Abstract
MicroRNAs (miRNAs) play a vital role in improving meat quality by binding to messenger RNAs (mRNAs). We performed an integrated analysis of miRNA and mRNA expression profiling between bulls and steers based on the differences in meat quality traits. Fat and fatty acids are the major phenotypic indices of meat quality traits to estimate between-group variance. In the present study, 90 differentially expressed mRNAs (DEGs) and 18 differentially expressed miRNAs (DEMs) were identified. Eighty-three potential DEG targets and 18 DEMs were used to structure a negative interaction network, and 75 matching target genes were shown in this network. Twenty-six target genes were designated as intersection genes, screened from 18 DEMs, and overlapped with the DEGs. Seventeen of these genes enriched to 19 terms involved in lipid metabolism. Subsequently, 13 DEGs and nine DEMs were validated using quantitative real-time PCR, and seven critical genes were selected to explore the influence of fat and fatty acids through hub genes and predict functional association. A dual-luciferase reporter and Western blot assays confirmed a predicted miRNA target (bta-miR-409a and PLIN5). These findings provide substantial evidence for molecular genetic controls and interaction among genes in cattle.
Similar content being viewed by others
Introduction
Meat quality traits are complex genetic traits, which are crucial in meat quality improvement. The studies demonstrate that meat fat and fatty acid composition are influenced by genetic factors and are closely related to meat quality traits, including appearance, texture, flavor, juiciness, tenderness, and hardness1,2,3,4,5,6. It implies that fats and fatty acids may reflect complicated underlying genetic controls. These two factors may be considered major phenotypic indices for meat quality evaluation, aided by understanding its molecular genetic controls.
MicroRNAs (miRNAs) are noncoded small molecule RNAs widely found in eukaryotic organisms and approximately 19–24 nucleotides in length. MiRNAs are essential post-transcription regulatory factors and negatively modulate gene expression in animals at a post-transcriptional level through cleavage or translational inhibition7. RNAs use mature miRNAs to induce silencing complexes (RISCs) to modulate target gene messenger RNAs (mRNAs)8. When a miRNA is entirely complementary to a target mRNA, a miRISC directly cuts the target mRNA to reduce the level of gene expression. When a miRNA is paired incompletely with a target mRNA, the target gene translation is suppressible, and binding sites are mainly in the 3′ untranslated regions (UTR) of the target mRNA9,10. miRNA sequences are highly conserved in various species, from nematodes to cattle and humans, and thus considerably crucial to biology and developmental decisions11,12,13,14,15. The conserved Wat-son–Crick is pairing to the miRNA's 5′ region is called the miRNA "seed" 16. However, miRNA target sites from the multitude of 3ʹ-UTR segments exist and do not depend on seed sequence17. miRNAs also play an essential regulatory role in several biological processes18,19,20, such as cell proliferation21, differentiation22, and apoptosis23, as well as epigenetic changes24,25.
In cattle, miRNAs are considered to relate to embryonic development26,27,28, skeletal muscle function29,30,31, adipose32,33,34, and fat cell metabolism35,36,37. For example, miR-378 promotes the differentiation of bovine preadipocytes38; miR-2373-5p and miR-23b-3p are expressed highly in intramuscular fat39; miR-1 and miR-133 are muscle-specific and involved in the modulation of muscle proliferation40. Fluctuating hormones substantially affect gene activity and cause fat deposits in mammals41,42,43. In a castrated bull, the amount of testosterone in the body decreases or disappears, which reduces protein assimilation in the body, decreases muscle growth, and increases fat deposition. As the changing increases, the meat of steer exhibits increased fat content, improved taste, and enhanced tenderness44,45,46,47,48,49. In brief, hormonal changes between bull and steer cause the difference in meat quality and affect muscle and fat composition. Gene expression profiles help understand phenotypic differences, phenotypic effects, and underlying evolutionary mechanisms for individual genes50. However, differences in gene expression are especially worth exploring between bull and steer in the phenotypic traits of mammalian tissues.
The molecular mechanism of influences on meat quality traits relative to fat and fatty acid in cattle was explored in the present study. The experimental animals were divided into two groups based on the bull and steer. We assume a difference between the bull and steer selected to investigate the expression profiles of miRNAs and mRNAs. We conducted the integrated analyses of differentially expressed miRNAs (DEMs) and differentially expressed genes (DEGs) and identified the significant differential expression of miRNAs and mRNAs associated with meat quality in the Longissimus dorsi (LD). Subsequently, we performed the expression pattern and co-expression analyses on differentially expressed mRNAs. Moreover, an mRNA–miRNA interaction study was performed using computational prediction and expression relationship analysis. This primary focus is the in-depth analysis of critical genes and miRNAs related to fats and fatty acids to obtain a comprehensive view, reveal their molecular functions in lipid metabolism, and identify related regulatory pathways.
Results
Integrated analysis for DEMs and DEGs
A total of 18 DEMs and 90 DEGs were screened at rigorous threshold FC ≥ 2.0 and P ≤ 0.05 between bulls and steers, respectively (Supplementary Tables S6 and S7). The description of DEMs and mRNA was visualized using a volcano plot (Supplementary Fig. S1). Hierarchical clustering may have similar biological functions in the same cluster, as shown in Supplementary Figs. S2 and S3. The actual variations of expression are described in Supplementary Fig. S4. In this study, the target genes of miRNAs were predicted based on the transcript of Bos Taurus UMD 3.1.1 by using miRanda at 140 default threshold scores. A total of 5688 target genes for 18 DEMs were identified.
Pathway, GO, and UniProt analysis for intersection genes
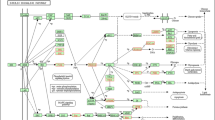
The significantly enriched GO, Pathway, and UniProt based on the 26 intersection genes were primarily involved in lipid metabolism, shown in Fig. 1. Some metabolic processes assessed were lipid droplets, identical protein binding, lipid homeostasis, the negative regulation of lipase activity, protein phosphorylation, lipid localization, lipid storage, triglyceride homeostasis, and response to lipid and muscle contraction. Exactly 17 genes of the 26 enriched in 19 terms were related to lipid metabolism (CIDEC, DGAT2, PLIN5, ABAT, ACVR1, ANGPTL4, DDIT4, HES1, ROCK2, S100A14, SCN4B, USP15, CSNK1A1, FABP4, ATG4A, CCNE2, and IRS1). Several genes were co-presented in multiple terms (Supplementary Fig. S5). Few genes enriched in the characterization of candidate canonical Pathway and UniProt. Significantly enriched pathways included the peroxisome proliferator-activated receptor (PPAR) signaling pathways (ANGPTL4, FABP4, and PLIN5) and the regulation of lipolysis in adipocytes (FABP4 and IRS1). Significantly enriched UniProt included the lipid droplet (CIDEC, DGAT2, and PLIN5).
miRNA-mRNA interaction network analysis
Using expression correlation and computation prediction (∣r∣ ≥ 0.8, P ≤ 0.05), we identified 83 potential mRNA targets for 18 miRNAs based on paired negative miRNA–mRNA expression profiling. The negative interaction network classed two subnetworks, including four miRNAs with regulated mRNAs (FABP4, CIDEC, ROCK2, THRSP, ANGPTL4, PLIN5, DGAT2, and CIDEC), and another included IRS1 with corresponding miRNAs (Fig. 2 and Supplementary Table S8).
A total of 75 matching target genes has been shown in the expression network. Among them, 26 target genes overlapped between DEGs and negative co-expression genes. These genes were an intersection gene for integration, as presented in Supplementary Fig. S6. In the 26 intersection genes, four down-regulated miRNAs were associated, and the relative expression of 12 genes increased, whereas the expression of 14 genes decreased in the bulls compared with steers. Most (14/26) genes associated with the 11 up-regulated miRNAs showed relatively increased abundances in steers, but the transcripts of 12 genes were down-regulated (Supplementary Table S9).
Intragroup co-expression analysis
The bull group (Fig. 3a) contains crucial co-expression genes, namely, ATG4A, ANGPTL4, FABP4, PIGO, ICA1, SSX21P, DDIT4 CIDEC, SCN4B, NMT2, GSTO1, COL21A1, CCNE2, and ABAT, were highly observed. The genes provide excellent contribution degrees and are indispensable for stable networks. Among them, CIDEC, FANBP4, ANGPTL4, ATG4A, and ABAT interacted with several genes. In the steer group (Fig. 3b), these crucial co-expression genes, namely, ATG4A, PER2, CHCHD7, MAP1A, PIGB, DDIT4, CIDEC, SCN4B, NMT2, CRAT, USP15, POLH, CYP1A1, EVL, DYNC1H1, KIAA0101, AGEF1, THRSP, COL21A1, CCNE2, PLIN5, ABAT, and DDIT4, were highly focused. These genes provided excellent contribution degrees and were indispensable for stable networks. CCNE2, DDIT4, CIDEC, EVL, ABAT, SCN4B, and ATG4A interacted with many genes.
Hub genes and Co-expression predict the functional association
Four intersection genes, namely PLIN5, ANGPTL4, FABP4, and IRS1, were primarily observed in fat and fatty acid significantly enriched pathways. To further understand the intersection genes' function, we analyzed the top 10 hub genes using the cytoHubba calculation module of Cytoscape (Version 3.6.1). We observed six hub genes, namely DGAT2, FABP4, CIDEC, PLIN5, THRSP, and ANGPTL4, classed in a cluster and descending order (Supplementary Fig. S7).
In the triangle matrices above, the intensity of the color indicates the level of confidence that two proteins are functionally associated, given the organism's overall expression data based on STRING (Version 11.0). Figure 4 shows the red genes representing strong confidence, their associations were specific and meaningful, and these genes jointly contributed to a shared function.
A total of seven genes were critical genes, namely, PLIN5, DGAT2, ANGPTL4, CIDEC, THRSP, FABP4, and IRS1. Although DGAT2, CIDEC, and THRSP were not found in the pathways, they presented in the interaction network, intragroup co-expression, and UniProt. To visualize and understand the interaction between them and their corresponding DEMs, we constructed an interaction network between miRNAs and genes in Fig. 5, and the involved genes included in IRSI were further validated by qRT-PCR (Fig. 6).
Interaction network for six critical genes with corresponding DEMs. Round and square indicate up- and down-regulation, respectively; Diamond and dash-dot indicate they involved pathways and enriched genes in the pathways, respectively; Vertical slash indicates the functional association between genes; Solid indicates a regulated relationship between gene and miRNA; Orange indicates the most strong closeness centrality, followed by yellow, green, and blue; Size is influenced by radiality; Line thickness is influenced by edge betweenness.
According to the predicted binding sites on the miRNA regulation of target genes, the transcripts of the PLIN5 gene may be targets consistent with miRNAs. This finding was explored by the co-transfection of luciferase reporter vectors containing the wild-type or mutant 3′ UTR of genes. As shown in Fig. 7, the luciferase activities of the wild-type gene reporter co-transfected with the miRNA mimic were reduced significantly compared with that co-transfected with the negative control mimic or in mutated-type reporters co-transfected with the miRNA mimic. Compared with the negative control, the mRNA level significantly decreased in bovine adipocytes at 48 h after transfection with the miRNA mimic.
Validation of miRNA endogenous targets via dual-luciferase reporter and Western blot assay. (a) Showed the activity of PLIN5 and bta-miR-409a by binding assay. (b) Presented the validating of PLIN5 as a target of bta-miR-209a at the protein level using western blot. Three tracks were captured from the original full-length membrane image and were presented in the same order consistent with (a). (c) is nucleotide sequences of binding sites located in the 3′ UTR of PLIN5. The sample derives from the same experiment, and that blots were processed in parallel. The party of the full-length membrane image is presented in Supplementary Fig. S8.
Discussion
MiRNA, fat and fatty acid in mammals
MiRNAs are small RNAs that regulate gene expression post-transcriptionally by repressing the translation and promoting the degradation of target mRNAs51. A miRNA typically has hundreds of evolutionarily conserved target sites, yet only very few predicted targets are down‐regulated more than twofold in miRNA transfection experiments51,52,53. Thus identifying target mRNAs is crucial to the understanding of the biological functions of miRNAs. At present, microarray-based techniques have been used in identifying mRNA–miRNA interactions, and negative expression correlations between miRNAs and their target mRNAs can be determined54,55. This strategy can significantly decrease the false-positive rate for identifying the miRNA with mRNA targets and improve laborious and time-consuming validation processes56. To date, the expression of miRNAs related to fat and fatty acid is still rarely reported between steers and bulls58.
The fat and fatty acid showed a significant difference between bulls and steers (Supplementary Tables S1, S2). It provides strong evidence to support the data of the experiment group that can be used for further analysis. The DEMs quantified, bta-miR-365-3p, bta-miR-122, bta-miR-200c, bta-miR-374b, bta-miR-15a, and bta-miR-671 were up-regulated in the LD of steers, and RT-qPCR confirmed the results. These significantly expressed miRNAs also were frequently reported in mammals. For example, A study identifies that activin A receptor type I (ACVR1) could be a direct target of miR-365-3p and further elucidate that miR-365-3p inhibits proliferation but promotes differentiation of primary bovine myoblasts by targeting the activin A receptor type I59. In human overlapping QTL for obesity, miR-365-3p is recognized in brown adipose tissue60. MiR-122 represents about 70% of the total miRNA in the liver, and its down- or up-regulation can modify fatty acids and cholesterol metabolism61,62. MiR-122 also associates lipid metabolism and adipocyte differentiation in cattle63,64,65, regulated PPAR-γ signaling and adipocyte differentiation in vitro and human adipose tissue66. MiR-200c is mentioned high expression in the mammary gland and present in milk whey67,68. MiR-374b has higher expression in the Longissimus lumborum muscle of grazing cattle than grain-fed Japanese Black cattle, as analyzed by qRT-PCR69. MiR-15a is reported more intensively in bovine mastitis. The studies indicate that miR-15a constitutes potential miRNA-mRNA regulatory pairs with target gene (IRAK2) for use as biomarkers to predict a mastitis response or indirectly affects the expression of CD163 gene in E. coli-infected mastitis cows70,71,72. MiR-671 and miR-7 largely interact with circRNA CDR1 antisense RNA (CDR1as; also known as ciRS-7)73,74. MiR-671 is up-regulated in the tissues from cows with mastitis75 and highly expressed in intermuscular fat65. MiR-7 and miR-15a may play a role in the variation of residual feed intake76.
In summary, these findings indicated that they might play essential or potential regulatory roles in lipid mechanisms in cattle. In comparison, changes in the three lipid-related DEMs (bta-miR-216b, bta-miR-409a, and bta-miR-2407) identified by miRNA profile and confirmed by qRT-PCR were down-regulated in the LD of steers. MiR-216b inhibits heat stress-induced cell apoptosis by targeting Fas in bovine mammary epithelial cells and modulates cell proliferation during early embryo development via K-RAS in cattle77,78. MiR-409a is up-regulated in bovine follicular atresia relative to healthy follicles79 and significantly down-regulated in preovulatory dominant follicles80. However, miR-409a related to lipid metabolism was down-regulated in the LD of steers in this study. An extensive literature search in PubMed and other databases did not retrieve any published results for miR-2407 in cattle.
Critical genes, fat and fatty acids in mammals
Seven critical genes were identified, four of which are involved in lipid metabolism pathways. The roles of ANGPTL4, FABP4, THRSP, and DGAT2 were elucidated. DGAT2and PLIN5 enriched in lipid droplet (GO:0005811), lipid storage (GO:0019915), and lipid droplet (KW-0551). ANGPTL4, FABP4, and PLIN5 were significant in PPAR signaling pathway (bta03320). FABP4 and IRS1 appeared in the regulation of lipolysis in the adipocytes pathway (bta04923). Bovine ANGPTL4 is a critical enzyme in lipolysis that stimulates the oxidation of fatty acids and inhibits fat accumulation by inhibiting lipoprotein lipase activity and is high in subcutaneous adipose tissue81. DGAT2 gene acts in the deposition of saturated fat in the adipose tissue82 and is identified as a functional candidate gene affecting milk production, especially for fat content in milk83. THRSP is expressed in mature adipocytes rather than in the early stages of adipogenesis, demonstrate that an increased expression of THRSP in Longissimus dorsi is a consequence of but not the reason for a higher number of intramuscular adipocytes in cattle with enhanced intramuscular fat deposition84. FABP4 is identified as a lipid transport protein in adipocytes and belongs to the FABP family, is a relevant candidate gene for beef quality. It correlates with intramuscular fat content85, fatty acid compositions86,87, and inhibits the expression by miR-130a/b in adipocyte differentiation 88.
Among the other rarely reported critical genes, CIDEC was mentioned maybe a potential earlier predictor of the marbling potential of differential intramuscular fat89 and lipid droplet90. IRS1 may be related to yak milk protein synthesis during the lactation cycle91. PLIN5 involved the regulation of lipid and was widely reported to relate to maintaining the balance between lipogenesis and lipolysis in humans92. A study suggested that PLIN5 may play a crucial role in regulating lipids deposition as code for proteins coating intracellular lipid droplets surfaces in pigs, and further pointed out that variations in the PLIN5 sequence may be linked to Hormone-sensitive lipase (LIPE) gene expression through a still poorly known regulative molecular process93. However, the knowledge of the PLIN5 gene in the lipid metabolism of cattle is still incomplete94. Given that they displayed significant differential expression except for critical genes, these genes must be further studied. Additional genes identified here include ATG4A, CCNE2, CSNK1A1, ABAT, HES1, and ROCK2. The effect of these genes on lipid metabolism or fat accumulation is not apparent; thus, these genes require further study in bovine fat and fatty acids.
In the interaction network of miRNA–mRNA, three DEMs, namely, bta-miR-2407, bta-miR-409a, and bta-miR-216b, up-regulated the critical genes. They may be strong candidates for regulating fats and fatty acids because independent qRT-PCRs between bulls and steers differentially express these three DEMs. Under the influence of bta-miR-409a, the activity of the wild-type of PLIN5 decreased in the dual-luciferase reporter assay. After the 3′ UTRs of genes were mutated artificially, the mutated-type activity was higher than that of the wild-type. This finding indicated that mutations were essential for miRNAs binding. Therefore, we supposed that PLIN5 is perhaps involved in lipid transport and storage protein in adipocytes or has aroused binding to lipid droplets, regulates their enlargement, and promotes incorporating endogenously synthesized fatty acids into triglycerides by down-regulating bta-miR-409a in cattle95,96,97. In brief, FABP4 and DGAT2 may play a central role in regulates lipid adipocytes and fatty acids. CIDEC and PLIN5 may be closely associated with lipid droplets and regulate their enlargement, highlighted in intragroup co-expression of steers and UniPort. In addition, PLIN5 is probably involved in lipid droplet homeostasis by regulating the storage of fatty acids in the PPAR signaling pathway (Fig. 1 and Supplementary Fig. S5)95. A close interaction relationship may exist among the four genes from hub genes, co-expression predicts, and intragroup co-expression. However, because part of anti-gene antibodies suited for bovine were lacking, changes in the level of some protein expression were not measured. Therefore, these actual results showed in Fig. 7.
In conclusion, differences between steers and bulls in the mRNAs and miRNAs related to fats and fatty acids were demonstrated. The integrated analysis of DEMs and DEGs suggests that three miRNAs (bta-miR-409a, bta-miR-2407, and bta-miR-216b) and seven critical genes (FABP4, IRS1, ANGPTL4, THRSP, CIDEC, DGAT2, and PLIN5) are strong candidate miRNAs and genes involved in regulating the fat and fatty acids in cattle. Potential miRNAs (bta-miR-365-3p, bta-miR-122, bta-miR-200c, bta-miR-374b, bta-miR-15a and bta-miR-671,) and genes (ATG4A, CCNE2, CSNK1A1, ABAT, HES1, and ROCK2) related to lipid metabolism were also identified. The study results indicate that bta-miR-409a that interacted with PLIN5 may play an important role in lipid droplets and fatty acid composition. The challenge for future studies is to identify the other relevant targets of miRNAs and determine the interaction among genes and contribute to the regulation of lipid droplets and fatty acid compositions.
Materials and methods
Ethics declarations
Following the protocols approved by Jilin Province, P. R. China for Biological Studies Animal Care and Use Committee, all experimental procedures were approved by the Animal Ethics Committee of Yanbian University and conducted in strict compliance with the recommendations (approval number: 2018062815-4). All efforts were made to minimize animal suffering.
Animals
Six-month-old Yanbian cattle were selected from a Jixing farm in Jilin province. After 30 days of fattening, 60 bulls were selected for castration. All experimental cattle in the same feeding conditions (Supplementary Table S10) were fattened to slaughter under the commercial standard at 36 months. The carcass was stored at 0–4 °C and bio-acid treated for three days. The 60 bull and 60 steer samples were obtained from LD between the 12th and 13th right ribs and stored at − 20 °C for meat quality determination. Three bull and three steer fresh tissue samples were randomly selected and cut into small 0.3 cm3 blocks, immediately placed in frozen tubes, and stored in liquid nitrogen to further detect miRNA and mRNA expression profiling.
RNA extraction
Total RNA was isolated from frozen tissue samples (n = 6) using mirVana™ RNA Isolation Kit (Applied Biosystem, Invitrogen) according to the manufacturer's instructions and treated with RNase-free DNase I to remove genomic DNA contamination. RNA integrity (RIN ≥ 7, 28S/18S ≥ 0.7) was assessed using an Agilent 2100 Bioanalyzer Lab-on-chip system (Agilent Technologies, USA). The same samples were used in all experiments.
Fat and fatty acids
Crude protein and crude fat were analyzed based on the semi-micro Kjeldahl method and the classic Soxhlet method. The composition analyses of fatty acid (2 g of freeze-dried sample collected from LD) were performed with a gas chromatograph (Agilent Technologies 7890A, Wilmington, DE, USA). The conditions of chromatography are as follows: column, Supelco sp-2560, 100 m × 250 μm × 0.2 μm; starting temperature, 130 °C, maintained for 3 min; after 5 min, the temperature increased to 240 °C at 4 °C/min for 50 min; and inlet temperature, 240 °C. Statistical analyses were performed with SAS 9.4. The phenotypic data of meat quality was estimated using ANOVA and expressed by mean ± standard deviation (Supplementary Tables S1 and S2). To observe genes that we predicted and cloned in advance, we preliminarily showed that the expression levels of ANGPTL4, FABP4, and DGAT2 genes in the LD are significantly higher than in bulls on predictions for previous work (15 bulls and 15 steers). The results are consistent with this study (Supplementary Table S3 and Fig. 8).
miRNA and mRNA expression profiling arrays
miRNA and mRNA profiling using microarrays was performed on individual RNA samples (n = 3) isolated from the bull and steer, respectively. Sample labeling, microarray hybridization, and washing were performed based on the standard protocols of the manufacturer.
GeneChip™ miRNA 3.0 Array (Affymetrix, miRNA Profiling) was used to perform miRNA expression profiling. In brief, total RNA was tallied with Poly A and then labeled with biotin. The labeled RNAs were hybridized onto the microarray. The slides were washed and stained, and the arrays were scanned using a GeneChip Scanner 3000 TG system (Thermo Fisher Scientific, USA). The GeneChip Command Console software (version4.0, Affymetrix) was used to analyze array images for capturing raw data. Expression console (version1.3.1, Affymetrix) was used in RMA normalization. The Genespring software (version 12.5; Agilent Technologies) was used for probe filtration. DEMs were identified through fold change (FC). P-value (P) was calculated using the t-test. The threshold of significance for the differential expression set was FC ≥ 2.0 and P ≤ 0.05. Hierarchical clustering was performed to show the distinguishable miRNA expression pattern among samples.
GeneChip™ Bovine Genome Array (Affymetrix, Transcriptome Profiling) was applied to analyze mRNAs expression. The total RNA of mRNA samples was primarily transcribed to double-stranded cDNA, and cRNA was synthesized and labeled with biotin compared with the miRNA profiling procedure. The labeled cRNAs were hybridized onto the microarray. The processing of sample labeling, microarray hybridization, and washing was consistent with miRNA profiling.
The Affymetrix GeneChip Command Console (Version 4.0, Affymetrix) and Genespring software (Version 12.5; Agilent Technologies) were employed to capture the raw data for Probe filtration. The raw data was normalized with the MAS5 algorithm by the Genespring software (Version 12.5; Agilent Technologies). The other analysis procedures, such as FC, significant difference (P-value), and hierarchical clustering, were the same as miRNA profiling.
Quantitative real-time PCR (qRT-PCR)
Part of the miRNA and intersection genes associated with fats and fatty acids were selected for verification. qRT-PCR was performed with a PCR Kit (LightCycler® 480 SYBR Green I Master, USA), and a miRNA-specific primer was used in quantifying nine relevant DEMs (Fig. 8a). U6 was selected as an internal control for the correction of analytical variations. Each primer was 10 μmol/μL. Then differentially regulated mRNAs as integration genes were used for validation (Fig. 8b). The primers used are shown in Supplementary Tables S4 and S5. The reacting system was 20 μL and used with an SYBR Premix Ex Taq™ II kit (Tli RNaseH Plus, Japan). The universal reverse primer that provided GAPDH was selected as a control for the correction of analytical variations. The final concentration of each primer was 10 μmol/μL. qRT-PCR experiments were performed in triplicate for each sample as described above. The relative expression was calculated using the 2–ΔΔCt method98. Differences between groups were analyzed using the Student's t-tests for independent samples and visualized as a histogram in R (Version 3.5.3).
Bioinformatic analysis
The miRanda algorithm was employed to predict the potential targets of all the DEMs. The transcript paired with miRNA information was extracted from the prediction results, and the annotation was obtained from the NCBI bovine (Bos taurus) database. Pathway, GO, and UniPort terms were analyzed using STRING 11.0 (https://string-db.org/). Intragroup co-expression analysis of mRNA was performed using the Pearson algorithm and Cytoscape (Version 3.6.1)99 to evaluate the co-expression genes in bulls and steers. The interaction networks of miRNA and mRNA were constructed using the Cytoscape software. MiRNAs from the most significant up- and down-regulated clusters were selected to construct the co-expression network100. Subsequently, the critical genes from negative correlation results in co-expression and related to significant lipid metabolism pathway were integrated and verified.
Vector construction
The wild-type construct 3′ UTR of genes containing the targets of miRNA binding site was amplified from bovine (Bos taurus) genomic DNA by PCR, and the primers are shown in Supplementary Table S2. PCR products were cloned into pmirGLO Dual-Luciferase miRNA Target Expression Vector (Promega Corporation, Madison, WI, USA) by using the Nhe I and Xho I restriction sites. The mutated-type construct 3′ UTR sequences of PLIN5 share a substitution (GUGGAGUUG → CGUUUCAAC) in the binding site.
Luciferase reporter assays
A dual-luciferase reporter assay was performed for detecting the interactions between the target genes and miRNAs. Bovine pre-adipocytes cells (BPCs) were seeded in 24-well plates at 2 × 104 cells/well and cultured under routine conditions with 10% fetal bovine serum (Ausbian). When the cells reached 60%–70% confluence, pmirGLO-3′ UTR (1 µg) was co-transfected with 60 nM negative control or miRNA mimic (both from RiboBio) by using 2 μL of X-tremegene HP (Promega) according to the manufacturer's instructions. The cells with plasmid and X-tremegene HP were cultured at 37 °C in an incubator supplemented with 5% CO2. The relative luciferase activity was measured 24–48 h after transfection by Dual-Luciferase® Reporter Assay system101.
Cell proliferation and differentiation assay
BPC model was established (Laboratory of Animal Medicine, Yanbian University, China), and cells were isolated from fatty tissues from calves. The filtered and rinsed cells were cultured at 37 °C in an incubator supplemented with 5% CO2. At 70% confluence, differentiation was induced with induced liquid IDI (0.5 mmol/L IBMX, 1 μmol/L DEX, and 10 μg/mL Insulin). After 48 h, the induced liquid was removed and changed to insulin-containing IDI.
Western blotting analysis
BPC lysed by RIPA Lysis and Extraction Buffer (ThermoFisher Technology, Beijing, China). Protein content was determined by the BCA protein assay reagent (Betotime Biotechnology, China), and 20 μg of each sample was subjected to polyacrylamide gel electrophoresis. The separated proteins were transferred to polyvinylidene fluoride (PVDF) membranes under 200 mA for 90 min. PVDF membranes were incubated with primary antibody (1:1000 dilution; PAB12542, Abnova Diagnostics, Dongguan, China), followed by incubation with horseradish peroxidase-conjugated secondary antibody (1:10,000 dilution; Zymed, San Diego, CA, USA). The membrane was re-probed with a primary antibody against GAPDH (1:3000 dilutions; Santa Cruz Biotech., Santa Cruz, CA, USA) as a control56. The assay was repeated to confirm the result.
Ethical approval
All methods were carried out in accordance with ARRIVE guidelines and regulations.
References
Gagaoua, M., Bonnet, M. & Picard, B. Protein array-based approach to evaluate biomarkers of beef tenderness and marbling in cows: understanding of the underlying mechanisms and prediction. Foods 9(9), 1180 (2020).
Pewan, S. B. et al. Genetics of omega-3 long-chain polyunsaturated fatty acid metabolism and meat eating quality in tattykeel australian white lambs. Genes (Basel) 11(5), 587 (2020).
Ekine-Dzivenu, C. et al. Phenotypic and genetic correlations of fatty acid composition in subcutaneous adipose tissue with carcass merit and meat tenderness traits in Canadian beef cattle. J. Anim. Sci. 95, 5184–5196 (2017).
Feitosa, F. L. et al. Genetic correlation estimates between beef fatty acid profile with meat and carcass traits in Nellore cattle finished in feedlot. J. Appl. Genet. 58, 123–132 (2017).
Ros-Freixedes, R., Reixach, J., Bosch, L., Tor, M. & Estany, J. Genetic correlations of intramuscular fat content and fatty acid composition among muscles and with subcutaneous fat in Duroc pigs. J. Anim. Sci. 92, 5417–5425 (2014).
Ibanez-Escriche, N., Magallon, E., Gonzalez, E., Tejeda, J. F. & Noguera, J. L. Genetic parameters and crossbreeding effects of fat deposition and fatty acid profiles in Iberian pig lines. J. Anim. Sci. 94, 28–37 (2016).
Liu, H. C., Hicks, J. A., Trakooljul, N. & Zhao, S. H. Current knowledge of microRNA characterization in agricultural animals. Anim. Genet. 41, 225–231 (2010).
Kawamata, T. & Tomari, Y. Making RISC. Trends Biochem Sci. 35, 368–376 (2010).
Bartel, D. P. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116, 281–297 (2004).
Ambros, V. The functions of animal microRNAs. Nature 431, 350–355 (2004).
Kedde, M. & Agami, R. Interplay between microRNAs and RNA-binding proteins determines developmental processes. Cell Cycle 7, 899–903 (2008).
McDaneld, T. G. MicroRNA: mechanism of gene regulation and application to livestock. J. Anim. Sci. 87, E21-28 (2009).
Ibanez-Ventoso, C., Vora, M. & Driscoll, M. Sequence relationships among C. elegans, D. melanogaster and human microRNAs highlight the extensive conservation of microRNAs in biology. PLoS ONE 3, e2818 (2008).
Friedman, R. C., Farh, K. K., Burge, C. B. & Bartel, D. P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 19, 92–105 (2009).
Takane, K. et al. Computational prediction and experimental validation of evolutionarily conserved microRNA target genes in bilaterian animals. BMC Genom. 11, 101 (2010).
Lewis, B. P., Shih, I. H., Jones-Rhoades, M. W., Bartel, D. P. & Burge, C. B. Prediction of mammalian microRNA targets. Cell 115, 787–798 (2003).
Bartel, D. P. MicroRNAs: target recognition and regulatory functions. Cell 136, 215–233 (2009).
Mathew, R., Mattei, V., Al-Hashmi, M. & Tomei, S. Updates on the current technologies for microRNA profiling. Microrna 9, 17–24 (2020).
Gao, L. & Jiang, F. MicroRNA (miRNA) profiling. Methods Mol. Biol. 1381, 151–161 (2016).
Saliminejad, K., Khorshid, H. R. K., Fard, S. S. & Ghaffari, S. H. An overview of microRNAs: biology, functions, therapeutics, and analysis methods. J. Cell Physiol. 234, 5451–5465 (2019).
Wang, J. et al. MiR-208b regulates cell cycle and promotes skeletal muscle cell proliferation by targeting CDKN1A. J. Cell Physiol. 234, 3720–3729 (2019).
Song, C. et al. miR-483 inhibits bovine myoblast cell proliferation and differentiation via IGF1/PI3K/AKT signal pathway. J. Cell Physiol. 234, 9839–9848 (2019).
Zhang, L. et al. Bta-miR-34b inhibits proliferation and promotes apoptosis via the MEK/ERK pathway by targeting MAP2K1 in bovine primary Sertoli cells. J Anim Sci. 98(10), skaa313 (2020).
Fridrichova, I. & Zmetakova, I. MicroRNAs contribute to breast cancer invasiveness. Cells 8(11), 1361 (2019).
Melnik, B. C. & Schmitz, G. MicroRNAs: Milk’s epigenetic regulators. Best Pract. Res. Clin. Endocrinol. Metab. 31, 427–442 (2017).
Scheel, T. K. H. et al. Global mapping of miRNA-target interactions in cattle (Bos taurus). Sci. Rep. 7, 8190 (2017).
Wang, L., Hand, J. M., Fu, L., Smith, G. W. & Yao, J. DNA methylation and miRNA-1296 act in concert to mediate spatiotemporal expression of KPNA7 during bovine oocyte and early embryonic development. BMC Dev. Biol. 19, 23 (2019).
Wu, C., Blondin, P., Vigneault, C., Labrecque, R. & Sirard, M. A. Sperm miRNAs- potential mediators of bull age and early embryo development. BMC Genom. 21, 798 (2020).
Ji, H. et al. Differential expression profile of microRNA in yak skeletal muscle and adipose tissue during development. Genes Genom.. 42, 1347–1359 (2020).
Carvalho, E. B. et al. Differentially expressed mRNAs, proteins and miRNAs associated to energy metabolism in skeletal muscle of beef cattle identified for low and high residual feed intake. BMC Genom. 20, 501 (2019).
Kappeler, B. I. G. et al. MiRNAs differentially expressed in skeletal muscle of animals with divergent estimated breeding values for beef tenderness. BMC Mol. Biol. 20, 1 (2019).
Pratt, S. L., Burns, T. A., Owens, M. D. & Duckett, S. K. Isolation of total RNA and detection procedures for miRNA present in bovine-cultured adipocytes and adipose tissues. Methods Mol. Biol. 936, 181–194 (2013).
Muroya, S. et al. Coordinated alteration of mRNA-microRNA transcriptomes associated with exosomes and fatty acid metabolism in adipose tissue and skeletal muscle in grazing cattle. Asian-Austr. J. Anim. Sci. 33, 1824–1836 (2020).
Huang, J. et al. miRNA transcriptome comparison between muscle and adipose tissues indicates potential miRNAs associated with intramuscular fat in Chinese swamp buffalo. Genome 62, 729–738 (2019).
Zhang, J. S. et al. Integrated microRNA-mRNA analysis reveals the roles of microRNAs in the muscle fat metabolism of Yanbian cattle. Anim. Genet. 52, 598–607 (2021).
Yu, X. et al. Isolation and identification of bovine preadipocytes and screening of MicroRNAs associated with adipogenesis. Animals (Basel) 10(5), 818 (2020).
Raza, S. H. A. et al. The role of MicroRNAs in muscle tissue development in beef cattle. Genes (Basel) 11(3), 295 (2020).
Liu, S. Y. et al. MiR-378 Plays an Important Role in the Differentiation of Bovine Preadipocytes. Cell Physiol. Biochem. 36, 1552–1562 (2015).
Wang, H., Zheng, Y., Wang, G. & Li, H. Identification of microRNA and bioinformatics target gene analysis in beef cattle intramuscular fat and subcutaneous fat. Mol. Biosyst. 9, 2154–2162 (2013).
Chen, J. F. et al. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat. Genet. 38, 228–233 (2006).
Christoffersen, B. O., Gade, L. P., Golozoubova, V., Svendsen, O. & Raun, K. Influence of castration-induced testosterone and estradiol deficiency on obesity and glucose metabolism in male Gottingen minipigs. Steroids 75, 676–684 (2010).
Gui, L. S. et al. Association of hormone-sensitive lipase (HSL) gene polymorphisms with the intramuscular fat content in two Chinese beef cattle breeds. Genomics 112, 3883–3889 (2020).
Daix, M. et al. Relationship between leptin content, metabolic hormones and fat deposition in three beef cattle breeds. Vet. J. 177, 273–278 (2008).
Choat, W. T. et al. The effects of cattle sex on carcass characteristics and Longissimus muscle palatability. J. Anim. Sci. 84, 1820–1826 (2006).
Guo, Y., Zhang, X., Huang, W. & Miao, X. Identification and characterization of differentially expressed miRNAs in subcutaneous adipose between Wagyu and Holstein cattle. Sci. Rep. 7, 44026 (2017).
Fritsche, S. & Steinhart, H. Differences in natural steroid hormone patterns of beef from bulls and steers. J. Anim. Sci. 76, 1621–1625 (1998).
Mersmann, H. J. Effect of sex on lipogenic activity in swine adipose tissue. J. Anim. Sci. 58, 600–604 (1984).
Mueller, L. F. et al. Gender status effect on carcass and meat quality traits of feedlot Angus x Nellore cattle. Anim. Sci. J. 90, 1078–1089 (2019).
Cafferky, J. et al. Effect of Breed and Gender on Meat Quality of M. Longissimus thoracis et Lumborum Muscle from Crossbred Beef Bulls and Steers. Foods. 8(5), 173 (2019).
Naqvi, S. et al. Conservation, acquisition, and functional impact of sex-biased gene expression in mammals. Science 365(6450), p.eaaw7317 (2019).
Rzepiela, A. J. et al. Single-cell mRNA profiling reveals the hierarchical response of miRNA targets to miRNA induction. Mol. Syst. Biol. 14, e8266 (2018).
Gaidatzis, D., van Nimwegen, E., Hausser, J. & Zavolan, M. Inference of miRNA targets using evolutionary conservation and pathway analysis. BMC Bioinf. 8, 69 (2007).
Hausser, J. & Zavolan, M. Identification and consequences of miRNA-target interactions–beyond repression of gene expression. Nat. Rev. Genet. 15, 599–612 (2014).
Ruike, Y. et al. Global correlation analysis for micro-RNA and mRNA expression profiles in human cell lines. J. Hum. Genet. 53, 515–523 (2008).
Van der Auwera, I. et al. Integrated miRNA and mRNA expression profiling of the inflammatory breast cancer subtype. Br. J. Cancer. 103, 532–541 (2010).
Tang, Z. et al. Integrated analysis of miRNA and mRNA paired expression profiling of prenatal skeletal muscle development in three genotype pigs. Sci. Rep. 5, 15544 (2015).
Zhang, Y. Y. et al. Transcriptome analysis of mRNA and microRNAs in intramuscular fat tissues of castrated and intact male Chinese Qinchuan cattle. PLoS ONE 12, e0185961 (2017).
Wang, Y. et al. Fatty acid-binding protein 1 increases steer fat deposition by facilitating the synthesis and secretion of triacylglycerol in liver. PLoS ONE 14, e0214144 (2019).
Hao, D. et al. MicroRNA bta-miR-365-3p inhibits proliferation but promotes differentiation of primary bovine myoblasts by targeting the activin A receptor type I. J. Anim. Sci. Biotechnol. 12, 16 (2021).
Gottmann, P. et al. A computational biology approach of a genome-wide screen connected miRNAs to obesity and type 2 diabetes. Mol. Metab. 11, 145–159 (2018).
Baldini, F. et al. Aquaporin-9 is involved in the lipid-lowering activity of the nutraceutical silybin on hepatocytes through modulation of autophagy and lipid droplets composition. Biochim. Biophys. Acta Mol. Cell Biol. Lipids. 1865, 158586 (2020).
Jin, J. C. et al. MicroRNA122 regulation of the morphology and cytoarchitecture of hepatoma carcinoma cells. Mol. Med. Rep. 9, 1376–1380 (2014).
Sun, J. et al. Discovery of novel and differentially expressed microRNAs between fetal and adult backfat in cattle. PLoS ONE 9, e90244 (2014).
Li, N. et al. Differential expression of mRNA-miRNAs related to intramuscular fat content in the Longissimus dorsi in Xinjiang brown cattle. PLoS ONE 13, e0206757 (2018).
Oliveira, G. B. et al. Integrative analysis of microRNAs and mRNAs revealed regulation of composition and metabolism in Nelore cattle. BMC Genom. 19, 126 (2018).
Liao, C. H. et al. MiR-122 marks the differences between subcutaneous and visceral adipose tissues and associates with the outcome of bariatric surgery. Obes. Res. Clin. Pract. 12, 570–577 (2018).
Sanchez, M. P. et al. Sequence-based GWAS, network and pathway analyses reveal genes co-associated with milk cheese-making properties and milk composition in Montbeliarde cows. Genet. Sel. Evol. 51, 34 (2019).
Chen, X. et al. Identification and characterization of microRNAs in raw milk during different periods of lactation, commercial fluid, and powdered milk products. Cell Res. 20, 1128–1137 (2010).
Muroya, S., Shibata, M., Hayashi, M., Oe, M. & Ojima, K. Differences in circulating microRNAs between grazing and grain-fed wagyu cattle are associated with altered expression of intramuscular microRNA, the potential target PTEN, and lipogenic genes. PLoS ONE 11, e0162496 (2016).
Li, H. M., Wang, C. M., Li, Q. Z. & Gao, X. J. MiR-15a decreases bovine mammary epithelial cell viability and lactation and regulates growth hormone receptor expression. Molecules 17, 12037–12048 (2012).
Chen, Z. et al. Screening candidate microR-15a- IRAK2 regulatory pairs for predicting the response to Staphylococcus aureus-induced mastitis in dairy cows. J. Dairy Res. 86, 425–431 (2019).
Ju, Z. et al. Genome-wide methylation and transcriptome of blood neutrophils reveal the roles of DNA methylation in affecting transcription of protein-coding genes and miRNAs in E coli-infected mastitis cows. BMC Genom. 21, 102 (2020).
Hansen, T. B. et al. Natural RNA circles function as efficient microRNA sponges. Nature 495, 384–388 (2013).
Gebert, L. F. R. & MacRae, I. J. Regulation of microRNA function in animals. Nat. Rev. Mol. Cell Biol. 20, 21–37 (2019).
Hou, Q. et al. Identification of splice variants, targeted microRNAs and functional single nucleotide polymorphisms of the BOLA-DQA2 gene in dairy cattle. DNA Cell Biol. 31, 739–744 (2012).
De Oliveira, P. S. N. et al. An integrative transcriptome analysis indicates regulatory mRNA-miRNA networks for residual feed intake in Nelore cattle. Sci. Rep. 8, 17072 (2018).
Alves, M. B. R. et al. Sperm-borne miR-216b modulates cell proliferation during early embryo development via K-RAS. Sci. Rep. 9, 10358 (2019).
Cai, M. et al. MicroRNA-216b inhibits heat stress-induced cell apoptosis by targeting Fas in bovine mammary epithelial cells. Cell Stress Chaperones. 23, 921–931 (2018).
Donadeu, F. X., Mohammed, B. T. & Ioannidis, J. A miRNA target network putatively involved in follicular atresia. Domest Anim. Endocrinol. 58, 76–83 (2017).
Gebremedhn, S. et al. MicroRNA expression profile in bovine granulosa cells of preovulatory dominant and subordinate follicles during the late follicular phase of the estrous cycle. PLoS ONE 10, e0125912 (2015).
Mi, X. et al. GR and Foxa1 promote the transcription of ANGPTL4 in bovine adipocytes. Mol. Cell Prob. 48, 101443 (2019).
Berton, M. P. et al. Gene expression profile of intramuscular muscle in Nellore cattle with extreme values of fatty acid. BMC Genom. 17, 972 (2016).
Liu, J., Wang, Z., Li, J., Li, H. & Yang, L. Genome-wide identification of diacylglycerol acyltransferases (DGAT) family genes influencing Milk production in Buffalo. BMC Genet. 21, 26 (2020).
Schering, L., Albrecht, E., Komolka, K., Kuhn, C. & Maak, S. Increased expression of thyroid hormone responsive protein (THRSP) is the result but not the cause of higher intramuscular fat content in cattle. Int. J. Biol. Sci. 13, 532–544 (2017).
Barton, L., Bures, D., Kott, T. & Rehak, D. Associations of polymorphisms in bovine DGAT1, FABP4, FASN, and PPARGC1A genes with intramuscular fat content and the fatty acid composition of muscle and subcutaneous fat in Fleckvieh bulls. Meat. Sci. 114, 18–23 (2016).
Li, H., Hao, X., Liu, Y., Li, Y. & Jin, Z. ZnxCd1-xS nanoparticles dispersed on CoAl-layered double hydroxide in 2D heterostructure for enhanced photocatalytic hydrogen evolution. J. Colloid Interface Sci. 572, 62–73 (2020).
Nafikov, R. A. et al. Association of polymorphisms in solute carrier family 27, isoform A6 (SLC27A6) and fatty acid-binding protein-3 and fatty acid-binding protein-4 (FABP3 and FABP4) with fatty acid composition of bovine milk. J. Dairy Sci. 96, 6007–6021 (2013).
Ma, X. et al. Bta-miR-130a/b regulates preadipocyte differentiation by targeting PPARG and CYP2U1 in beef cattle. Mol. Cell Probes. 42, 10–17 (2018).
Hudson, N. J. et al. Longitudinal muscle gene expression patterns associated with differential intramuscular fat in cattle. Animal 9, 650–659 (2015).
Wang, J. et al. Haplotypes in the promoter region of the CIDEC gene associated with growth traits in Nanyang cattle. Sci. Rep. 5, 12075 (2015).
Xia, W., Osorio, J. S., Yang, Y., Liu, D. & Jiang, M. F. Short communication: characterization of gene expression profiles related to yak milk protein synthesis during the lactation cycle. J. Dairy Sci. 101, 11150–11158 (2018).
Mass Sanchez, P. B., Krizanac, M., Weiskirchen, R. & Asimakopoulos, A. Understanding the role of perilipin 5 in non-alcoholic fatty liver disease and its role in hepatocellular carcinoma: a review of novel insights. Int. J. Mol. Sci. 22(10), 5284 (2021).
Zappaterra, M., Mazzoni, M., Zambonelli, P. & Davoli, R. Investigation of the Perilipin 5 gene expression and association study of its sequence polymorphism with meat and carcass quality traits in different pig breeds. Animal 12, 1135–1143 (2018).
Cesar, A. S. et al. Differences in the skeletal muscle transcriptome profile associated with extreme values of fatty acids content. BMC Genom. 17, 961 (2016).
Bindesboll, C., Berg, O., Arntsen, B., Nebb, H. I. & Dalen, K. T. Fatty acids regulate perilipin5 in muscle by activating PPARdelta. J. Lipid Res. 54, 1949–1963 (2013).
Jia, H. et al. Perilipin 5 promotes hepatic steatosis in dairy cows through increasing lipid synthesis and decreasing very low density lipoprotein assembly. J. Dairy Sci. 102, 833–845 (2019).
Najt, C. P. et al. Lipid droplet-derived monounsaturated fatty acids traffic via PLIN5 to allosterically activate SIRT1. Mol. Cell. 77, 810–824 (2020).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Shannon, P. et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504 (2003).
Assenov, Y., Ramirez, F., Schelhorn, S. E., Lengauer, T. & Albrecht, M. Computing topological parameters of biological networks. Bioinformatics 24, 282–284 (2008).
Shimono, Y. et al. Downregulation of miRNA-200c links breast cancer stem cells with normal stem cells. Cell 138, 592–603 (2009).
Acknowledgements
This work is supported by Regions of the National Natural Science, Natural Science Foundation of China, NO. 31160442; Higher Education Discipline Innovation 111 project, NO. D20034; Next-Generation BioGreen 21 Program, Rural Development Administration, Republic of Korea, No. PJ013222022020.
Author information
Authors and Affiliations
Contributions
X.J. and K.S. conceived the idea, supervised the project; Y.L. performed the analyses and drafted the manuscript; M.Y. assisted in drafting the manuscript. A.L., G.X., and C.R., collected and recorded phenotype data. M.Y., J.Y., A.L., and X.L. carried out cell assay, qRT-PCR, western blotting, and dual-luciferase reporter assay experiments, respectively. D.Y., K.N., and H.J. assisted with experiments, reviewed the manuscript, and provided critical comments. All authors reviewed the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Y., Yang, M., Lou, A. et al. Integrated analysis of expression profiles with meat quality traits in cattle. Sci Rep 12, 5926 (2022). https://doi.org/10.1038/s41598-022-09998-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-09998-w
- Springer Nature Limited