21. In clinical routine, as most reference values are based on the LL convention, TTE measurements are primarily performed using the LL convention22. However, in recent years technical upgrades and digital post-processing have led to major improvements in spatial resolution of transthoracic ultrasound. Thereby, the depiction of the thin aortic wall does not any longer limit the reproducibility of measurements23. Our results suggest a reconsideration of the LL-convention in TTE for improving comparability of SoV and STJ measurements with CMR.
Summarising, measurements of the SoV and AoAn highly correlated between TTE and CMR. Although we assessed a cohort with a low burden of aortic root dilatation, our data support an interchangeable clinical application of TTE and CMR for screening of incidental aortic root dilatation. However, CMR is still limited by lower availability, higher costs and more time-consuming examinations compared to TTE.
CMR and TTE measurements of the aortic root are highly reproducible
Echocardiographic evaluation of the aortic root is a well-established method in clinical routine and recommended in patients with suspected aortic root disease14,15. Cine CMR is widely and increasingly adapted in routine clinical practice allowing for accurate measurements of the aortic root as well. However, data on reproducibility are derived from small cohorts or measurements limited to a single diameter of the ascending thoracic aorta1,8. In our population-based study, derived from 741 subjects, both methods demonstrated a very high reproducibility, with CMR even exceeding TTE, for measuring aortic root diameters especially for SoV and STJ.
The complex structure of the AoAn limits reliable measurements and comparability
In contrast to measurements of SoV and STJ, diameters of the AoAn are routinely assessed according to II convention. However, both reproducibility and correlation of CMR and TTE measurements are reduced compared to measurements of SoV and STJ. The AoAn is an entity without a visible anatomic structure only virtually defined by the hinge-points of the three aortic valve leaflets14,24. This complex anatomy of the AoAn with an ellipsoid structure, in contrast to the more circular SoV and STJ, makes the exact measurement of the AoAn a challenging and error-prone process25. Only small alterations of the imaging plane, result in major differences of the measured diameter8. Individual alterations in dimension during the cardiac cycle further impair the reproducibility of AoAn assessment. Hence, our data suggest that measurements of the aortic annulus should only be performed in 3D CMR or computed tomography (CT) datasets with specifically angulated CMR planes.
Limitations
As the study sample origins from the general population of Hamburg, most subjects are of Caucasian ascend and represent a predominantly healthy population. Hence, the translation of our findings into other populations, especially the typical patient collective in aortic root surgery, is limited.
Another limitation of this study is that 3D reconstructed CT scans, as the recommended standard of reference for the measurement of aortic root dimensions, were not performed5. No conclusions can be drawn about the accuracy of each the modalities. Nevertheless, the aim of our study was to investigate whether measurements of the aortic root in standard planes of a regular cine-CMR are comparable to state-of-the-art TTE measurements. By showing the high correlation of both imaging techniques, these additional CMR measurements show high validity when compared to TTE and could be implemented in clinical routine for screening purposes and prevent or trigger further imaging.
The ascending aorta was not measured, hence no conclusions can be drawn regarding dilatations above the aortic root. Two further limitations have to be considered: First, since no blood pressure measurements were performed during TTE and CMR examinations, a possible impact of blood pressure fluctuations on aortic root dimensions, e.g. a raised blood pressure during CMR examination, was not considered. Second, TTE and CMR examinations were not performed on the same day. However, the median time interval between the two examinations was only 28 days. Consequently, it is highly unlikely that during this short period, aortic root diameters significantly changed.
Conclusion
In this study, derived from a large sample of the general population, CMR measurements of the aortic root were comparable to standard of care TTE measurements. Reproducibility and agreement were excellent for the SoV and STJ, while they were only fair for the AoAn. Comparability improved if the II instead of the LL convention was used, challenging the established LL-convention in aortic root echocardiography. These results suggest that cine CMR and TTE might be applied interchangeably in clinical routine thereby possibly reducing redundant multimodality imaging.
Data availability
The data underlying this article cannot be shared publicly due to the privacy of individuals that participated in the study. The data will be shared on reasonable request to the corresponding author.
Abbreviations
- AF:
-
Atrial fibrillation
- AoAn:
-
Aortic annulus
- ASE:
-
American Society of Echocardiography
- BMI:
-
Body mass index
- CMR:
-
Cardiac magnetic resonance imaging
- GFR:
-
Glomerular filtration rate
- EACVI:
-
European Association of Cardiovascular Imaging
- ESC:
-
European Society of Cardiology
- HCHS:
-
Hamburg City Health Study
- LAVI:
-
Left atrial systolic volume indexed to body surface area
- LAX:
-
Long axis
- LVEF:
-
Left ventricular ejection fraction
- LVMI:
-
Left ventricular mass index
- LVOT:
-
Left ventricular outflow tract
- NT-proBNP:
-
N-terminal pro-B-type natriuretic peptide
- SCMR:
-
Society for Cardiovascular Magnetic Resonance
- SoV:
-
Sinus of Valsalva
- STJ:
-
Sinotubular junction
- TR Vmax:
-
Maximal tricuspid regurgitation velocity
- TTE:
-
Transthoracic echocardiography
References
Turkbey, E. B. et al. Determinants and normal values of ascending aortic diameter by age, gender, and race/ethnicity in the Multi-Ethnic Study of Atherosclerosis (MESA). J. Magn. Reson. Imaging 39, 360–368 (2014).
Wenzel, J. P., Petersen, E., Nikorowitsch, J., Müller, J. & Kölbel, T. Aortic root dimensions as a correlate for aortic regurgitation’s severity. Int. J. Cardiovasc. Imaging 37, 3439–3449 (2021).
Guo, M. H. et al. Association of mortality and acute aortic events with ascending aortic aneurysm: A systematic review and meta-analysis. JAMA Netw. Open 1, 181281 (2018).
Gardin, J. M. et al. Usefulness of aortic root dimension in persons ≥ 65 years of age in predicting heart failure, stroke, cardiovascular mortality, all-cause mortality and acute myocardial infarction (from the Cardiovascular Health Study). Am. J. Cardiol. 97, 270–275 (2006).
Erbel, R. et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European. Eur. Heart J. 35, 2873–2926 (2014).
Schulz-Menger, J. et al. Standardized image interpretation and post-processing in cardiovascular magnetic resonance—2020 update: Society for Cardiovascular Magnetic Resonance (SCMR): Board of Trustees Task Force on standardized post-processing. J. Cardiovasc. Magn. Reson. 22, 19 (2020).
Gulati, M. et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain: A report of the American College of Cardiology/American Heart Association Joint Committee on Clinical practice guidelines. Circulation 144, e368 (2021).
Burman, E. D., Keegan, J. & Kilner, P. J. Aortic root measurement by cardiovascular magnetic resonance: Specification of planes and lines of measurement and corresponding normal values. Circ. Cardiovasc. Imaging 1, 104–113 (2008).
Rodríguez-Palomares, J. F. et al. Multimodality assessment of ascending aortic diameters: Comparison of different measurement methods. J. Am. Soc. Echocardiogr. 29, 819–826 (2016).
van Hout, M. J. et al. How to measure the aorta using MRI: A practical guide. J. Magn. Reson. Imaging 52, 971–977 (2020).
Kramer, C. M. et al. Standardized cardiovascular magnetic resonance imaging (CMR) protocols: 2020 update. J. Cardiovasc. Magn. Reson. 22, 17 (2020).
Jagodzinski, A. et al. Rationale and design of the Hamburg City health study. Eur. J. Epidemiol. 35, 169–181 (2020).
Bohnen, S. et al. Cardiovascular magnetic resonance imaging in the prospective, population-based, Hamburg City Health cohort study: Objectives and design. J. Cardiovasc. Magn. Reson. 20, 1–11 (2018).
Lang, R. M. et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur. Hear. J. Cardiovasc. Imaging 16, 233–271 (2015).
Goldstein, S. A. et al. Multimodality imaging of diseases of the thoracic aorta in adults: From the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 28, 119–182 (2015).
Wenzel, J.-P. et al. Transthoracic echocardiographic reference values of the aortic root: Results from the Hamburg City Health Study. Int. J. Cardiovasc. Imaging 37, 3513–3524 (2021).
Bannas, P. et al. Comparison of Cine-MRI and transthoracic echocardiography for the assessment of aortic root diameters in patients with suspected Marfan syndrome. Rofo 187, 1022–1028 (2015).
Hoey, E. T. D., Pakala, V., Kassamali, R. H. & Ganeshan, A. A comparative analysis of ECG-gated steady state free precession magnetic resonance imaging versus transthoracic echocardiography for evaluation of aortic root dimensions. Quant. Imaging Med. Surg. 4, 300–306 (2014).
Leone, D. et al. Accuracy of transthoracic echocardiography in the assessment of proximal aortic diameter in hypertensive patients: Comparison with cardiac magnetic resonance. J. Hypertens. 35, 1626–1634 (2017).
Lopez, L. et al. Recommendations for quantification methods during the performance of a pediatric echocardiogram: A report from the pediatric measurements writing Group of the American Society of Echocardiography Pediatric and Congenital Heart Disease Council. J. Am. Soc. Echocardiogr. 23, 465–495 (2010).
Hiratzka, L. F. et al. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with Thoracic Aortic Disease: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Association. Circulation 121, 266–369 (2010).
Saura, D. et al. Two-dimensional transthoracic echocardiographic normal reference ranges for proximal aorta dimensions: Results from the EACVI NORRE study. Eur. Heart J. Cardiovasc. Imaging 18, 167–179 (2017).
Servato, M. L. et al. Are aortic root and ascending aorta diameters measured by the pediatric versus the adult American Society of Echocardiography Guidelines interchangeable? J. Clin. Med. 10, 4–11 (2021).
Boccalini, S. et al. Bicuspid aortic valve annulus: Assessment of geometry and size changes during the cardiac cycle as measured with a standardized method to define the annular plane. Eur. Radiol. 31, 8116–8129 (2021).
Suchá, D. et al. Does the aortic annulus undergo conformational change throughout the cardiac cycle? A systematic review. Eur. Heart J. Cardiovasc. Imaging 16, 1307–1317 (2015).
Acknowledgements
The authors acknowledge the participants of the Hamburg City Health Study, the staff at the Epidemiological Study Center, cooperation partners, patrons and the Deanery from the University Medical Center Hamburg.
Funding
Open Access funding enabled and organized by Projekt DEAL. The study was funded by the German Foundation of Heart Research (Grant Number F/29/19). The HCHS is supported by the Innovative medicine initiative [Grant Number 116074], by the Foundation Leducq [Grant Number 16 CVD 03], by the euCanSHare Grant Agreement [Grant Number 825903-euCanSHare H2020], and the Deutsche Forschungsgemeinschaft [Grant Number TH1106/5-1; AA93/2-1]. Furthermore, it is supported by the participating institutes and departments from the University Medical Centre Hamburg-Eppendorf, which contribute with individual and scaled budgets to the overall funding. Technical equipment is provided by SIEMENS according to a contract for 12 years, the Schiller AG on a loan basis for 6 years, and Topcon on a loan basis from 2017 until 2022. The Hamburg City Health Study is additionally supported by an unrestricted Grant (2017 to 2022) by Bayer. Project-related analyses are supported by Amgen, Astra Zeneca, BASF, Deutsche Gesetzliche Unfallversicherung (DGUV), Deutsches Krebsforschungszentrum (DKFZ), Deutsches Zentrum für Herz-Kreislauf-Forschung (DZHK), Deutsche Stiftung für Herzforschung, Novartis, Seefried Stiftung, and Unilever. The study is further supported by donations from the “Förderverein zur Förderung der HCHS e.V.”, TePe® (2014) and Boston Scientific (2016). A current list of the supporters is online available on www.uke.de/hchs. Sponsor funding has in no way influenced the content or management of this study.
Author information
Authors and Affiliations
Contributions
J.P.W., J.N., and J.W. conceived, designed, and supervised the study. J.P.W., J.N., and J.W. wrote the manuscript. J.P.W., L.D., and J.W. performed data collection. R.b.d.K. performed data and statistical analysis. M.K. wrote the code for a program to extract text data from measurements of the aortic root and reviewed the manuscript. E.G., G.L., P.B., S.B., K.M., E.C., T.K., and G.A. reviewed the study. All authors contributed to manuscript revision, read, and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wenzel, JP., Nikorowitsch, J., bei der Kellen, R. et al. Comparison of cine cardiac magnetic resonance and echocardiography derived diameters of the aortic root in a large population-based cohort. Sci Rep 12, 15307 (2022). https://doi.org/10.1038/s41598-022-19461-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-19461-5
- Springer Nature Limited