Abstract
Gastrointestinal helminths infect livestock causing health problems including severe diarrhoea. To explore the underlying biological mechanisms relating to development and control of diarrhoea, we compared 4 sheep that were susceptible to development of diarrhoea with 4 sheep that were diarrhoea-resistant. Transcriptomes in the tissues where the parasites were located were analyzed using RNASeq. By considering low-diarrhoea sheep as control, we identified 114 genes that were down-regulated and 552 genes that were up-regulated genes in the high-diarrhoea phenotype. Functional analysis of DEGs and PPI sub-network analysis showed that down-regulated genes in the high-diarrhoea phenotype were linked to biological processes and pathways that include suppression of ‘antigen processing and presentation’, ‘immune response’, and a list of biological functional terms related to ‘suppression in immune tolerance’. On the other hand, up-regulated genes in the high-diarrhoea phenotype probably contribute to repair processes associated with tissue damage, including ‘extracellular matrix organization’, ‘collagen fibril organization’, ‘tissue morphogenesis’, ‘circulatory system development’, ‘morphogenesis of an epithelium’, and ‘focal adhesion’. The genes with important roles in the responses to helminth infection could be targeted in breeding programs to prevent diarrhoea.
Similar content being viewed by others
Introduction
The sheep is globally relevant to production of milk, meat, and wool for human consumption. These industries contribute significantly to the world economy and are particularly important for Australia, one of the largest sheep-producing countries in the world (www.fao.org)1. One major challenge is the disease caused by parasites2. For example, in south-western Australia, owing to its Mediterranean climate, the winter rainfall season provides conditions that allow the larvae of gastrointestinal helminths to flourish on pasture, and thus infect grazing sheep. The major helminth species in this region, Teladorsagia circumcinta and Trichostrongylus colubriformis3, are responsible for significant losses in productivity and they also cause diarrhoea that predisposes the host to flystrike, a serious concern for animal welfare.
As licensed vaccines are not available for T. circumcinta or T. colubriformis4,5, the dominant approach for many decades has been to use anthelmintic medications6. However, the helminths are becoming increasingly resistant to them4. Moreover, there is an increase in consumer demand for residue-free and chemical-free products, placing extra pressure on this option7. The most promising long-term solution seems to be genetic selection of sheep that resist helminths8, a path taken in 1988 by the Department of Primary Industries and Regional Development, Western Australia. The outcome has been the ‘Rylington’ flock of Merino sheep that are highly resistant to helminths, with very low faecal egg counts (FEC) for the dominant Teladorsagia and Trichostrongylus species8,9,10. However, breeding for helminth resistance appears to have led to an increase in susceptibility to diarrhoea11,12. To further address the diarrhoea problem, a ‘breech-strike flock’ was developed by selecting for the low-diarrhoea phenotype. In both flocks, diarrhoea has been assessed by faecal consistency score (or the more colloquial ‘dag’ score), a subjective measure of the amount of faecal matter accumulated around the anus that is a heritable trait13.
Helminths evoke both adaptive and innate immune responses14. The adaptive (long-term) response is initiated by the binding of helminth antigen to CD4+ T cells through molecules of the major histocompatibility complex class II (MHC-II), eliciting a Th2 antibody-mediated response. This leads to the secretion of Th2-type cytokines (IL-13, IL-5, IL-4), infiltration of mast cells and eosinophils, and production of antibodies (IgE and/or IgA15,16). The innate immune response, on the other hand, comprises the physical barriers (gut mucus layer, defensins, trefoil factors, enteric muscle contractility)17,18, pattern recognition receptors (C-type lectin receptors and Toll-like receptors expressed by antigen-presenting immune cells in tissues)19, proinflammatory and cytotoxic cells (eosinophils, mast cells)20, and various chemo-attractants (such as IL-5, eotaxin family of chemokines CCL26, CCL24, CCL11)21. These innate responses act to expel the helminths from the gastrointestinal tract, and often involve diarrhoea. However, diarrhoea can also be caused by damage to gastrointestinal mucosa by the helminths, increased gut permeability, changes in gut motility22,23 and, in some cases, ‘hypersensitivity scouring/diarrhoea’– a heightened immune response to the larvae12,24,25. A number of studies have linked susceptibility to helminths in sheep to a Th1-mediated immune response, involving Th1-type cytokines (IL-12, INF-γ) production and proliferation of CD8+ cytotoxic T cells26. The diarrhoea is thus a complex problem and does not depend upon any single factor25.
To resolve the problem, we need to go beyond the measurements of environmental factors and phenotypic aspects (FEC, diarrhoea score), and develop an understanding of the biological mechanisms that underlay the helminth-diarrhoea relationship. Most of the physiological mechanisms are controlled by normal gene expression and most of the pathological processes can be explained by disrupted gene expression27. Therefore, analysis of gene expression and the associated molecular pathways should be useful for studying and grouping genes that are responsible for a phenotype. Such studies have already provided promising gene candidates with respect to helminth resistance and diarrhoea control28. For example, IL-13, IL-4 and IL-5, the humoral immune response, protein synthesis and the inflammatory response, were all found to be central to a phenotype for resistance to T. circumcinta infection in the abomasal lymph node transcriptome29. In another study of abomasal mucosa and abomasal lymph nodes, ITLN2, CLAC1, galectins, the PPARG signaling pathway and the cytokine-mediated immune response, were associated with resistance to T. circumcinta infection in sheep28. Moreover, resistance to helminths has been associated with allelic forms of candidate genes, including those for MHC Class I and II30 and MHC-DRB31.
So far, the genetic selection of animals has relied on two phenotypic traits—FEC (described above) and an indictor trait for diarrhoea32—and has not explored molecular markers that could be more reliable for high resolution selection33. The three ways to identify such markers include genome-wide association studies (GWAS), the mapping of quantitative trait loci (QTL) and potential gene expression analysis (e.g., RNASeq), with the latter being more practical for evaluating links between variation in gene expression and phenotypic traits29,34. Generally, the relationship between helminths and diarrhoea has been considered an outcome of the infection35 but it is clear that the two can be independent, as shown by studies in sheep where the level of diarrhoea is often not related to FEC, total helminth burden, or the number of infective larvae administered36,37,38. We have therefore studied animals that are susceptible and not susceptible to development of diarrhoea. In addition, most studies on the molecular links to low- and high-diarrhoea phenotypes have been studied under controlled conditions, using tissues other than the duodenum (the site of infection). For example, in our previous study of Merino sheep that were genetically prone or not prone to develop diarrhoea, and genetically resistant and susceptible to helminth infection, in a winter rainfall region, we assessed haematology (haemoglobin, red blood cell count, packed cell volume, white blood cell count) and found no significant differences among the groups39. However, haematology profiles are still useful as indicators of Haemonchus contortus infestations in summer rainfall regions40.
In the current study, we therefore investigated the molecular mechanisms and pathways in the duodenum through differential gene expression analysis in sheep that were susceptible or resistant to the development of diarrhoea. We tested whether tendency for diarrhoea is caused by up-regulation of genes related to the innate and adaptive immune responses. Specifically, we tested the hypothesis that diarrhoea is caused by up-regulation of genes related to the inflammatory/hypersensitive immune response.
Materials and methods
Animals, location, experimental design, and ethics statement
All of the experimental work was approved by the Animal Ethics Committee of the Department of Primary Industries and Regional Development, Western Australia (AEC No.17-1-02). Lambs were selected on the basis of Australian Sheep Breeding Values (ASBVs) and sourced from the flocks that had been maintained at the Katanning Research Facility (Western Australia) since 2015 as part of a large, long-term study addressing the helminth-diarrhoea-flystrike complex. The Katanning facility is located at an elevation 300 m, longitude 117.5 5°E and latitude of 33.7°S, with an average yearly rainfall of 480 mm, predominantly in winter, typical of a Mediterranean climatic zone.
The experiment started with a total of 986 lambs. Faeces were sampled from them all at weaning (100 days of age) when they were also given a broad-spectrum anthelmintic (monepantel; 1 mL/10 kg body weight) orally in November 2016. For all lambs, diarrhoea scores at weaning and completed pedigrees had been submitted to Sheep Genetics (www.sheepgenetics.org.au) for the estimation of Australian Sheep Breeding Values (ASBVs). Individuals with extreme high and low breeding values for diarrhoea score were then identified, and 100 males and 100 females were selected.
From November 2016 until September 2017, the lambs were kept in single-sex flocks in two fields, but otherwise managed similarly, where they grazed an annual grass clover pasture with capeweed (Arctotheca calendula). The stocking rate was kept at 10 lambs per hectare. The pasture had not been grazed for 4 months before the experiment began in March 2017, allowing the lambs to acquire a natural helminth infection under natural conditions. The pastures were kept green by unusual rain in February and subsequent rainfalls (in March, April and May), so the sheep had access to green fodder throughout the experiment, from February until the end in September, in contrast to normal season when green fodder does not become available until May. The dominant parasite species at the start of experiment were Teladorsagia spp., then Trichostrongylus spp., followed by Chabertia ovina and Oesophagostomum venulosum, and a small fraction of Haemonchus contortus. This species composition changed by the end of experiment as Trichostrongylus spp. became dominant, followed by C. ovina, O. venulosum, Teladorsagia spp. and a small fraction of H. contortus39.
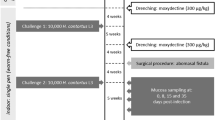
In March, May June, August, and September (Fig. 1), all 200 lambs were scored for diarrhoea, using a scale of 1 to 5 (low to high) based on the amount of faecal material accumulated on the hind quarters, between the anus and the hock joint. At the same time, FEC was measured. Note that FEC; ≥ 500 helminth eggs per gram faeces is considered as high under these conditions. In September 2017, 18 diarrhoea-resistant (low-diarrhoea; LD) and 20 diarrhoea-susceptible (high-diarrhoea; HD) lambs were slaughtered for the collection of duodenal tissues. From these 38 lambs, four replicates from each of the two groups were selected for the present study. Each HD or LD consisted of two males and two females. The tissues were kept in RNAlater solution (Sigma-Aldrich, St. Louis, United States) and stored as per manufacturer’s instructions at − 80 °C before RNA extraction.
Schema of the experimental protocol, showing timing of measurements (FEC and diarrhoea score sheep) from March (autumn) through to September (spring). For the 4-month pre-experimental period leading up the first measurement, the pasture was not used for grazing to avoid contamination with helminth eggs. In this environment, helminth infection is usually associated with the onset of winter rains in April–May.
RNA extraction, library preparation, quality control and sequencing
From each of the eight selected lambs, approximately 30 mg tissue was sampled from the duodenum. Total RNA was extracted using the RNeasy mini plus kit (Qiagen, Hilden, Germany) according to manufacturer’s instructions with minor modifications. Briefly, after tissue homogenization in lysis buffer, tubes were centrifuged at 14,000×g for 3 min. The supernatant was loaded onto the gDNA removal column and centrifuged at 10,000×g for one min, following which an equal volume of 70% (v/v) ethanol was added to the flow-through fraction and mixed by pipetting. The mixture was loaded onto the RNA binding column and centrifuged at 10,000×g for 15 s. The column was washed once with RW1 wash buffer, prior to adding 80 µL of DNase-I (0.34 Kunitz/µL) (Qiagen, Hilden, Germany) for 15 min to ensure complete gDNA removal. Subsequently, the column was washed again with RW1 buffer and twice with RPE buffer before eluting the RNA sample with RNAse free water.
The RNA content was quantified with a Qubit fluorometer using an RNA-BR kit and the purity was checked using NanoDrop. Quality was checked on a 2% agarose gel by assessing the 28S/18S ribosomal RNA ratio. Only samples with an OD 260/280 ratio of greater than 2 and a 260/230 ratio of 1.8 or greater were sent to BGI Genomics in Hong Kong for library preparation and sequencing. The sequencing libraries prepared from RNA samples with RIN (RNA integrity number) values ≥ 7 were sequenced using the 100 bp paired-end chemistry on DNBseq™ platform, with a depth of ≥ 20 million reads/sample.
Data processing
The quality of the reads was checked by FastQC and they all scored above 30. The reads were then aligned with the reference genome (Oar_rambouillet_v1.0) using STAR (v2.7.3a)41 with default parameters. After alignment, the featureCounts function in the Subread (version 2.0.0)42 software was used to enumerate the number of reads mapping to each gene using the following parameters; -p –primary -t gene. Differential gene expression analysis was aided by the DESeq2 R package, using the low-diarrhoea samples as the reference group43. A differentially expressed gene (DEG) was defined as a gene with a false discovery rate (FDR) < 0.05 and a log2 fold change > 1. To estimate and visualize the variations between samples, a principal component analysis (PCA) was performed on the DESeq2-normalised count data after regularized log transformation.
Functional enrichment analysis
Functional enrichment analysis using the up-regulated and down-regulated DEGs was performed using the web-based tool, DAVID (The Database for Annotation, Visualization and Integrated Discovery)44. Additionally, the clusterProfiler (v3.18.1) R package was used for Gene Set Enrichment Analysis (GSEA) with org.Bt.eg.db (Bos taurus) being the source of annotation as the genome wide annotation list for sheep was not available. Significant Gene Ontology (GO) terms for biological processes and KEGG pathways with an FDR < 0.05 are reported.
To highlight functional interactions among the DEGs, the web-based Search Tool for the Retrieval of Interacting Genes/Proteins (STRING)45 database (v11), was used with the following options selected: ‘disconnected nodes hidden’; ‘interactions discarded with a confidence score < 0.4’; ‘no more than 5 interactors to show in 1st and 2nd shell’. Gene sub-networks that each contain at least five genes and a p-value < 0.05 were generated in Cytoscape (v 3.7.2) using the ClusterONE plugin that clusters genes by functional relevance. Enrichr, another web-based tool, was used to study the biological functions and pathways associated with genes in the sub-network analysis46.
Ethics declarations
The authors confirm that the study was carried out in compliance with the ARRIVE guidelines. All animal experimentation was approved by the Animal Ethics Committee of the WA Department of Primary Industry and Rural Development, under the guidelines of the National Health and Medical Research Council’s Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (approval no: AEC 17-1-02).
Results
Diarrhoea scores and FEC
The groups differed significantly for diarrhoea score (p-value = 0.02) but not for FEC (p-value = 0.85). The difference in diarrhoea score, along with the ASBV data used to select the animals studied, supports the use of gene expression analysis to investigate the genes associated with the tendency to develop diarrhoea.
RNA-Seq data description and PCA outcome
In this study, a total of 187,757,422 paired-end reads were generated, ranging from 21,654,075 to 25,460,771 paired-end reads per sample, with a minimum of 21,048,774 reads were successfully mapped against reference genome. The average unique mapping rate was 77% for the HD group and 81% for the LD group (Fig. 2). Next, our PCA plot (Fig. 3a) showed a tight clustering of the LD samples, whereas the HD samples were irregularly distributed, with PC1 representing 56.2% of the total variance and PC2 representing 17.8% of the total variance, indicating that there are inherent gene expression differences between the LD and HD duodenal samples.
Differential gene expression and functional enrichment analyses
In the HD sheep samples, we identified 666 DEGs, of which 114 were down-regulated and 552 were up-regulated (Fig. 3b). For the LD samples, the control group in this study, the list of identified DEGs is presented in Supplementary File S1. A heatmap illustrating the top 100 most significant DEGs is presented in Supplementary Fig. S1. The 10 most significant up-regulated and down-regulated DEGs are shown in Fig. 4. Among the up-regulated DEGs were IGF-I, SEMA3C, EML1, DUSP27, PRDM5, SLC8A1, RYR2, SORBS1, PRUNE2 and CHRM2. Among the down-regulated DEGs were IDO2, TTI2, and several genes with functions that are yet to be determined.
Functional enrichment analyses, for identification of significantly enriched GO biological process terms and KEGG pathways, were performed using DAVID and the GSEA approach available in clusterProfiler. Importantly, the enrichment outcomes from both methods were similar. For the GO terms significantly enriched in the HD samples, both methods reported the up-regulation of ‘mesenchyme development’ and ‘organ development’, and the down-regulation of ‘immune response’-related biological processes (Fig. 5). The enrichment analysis based on DAVID revealed 13 significant KEGG pathways and the analysis GSEA revealed 63 significant KEGG pathways. The up-regulated KEGG pathways found in both analyses included ‘focal adhesion’, ‘PI3K-Akt signaling pathway’ ‘hypertrophic cardiomyopathy’, ‘dilated cardiomyopathy’, ‘ECM-receptor interaction’, ‘vascular smooth muscle contraction’ and ‘oxytocin signaling pathway’ (Fig. 6). The common down-regulated KEGG pathways comprised ‘allograft rejection’ and ‘antigen processing and presentation’. It is important to note that on GSEA analysis showed significant suppression of the pathway for ‘intestinal immune network for IgA production’ in the HD samples.
Functional enrichment analysis for GO biological process terms in the HD samples. (a) Analysis with DAVID using the up-regulated DEGs and down-regulated DEGs as the input data; the values on the x-axis represent the number of genes associated with each significant GO biological process term. (b) Analysis with GSEA utilizing the entire DESeq2-normalised dataset; the top 20 significantly enriched GO biological process terms are shown.
Functional enrichment analysis for KEGG pathways in the HD samples. (a) Analysis with DAVID using the up-regulated DEGs and down-regulated DEGs as the input data; the values on the x-axis represent the number of genes associated with each significant KEGG pathway. (b) Analysis with GSEA utilizing the entire DESeq2-normalised dataset; the top 20 significantly enriched KEGG pathways are shown in this figure.
PPI network and sub-network analysis
The DEGs were further annotated using the online STRING database to determine the relationships among them and how they interact to form protein–protein interaction (PPI) networks. In the HD samples, 74 of 114 down-regulated genes and 481 of 552 up-regulated genes were annotated as significant (PPI enrichment p-value < 0.001). The up-regulated genes formed only one significant subnetwork (SN) and were mostly associated with GO biological processes for ‘vesicle organization and vesicle-mediated transport’ and ‘calcium ion-dependent exocytosis’, and KEGG pathways for ‘synaptic vesicle cycle’, ‘SNARE interactions in vesicular transport’ and ‘insulin secretion’. The subnetwork plot of the up-regulated genes is presented in Supplementary Fig. S2.
The down-regulated genes were clustered into five significant SNs, as shown in Fig. 7. The genes in SN1 were significantly associated with biological processes linked to ‘antigen processing and presentation of exogenous peptide antigen via MHC class II’, ‘regulation of complement activation’ and ‘regulation of humoral immune response’. The KEGG pathways associated with SN1 included ‘antigen processing and presentation’, ‘intestinal immune network for IgA production’ and ‘complement and coagulation cascades’. For SN2, the genes were significantly linked to the KEGG pathway ‘tryptophan metabolism’. The full list of biological processes and KEGG pathways associated with each SN cluster are presented in Supplementary File S2.
Discussion
The molecular processes and pathways that are activated by worm infection, and apparently influenced by genetic selection, differ clearly between sheep that are susceptible (HD) and resistant (LD) to the development of diarrhoea. The HD sheep tend to keep intact the physical barriers and promote tissue repair in an attempt to elicit diarrhoea to expel the helminths, whereas the LD sheep tackle the diarrhoea through innate and adaptive (Th2-mediated) immune responses (activated in low-diarrhoea and suppressed in high-diarrhoea). Following a natural helminth infection, a number of DEGs were up-regulated in HD duodenum that are related to repair mechanisms, whereas the down-regulated DEGs were mostly linked with a required immune response, not a hypersensitive immune response. These down-regulated DEGs were up-regulated in LD duodenum, explaining why fewer but more specific genes are up-regulated in specific biological processes and pathways that control diarrhoea. While some aspects of these issues could be better explored in a fully controlled laboratory experiment, the field study that we used offered an opportunity to select extreme genotypes from a large number of animals that had taken decades to develop, with outcomes that are relevant to real-world sheep management. Despite the limitations, we were able to reveal clear and strong relationships related to diarrhoea that are consistent with the literature and with the theoretical basis of immune responses to helminth infection.
In HD sheep, the biological processes and pathways associated with up-regulated genes indicate activation of repair mechanisms rather than activation of the immune system, specifically ‘tissue morphogenesis’, ‘focal adhesion’, ‘ECM-receptor interaction’, ‘cardiovascular system development’, ‘sensory organ development’, ‘vasculature development’, ‘vascular smooth muscle contraction’ and ‘PI3K-Akt signaling pathway’. Tissue morphogenesis is an important aspect not only during embryonic morphogenesis but during tissue repair. The response to tissue injury is a dynamic process, involving biochemical signals and mechanical forces leading to the formation of a fibrotic scar. The extracellular matrix (Fig. 8) links to the intracellular cytoskeleton through ‘focal adhesions’, a collection of multiple-protein-complexes playing a vital role in the proliferation of initial mechanical signals into a wide-ranging network of bio-chemical signals.
(a) The ECM-receptor interaction47, an important KEGG pathway, indicated by up-regulated genes in the high diarrhoea group. (b) A category Netplot (CNET) showing the relationships among genes in ECM-receptor interaction pathway.
These biochemical signals result in extensive downstream effects including angiogenesis (development of blood vessels, as indicated by some of biological processes), collagen biosynthesis and fibroblast propagation48. The epithelial transition to mesenchymal cells occurs during tissue/organ fibrosis as a result of injury and ‘PI3K-Akt signaling pathway’ mediates the process of this transition49. Some of the up-regulated genes included insulin-like growth factor-I (IGF-I), semaphorin 3C (SEMA3C), tenascin-C (TNC), transforming growth factor-β (TGFβ2), and genes for the families of fibroblast growth factors, integrins and collagens.
IGF-I and SEMA3C have a developmental role in tissue repair, regulation of epithelial junction and epithelium morphogenesis50,51. TNC is an extracellular-matrix (ECM) glycoprotein that is highly expressed during embryogenesis and re-appears in response to tissue injury, during wound healing, and with Crohn’s disease and ulcerative colitis52,53,54,55,56. TGF-β promotes the expression of TNC so both are linked to induction of tissue repair mechanisms with diarrhoea52. TGF-β is also important in the healing of intestinal epithelium, in preventing susceptibility to injury57 and in the promotion of tissue fibrosis. In the high-diarrhoea sheep, we observed several upregulated genes in the FGF family, including FGF5, FGF10, FGF11 and FGFR1, all of which play roles in tissue repair, homeostasis and organ development, including the gastrointestinal tract58. FGFR1 is a receptor for fibroblast growth factor 2 (FGF2), a factor that interacts with IL-17 in the repair of damaged intestinal epithelium59.
The organization of the ECM also involves genes that transcribe for collagen, including COL5A1, COL6A3, COL8A2 and COL27A1, also known for their role in tissue healing60 and are activated as a result of tissue damage. Similarly, the integrin family helps maintain the integrity of the ECM by promoting ECM adhesion, one of the most important biological process identified in our GO analysis61. Overall, therefore, we have shown that the fibroblast, integrin and collagen family genes are activated in the high-diarrhoea sheep, strongly indicating ongoing fibrosis. As they establish in the gut mucosa, the helminths promote fibrosis and are thus able evade the immune response mounted by the host. Fibrosis can also lead to alterations in the way the immune system responds, as seen in cystic fibrosis, where there is a decrease in the expression of MHC class II molecules62, a similar scenario observed in high diarrhoea animals (decreased expression of Ovar-DRB1 and DQA).
On the other hand, most of the genes that were down-regulated with high diarrhoea, compared low diarrhoea, are directly or indirectly involved in the immune response, the regulation of immune response and immune tolerance: ‘antigen processing and presentation’, ‘antigen processing and presentation of peptide or polysaccharide antigen via MHC class II’, ‘intestinal immune network for IgA production’, ‘primary immunodeficiency’ and ‘immune response’. The processing, conversion and presentation of helminth antigen through MHC-II molecules is critical in mounting a protective immune response so down-regulating this process can make the sheep susceptible to infection, leading to helminth establishment and possibly diarrhoea63,64. Immunoglobulin-A (IgA) has been associated with the length and fecundity of helminths65 so down-regulation of ‘intestinal immune network for IgA production’ suggests a failure to arrest helminth growth and reproduction, allowing establishment of a successful population and ultimately gut damage leading to diarrhoea.
Other DEGs that were down-regulated in the high-diarrhoea group include Ovar-DRB1, DQA, TREM2, TFF3, ITLN2, CD74, CCL5, and CCR3. Ovar-DRB1, a Major Histocompatibility Complex class II (MHC-II) gene, is widely associated with resistance to nematodes in sheep31,66,67 and underpins presentation of antigenic molecules to T-cells, an integral component of the adaptive immune response68. MHC-II molecules, present on antigen presenting cells, tend to present parasitic antigens to T-helper type 2 (TH2) cells to initiate the antibody-producing adaptive immune response that is required to control helminths14,69. In addition to its role in resistance to roundworms, Ovar-DRB1 has been associated with resistance to tapeworms in Chinese Merino sheep70. Carriers of Ovar-DRB1 show reductions in duodenal contractile force and FEC30,31,66 so down-regulation of Ovar-DRB1 may increase duodenal contractions71 and thus explain our high-diarrhoea phenotype. DQA is similar to Ovar-DRB1 in that it is a MHC-II molecule that plays an important role in adaptive immunity by presenting processed antigen to T-cells72,73.
The triggering receptor expressed on myeloid cells (TREM2), also found on intestinal macrophages and lamina-propria DCs, has inflammatory activities, so down-regulation of TREM2 would lead to immune suppression by decreasing pro-inflammatory cytokine production and T-cell activation74,75. Trefoil factor 3 (TFF3) and sheep intelectin-2 (ITLN2) normally help the host to combat helminths by altering luminal mucus76,77, but here we see a down-regulation of these genes with the high-diarrhoea phenotype, indicating a reduction of two important Th2-cytokines, IL-4 and IL-1378. Cluster of differentiation (CD74) is a class-II cell surface receptor for the macrophage migration inhibition factor (MIF) that is expressed by antigen-presenting cells (APCs) and epithelial cells79. Binding of MIF to CD74 induces intracellular cell survival cascades80 and regeneration of intestinal epithelial cells81 leading to healing of the mucosa.
Chemokine ligand 5 (CCL5; also known as RANTES), an important chemokine found in eosinophil granules, is required for normal T-cell function and for recruiting other lymphocytes, such as monocytes and T-cells, to the site of infection/inflammation82. CCL5 interacts with CCR3 (a chemokine receptor) leading to a Th2-polarised immune response83. Thus, the down-regulation of CCL5 in high-diarrhoea sheep complements an overall suppression of the immune response to helminth infection.
In addition to suppression of the immune system, the sub-network analysis also identified a set of down-regulated genes related to immune tolerance, a way of limiting the immune response after it has been initiated in response to a foreign antigen/pathogen. It is important for the response to subside, or at least be kept under control, so the host does not develop autoimmunity/hypersensitivity84. One such check is mediated by Indoleamine2,3-Dioxygenase (IDO), an important rate-limiting enzyme that catabolises tryptophan85,86 to kynurenine. This process is crucial in modulating an adaptive immune response by inhibiting effector T-cell proliferation and by diverting the immune response from effector T-cells to regulatory T-cells87. In inflammatory bowel syndrome (IBD) and murine models of colitis, the severity of disease is restrained by induction of IDO88. In general, therefore, IDO suppresses immune responses promoting immune tolerance and preventing autoimmunity89,90,91. The suppression of immune tolerance in the high-diarrhoea sheep could lead to a potential inflammatory/hypersensitive immune response but, overall, the down-regulation of the immune response outweighs the possibility that ‘suppressed immune tolerance’ could lead to a pathological hypersensitive immune response.
Conclusion
We have been able to identify pathways and biological process in the immune system that respond to helminth infection and might explain why some sheep are susceptible to the development of diarrhoea while others are not. Importantly, it has become clear that diarrhoea in the diarrhoea-susceptible sheep is not caused by an inflammatory/hypersensitive immune response because most of the up-regulated genes are related to repair mechanisms in the duodenum and most of the down-regulated genes are related to different components of immune response. The literature shows that Ovar-DRB1 is associated with helminth resistance and, in our study, down-regulation Ovar-DRB1 was associated with diarrhoea rather than the parasitism itself. The immune tolerance due to tryptophan catabolism by IDO shows how the immune system is regulated and kept in check in low-diarrhoea sheep. Several up-regulated genes in the high-diarrhoea group are related to ECM organization leading to fibrosis, perhaps explaining why these animals have a high-diarrhoea, because the focus of these genes is repair rather than addressing the primary cause. The organization of ECM could also be linked to the maintenance of physical barriers as another way to resist the helminths and maintain tissue integrity. With diarrhoea or helminth infection, injury could change gut morphology for a certain period, perhaps affecting subsequent immune responses. The extent of fibrosis, and its resolution, are yet to be determined but our observations lead us to suggest a ‘remodeled immune’ response and fibrosis that probably provides a ‘helminth hideout’. Some of the biological processes and pathways that we have revealed could explain how diarrhoea develops, but we are still not really sure how some sheep resist diarrhoea while others readily develop it.
Data availability
The raw sequencing reads generated in this study have been submitted to NCBI Gene Expression Omnibus (GEO) database under the BioProject accession number PRJNA736720.
References
Sackett, D., Holmes, P., Abbott, K., Jephcott, S. & Barber, M. Assessing the economic cost of endemic disease on the profitability of Australian beef cattle and sheep producers. MLA Rep. AHW 87, (2006).
Taylor, M. A. Emerging parasitic diseases of sheep. Vet. Parasitol. 189, 2–7 (2012).
Jacobson, C., Larsen, J. W., Besier, R. B., Lloyd, J. B. & Kahn, L. P. Diarrhoea associated with gastrointestinal parasites in grazing sheep. Vet. Parasitol. 282, 109139 (2020).
Williams, A. R. Immune-mediated pathology of nematode infection in sheep–is immunity beneficial to the animal?. Parasitology 138, 547–556 (2011).
Claerebout, E. & Geldhof, P. Helminth vaccines in ruminants. Vet. Clin. North Am. Food Anim. Pract. 36, 159–171 (2020).
Karlsson, L. J. & Greeff, J. C. Genetic aspects of sheep parasitic diseases. Vet. Parasitol. 189, 104–112 (2012).
Cooper, K. M., Whelan, M., Danaher, M. & Kennedy, D. G. Stability during cooking of anthelmintic veterinary drug residues in beef. Food Addit. Contam. Part A 28, 155–165 (2011).
Karlsson, L. J. E. & Greeff, J. C. Selection response in fecal worm egg counts in the Rylington Merino parasite resistant flock. Aust. J. Exp. Agric. 46, 809–811 (2006).
Karlsson, L., Greeff, J. C. & Harris, J. F. Genetic trends in a selection line for low faecal worm egg count. Aust. Assoc. Anim. Breed. Genet. 11, 122–125 (1995).
Bisset, S. A. & Morris, C. A. Feasibility and implications of breeding sheep for resilience to nematode challenge. Int. J. Parasitol. 26, 857–868 (1996).
Larsen, J. W. A., Anderson, N. & Vizard, A. L. The pathogenesis and control of diarrhoea and breech soiling in adult Merino sheep. Int. J. Parasitol. 29, 893–902 (1999).
Karlsson, L. J. E., Greeff, J. C., Eady, S. J. & Pollott, G. E. Nematode resistance & hypersensitivity diarrhoea. (2005).
Greeff, J. C., Karlsson, L. J. E. & Schlink, A. C. Identifying indicator traits for breech strike in Merino sheep in a Mediterranean environment. Anim. Prod. Sci. 54, 125 (2014).
McRae, K. M., Stear, M. J., Good, B. & Keane, O. M. The host immune response to gastrointestinal nematode infection in sheep. Parasite Immunol 37, 605–613 (2015).
Anthony, R. M., Rutitzky, L. I., Urban, J. F., Stadecker, M. J. & Gause, W. C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 7, 975–987 (2007).
Paul, W. E. & Zhu, J. How are TH2-type immune responses initiated and amplified?. Nat. Rev. Immunol. 10, 225–235 (2010).
Douch, P. G. C., Harrison, G. B. L., Buchanan, L. L. & Brunsdon, R. V. Relationship of histamine in tissues and antiparasitic substances in gastrointestinal mucus to the development of resistance to trichostrongyle infections in young sheep. Vet. Parasitol. 16, 273–288 (1984).
Harrison, G. B. L. et al. Immune rejection of Trichostrongylus colubriformis in sheep; a possible role for intestinal mucus antibody against an L3-specific surface antigen. Parasite Immunol. 25, 45–53 (2003).
Ingham, A., Reverter, A., Windon, R., Hunt, P. & Menzies, M. Gastrointestinal nematode challenge induces some conserved gene expression changes in the gut mucosa of genetically resistant sheep. Int. J. Parasitol. 38, 431–442 (2008).
Henderson, N. G. & Stear, M. J. Eosinophil and IgA responses in sheep infected with Teladorsagia circumcincta. Vet. Immunol. Immunopathol. 112, 62–66 (2006).
Rosenberg, H. F., Dyer, K. D. & Foster, P. S. Eosinophils: changing perspectives in health and disease. Nat. Rev. Immunol. 13, 9–22 (2013).
Williams, A. R., Palmer, D. G., Williams, I. H., Vercoe, P. E. & Karlsson, L. J. Faecal dry matter, inflammatory cells and antibodies in parasite-resistant sheep challenged with either Trichostrongylus colubriformis or Teladorsagia circumcincta. Vet. Parasitol. 170, 230–237 (2010).
Williams, A. R. & Palmer, D. G. Interactions between gastrointestinal nematode parasites and diarrhoea in sheep: Pathogenesis and control. Vet. J. 192, 279–285 (2012).
Larsen, J., Vizard, A. & Anderson, N. Production losses in Merino ewes and financial penalties caused by trichostrongylid infections during winter and spring. Aust. Vet. J. 72, 58–63 (1995).
Stanger, K. J., McGregor, H. & Larsen, J. W. A. Outbreaks of diarrhoea (‘winter scours’) in weaned Merino sheep in south-eastern Australia. Aust. Vet. J. 96, 176–183 (2018).
Finkelman, F. D. Effects of interleukin 12 on immune responses and host protection in mice infected with intestinal nematode parasites. J. Exp. Med. 179, 1563–1572 (1994).
Staudt, L. M. Gene expression physiology and pathophysiology of the immune system. Trends Immunol. 6, 35–40 (2001).
Chitneedi, P. K., Suarez-Vega, A., Martinez-Valladares, M., Arranz, J. J. & Gutierrez-Gil, B. Exploring the mechanisms of resistance to Teladorsagia circumcincta infection in sheep through transcriptome analysis of abomasal mucosa and abomasal lymph nodes. Vet. Res. 49, 39 (2018).
Gossner, A., Wilkie, H., Joshi, A. & Hopkins, J. Exploring the abomasal lymph node transcriptome for genes associated with resistance to the sheep nematode Teladorsagia circumcincta. Vet. Res. 44, 68 (2013).
Schwaiger, F.-W. et al. An ovine Major histocompatibility complex DRB1 allele is associated with low faecal egg counts following natural, predominantly Ostertagia circumcincta infection. Int. J. Parasitol. 25, 815–822 (1995).
Hassan, M. et al. The dynamic influence of the DRB1*1101 allele on the resistance of sheep to experimental Teladorsagia circumcincta infection. Vet. Res. 42, 46 (2011).
visual-sheep-scores-producer-version-2019.pdf.
Davies, G. et al. Quantitative trait loci associated with parasitic infection in Scottish blackface sheep. Heredity (Edinb) 96, 252–258 (2006).
Knight, P. A. et al. Novel gene expression responses in the ovine abomasal mucosa to infection with the gastric nematode Teladorsagia circumcincta. Vet. Res. 42, 78 (2011).
Anderson, N. Trichostrongylid infections of sheep in a winter rainfall region. I. Epizootiological studies in the Western District of Victoria, 1966–67. Aust. J. Agric. Res. 23, 1113–1129 (1972).
Larsen, J. W., Anderson, N., Vizard, A. L., Anderson, G. A. & Hoste, H. Diarrhoea in merino ewes during winter: Association with trichostrongylid larvae. Aust. Vet. J. 71, 365–372 (1994).
Larsen, J. W., Vizard, A. L. & Anderson, N. Role of larval nematode infection in lamb diarrhoea. Vet. Rec. 137, 572 (1995).
Larsen, J. W. A. & Anderson, N. The relationship between the rate of intake of trichostrongylid larvae and the occurrence of diarrhoea and breech soiling in adult Merino sheep. Aust. Vet. J. 78, 112–116 (2000).
Greeff, J. C. et al. Temporal changes in circulatory blood cell parameters of sheep genetically different for faecal worm egg count and diarrhoea from late summer to spring in a Mediterranean environment. Anim. Prod. Sci. 60, 1630–1642 (2020).
Hunt, P. W., Knox, M. R., Le Jambre, L. F., McNally, J. & Anderson, L. J. Genetic and phenotypic differences between isolates of Haemonchus contortus in Australia. Int. J. Parasitol. 38, 885–900 (2008).
Dobin, A. et al. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: An efficient general-purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Huang, D. W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2009).
Szklarczyk, D. et al. STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 47, D607–D613 (2019).
Chen, E. Y. et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 14, 128 (2013).
Kanehisa, M., Furumichi, M., Sato, Y., Ishiguro-Watanabe, M. & Tanabe, M. KEGG: Integrating viruses and cellular organisms. Nucleic Acids Res. 49, D545–D551 (2020).
Rustad, K. C., Wong, V. W. & Gurtner, G. C. The role of focal adhesion complexes in fibroblast mechanotransduction during scar formation. Differentiation 86, 87–91 (2013).
Xu, W., Yang, Z. & Lu, N. A new role for the PI3K/Akt signaling pathway in the epithelial-mesenchymal transition. Cell Adhes. Migr. 9, 317–324 (2015).
Schmidt, M. B., Chen, E. H. & Lynch, S. E. A review of the effects of insulin-like growth factor and platelet derived growth factor on in vivo cartilage healing and repair. Osteoarthritis Cartilage 14, 403–412 (2006).
Treps, L., Le Guelte, A. & Gavard, J. Emerging roles of Semaphorins in the regulation of epithelial and endothelial junctions. Tissue Barriers 1, e23272 (2013).
Islam, M. S. et al. PDGF and TGF-β promote tenascin-C expression in subepithelial myofibroblasts and contribute to intestinal mucosal protection in mice. Br. J. Pharmacol. 171, 375–388 (2014).
Jones, F. S. & Jones, P. L. The tenascin family of ECM glycoproteins: Structure, function, and regulation during embryonic development and tissue remodeling. Dev. Dyn. 218, 235–259 (2000).
Riedl, S. et al. Serum tenascin-C is an indicator of inflammatory bowel disease activity. Int. J. Colorectal Dis. 16, 285–291 (2001).
Salas, A. et al. Subepithelial myofibroblasts and tenascin expression in microscopic colitis. Histopathology 43, 48–54 (2003).
Brain, O., Rajaguru, C., Warren, B., Booth, J. & Travis, S. Collagenous gastritis: Reports and systematic review. Eur. J. Gastroenterol. Hepatol. 21, 1419–1424 (2009).
Beck, P. L. et al. Transforming growth factor-β mediates intestinal healing and susceptibility to injury in vitro and in vivo through epithelial cells. Am. J. Pathol. 162, 597–608 (2003).
Danopoulos, S., Schlieve, C. R., Grikscheit, T. C. & Alam, D. A. Fibroblast growth factors in the gastrointestinal tract: Twists and turns. Dev. Dyn. 246, 344–352 (2017).
Song, X. et al. Growth factor FGF2 cooperates with interleukin-17 to repair intestinal epithelial damage. Immunity 43, 488–501 (2015).
Mutsaers, S. E., Bishop, J. E., McGrouther, G. & Laurent, G. J. Mechanisms of tissue repair: From wound healing to fibrosis. Int. J. Biochem. Cell Biol. 29, 5–17 (1997).
Barczyk, M., Carracedo, S. & Gullberg, D. Integrins. Cell Tissue Res. 339, 269–280 (2010).
Hofer, T. P. et al. Decreased expression of HLA-DQ and HLA-DR on cells of the monocytic lineage in cystic fibrosis. J. Mol. Med. 92, 1293–1304 (2014).
Blum, J. S., Wearsch, P. A. & Cresswell, P. Pathways of antigen processing. Annu. Rev. Immunol. 31, 443–473 (2013).
Lee, C. Y. et al. The influence of MHC and immunoglobulins A and E on host resistance to gastrointestinal nematodes in sheep. J. Parasitol. Res. 2011, 1–11 (2011).
Stear, M. J. et al. Regulation of egg production, worm burden, worm length and worm fecundity by host responses in sheep infected with Ostertagia circumcincta. Parasite Immunol. 17, 643–652 (1995).
Sayers, G. et al. Major histocompatibility complex DRB1 gene: Its role in nematode resistance in Suffolk and Texel sheep breeds. Parasitology 131, 403–409 (2005).
Hassan, M., Hanrahan, J. P., Good, B., Mulcahy, G. & Sweeney, T. A differential interplay between the expression of Th1/Th2/Treg related cytokine genes in Teladorsagia circumcincta infected DRB1*1101 carrier lambs. Vet. Res. 42, 45 (2011).
Choi, N. M., Majumder, P. & Boss, J. M. Regulation of major histocompatibility complex class II genes. Curr. Opin. Immunol. 23, 81–87 (2011).
Neefjes, J., Jongsma, M. L. M., Paul, P. & Bakke, O. Towards a systems understanding of MHC class I and MHC class II antigen presentation. Nat. Rev. Immunol. 11, 823–836 (2011).
Shen, H., Han, G., Jia, B., Jiang, S. & Du, Y. MHC-DRB1/DQB1 gene polymorphism and its association with resistance/susceptibility to cystic echinococcosis in Chinese merino sheep. J. Parasitol. Res. 2014, 1–7 (2014).
Spiller, R. Role of motility in chronic diarrhoea. Neurogastroenterol. Motil. 18, 1045–1055 (2006).
Amills, M., Ramírez, O., Tomàs, A., Obexer-Ruff, G. & Vidal, O. Positive selection on mammalian MHC-DQ genes revisited from a multispecies perspective. Genes Immun. 9, 651–658 (2008).
Vandre, R., Gowane, G., Sharma, A. & Tomar, S. Immune responsive role of MHC class II DQA1 gene in livestock. Liver Res. Int. 2, 7 (2014).
Genua, M., Rutella, S., Correale, C. & Danese, S. The triggering receptor expressed on myeloid cells (TREM) in inflammatory bowel disease pathogenesis. J. Transl. Med. 12, 1–12 (2014).
Correale, C. et al. Bacterial sensor triggering receptor expressed on myeloid cells-2 regulates the mucosal inflammatory response. Gastroenterology 144, 346-356.e3 (2013).
Tsuji, S. et al. Human intelectin is a novel soluble lectin that recognizes galactofuranose in carbohydrate chains of bacterial cell wall. J. Biol. Chem. 276, 23456–23463 (2001).
Datta, R. et al. Identification of novel genes in intestinal tissue that are regulated after infection with an intestinal nematode parasite. IAI 73, 4025–4033 (2005).
Blanchard, C. et al. IL-4 and IL-13 up-regulate intestinal trefoil factor expression: Requirement for STAT6 and de novo protein synthesis. J. Immunol. 172, 3775–3783 (2004).
Beswick, E. J. & Reyes, V. E. CD74 in antigen presentation, inflammation, and cancers of the gastrointestinal tract. World J. Gastroenterol. 15, 2855–2861 (2009).
Gil-Yarom, N. et al. CD74 is a novel transcription regulator. Proc. Natl. Acad. Sci. USA 114, 562–567 (2017).
Farr, L. et al. CD74 Signaling links inflammation to intestinal epithelial cell regeneration and promotes mucosal healing. Cell Mol. Gastroenterol. Hepatol. 10, 101–112 (2020).
Impaired T Cell Function in RANTES-Deficient Mice—ScienceDirect. https://www.sciencedirect.com/science/article/pii/S1521661601951782?via%3Dihub.
Zhang, Q. et al. CCL5-Mediated Th2 immune polarization promotes metastasis in luminal breast cancer. Cancer Res. 75, 4312–4321 (2015).
Viganò, S., Perreau, M., Pantaleo, G. & Harari, A. Positive and negative regulation of cellular immune responses in physiologic conditions and diseases. Clin. Dev. Immunol. 2012, 1–11 (2012).
Wu, H., Gong, J. & Liu, Y. Indoleamine 2, 3-dioxygenase regulation of immune response (Review). Mol. Med. Rep. 17, 4867–4873 (2018).
Mellor, A. L. & Munn, D. H. Tryptophan catabolism and regulation of adaptive immunity. J. Immunol. 170, 5809–5813 (2003).
Mellor, A. L., Keskin, D. B., Johnson, T., Chandler, P. & Munn, D. H. Cells expressing indoleamine 2,3-dioxygenase inhibit T cell responses. J. Immunol. 168, 3771–3776 (2002).
Ciorba, M. A. Indoleamine 2,3 dioxygenase (IDO) in intestinal disease. Curr. Opin. Gastroenterol. 29, 146–152 (2013).
Mbongue, J. C. et al. The role of indoleamine 2, 3-dioxygenase in immune suppression and autoimmunity. Vaccines (Basel) 3, 703–729 (2015).
Mellor, A. L. & Munn, D. H. Tryptophan catabolism and T-cell tolerance: immunosuppression by starvation?. Immunol. Today 20, 469–473 (1999).
Mellor, A. L., Lemos, H. & Huang, L. Indoleamine 2,3-dioxygenase and tolerance: Where are we now?. Front. Immunol. 8, 1360 (2017).
Acknowledgements
The technical staff (Geoff Cox, Tyler Loud and Nicola Stanwyck) at the Katanning Research facility are thanked for managing the flocks and collecting all the basic performance data, and also the postgraduate students at the University of Western Australia who assisted with sampling the animals during the experiment. We gratefully acknowledge the funding and resources provided by the University of Western Australia and the University of Agriculture Faisalabad Pakistan and additional computational resources and support from the Pawsey Supercomputing Centre with funding from the Australian Government and the Government of Western Australia.
Funding
S.U.H. received a joint scholarship from University of Agriculture Faisalabad (Pakistan) and University of Western Australia for PhD studies (reference number PS-2(11) FDP/17/8071). The Department of Primary Industries and Regional Development of Western Australia is also thanked for making funds available for this experiment.
Author information
Authors and Affiliations
Contributions
Conceptualization: S.U.H. and G.B.M. Computational analysis and data interpretation: S.U.H., P.K. and E.G.C. Funding acquisition: J.C.G. and G.B.M. Investigation; S.U.H., J.C.G., E.A.P., S.L., P.K. and C.Y.T. Writing (original draft); S.U.H. Writing (review and editing); G.B.M., J.C.G. and E.G.C. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hassan, S.U., Chua, E.G., Paz, E.A. et al. Investigating the development of diarrhoea through gene expression analysis in sheep genetically resistant to gastrointestinal helminth infection. Sci Rep 12, 2207 (2022). https://doi.org/10.1038/s41598-022-06001-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-06001-4
- Springer Nature Limited
This article is cited by
-
Contribution of the Immune Response in the Ileum to the Development of Diarrhoea caused by Helminth Infection: Studies with the Sheep Model
Functional & Integrative Genomics (2022)