Abstract
The aim of this study was to investigate the association between uric acid (UA) and cardiometabolic risk factors (CMRFs) by sex in youth with type 1 diabetes (T1D). Retrospective data collected from 1323 children and adolescents (5–18 years; 716 boys) with T1D recruited in 9 Italian Pediatric Diabetes Centers were analyzed. CMRFs included UA, HbA1c, blood pressure (BP), cholesterol (TC), HDL, triglycerides (TG), neutrophils (N) and lymphocytes (L) count, glomerular filtration rate (eGFR) (calculated using Schwartz-Lyon equation). In boys, we found a higher age, daily insulin dose, TG, TG/HDL ratio, TC/HDL ratio, systolic BP, N/L ratio and lower HDL, and eGFR across UA tertiles (p = 0.01–0.0001). Similar results were found in girls but not for TG and systolic BP. In boys, the odds ratio (OR) of high levels of TG/HDL ratio, TC/HDL ratio, BP and mildly reduced eGFR (MRGFR) increased for 0.5 mg/dL of UA. Instead, in girls an increased levels of 0.5 mg/dL of UA were associated with high OR of TC/HDL ratio, N/L ratio and MRGFR. Uric acid may represent a useful marker for identifying youth with T1D at high cardiometabolic risk, and this association appears to vary by sex.
Similar content being viewed by others
Introduction
Several cross-sectional and longitudinal studies have reported the independent role of uric acid (UA) as a predictor of cardiovascular morbidity, mortality and incident chronic kidney disease (CKD), in adult populations, especially with type 2 diabetes1,2,3,4. A robust association between UA and risk of incident cardiovascular events, mortality and an early onset of glomerular filtration rate (GFR) decline has also been reported in adults with type 1 diabetes (T1D)5,6,7. The mechanisms linking UA to cardio-renal disease risk are not fully elucidated yet. However, the effect of UA might be mediated by the activation of oxygen free radicals which in turn increase oxidative stress and inflammation1,8.
Several cross-sectional studies in pediatric populations, primarily performed in obese children, demonstrated that high UA levels are associated to metabolic syndrome and its individual components, nonalcoholic fatty liver disease, reduced estimated GFR (eGFR) and glucose dysmetabolism9,10,11.
In contrast, in adolescents with T1D, the relationship between UA levels and cardiometabolic risk factors (CMRFs) is controversial and little explored12,13. This is an important issue which deserves clarification since it is well known that cardiovascular prevention should start as early as possible, not only in children with obesity and type 2 diabetes but also in those with T1D14,15,16. Furthermore, a potential sex-related difference in the relationship between UA and CMRF has not been well explored so far12,13. Therefore, the aim of the present study was to evaluate the cardiometabolic risk (CMR) profile associated with UA levels in a large sample of children and adolescents with T1D. In addition, we assessed whether the UA-related CMR profile is different among the two sexes.
Results
The study population included 1323 individuals with T1D, 716 boys and 607 girls, with a mean (± SD) age of 13.5 ± 3.1 years. The clinical and biochemical characteristics of the study population are summarized in Table 1. There were no missing data. In boys, statistically significant differences were found across UA tertiles for age, BMI, daily insulin dose, HDL, TG, TG/HDL ratio, TC/HDL ratio, N/L ratio, systolic BP, eGFR, across tertiles of UA (p = 0.01–0.0001) (Table 2).
In girls, statistical differences between tertiles of UA were observed for age, daily insulin dose, HDL, TG/HDL ratio, TC/HDL ratio, N/L ratio and eGFR (p = 0.04–0.0001) (Table 3).
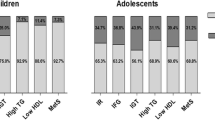
The age-adjusted proportions (mean and 95% CI) of youth with abnormal CMRFs across tertiles of UA both in boys and girls are shown in Fig. 1. A statistically significant increase across UA tertiles was found in both sexes for high TC/HDL ratio, and MRGFR. In boys, a higher prevalence of high blood pressure was found, whereas in the female group the prevalence of high N/L ratio varied across UA tertiles.
In boys, the odds ratio of high levels of TG/HDL ratio, TC/HDL ratio, BP and MRGFR increased for 0.5 mg/dL of UA; instead, in girls a 0.5 mg/dL increase in UA was associated with high odds ratio of TC/HDL ratio, N/L ratio and MRGFR (Table 4).
Discussion
Our study showed a potential association between UA and CMRFs in children and adolescents with T1D. This association suggests that UA could be a potential marker of cardiometabolic risk in this population. Furthermore, our study showed that the CMR risk profile associated with UA is worse in males than in females.
High levels of UA have been reported in adults with hypertension, obesity and metabolic syndrome1. Furthermore, prospective studies have shown the usefulness of UA in predicting incident cardio-renal events both in individuals with T1D and type 2 diabetes2,3,4,5,6,7.
In children, recent studies have shown a strong association between UA, nonalcoholic fatty liver disease mildly reduced eGFR and glucose dysmetabolism10,11. These studies have been performed in obese youth, whereas few and conflicting observations have been reported in youth with T1D12,13. Indeed, Lytvyn et al. could not detect any relationship between UA and cardio-renal abnormalities in their study conducted in small sample of 180 adolescents with T1D13. In contrast, Słomiński et al. demonstrated a positive association between UA and both nephropathy and subclinical inflammatory marker concentrations in boys but not in girls with T1D12. Differences findings between studies might be related to sample size, age range and other specific characteristics of the study populations, such as ethnicity.
In our study, we performed a comprehensive analysis including markers of low grade-inflammation and UA levels separately in the two sexes. To the best of our knowledge, our study demonstrates for the first time a robust association between CMRFs and UA in children and adolescents with T1D, which was more pronounced in boys than in girls.
Uric acid levels are higher in adult males than females, as well in obese boys than girls1,10,11. This difference is likely due to a distinct roles of sex hormones and a higher muscle mass in males17. In our study population, we did not observe any difference in UA levels between the two sexes, and this could be related most of participants being normal weight as well as to the specific age range.
In childhood, high values of UA are influenced by ethnicity, age and sex, so a universal definition of abnormal UA levels is lacking. In non-obese youth values above 6 or 7 mg/dL in boys and 5 or 6 mg/dL in girls were previously considered “elevated”18,19.
In children and adolescents with T1D levels of UA are lower than healthy individuals20. This result could lead to undermining the usefulness of assessing UA in clinical practice in youth with T1D. Despite this, our study showed a robust negative association between 'high normal' levels of UA and eGFR, in line with our previous studies21,22 and extends a positive association between UA and different CMRFs in young people with T1D. Interestingly, the results differed by sex, with boys having an increased risk for high TG/HDL, TC/HDL ratio, high BP and MRGFR, whereas girls showed a high risk of abnormal TC/HDL ratio, N/L ratio and MRGFR.
The different impact of UA in males vs females is still undefined. In our sample, we confirm in boys an association between high BP and high levels of UA as reported in a Caucasian sample of obese youth23. This suggests the link between UA, even in a high normal range, and elevated BP levels that may be useful to identify boys with T1D at high risk of hypertension.
In the present study, an association between the TG/HDL ratio and UA in boys was also detected. Insulin resistance may represent the link between the TG/HDL ratio and UA.
A further novel finding from our study is the potential association between high levels of UA and high TC/HDL ratio. This lipid ratio is considered a marker of atherogenic dyslipidemia24 and a more sensitive predictor of cardiovascular events in adults than total cholesterol25. Interestingly, the association between UA and high TC/HDL ratio is shared by the two sexes, independently of BMI. This further supports the usefulness of UA as marker of early atherosclerosis in both sexes.
In adults and obese children, the relationship between UA and MRGFR is well established1,10, whereas this association has been little explored in T1D, especially in young people12,13. The strong associations we found between UA and MRGFR in both boys and girls is in line with our previous reports21,22. The strength of this association sustains the evaluation of UA in clinical practice to identify youth with T1D at risk of eGFR decline in both sexes. Our results might have influenced by the specific formula used to calculate eGFR. At present, there is no consensus regarding the best method for estimating GFR in children and adolescents with T1D. We used a recently validated formula for the pediatric population, which relies on the use of creatinine and it is easily implemented into clinical practice26.
Of particular interest is the link between a high N/L ratio, as surrogate of low-grade inflammation, and high levels of UA in girls. The N/L ratio has been recently studied in several conditions, such as cardiac, vascular, and kidney disease where the low-grade inflammation was potentially involved27. In particular, recent studies demonstrated that a higher N/L ratio represents a useful marker to identify diabetic kidney disease28. In our sample we observed a robust association between N/L ratio and high levels of UA in girls, but not in boys. This association may be supported by higher percentage of girls with concomitant autoimmune diseases as compared to boys. This finding contrasts with those form Słomiński et al.12, who reported a positive association between UA and subclinical inflammatory marker concentrations only in boys but not in girls. The reasons for this discrepancy are unknown and warrant further study.
Some limitations of the present study need to be acknowledged. Firstly, being a retrospective multicenter, clinic-based study, data were collected and analyzed across different centers. However, all laboratories were standardized and used similar methods that were aligned. In addition, it is important to acknowledge that the study methods reflect real-word data collection and therefore closely reflect what can be implemented in daily clinical practice. The study population lacked diversity in terms of ethnicity, and this may limit the generalization of the study findings. However, despite these limitations, the present study, based on a large sample size and data collection, highlighted an important role of UA as potential early marker of cardiometabolic risk.
In conclusion, our study suggests that UA levels in youth with T1D could be useful to identify those at higher cardio-metabolic risk, although sex-related differences need to be taken into account. If confirmed by future prospective studies, the present findings could lead towards the implementation of UA as part of the set of investigations required in all youth with T1D from early stages of the disease to support prediction and prevention of cardio-metabolic complications.
Methods
This multicenter retrospective cross-sectional study including Caucasian children and adolescents with T1D consecutively recruited within 16 months from 1 January 2019 to 30 April 2020 in 9 Italian Pediatric Diabetes Centers that are part of the Diabetes Study Group of the Italian Society for Pediatric Endocrinology and Diabetology.
Medical history, biochemical and clinical data were retrieved from clinical records. Inclusion criteria were: availability of data regarding UA, diagnosis of T1D (at least 1 positive autoantibody), diabetes duration > 1 year, age 5–18 years, availability of anthropometric and biochemical data. Finally were analyzed records of 1323 youth. The study protocol was approved by the Ethics Committees of the participating Centers (Federico II University of Naples, coordinating center; Ancona; Bari; Campania Vanvitelli University of Naples; Parma; Verona; Bologna; Cagliari; Roma) and was conducted in accordance with the declaration of Helsinki and good clinical practice guidelines. Informed written consent was obtained from all the parents and/or patients before their inclusion in the study. The clinical and biochemical parameters were anonymously entered in a database using an alphanumeric and progressive code.
Measurements
For all study participants, the following data were recorded by each of the nine participating centers: age, sex, height, weight, body mass index (BMI), blood pressure (BP), serum creatinine (Scr), UA, HbA1c, total cholesterol (TC), triglycerides (TG), HDL, Neutrophil (N) and Lymphocyte (L) counts, albuminuria and insulin regimen (multiple daily injections vs continuous subcutaneous insulin infusion) and doses (IU/kg/day). Weight and height were collected by a single trained operator in each center. BMI-SDS was calculated based on the Italian BMI normative data29.
In all centers HbA1c was assessed by High-Performance Liquid Chromatography; TC, TG and HDL were determined by enzymatic methods; N and L counts were measured using an automated analyzer.
Scr was analyzed in 4 centers by enzymatic method and in 5 centers using Jaffé IDMS traceable method, while UA by the uricase method in all centers30. The relationship between Scr, age, sex, BMI and enzymatic method was explored by a linear regression analysis using Scr as dependent variable and age, sex, BMI and enzymatic method as covariate. We obtained a B coefficient for enzymatic method that was subtracted from the value of Scr assessed by Jaffé method. The value of Scr assessed by Jaffé method resulted identical to that obtained with enzymatic method as elsewhere described21.
The following parameters: TG to HDL (TG/HDL), TC to HDL (TC/HDL) and Neutrophil to Lymphocyte ratio (N/L) were calculated31.
Estimated glomerular filtration rate (eGFR) was calculated using Schwartz-Lyon equation31:
Evaluation of albuminuria was determined in 4 centers (n = 697) as albumin to creatinine ratio (ACR) (using an enzymatic method for assessment of Scr) on first-morning non-orthostatic urine samples, while in 5 centers (n = 626) as albumin excretion rate (AER) was assessed as timed urine collection, as recommended by the kidney disease: Improving Global Outcomes (KDIGO) clinical practice guideline32. Albuminuria was defined as normal to mildly increased (ACR < 30 mg/g; AER < 30 mg/d); moderately increased (ACR 30–300 mg/g; AER 30–300 mg/d); severely increased (ACR > 300 mg/g; AER > 300 mg/d)32. The presence of albuminuria was defined by positivity in at least two different measurements.
BP was measured following the recommendations by the European Society of Hypertension33. BP was measured, after 5 min of resting in a quiet room, using an appropriate sized arm cuff on the right arm and an aneroid sphygmomanometer. Three measurements were obtained every 2 min and the mean of the last two values were used in the analyses.
All blood samples for biochemical analyses were collected in each center after 12 h of fasting. Although laboratory analyses were performed in different laboratories, these are all part of the Italian National Health System and are certified according to International Standards ISO 9000 (www.iso9000.it/)34.
Definitions
Presence of puberty in all patients was identified as breast development in girls and testicular growth to at least 4 mL in volume in boys; pre-puberty in case of absence of any sign of puberty35. Autoimmune diseases were defined by the presence of autoimmune thyroiditis and/or celiac disease. Albuminuria (microalbuminuria) was defined as moderately increased (ACR 30–300 mg/g; AER 30–300 mg/d) or severely increased (ACR > 300 mg/g, > 300 mg/d) (macroalbuminuria)32. The presence of albuminuria was defined by positivity in at least two different measurements36.
High BP was defined using criteria proposed by the American Academy of Pediatrics: BP ≥ 95th percentile for age, sex and height in children aged less than 13 years or BP ≥ 130/80 mmHg in adolescents (age ≥ 13 years)37.
High TG/HDL ratio, TC/HDL ratio and N/L ratio were defined by the 75th percentile of the study population. Mildly reduced eGFR was defined by a value between 60–89 mL/min/1.73 m232.
Statistics
Data are expressed as mean ± standard deviation, median and interquartile range, or absolute and relative frequencies, unless otherwise stated. Given the skewed distribution of HbA1c, TG, TG/HDL ratio, TC/HDL ratio, N/L ratio and insulin dose, the statistical analysis of these variables was applied after log transformation and back transformation to natural units to allow presentation in the text and tables. Between groups differences in continuous variables were assessed with Student’s t-test. Analysis of variance (ANOVA) was applied to evaluate differences across tertiles of UA. The χ2 and Fisher’s exact tests, as appropriate, were used to compare categorical variables. Exact tests were performed using the Monte Carlo method. To evaluate the association (Odds ratio, 95% CI) between the CMRFs and UA, a logistic regression analysis was performed using the high levels of each CMRF as dependent variable and centers, age, insulin dose (IU/kg/day), BMI-SDS and 0.5 mg/dL of UA as covariates. Statistical analysis was performed using the IBM SPSS Statistics for Windows, Version 27. A p-value < 0.05 was considered statistically significant.
Ethics approval
The study protocol was approved by the ethics committees of all participating centers and was conducted in accordance with the declaration of Helsinki and good clinical practice guidelines.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sharaf El Din, U. A. A., Salem, M. M. & Abdulazim, D. O. Uric acid in the pathogenesis of metabolic, renal, and cardiovascular diseases: a review. J. Adv. Res. 8, 537–548. https://doi.org/10.1016/j.jare.2016.11.004 (2017).
Rodrigues, T. C. et al. Serum uric acid predicts progression of subclinical coronary atherosclerosis in individuals without renal disease. Diabetes Care 33, 2471–2473. https://doi.org/10.2337/dc10-1007 (2010).
Cang, Y. et al. Serum uric acid revealed a U-shaped relationship with all-cause mortality and cardiovascular mortality in high atherosclerosis risk patients: the ASSURE Study. Front. Cardiovasc. Med. 8, 641513. https://doi.org/10.3389/fcvm.2021.641513 (2021).
Zoppini, G. et al. Serum uric acid levels and incident chronic kidney disease in patients with type 2 diabetes and preserved kidney function. Diabetes Care 35, 99–104. https://doi.org/10.2337/dc11-1346 (2012).
Pilemann-Lyberg, S. et al. Uric acid is an independent risk factor for decline in kidney function, cardiovascular events, and mortality in patients with type 1 diabetes. Diabetes Care 42, 1088–1094. https://doi.org/10.2337/dc18-2173 (2019).
Bjornstad, P. et al. Serum uric acid predicts vascular complications in adults with type 1 diabetes: the coronary artery calcification in type 1 diabetes study. Acta Diabetol. 51, 783–791. https://doi.org/10.1007/s00592-014-0611-1 (2014).
Ficociello, L. H. et al. High-normal serum uric acid increases risk of early progressive renal function loss in type 1 diabetes. Diabetes Care 33, 1337–1343. https://doi.org/10.2337/dc10-0227 (2010).
Doehner, W., Jankowska, E. A., Springer, J., Lainscak, M. & Anker, S. D. Uric acid and xanthine oxidase in heart failure—emerging data and therapeutic implications. Int. J. Cardiol. 15, 15–19 (2016).
Moulin-Mares, S. R. A., Polyana, R., Oliosa, E. R., Zago-Gomes, M. P. & Mill, J. G. Association of uric acid with cardiovascular risk in Brazilian children and adolescents. Nutr. Metab. Cardiovasc. Dis. 31, 314–321. https://doi.org/10.1016/j.numecd.2020.09.012 (2021).
Di Bonito, P. et al. High uric acid, reduced glomerular filtration rate and non-alcoholic fatty liver in young people with obesity. J. Endocrinol. Investig. 43, 461–468. https://doi.org/10.1007/s40618-019-01130-6 (2020).
Di Bonito, P. et al. Uric acid, impaired fasting glucose and impaired glucose tolerance in youth with overweight and obesity. Nutr. Metab. Cardiovasc. Dis. 31, 675–680. https://doi.org/10.1016/j.numecd.2020.10.007 (2021).
Słomiński, B., Skrzypkowska, M., Ryba-Stanisławowska, M. & Brandtet, A. Sex-related association of serum uric acid with inflammation, kidney function and blood pressure in type 1 diabetic patients. Pediatr. Diabetes 19, 1014–1019. https://doi.org/10.1111/pedi.12670 (2018).
Lytvyn, Y. et al. Association between plasma uric acid levels and cardiorenal function in adolescents with type 1 diabetes. Diabetes Care 39, 611–616. https://doi.org/10.2337/dc15-2345 (2016).
De Ferranti, S. D. et al. Cardiovascular risk reduction in high-risk pediatric patients: a scientific statement from the American Heart Association. Circulation 139, e603–e634. https://doi.org/10.1161/CIR.0000000000000618 (2019).
Niechciał, E. & Marcovecchio, M. L. Treatment of cardiometabolic risk factors in patients with type 1 diabetes. Curr. Opin. Pediatr. 32, 589–594. https://doi.org/10.1097/MOP.0000000000000915 (2020).
Fornari, E. et al. Cardiovascular risk factors in children and adolescents with type 1 diabetes in Italy: a multicentric observational study. Pediatr. Diabetes 21, 1546–1555. https://doi.org/10.1111/pedi.13123 (2020).
Alvim, R. O. et al. Influence of muscle mass on the serum uric acid levels in children and adolescents. Nutr. Metab. Cardiovasc. Dis. 10(30), 300–305. https://doi.org/10.1016/j.numecd.2019.08.019 (2020).
Moulin-Mares, S. R. A. et al. Uric acid reference values: report on 1750 healthy Brazilian children and adolescents. Pediatr. Res. 89, 1855–1860 (2021).
Dai, C. et al. Age and gender-specific reference intervals for uric acid level in children aged 5–14 years in Southeast Zhejiang Province of China: Hyperuricemia in children may need redefinition. Front. Pediatr. 10(9), 560720. https://doi.org/10.3389/fped.2021.560720 (2021).
Lytvyn, Y. et al. Glycosuria-mediated urinary uric acid excretion in patients with uncomplicated type 1 diabetes mellitus. Am. J. Physiol. Renal. Physiol. 308, F77–F83. https://doi.org/10.1152/ajprenal.00555.2014 (2015).
Di Bonito, P. et al. “The DIAbetic kidney disease in youth with type 1 diabetes in ITALY” (DIANITALY) Study Group. Albuminuric and non-albuminuric reduced eGFR phenotypes in youth with type 1 diabetes: Factors associated with cardiometabolic risk. Nutr. Metab. Cardiovasc. Dis. 31, 2033–2041. https://doi.org/10.1016/j.numecd.2021.03.019 (2021).
Di Bonito, P. et al. Non-albuminuric reduced eGFR phenotype in children and adolescents with type 1 diabetes. Diabetes Res. Clin. Pract. 155, 107781. https://doi.org/10.1016/j.diabres.2019.07.005 (2019).
Lurbe, E. et al. Uric acid is linked to cardiometabolic risk factors in overweight and obese youths. J. Hypertens 36, 1840–1846. https://doi.org/10.1097/HJH.0000000000001814 (2018).
Millán, J. et al. Lipoprotein ratios: Physiological significance and clinical usefulness in cardiovascular prevention. Vasc. Health Risk Manag. 5, 757–765 (2009).
Genest, J., Frohlich, J., Fodor, G. & McPherson, R. The Working Group on Hypercholesterolemia and Other Dyslipidemias. Recommendations for the management of dyslipidemia and the prevention of cardiovascular disease: 2003 update. CMAJ 169, 921–924 (2003).
Björk, J., Nyman, U., Berg, U., Delanaye, P. & Dubourg, L. Validation of standardized creatinine and cystatin C GFR estimating equations in a large multicentre European cohort of children. Pediatr. Nephrol. 34(6), 1087–1098. https://doi.org/10.1007/s00467-018-4185-y (2019).
Bhat, T. et al. Neutrophil to lymphocyte ratio and cardiovascular diseases: a review. Expert Rev. Cardiovasc. Ther. 11, 55–59. https://doi.org/10.1586/erc.12.159 (2013).
Khandare, S. A., Chittawar, S., Nahar, N., Dubey, T. N. & Qureshi, Z. Study of neutrophil-lymphocyte ratio as novel marker for diabetic nephropathy in type 2 diabetes. Indian J. Endocrinol. Metab. 21(3), 387–392. https://doi.org/10.4103/ijem.IJEM_476_16 (2017).
Cacciari, E. et al. Italian cross-sectional growth charts for height, weight and BMI (2 to 20 yr). J. Endocrinol. Investig. 29, 581–593. https://doi.org/10.1007/BF03344156 (2006).
Liao, F. et al. Evaluation of a kinetic uricase method for serum uric acid assay by predicting background absorbance of uricase reaction solution with an integrated method. J. Zhejiang Univ. Sci. B 7(6), 497–502. https://doi.org/10.1631/jzus.2006.B0497 (2006).
De Souza, V. C. et al. Schwartz formula: is one k-coefficient adequate for all children?. PLoS ONE 7, e53439 (2021).
Levey, A. S. et al. Nomenclature for kidney function and disease: report of a kidney disease: improving global outcomes (KDIGO) consensus conference. Kidney Int. 97, 1117e29. https://doi.org/10.1016/j.kint.2020.02.010 (2020).
Lurbe, E. et al. European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J. Hypertens 34, 1887–1920 (2016).
Montano, D. Certifying leaders? High-quality management practices and healthy organizations: an ISO-9000 based standardization approach. Ind. Health 54, 324–336. https://doi.org/10.2486/indhealth.2015-0178 (2016).
Tanner, J. M. & Whitehouse, R. H. Clinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch Dis. Child 51(3), 170–179. https://doi.org/10.1136/adc.51.3.170 (1976).
Donaghue, K. C. et al. ISPAD Clinical Practice Consensus Guidelines 2018: Microvascular and macrovascular complications in children and adolescents. Pediatr. Diabetes 19, 262–274. https://doi.org/10.1111/j.1399-5448.2009.00576.x (2018).
Flynn, J. T. et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics 140, e20171904 (2017).
Acknowledgements
We kindly thank the patients and their families who participated in the study.
Author information
Authors and Affiliations
Contributions
P.D.B. and E.M. wrote the manuscript. M.L.M. suggested statistical analysis and P.D.B. oversaw the statistical analysis. E.M. and F.M.R. edited the manuscript. P.D.B., M.L.M., A.F., E.M. reviewed the manuscript. E.M., F.M.R., V.C., M.D., F.D.C., D.I., B.I., C.M., G.M., C.R., E.P., C.A.P., M.R.R., R.S., T.S. collected data. E.M. and P.D.B. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors were responsible for drafting the article and revising it critically for important intellectual content. All authors approved the version to be published.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Di Bonito, P., Rosanio, F.M., Marcovecchio, M.L. et al. Uric acid and cardiometabolic risk by gender in youth with type 1 diabetes. Sci Rep 12, 12153 (2022). https://doi.org/10.1038/s41598-022-15484-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-15484-0
- Springer Nature Limited