Abstract
The dynamic synergy of genes and pathways in muscles in relation to age affects the muscle characteristics. Investigating the temporal changes in gene expression will help illustrate the molecular mechanisms underlying muscle development. Here we report the gene expression changes in skeletal muscles through successive age groups in Bandur, a meat type sheep of India. RNA sequencing data was generated from the longissimus thoracis muscles from four age groups, ranging from lamb to adult. Analysis of 20 highest expressed genes common across the groups revealed muscle protein, phosphorylation, acetylation, metal binding and transport as significant functions. Maximum differentiation was observed after 2.5–3 years on transition from lambs to adult. Transcriptional regulation by the TFAP2 transcription factors, IL-6 signaling and PI3K/AKT signaling pathways were enriched in younger animals. The gene-protein network demarcated key interactive genes involved in muscle development and proliferation that can be used as candidates for future research on improvement of muscle characteristics.
Similar content being viewed by others
Introduction
Livestock sector plays an important role in the rural economy of India. Sheep and goats contribute substantially to the livelihood of the small and marginal farmers. Small ruminants provide 21.89% of the total meat produced, of which 8.36% is from sheep1. Meat quality is an amalgamation of the effects of nutrition, environment and genetic capability. Factors like growth and development of skeletal muscle directly affect the quality and quantity of meat. The palatability of meat is influenced by tenderness and fat content, which are in turn affected by nutrition, muscle characteristics, post mortem events, genetics and the age of the animal2. Several studies have evaluated the impact of age on the tenderness and fatty acid profile of muscles in various species3,4. The dynamic synergy of genes and pathways in muscles in relation to age of the animal has also been explored in several species5,6,7. In sheep however, there is dearth of related information on lambs and mature animals.
Deep sequencing techniques have significantly broadened our knowledge of global gene expression patterns and regulatory mechanisms in several tissues. Transcriptomic studies have identified regulatory factors involved in muscle growth and meat quality in livestock species8,9. The genetic mechanisms underlying growth and development of skeletal muscles of sheep have been investigated using next generation sequencing techniques5. Differentially expressed genes for contrasting characteristics have been detected in small as well as large ruminants8,10. Such efforts have led to identification of biomarkers for meat quality11.
Consumer preferences for Bandur sheep in India led to the investigation that established tenderness and higher backfat content in muscles of Bandur sheep as compared to local sheep found in the same area12. Our previous studies have attempted to understand the regulatory molecular mechanisms underlying the muscle traits in Bandur sheep breed in comparison to the local sheep12,13. These studies have identified the genes and pathways that may be associated with the muscling traits. Previous studies on goat6 and pigs14 have provided some insight on the temporal pattern of expression of genes through developmental stages. However, lack of information on the effect of age on the expression of genes in skeletal muscles of sheep has prompted this investigation. The aim of the present study was to compare the gene expression pattern in skeletal muscles through successive age groups in Bandur sheep, a meat type breed of India. The temporal changes in gene expression will help illustrate the molecular mechanisms underlying muscle development in sheep.
Results
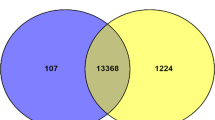
Quality control and filtering of raw data resulted in an average of 93,434,064 reads for each library. The processed reads were aligned to the Ovis aries reference genome Oar_v4.0 (SAMN00116405). An overall mapping of 94.6% was observed across samples of all age groups (Table S1) while 79–90% of the reads were uniquely mapped. Comparison of the transcript profile revealed 85.4% transcripts common to all groups. The unique transcripts in each group accounted for only 1–1.8% (Fig. 1). The total number of known genes discovered with a minimum threshold of RPKM > 0.01, was 11045, 11168, 10907 and 10514 in group 1, 2, 3 and 4, respectively (Table S2).
Abundantly expressed genes and their functions
Twenty genes common across all four groups, with expression > 2000 RPKM are shown in Fig. 2. Analysis of the 20 highest expressed genes common across the groups revealed muscle protein, phosphorylation, acetylation, metal binding and transport as significant (p ≤ 0.05) functions. The major biological processes associated with these genes included skeletal muscle contraction, regulation of muscle contraction, muscle tissue morphogenesis, transition between fast and slow fiber, etc. Significant cellular component were troponin and myosin complex, myofibril actin filament and muscle thin filament tropomyosin while actin filament binding, structural constituent of muscle, calcium ion binding were the major molecular functions common in all age groups (Fig. 3). Among these genes, the expression of ACTA1, COX3, TPM2, MYL2, CYTB2, MYH7, ND4 and TNNCI decreased with progression of age. On the other hand, expression of TNNI2, TPM1 and ENO3 genes increased with age. Enriched pathways associated with these genes were muscle contraction, glucose metabolism, respiratory electron transport and creatine metabolism (Table S3).
The expression profile of some genes previously associated with meat quality in livestock species11 like HSPB1 and HSPB8 conspicuously increased after 3 years of age. Expression of DNAJB5 showed a mild increase whereas that of HSPA6 decreased with age. Transcription of CAPN3 also exhibited a very slight increase with advancement of age in investigated animals. Other genes like FABP3, CAPN1 and CAST did not express prominent difference across the groups. Similarly, collagen genes are also known to influence meat quality15. Although the expression of some collagen genes, namely, COL15A1, COL23A1, COL4A2, COL6A2, COL1A1, COL4A1, COL1A2 and COL3A1 was observed to be quite low (≤ 7.3 RPKM), their expression was highest in young animals and decreased considerably with age.
Differentially expressed genes and enriched pathways
Differential expression of genes was analyzed across all combinations of the four age groups. Considerably larger number of genes was observed to be differentially expressed between Group1–3 and Group1–4 only, which were therefore used for further analysis (Fig. 4). There were 301 significantly differentially expressed genes in Group 1 versus 3 (padj < 0.05; FDR < 0.05), followed by 141 in Group 1 versus 4. The up- and down-regulated genes observed in Group 1 versus 3 were 222 and 79, respectively, whereas 98 up-regulated and 43 down-regulated genes were identified in Group 1 versus 4 (Table S4). Gene ontology analysis revealed negative regulation of cell proliferation, response to hypoxia, positive regulation of GTPase activity, negative regulation of cysteine-type endopeptidase activity involved in apoptotic process, calcium ion binding etc., as significant functions (padj < 0.05) associated with differentially expressed genes in Group 1 versus 3 (Fig. 5a). Significant pathways associated with the up-regulated genes were transcriptional regulation by the TFAP2 (AP-2) family of transcription factors, IL-6 signaling, PI3K/AKT signaling, ECM proteoglycans, muscle contraction etc. (Table S5), while down-regulated genes were related with transcriptional regulation by RUNX1 and RUNX3. Similarly, enriched functions in Group 1 versus 4 included regulation of transcription from RNA polymerase II promoter, myoblast fusion, regulation of epithelial to mesenchymal transition and protein phosphatase regulator activity (Fig. 5b). The top canonical pathways identified again were transcriptional regulation by the TFAP2 (AP-2) family of transcription factors, IL-6 signaling, PI3K/AKT signaling. Other important pathways included MAPK1 and MAPK3 activation, DAG and IP3 signaling and PKA activation (Table S6).
Validation of RNAseq data by RT-qPCR
Differentially expression of genes in Group 1 versus Group 3 and Group 1 versus Group 4 was validated by RT-qPCR. The magnitude of relative expression of the selected genes namely ACTA1, FABP3, COL1A1, COL1A2, HSPA6, HSPB1, IL17A, MYOD, RUNX1 and SOCS3 was observed to be similar to that derived from RNAseq data (Figs. 6 and 7).
Gene—protein interactions
To identify highly connected genes from the set of differentially expressed ones, a gene—protein network was constructed for each comparative Group 1 versus 3 and Group 1 versus 4 (Figs. 8 and 9). The central genes with 5 or more interactions identified for Group1 versus 3 included COL1A2, SOCS3, IL17A, COL1A1, IFIT1, NR4A1, RUNX1, CDKL2, CBLB and VIPR1. Whereas ESR1, CCNE1, MYOD1, HSPB1, COL1A1, NR4A2, TWIST1, ISG15, PER1 and TLE3 were the most interactive hub genes identified in the network for Group 1 versus 4. Among these genes COL1A2, IL17A, COL1A1, NR4A1, VIPR1, ESR1, CCNE1, TWIST1 and ISG15 were up-regulated, while the remaining were down-regulated. COL1A1 was common in both the networks. Some other important genes identified in the network were HSPA6, LPAR1, EIF5A and PPARGC1A.
Discussion
Muscle traits relevant to meat quality are modulated by age, nutrition, genetics and environmental factors which in turn affect the structure and composition of muscle fibres as well as intramuscular fat. Differential expression of genes in muscles between different breeds in relation to feed, carcass and muscle properties has been examined by several studies8,9,10,12. It is well established that muscle characteristics are affected by age16. However, investigating the simultaneous change in gene expression with age in skeletal muscles of sheep has not garnered much interest. The present study therefore, explored the pattern of gene expression with progression of age in skeletal muscles of Bandur sheep. The sarcomere proteins, mainly alpha-actinins, myosin heavy chains and Z disc form the basic units of muscle fibres and are coded by ACTA1, ACTN2, ACTN3, MYH4, MYH7 and MYOZ genes17,18. The troponin complex modulates the interaction between actin and myosin during muscle contraction19. TNNI2 and TNNC2 encode for their respective troponin isoforms and are known to play a key role in muscle composition and aging20. The genes mainly involved in muscle contraction and muscle fibre composition (ACTA1, ACTN2, ACTN3, ANKRD1, TCAP, NRAP, MYH4, MYH8, TNNI2, TNNC2, MYOZ1, LDB3, TPM1) were abundantly expressed irrespective of age group in our study. Earlier investigations have revealed no significant effect of sex on muscle fibre type, however, the fibre diameter was affected21. Hence, further research on gene expression in longissimus thoracis muscles, across both rams and ewes is warranted. Besides structural components of muscles the highly expressed genes also represented glycogen metabolism and storage (LDHA, PYGM, PGAM2) as well as energy metabolism (COX1, COX3, CKM, ND4). The calpains are another family of genes associated with muscle tenderization22. The calpain-calpastatin activity was reported to increase with age in beef cattle23. It is well established that the activity of calpains is enhanced during aging24. A slight increase in the expression of CAPN3 with age was also detected in Bandur sheep. The domination of expression of these genes in our study reinforces their fundamental role in muscle physiology.
Major pathways identified by differential analyses also had relevance to myogenesis. During muscle development, the transition from epithelial to mesenchymal cells is known to be regulated by TFAP225. Studies on mice have demonstrated that cytokines of the IL-6 family play a crucial role in regulating myogenesis26. It is interesting to note that this activity is in turn dependent on MAPK and NFκβ signaling27. Recent studies on Bandur sheep have also reported the enrichment of the PI3K-Akt and MAPK pathways that are associated with hypertrophy as well muscle differentiation12,13. These pathways were enriched in the young animals.
As expected, lesser differentiation of gene expression was observed between successive age groups. Differential expression was prominent after 2.5–3 years in Bandur sheep reflecting transition from lamb to adult sheep. Major differences observed between lambs and adult sheep muscles were evident in the expression of genes associated with muscle structure, growth and lipid metabolism. The gene-protein network demarcated important interactive hub genes that may be involved in age related muscle development in sheep. The hub genes enriched in younger animals included COL1A2, COL1A1, TWIST1, NR4A1, VIPR1, CCNE1, ESR1, IL17A and ISG15.It is worth mentioning that these highly connected genes identified in our study are known to be involved in muscle growth and regeneration. Collagens form the major connective tissue that imparts structural strength to muscles28. The crosslinking between the collagen molecules increases with age resulting in increased toughness of meat in adult animals18. The expression of COL1A1 and COL1A2 was observed to be decreased in bovine fetuses that were myostatin deficient29. A decrease of collagen expression in older animals was also observed in mice30. Several studies have associated the significance of expression of collagen type 1 gene in muscle composition and regulation of intra muscular fat in pork31,32. Age affects the structure and distribution of collagen in the extracellular matrix of skeletal muscle15, which is also reflected in the change in expression of collagen precursor genes in skeletal muscles of Bandur sheep. TWIST1 is another gene encoding a transcription factor that is involved in the early growth of skeletal muscles but its function in adults is less explored33. It is also known to inhibit DNA binding of MYOD1 during myotube formation34. Other genes with higher expression in lambs that are associated with muscle growth include NR4A1 and VIPR1. Experimental evidence using NR4A1 knockout mice suggests its association with growth and proliferation of muscle cells35. Studies on mouse myoblasts revealed NR4A1 as an important factor regulating myoblast differentiation36. VIPR1 is a G-protein coupled receptor expressed in the skeletal muscles that may be involved in muscle atrophy/hypertrophy37. Further, experimental evidence supports the role of the estrogen receptor (ESR1) in muscle strength by modulation of myosin regulatory light chain in mice38. The ESR1 gene regulates expression via the estrogen signaling pathway. The IL17A gene, on the other hand has been reported to activate PPARγ which is involved in increased adipogenesis39. In addition, NR4A receptor family in conjunction with CRTC2 and PPARγ assists in myogenesis and adipogenesis via the CREB pathway40. Thus, most of the highly connected genes identified in Bandur lambs were associated with growth, proliferation and adipogenesis.
Notable hub genes in older or mature animals included MYOD1, RUNX1, TLE3. SOCS3, CBLB, HSPB1, IFIT1, CDLK2, NR4A2 and PER1. Myoblast differentiation is orchestrated by an assortment of muscle-specific regulatory factors. MYOD1, a myogenic regulatory factor and an important marker for myoblast development, facilitates myogenesis during embryogenesis. MYOD1 along with RUNX1 and TFAP1 transcription factors, has been associated with myoblast regeneration or proliferation41. Although the function of MYOD1 in adult muscle is less investigated, there are some reports that have established its role in growth and proliferation in adult muscles42,43. RUNX1 has also been suggested to contribute to myotube generation in humans44 and its expression is activated by muscle damage41. TLE is a family of transcriptional co-factors that are involved in cell differentiation. The transcriptional activity of MyoD has also been reported to be regulated by TLE345. Another important gene SOCS3, identified in this study has been linked to myogenic differentiation46. Previous reports suggest the role of CBLB gene in skeletal muscle atrophy in mice by negative regulation of growth factors during cell differentiation and development47.
The small heat shock proteins (Hsps) long associated with heat stress, are being increasingly implicated in cellular senescence and apoptosis48. The HSPB1 is highly expressed in muscle tissues and codes for Hsps49. The Hsps are anti-apoptotic and prevent the muscles from denaturation, which has implications in meat tenderness50. Several studies have reported the involvement of HSPB1 in meat tenderness in cattle51,52. Age related expression of genes associated with meat quality detected considerable increase in the expression of HSPB1 and HSPB8 in older age (3.5–4 years) in Bandur sheep. A similar increase in hspb1 protein was observed in the skeletal muscles of chicken53. Although much information on HSPB8 and aging are not available, it is known to be involved in protein damage through translational arrest and autophagy54. Previous studies on sheep have reported no significant effect of sex on the tenderness of longissimus thoracis muscles55. Therefore, it may be speculated that a similar expression of hsps could be expected across both sexes.
The role of some of the identified hub genes is not very well defined in skeletal muscles. These include the interferon stimulated genes ISG15 and IFIT1 that are suspected to be involved in muscular diseases56. Although inadequate information is available on CCNE1 (Cyclin E1) and Cyclin Dependent Kinase (CDK) gene in muscle tissues, both participate in the cell growth during the cell cycle57,58. CDKL2 is known to be involved in cell proliferation in human cancer cells59,60, while PER1 gene of the circadian clock is known to regulate cell proliferation and apoptosis in association with human cancer cells61. Further investigations are required to determine the precise role of these genes in sheep muscle development.
Conclusion
This study is an attempt to gain an insight into the dynamics of gene expression with progression of age in sheep skeletal muscles. Our results highlight the temporal changes in gene expression in skeletal muscles from lambs to adult sheep. Noteworthy demarcation between lambs and adult sheep was observed in the expression of genes associated with muscle growth, adipogenesis and apoptosis. The enrichment of PI3K-Akt and MAPK pathways in lambs suggests their relevance in muscle growth. This differential expression analysis will contribute towards understanding the genetic basis of physiological changes in muscles with age. Several highly connected genes identified in our study are known to be involved in muscle development and proliferation and may serve as candidates for future research in myogenesis.
Materials and methods
Samples
Healthy animals were selected in coordination with the sheep rearers and butchers from Mandya district, Karnataka. As the animals were selected from the field, they were considered healthy if they did not show any visible signs of infection or ailment and were inspected by a veterinary officer. Care was taken to select unrelated male animals from flocks reared under the same management and feed. Since no records of the animals were maintained, the age of the animals was determined by information provided by the rearers as well as the dentine pattern. A two-tooth stage confirmed the age about 12 months (Group 1), 4-tooth between 18 and 24 months (Group 2), 6-tooth between 30 and 36 months (Group 3) and 8-tooth between 42 and 48 months (Group 4). Four animals in each group were selected. The animals were slaughtered according to standard commercial ‘halal’ procedures.
RNA sequencing
The longissimus thoracis muscles were collected in RNALater (Thermo Fisher Scientific Baltics, UAB, Vilnius, Lithuania) solution. RNeasy kit (Qiagen, Hilden, Germany) was used for extraction and purification of total RNA. Samples with RIN value ≥ 7.0 (Agilent Bioanalyzer) were selected for further processing. Libraries of four biological replicates from each age group were prepared by TruSeq RNA Library Prep Kit v2 (Illumina, San Diego, CA, USA). Paired end sequencing (150 bp) was performed on Illumina HiSeq-2000 Platform.
Data analysis
FastQC (v 0.11.5) was used for assessment of the quality of samples62. The raw reads were trimmed and filtered using FastXToolKit. CLS Genomics Workbench 6.5.1 (CLC Bio, Aarhus, Denmark) was used for data analysis. The reads were mapped against the ovine genome assembly (Oar_v4.0). The RNA sequencing data have been deposited in the NCBI Bio Project PRJNA416678 with accession numbers SAMN16191735-750. Normalization of reads was done as reads per kilobase million (RPKM) and reads with RPKM values > 0.01 were included in the study. The CLC transcriptomics analysis tool was used for differential expression analysis across the different age groups.
Validation by real time quantitative PCR (RT qPCR)
Ten differentially expressed genes were selected for RT qPCR analysis (Table S7). Primers for selected genes were designed by Primer 3 software63 or taken from published sequences64,65,66. cDNA was synthesized from 2 µg of purified RNA, using Super Script III Reverse Transcriptase (Thermo Fisher Scientific, Carlsbad, CA), as per manufacturer’s protocol. Each sample was analyzed in triplicate qPCR reactions. The final reaction volume of 10 µl contained 2 µl of cDNA, 8 µl of qPCR master mix (5 µl of SYBR Green Real-Time master mix (Applied Biosystems,Vilnius, Lithuania), 0.3 µl of each primer, 2.4 µl of DNA/RNA-free water). The samples were run on QuantStudio 5 Real-Time PCR System (Applied Biosystems). Standard curve calculation using four points of cDNA serial dilutions was used to estimate the PCR efficiency.
Statistical analysis
Differentially expressed genes with log2 fold change ≥ 2.0 and p value (padj) < 0.05 were used for further analyses. DAVID67, Consensus Pathway Data Base68 and Reactome were utilized for the functional and pathways analysis69. Cytoscapever 3.6.070 along with cytoHubba app was utilized for gene-protein network analysis71. For the RT qPCR the mean cycle threshold (Ct) values of the genes were normalized to geometric mean of the reference genes PPIB and β-ACTN72. The 2−ΔΔCT method was used for data analysis73.
Ethics approval
Animal samples were purchased from local butchers. All ethical norms and guidelines were followed, with approval from Institutional Animal Ethics Committee, ICAR-National Bureau of Animal Genetic Resources, Karnal, Haryana, India (F.No. NBAGR/IAEC/2017, dated 21.01.2017).
Data availability
Data supporting this paper was generated by ICAR-NBAGR. The dataset generated in the study has been deposited in the NCBI (PRJNA416678).
References
BAHS-Basic Animal Husbandry & Fisheries Statistics, Government of India, Ministry of Agriculture, Department of Animal Husbandry, dairying & Fisheries, Krishi Bhavan, New Delhi, 1–132 (2019).
Mullen, A. M., Stapleton, P. C., Corcoran, D., Hamill, R. M. & White, A. Understanding meat quality through the application of genomic and proteomic approaches. Meat. Sci. 74(1), 3–16. https://doi.org/10.1016/j.meatsci.2006.04.015 (2006).
Kopuzlu, S. et al. Effects of slaughter age and muscle type on meat quality characteristics of Eastern Anatolian Red bulls. Arch. Anim. Breed 61(4), 497–504. https://doi.org/10.5194/aab-61-497-2018 (2018).
Li, Q. et al. Effects of age on slaughter performance and meat quality of Binlangjang male buffalo. Saudi J. Biol. Sci. 25(2), 248–252. https://doi.org/10.1016/j.sjbs.2017.10.001 (2018).
Byrne, K. et al. A gene network switch enhances the oxidative capacity of ovine skeletal muscle during late fetal development. BMC Genomics 11, 378. https://doi.org/10.1186/1471-2164-11-378 (2010).
Lin, Y., Zhu, J., Wang, Y., Li, Q. & Lin, S. Identification of differentially expressed genes through RNA sequencing in goats (Capra hircus) at different postnatal stages. PLoS ONE 12(8), e0182602. https://doi.org/10.1371/journal.pone.0182602 (2017).
Sadkowski, T., Jank, M., Oprzadek, J. & Motyl, T. Age-dependent changes in bovine skeletal muscle transcriptomic profile. J. Physiol Pharmacol. 57(Suppl 7), 95–110 (2006).
Bongiorni, S. et al. Transcriptomic investigation of meat tenderness in two Italian cattle breeds. Anim Genet. 47(3), 273–287. https://doi.org/10.1111/age.12418 (2016).
Ayuso, M. et al. Comparative analysis of muscle transcriptome between pig genotypes identifies genes and regulatory mechanisms associated to growth, fatness and metabolism. PLoS ONE 10(12), e0145162. https://doi.org/10.1371/journal.pone.0145162 (2015).
Kumar, A. et al. Transcriptomic diversity in longissimus thoracis muscles of Barbari and Changthangi goat breeds of India. Genomics S0888–7543(21), 00146–00154. https://doi.org/10.1016/j.ygeno.2021.04.019 (2021).
Hocquette, J. F. et al. The GENOTEND chip: A new tool to analyse gene expression in muscles of beef cattle for beef quality prediction. BMC Vet. Res. 8, 135. https://doi.org/10.1186/1746-6148-8-135 (2012).
Arora, R. et al. Transcriptome profiling of longissimus thoracis muscles identifies highly connected differentially expressed genes in meat type sheep of India. PLoS ONE 14(6), e0217461. https://doi.org/10.1371/journal.pone.0217461 (2019).
Kaur, M. et al. Differential expression of miRNAs in skeletal muscles of Indian sheep with diverse carcass and muscle traits. Sci. Rep. 10(1), 16332. https://doi.org/10.1038/s41598-020-73071-7 (2020).
Zhao, Y. et al. Dynamic transcriptome profiles of skeletal muscle tissue across 11 developmental stages for both Tongcheng and Yorkshire pigs. BMC Genomics 16, 377. https://doi.org/10.1186/s12864-015-1580-7 (2015).
Weston, A. R., Rogers Pas, R. W. & Althen, T. G. The role of collagen in meat tenderness. Profess. Anim. Sci. 18, 107–111 (2002).
Polidori, P., Pucciarelli, S., Cammertoni, N., Polzonetti, V. & Vincenzetti, S. The effects of slaughter age on carcass and meat quality of Fabrianese lambs. Small Rumin. Res. 155, 12–15 (2017).
Hogarth, M. W. et al. Analysis of the ACTN3 heterozygous genotype suggests that α-actinin-3 controls sarcomeric composition and muscle function in a dose-dependent fashion. Hum. Mol. Genet. 25(5), 866–877. https://doi.org/10.1093/hmg/ddv613 (2016).
Lee, L. A., Karabina, A., Broadwell, L. J. & Leinwand, L. A. The ancient sarcomeric myosins found in specialized muscles. Skelet. Muscle. 9(1), 7. https://doi.org/10.1186/s13395-019-0192-3 (2019).
Gomes, A. V., Potter, J. D. & Szczesna-Cordary, D. The role of troponins in muscle contraction. IUBMB Life 54(6), 323–333. https://doi.org/10.1080/15216540216037 (2002).
Johnston, J. R., Chase, P. B. & Pinto, J. R. Troponin through the looking-glass: Emerging roles beyond regulation of striated muscle contraction. Oncotarget 9, 1461–1482 (2018).
Wojtysiak, D., Kaczor, U., Połtowicz, K. & Krzysztoforski, K. The effects of sex and slaughter weight on muscle fibre characteristics and physico-chemical properties of lamb longissimus thoracis muscle. Anim. Sci. Papers Rep. 28(1), 61–69 (2010).
Lian, T., Wang, L. & Liu, Y. A new insight into the role of calpains in post-mortem meat tenderization in domestic animals: A review. Asian-Australas. J. Anim. Sci. 26(3), 443–454. https://doi.org/10.5713/ajas.2012.12365 (2013).
Cruzen, S. M., Paulino, P. V., Lonergan, S. M. & Huff-Lonergan, E. Postmortem proteolysis in three muscles from growing and mature beef cattle. Meat Sci. 96(2 Pt A), 854–861. https://doi.org/10.1016/j.meatsci.2013.09.021 (2014).
Nixon, R. A. The calpains in aging and aging-related diseases. Ageing Res Rev. 2(4), 407–418. https://doi.org/10.1016/s1568-1637(03)00029-1 (2003).
Dimitrova, Y. et al. TFAP2A is a component of the ZEB1/2 network that regulates TGFB1-induced epithelial to mesenchymal transition. Biol. Direct. https://doi.org/10.1186/s13062-017-0180-7 (2017).
Muñoz-Cánoves, P., Scheele, C., Pedersen, B. K. & Serrano, A. L. Interleukin-6 myokine signaling in skeletal muscle: A double-edged sword?. FEBS J. 280(17), 4131–4148. https://doi.org/10.1111/febs.12338 (2013).
Baeza-Raja, B. & Munoz-Canoves, P. p38 MAPK-induced nuclear factor-kappa B activity is required for skeletal muscle differentiation: Role of interleukin-6. Mol. Biol Cell. 15, 2013–2026 (2004).
Mukund, K. & Subramaniam, S. Skeletal muscle: A review of molecular structure and function, in health and disease. Wiley Interdiscip. Rev. Syst. Biol. Med. 12(1), e1462. https://doi.org/10.1002/wsbm.1462 (2020).
Cassar-Malek, I., Passelaigue, F., Bernard, C., Léger, J. & Hocquette, J. F. Target genes of myostatin loss-of-function in muscles of late bovine fetuses. BMC Genomics https://doi.org/10.1186/1471-2164-8-63 (2007).
Goldspink, G., Fernandes, K., Williams, P. E. & Wells, D. J. Age-related changes in collagen gene expression in the muscles of mdx dystrophic and normal mice. Neuromuscul. Disord. 4(3), 183–191. https://doi.org/10.1016/0960-8966(94)90019-1 (1994).
McBryan, J., Hamill, R. M., Davey, G., Lawlor, P. & Mullen, A. M. Identification of suitable reference genes for gene expression analysis of pork meat quality and analysis of candidate genes associated with the trait drip loss. Meat Sci. 86(2), 436–439. https://doi.org/10.1016/j.meatsci.2010.05.030 (2010).
Hamill, R. M. et al. Transcriptome analysis of porcine M semimembranosus divergent in intramuscular fat as a consequence of dietary protein restriction. BMC Genomics 14, 453. https://doi.org/10.1186/1471-2164-14-453 (2013).
Mudry, J. M., Massart, J., Szekeres, F. L. & Krook, A. TWIST1 and TWIST2 regulate glycogen storage and inflammatory genes in skeletal muscle. J. Endocrinol. 224(3), 303–313. https://doi.org/10.1530/JOE-14-0474.Erratum.In:J.Endocrinol.2015Sep;226(3):X1 (2015).
Miraoui, H. & Marie, P. J. Pivotal role of twist in skeletal biology and pathology. Gene 468, 1–7. https://doi.org/10.1016/j.gene.2010.07.013 (2010).
Cortez-Toledo, O., Schnair, C., Sangngern, P., Metzger, D. & Chao, L. C. Nur77 deletion impairs muscle growth during developmental myogenesis and muscle regeneration in mice. PLoS ONE 12(2), e0171268 (2017).
Pan, X. et al. Nr4a1 as a myogenic factor is upregulated in satellite cells/myoblast under proliferation and differentiation state. Biochem. Biophys. Res. Commun. 513(3), 573–581. https://doi.org/10.1016/j.bbrc.2019.04.026 (2019).
Jean-Baptiste, G., Yang, Z., Khoury, C., Gaudio, S. & Greenwood, M. T. Peptide and non-peptide G-protein coupled receptors (GPCRs) in skeletal muscle. Peptides 26(8), 1528–1536. https://doi.org/10.1016/j.peptides.2005.03.011 (2005).
Collins, B. C. et al. Deletion of estrogen receptor α in skeletal muscle results in impaired contractility in female mice. J. Appl. Physiol. 124(4), 980–992. https://doi.org/10.1152/japplphysiol.00864.2017 (2018).
Lee, S. J. et al. IL-17A promotes trans differentiation of mouse myoblast cells (C2C12) into adipocytes by increasing the expression of peroxisome proliferator-activated receptor γ through CAAT/enhancer binding protein β signaling. Biotechnol. Lett. 33(2), 229–235. https://doi.org/10.1007/s10529-010-0440-4 (2011).
Khan, R. et al. Genetic variants in the TORC2 gene promoter and their association with body measurement and carcass quality traits in Qinchuan cattle. PLoS ONE 15(2), e0227254. https://doi.org/10.1371/journal.pone.0227254 (2020).
Umansky, K. B. et al. Runx1 transcription factor is required for myoblasts proliferation during muscle regeneration. PLoS Genet. 11(8), e1005457. https://doi.org/10.1371/journal.pgen.1005457 (2015).
Kuang, S., Kuroda, K., Le Grand, F. & Rudnicki, M. A. Asymmetric self-renewal and commitment of satellite stem cells in muscle. Cell 129, 999–1010 (2007).
Ganassi, M., Badodi, S., Wanders, K., Zammit, P. S. & Hughes, S. M. Myogenin is an essential regulator of adult myofibre growth and muscle stem cell homeostasis. Elife 9, e60445 (2020).
Choi, I. Y. et al. Transcriptional landscape of myogenesis from human pluripotent stem cells reveals a key role of TWIST1 in maintenance of skeletal muscle progenitors. Elife 9, e46981. https://doi.org/10.7554/eLife.46981 (2020).
Kokabu, S. et al. The transcriptional co-repressor TLE3 regulates myogenic differentiation by repressing the activity of the MyoD transcription factor. J. Biol. Chem. 292(31), 12885–12894. https://doi.org/10.1074/jbc.M116.774570 (2017).
Swiderski, K. et al. Deletion of suppressor of cytokine signaling 3 (SOCS3) in muscle stem cells does not alter muscle regeneration in mice after injury. PLoS ONE 14(2), e0212880. https://doi.org/10.1371/journal.pone.0212880 (2019).
Nikawa, T. & Ishidoh, K. Ubiquitin ligase Cbl-b and inhibitory Cblin peptides. Biochim. Biophys. Acta Proteins Proteom. 1868(11), 140495. https://doi.org/10.1016/j.bbapap.2020.140495 (2020).
Tower, J. Hsps and aging. Trends Endocrinol. Metab. 20(5), 216–222. https://doi.org/10.1016/j.tem.2008.12.005 (2009).
Mymrikov, E. V., Seit-Nebi, A. S. & Gusev, N. B. Large potentials of small heat shock proteins. Physiol. Rev. 91(4), 1123–1159. https://doi.org/10.1152/physrev.00023.2010 (2011).
Malheiros, J. M. et al. Association of CAST2, HSP90AA1, DNAJA1 and HSPB1 genes with meat tenderness in Nellore cattle. Meat Sci. https://doi.org/10.1016/j.meatsci.2018.01.003 (2018).
Picard, B. et al. Skeletal muscle proteomics in livestock production. Brief Funct. Genomics 9(3), 259–278. https://doi.org/10.1093/bfgp/elq005 (2010).
Guillemin, N. et al. Variations in the abundance of 24 protein biomarkers of beef tenderness according to muscle and animal type. Animal 5(6), 885–894. https://doi.org/10.1017/S1751731110002612 (2011).
Ueda, S. et al. Chicken heat shock protein HSPB1 increases and interacts with αB-crystallin in aged skeletal muscle. Biosci. Biotechnol. Biochem. 79(11), 1867–1875. https://doi.org/10.1080/09168451.2015.1061419 (2015).
Carra, S., Seguin, S. J. & Landry, J. HspB8 and Bag3: A new chaperone complex targeting misfolded proteins to macro autophagy. Autophagy 4, 237–239 (2008).
Miguel, E., Blázquez, B. & Ruiz de Huidobro, F. Live weight and sex effects on instrumental meat quality of Rubia de El Molar autochthonous ovine breed. Animals 11, 1323. https://doi.org/10.3390/ani11051323 (2021).
Walsh, R. J. et al. Type I interferon-inducible gene expression in blood is present and reflects disease activity in dermato myositis and poly myositis. Arthritis Rheum. 56(11), 3784–3792. https://doi.org/10.1002/art.22928 (2007).
Morgan, D. O. Principles of CDK regulation. Nature 374(6518), 131–134. https://doi.org/10.1038/374131a0 (1995).
Winey, M. Cell cycle: Driving the centrosome cycle. Curr Biol. 9(12), R449–R452. https://doi.org/10.1016/s0960-9822(99)80279-6 (1999).
Li, L. et al. CDKL2 promotes epithelial-mesenchymal transition and breast cancer progression. Oncotarget 5(21), 10840–10853. https://doi.org/10.18632/oncotarget.2535 (2014).
Fang, C. L. et al. Loss of cyclin-dependent kinase-like 2 predicts poor prognosis in gastric cancer, and its overexpression suppresses cells growth and invasion. Cancer Med. 7(7), 2993–3002. https://doi.org/10.1002/cam4.1577 (2018).
Gong, X., Tang, H. & Yang, K. PER1 suppresses glycolysis and cell proliferation in oral squamous cell carcinoma via the PER1/RACK1/PI3K signaling complex. Cell Death Dis. 12(3), 276. https://doi.org/10.1038/s41419-021-03563-5 (2021).
Andrews, S. FastQC: A quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc (2010).
Untergasser, A. et al. Primer3-new capabilities and interfaces. Nucleic Acids Res. 40(15), e115. https://doi.org/10.1093/nar/gks596 (2012).
Banerjee, D. et al. Seasonal variation in expression pattern of genes under HSP70 family in heat- and cold-adapted goats (Capra hircus). Cell Stress Chaperones 19(3), 401–408. https://doi.org/10.1007/s12192-013-0469-0 (2014).
Bernard, C. et al. New indicators of beef sensory quality revealed by expression of specific genes. J. Agric. Food Chem. 55(13), 5229–5237. https://doi.org/10.1021/jf063372l (2007).
Zhu, W., Lin, Y., Liao, H. & Wang, Y. Selection of reference genes for gene expression studies related to intramuscular fat deposition in Capra hircus skeletal muscle. PLoS ONE 10(3), e0121280. https://doi.org/10.1371/journal.pone.0121280 (2015).
Huang, D. W., Sherman, B. T. & Lempicki, R. A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4(1), 44–57 (2009).
Kamburov, A. et al. ConsensusPathDB: Toward a more complete picture of cell biology. Nucleic Acids Res. 39, D712-717 (2011).
Jassal, B. et al. The reactome pathway knowledge base. Nucleic Acids Res. 8(48), D498–D503. https://doi.org/10.1093/nar/gkz1031 (2020).
Shannon, P. et al. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 13(11), 2498–2504. https://doi.org/10.1101/gr.1239303 (2003).
Chin, C. H. et al. cytoHubba: Identifying hub objects and sub-networks from complex interactome. BMC Syst. Biol. 8(Suppl 4), S11. https://doi.org/10.1186/1752-0509-8-S4-S11 (2014).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3(7), RESEARCH0034. https://doi.org/10.1186/gb-2002-3-7-research0034 (2002).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta deltaC(T)) method. Methods 25, 402–408 (2001).
Acknowledgements
This work was financially supported by ICAR-Consortium Research Platform-Genomics (Animal Science). We are grateful to Director, ICAR-National Bureau of Animal Genetic Resources (NBAGR), Karnal and Indian Council of Agricultural Research (ICAR), New Delhi for providing necessary facilities.
Funding
This work was financially supported by ICAR-Consortium Research Platform—Genomics (Animal Science).
Author information
Authors and Affiliations
Contributions
RA: conceptualization and supervision. MNF, NKS, SS, MSS and SRM: resources. AK, MK and SA: data curation. AK, PC, RKV and RA: formal analyses; RA: writing—review & editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Arora, R., Siddaraju, N.K., Manjunatha, S.S. et al. Muscle transcriptome provides the first insight into the dynamics of gene expression with progression of age in sheep. Sci Rep 11, 22360 (2021). https://doi.org/10.1038/s41598-021-01848-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-01848-5
- Springer Nature Limited
This article is cited by
-
Skeletal muscle transcriptomics of sheep acclimated to cold desert and tropical regions identifies genes and pathways accentuating their diversity
International Journal of Biometeorology (2024)
-
Molecular insights into Pashmina fiber production: comparative skin transcriptomic analysis of Changthangi goats and sheep
Mammalian Genome (2024)