Abstract
Milk fat globule (MFG) size is a milk production trait characteristic to the individual animal and has important effects on the functional and nutritional properties of milk. Although the regulation of MFG size in the mammary epithelial cell is not fully understood, lipid droplet (LD) fusion prior to secretion is believed to play a role. We selected cows that consistently produced milk with predominantly small or large MFGs to compare their lipidomic profiles, with focus on the polar lipid fraction. The polar lipid composition of the monolayer surrounding the LD is believed to either promote or prevent LD fusion. Using a targeted LC–MS/MS approach we studied the relative abundance of 301 detected species and found significant differences between the studied groups. Here we show that the lipidomic profile of milk from small MFG cows is characterised by higher phosphatidylcholine to phosphatidylethanolamine ratios. In contrast, the milk from large MFG cows contained more ether-phosphatidylethanolamine species. This is the first time that a potential role for ether-phosphatidylethanolamine in MFG size development has been suggested.
Similar content being viewed by others
Introduction
Milk lipids are secreted from the mammary epithelial cell as spherical structures termed milk fat globules (MFGs). They comprise a neutral lipid core surrounded by a membrane derived partially from the endoplasmic reticulum (ER, innermost layer) and the apical plasma membrane (two outer layers)1. The MFG membrane (MFGM) keeps lipids solubilised in milk, but also has several health benefits for infant and adult consumers. For example, beneficial effects on cancer, hypercholesterolemia, diabetes and cognitive function have been attributed to the protein, glycerophospholipid (PL) and sphingolipid (SL) components of the MFGM in bovine milk2. The amount of MFGM material in milk depends on the fat content and MFG size, with the greater surface area in smaller globules providing more MFGM material per unit of fat3. Moreover, the processing of milk into butter and cheese is influenced by the MFG size. For example, larger MFGs are more prone to coalescence and can increase the efficiency of butter manufacturing, while cheese manufactured from milk with small MFGs can have improved sensory properties4.
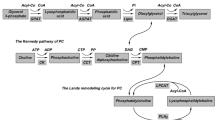
The formation of MFGs within the mammary epithelial cell starts with the accumulation of neutral lipids (mainly triacylglycerols (TG)) in the ER1. This results in the formation of a lipid droplet (LD), the intracellular precursor of the MFG, which is released from the ER surrounded by a single layer of polar lipids and membrane proteins. The ultimate size of individual MFGs is determined by two growth mechanisms inside the mammary epithelial cell: either via local TG synthesis by enzymes residing in the monolayer surrounding the LD or by LD fusion5,6. The exact mechanisms behind LD fusion remain elusive but may depend on the PL and fatty acid (FA) composition of the monolayer. PLs with smaller headgroups, such as phosphatidylethanolamine (PE) and phosphatidic acid are predicted to enhance fusion of LD, while phosphatidylcholine (PC), which has a more cylindrical shape, can protect LDs from fusion7. Although most of the available evidence in the literature is based on studies in non-mammary cells, this important role of the polar lipid composition of the LD monolayer in the regulation of LD fusion has also been demonstrated in mammary epithelial cells8. Moreover, not only the type but also the FA composition of the polar lipids residing in the monolayer surrounding the LD determine its size9. Apart from sphingomyelin (SM), which contains predominantly saturated FAs (SFAs), most PLs are rich in unsaturated FAs (UFAs), although PC is less unsaturated compared to PE, phosphatidylinositol (PI) and phosphatidylserine (PS)10. Using a cell line engineered to produce larger LDs, it has been shown that LDs extracted from these cells contained less UFAs in the PE and PC fractions of the LD monolayer compared to control cells with smaller LDs9. Figure 1 illustrates the suggested impact of several metabolic pathways on LD and ultimately MFG size, based on evidence from the literature combined with hypothetical explanations for the differences in lipidomic profiles reported in the current study.
Possible mechanisms through which major metabolic pathways contribute to the milk fat globule (MFG) size development in the mammary epithelial cell. This illustration is based on the currently available literature and provides hypothetical explanations for the observed differences in milk lipidomic profiles between the studied groups. Please note that the precise role of these metabolic pathways has not been established. Diacylglycerol (DG) plays a central role and connects phospholipid (PL), triacylglycerol (TG) and sphingolipid synthesis pathways. DG is utilised to either produce TG or the major PLs, phosphatidylethanolamine (PE) and phosphatidylcholine (PC). TG synthesis is catalysed by the enzyme diacylglycerol-acyltransferase (DGAT), in the form of two isoenzymes DGAT1 and DGAT2 which are located in the endoplasmic reticulum (ER) or on the surface of lipid droplets (LD), respectively5. The synthesis of PC and PE is catalysed by DG choline/ethanolamine phosphotransferase (CEPT) found in the ER61. This enzyme also catalyses the production of ether PLs (ePL) by transferring a choline or ethanolamine headgroup to alkylacylglycerol (AAG), which is derived from peroxisomal production of 1-O-alkyl-glycerol-3-phosphate (AGP)33. We suggest that when DG is more efficiently channelled towards TG synthesis, for example due to increased DGAT1 activity, diacyl-PL synthesis could be limited and ePL synthesis through CEPT in the ER could present a possible salvage pathway that is not reliant on DG as a precursor. This shift towards ePL synthesis could promote LD fusion and thus contribute to the large MFG (LMFG) phenotype. In the small MFG (SMFG) phenotype, DG could be more efficiently channelled towards PL synthesis, either due to decreased DGAT1 activity or possibly due to increased SM synthase 1 (SMS1) activity in the Golgi apparatus, although this hypothesis could not be confirmed by the current study. SMS1 catalyses the transferal of the choline headgroup from PC to ceramide (Cer) to produce SM and DG, thus contributing to the DG pool. Moreover, increased SMS1 activity can stimulate PC synthesis51. Another potential pathway contributing to the SMFG phenotype is the phosphatidylethanolamine N-methyltransferase (PEMT) pathway, which converts PE into PC and thus increases the PC/PE ratio. The PE used in the PEMT pathway is derived mainly through decarboxylation of phosphatidylserine (PS) in the mitochondrion by PS decarboxylase (PSD) and results in PC with a higher degree of unsaturation. Increased unsaturation within the monolayer surrounding the LD and increased PC/PE ratios could prevent LD fusion, resulting in the SMFG phenotype. Figure created with BioRender.com.
Variations in MFG size exist between species of mammals and between breeds of the same species11,12, but also between animals of the same breed of cows13,14. The size of MFGs in cow’s milk ranges from less than 1 µm to 15 µm15, with the average size ranging between 2.5 and 5.7 µm within a herd of Holstein–Friesian cows13. Moreover, the average MFG size changes throughout the lactation cycle depending on the days in milk since the last calving and throughout the year in response to changes in the diet16. Other suggested impact factors on MFG size are fat content17, fat-to-protein ratio18 and milking period16. MFG size also depends on the physiological state of the animal16,19 and the milk from cows producing small or large MFGs can differ in the FA composition14,18. However, the magnitude of the effects of parameters such as the physiological state of the animal and its milk production traits on MFG size is less than the overall variation that is seen between individual animals20.
Differences in the FA composition of MFGs from cows naturally producing milk with small or large average MFG sizes have previously been reported14 and were also demonstrated in our previous study using the same samples as in the current work21. Moreover, a role of the relative proportion of major polar lipid classes in milk fat, such as PE and PC, is believed to impact MFG size22,23,24, although this has only been shown using milk samples separated into small and large MFG fractions and is yet to be demonstrated using whole milk samples from cows that naturally produce milk with small or large average MFG sizes. We hypothesised that lipidomic differences between cows consistently producing small or large MFGs could extend beyond the major lipid classes and therefore aimed to study the role of minor lipid classes and individual lipid species on MFG size using triple quadrupole LC–MS/MS analysis. To the best of our knowledge, with 301 detected species, this study presents the most extensive milk lipidomic analysis yet and reports differences in the polar lipid species and their FA composition between cows producing small or large MFGs.
Results
Differences in milk production traits and MFG size between the studied groups
The average MFG size, expressed as the volume weighted mean diameter D4,3, was 1.63 µm larger in the milk from cows in the large MFG (LMFG) group compared to the small MFG (SMFG) group. The selected cows also differed significantly in the surface weighted mean diameter D3,2 (Table 1). Other production traits such as milk composition, the average number of milking sessions per day and concentrate intake were statistically similar between the studied groups (Table 1). However, there was a small difference in days in milk, representing the number of days since the last calving (25 days difference, Table 1). Furthermore, cows from the SMFG group included some primiparous cows (parity 1 to 3), while the LMFG group contained cows in later parities only (parity 3 to 5).
Lipidomic profile of milk from the studied groups
Lipidomic analysis was performed on whole raw milk samples which were frozen within a minute of sample collection and thus most accurately represent the milk lipid metabolite composition of each animal at the time of sampling. Using a targeted approach, we measured 445 compounds and detected 301 thereof in milk. Although previous reports have reported higher numbers of neutral lipid species25, to the best of our knowledge, this study presents the highest number of polar lipids detected in milk to date. These species were from a total of 14 lipid classes, many of which had not been previously the focus of milk lipidomics (Table 2). For example, we detected 23 and 19 species of plasmenyl-PE and plasmenyl-PC, respectively. Moreover, we detected 24 ceramide (Cer) species and five and seven additional dihydroceramide and hexosylceramide species. Detected lysophospholipid species included 18 lysophosphatidylcholine (LPC), six lysophosphatidylethanolamine (LPE) and three lysophosphatidylinositol (LPI) species (Table 2). A complete list of the identified lipid species is provided in the supplementary file of this article. Amongst the detected compounds, nine lipid species were significantly different in relative abundance (P < 0.05) and 18 species tended to differ (P < 0.10) between the SMFG and LMFG groups.
To date, about 400 lipid species have been discovered in milk, however the estimated total number is believed to be around 1,000 lipid species, with many low abundance compounds yet to be discovered26. The targeted method used in our study allowed us to identify the total carbon number (CN), number of double bonds and the FA composition of glycerophospholipids which was characterised by collision induced dissociation in positive ionization mode as reported previously27,28. However, cow’s milk contains short chain FAs with less than 14 carbons29 and TG species containing these FAs, resulting in a total CN < 48, which will not be detected by this approach. This represents a limitation of the present study in relation to TG analysis since a large proportion of TGs have total CNs < 48:1 which was the smallest CN of TG species detected in our study26. Therefore, the differences in the FA composition and origin of FAs between the studied groups were not the focus of the current study. The PL components of milk, on the other hand, contain mostly medium and long chain FAs and the lipidomic approach described here covers the major lipid species in this group. Therefore, the focus of this study was the identification of differences in polar lipid species.
Differences in neutral lipid species
The milk lipidomic profile contained six TG lipid species that were relatively more abundant in LMFG milk compared to SMFG milk (Fig. 2D). However, detailed analysis of the lipid species revealed that the TGs with higher relative abundance in whole milk of LMFG cows mainly contained FAs with one or no double bond (P = 0.132, Table 3). Although, the TG classes measured in this study were limited to species with CN ≥ 48, these results align with the results reported by others14. Therefore, the results of the milk lipidome in combinations with previously reported results14,18, indicate higher saturation in LMFG cows compared to SMFG cows, while fat yield and fat content were statistically similar between the groups (Table 1).
Differences in polar lipid species
Comparative analysis of the polar lipid species in raw milk from SMFG and LMFG cows revealed differences in the two major PL classes, PE and PC (Fig. 2B). Milk from SMFG cows contained relatively more of two PC species (PC 36:4 and PC-P 34:0) along with a trend towards higher relative abundance of total PC compared to LMFG cows (16% increase, P = 0.123, Fig. 3). The PC/PE ratio was also calculated to minimise the impact of intrinsic differences in PL abundance in small compared to large MFGs, due to a higher PL/TG ratio in smaller MFGs3. Indeed, in the SMFG group, PC/PE ratios were higher than in LMFG cows (21%, P = 0.001). Milk from LMFG cows, on the other hand, differed from SMFG milk in seven PE lipid species (five species with P < 0.05 and two species with P < 0.10, Fig. 2B). Some of the ePE species showed increases in relative abundance of over 45% (PE-P 34:3, PE-P 36:1, PE-P 34:1 and PE-O 34:2, all P < 0.01). This led to an increase in the plasmanyl-PE and plasmenyl-PE subclasses of 27% and 33% (P = 0.104 and P < 0.01, respectively). However, these differences did not lead to a change in the total relative abundance of the total PE lipid class between the groups (Fig. 3). These results suggest, for the first time, a possible link between ePE and MFG size.
Volcano plots showing the milk lipidome of cows with small (SMFG) or large (LMFG) average milk fat globule size. The abundance of lipid species from SMFG relative to LMFG milk are shown as (A) total lipid species, (B) phospholipid species, (C) sphingolipid species and (D) neutral lipid species with a total carbon number of 48 and over. The fold change, expressed as ln(SMFG)-ln(LMFG), when multiplied by 100 represents the change in %. Lipid species with positive percentages are more abundant in SMFG milk and lipid species with negative percentages are more abundant in LMFG milk. The horizontal lines denote statistical significance with lipid species above the upper line displaying adjusted P values < 0.05 and above the lower line displaying adjusted P values < 0.10. P values were adjusted according to the Benjamini and Hochberg58 procedure.
The relative difference in abundance of lipid classes and subclasses in milk from cows with small average milk fat globule size (SMFG) compared to cows with large average milk fat globule size (LMFG). Estimated mean difference and 95% confidence intervals are shown. Lipid classes shown in black (P < 0.10) or pink (P < 0.05) are different between the groups after adjusting the P values according to the Benjamini and Hochberg58 procedure.
Some differences were also observed within the Cer and SM classes (Fig. 2C). More specifically, the milk of LMFG cows contained more Cer (d18:1_14:0, P < 0.05) and Cer (d18:2_22:0, P < 0.10) than the milk of SMFG cows, while the relative abundance of total Cer was similar between the studied groups (Fig. 3). In contrast, SMFG milk contained a higher relative abundance of Hex2Cer (d18:1_22:0, P < 0.10) and two SM species (SM 38:1and SM 40:3, P < 0.10) compared to LMFG milk (Fig. 2C). Again, this was not related to a difference in total SM abundance (Fig. 3). However, SM/Cer ratios tended to be numerically higher in SMFG milk compared to the LMFG group, although this did not reach statistical significance (Fig. 3).
Degrees of saturation within the lipid classes
Lipid droplet fusion not only depends on the physical properties of the main PLs in its monolayer but is also influenced by the FA composition of the acyl chains and their impact on fluidity and packing density, with UFAs predicted to increase fluidity and prevent LD fusion9. To investigate possible differences in the degree of saturation within the lipid classes between the studied groups, the data was categorised into three groups containing no double bonds, a total of one double bond or a total of two or more double bonds in their acyl chains and the results are shown in Table 3. Numerically, the degree of unsaturation appeared to be higher in some of the polar lipid classes (PC, LPC and PI) along with a numerically reduced degree of saturation within the TG class in the milk from SMFG cows compared to the milk from LMFG cows (Table 3), but these results did not reach statistical significance after adjustment for multiple comparisons. The FA composition of the MFG core (mainly containing neutral lipids), was the subject of our previous study and also showed a higher proportion of SFAs in the core of MFGs form LMFG cows compared to SMFG cows21, suggesting that the FA composition of the neutral lipid core could impact MFG size. However, the polar lipids, which are found in the MFGM are more likely to affect LD fusion and in the current study, the degree of saturation within the polar lipid classes did not differ significantly between the studied groups.
Discussion
Comparative analysis of the lipidomic profiles of milk from the SMFG and LMFG groups showed several important differences, mainly within the PC and PE lipid classes. The milk lipidome from LMFG cows was characterised by a higher relative abundance of plasmanyl- and plasmenyl-ethanolamine lipid subclasses, while the milk from SMFG cows showed higher PC/PE ratios and tended to have a higher relative abundance of total PC species (Fig. 3). The biophysical properties of PC compared to PE result in its superior surfactant properties and increased PC/PE ratios in the monolayer surrounding the intracellular LD are predicted to limit LD fusion7,8. Moreover, ePL species and particularly plasmenyl-PE have been shown to reduce the fluidity of biological membranes and have been implicated in membrane fusion events30,31. Therefore, the milk lipidome from cows presenting small and large phenotypes provides several differences, which could explain their characteristic MFG phenotype.
A limitation of our study was that the selected cows differed slightly in days in milk and were significantly different in terms of their parities (Table 1). Both these parameters can affect MFG size and could be contributing factors to the observed difference. However, we previously estimated the effect of these parameters on MFG size within our herd20. Based on this analysis we estimate that the difference between the groups attributable to the difference in days in milk and parity is in the magnitude of 0.21 µm (when comparing SMFG cows to LMFG cows, a decrease of 0.05 µm can be attributed to 25 additional days in milk in average for SMFG cows; and an additional decrease in MFG size of 0.16 µm can be attributed to the difference in parity, with an average parity of 1.8 for SMFG cows compared to 3.8 in LMFG cows). The difference in MFG size between the selected groups (1.63 µm) is therefore approximately 8 times the estimated effect of the impact of days in milk and parity. However, days in milk and season were added to the statistical model as a fixed effect to control for their impact on MFG size. Nonetheless, due to the lack of studies using LC–MS/MS based lipidomics in milk, at this stage, we cannot determine, to what extend the difference in parity could have contributed to the differences in the lipidomic profiles seen between the studied groups.
The significance of our results lies in the physical properties of ePLs, particularly plasmenyl-PE, and their potential effect on LD fusion. Ether-PLs (ePLs) contain an ether linked fatty alcohol at the sn-1 position, where their diacyl equivalents contain an ester linked FA32. Among ePLs, there are two subcategories depending on the nature of the ether bond. The so-called plasmanyl-species contain a 1-O-alkyl ether bond, while plasmenyl-species have a vinyl-ether bond (cis double bond at the Δ1 position adjacent to the ether bond) and are also known as plasmalogens33. Plasmalogens are mainly found in the form of PE species and their concentrations are reported to reach up to 18% of the total PLs in membranes and can constitute up to 70% of the total PE lipid class depending on the tissue33. However, information on the abundance of plasmalogens in the lactating mammary gland is lacking and can only be estimated based on their abundance in milk PLs, which are derived from the plasma membrane of the mammary epithelial cell. These results suggest an abundance of plasmenyl-PE in cow’s milk of approximately 4.5% of total PLs.
Plasmenyl-PE with its vinyl-ether bond has been shown to increase the speed of fusion in vitro in small unilamellar vesicles30. This is attributed to the propensity of plasmenyl-PE to form so called non-bilayer structures at lower temperatures than plasmanyl- and diacyl-PE. Phase transition from the liquid-crystalline to the inverted hexagonal (non-bilayer) state promotes membrane fusion33. It is now known that LD fusion contributes to the ultimate MFG size in mammary cells, as shown by in vitro8 and in vivo time-lapse imaging6. The increased relative abundance of ePE in LMFG cows could also impact packing density and fluidity of the LD monolayer. Ether PLs are more lipophilic, which leads to tighter packing of ether lipids in biological membranes compared to their diacyl counterparts, decreasing membrane fluidity31. Membrane fluidity is also predicted to impact LD fusion34, with reduced fluidity of the monolayer promoting LD fusion. We therefore suggest that the higher relative abundance of plasmenyl-PE in the milk from LMFG cows, which is predicted to enhance LD fusion, could influence MFG size. This is the first time that a relationship between the ePE subclass and MFG size has been suggested. However, studies focusing on the pathophysiology of ePL deficiencies have revealed various important biological functions. For example, a possible role of plasmalogens in LD homeostasis has been suggested, characterised by smaller LDs in brown adipose tissue of plasmalogen deficient mice35.
In terms of the major polar lipid classes, where SMFG cows showed higher PC/PE ratios compared to LMFG cows, the different physical properties of PC compared to PE, as well as some in vitro evidence suggest that PE enhances LD fusion, while PC has a protective effect7,8,36. However, several studies on the PL composition of MFGs with different sizes have reported conflicting results22,23,24 or found no difference in the PL composition between small and large MFGs37. These studies separated MFGs from bulk raw milk or individual raw milk samples through gravity separation, microfiltration and centrifugation. We used samples from cows naturally different in MFG size distributions, with the aim to study the lipid metabolic differences between the small and large phenotype. Our results support the notion that increased relative PC abundance and more importantly the significantly increased PC/PE ratio are related to smaller MFG size. While the CDP-choline pathway is responsible for the bulk of PC synthesis (about 70%), the phosphatidylethanolamine N-methyltransferase (PEMT) pathway (Fig. 1) can make up the remaining proportion38 and the PC/PE ratio is regarded as an indicator for the PEMT pathway activity39. Notably, during this process PE is converted into PC, resulting in the production of PC at the expense of PE. The PE used during this reaction is predominantly derived from decarboxylation of PS as opposed to PE derived from the CDP-ethanolamine pathway40. Moreover, the PC species produced by the conversion of PE into PC contain more long chain and polyunsaturated FAs (PUFAs) compared to the PC species derived through the CDP-choline pathway41. Although we found a numerically higher relative abundance of PC species with two or more double bonds (P = 0.132, Table 3), this was not statistically significant and we cannot ascertain if the observed trend towards increased relative PC abundance in SMFG cows was due to an increase in unsaturated PC lipid species. Interestingly, it has also been reported that the concentration of PC derived through the PEMT pathway is higher in the LD monolayer compared to other cellular membrane components40. Furthermore, the PEMT enzyme has been localised on the surface of LDs or in close proximity to LDs, at sites where they are in close contact with the ER and mitochondria-associated membranes40. The contribution of this pathway to the total PC content in mammary tissue is still unknown, but in vitro studies on primary bovine mammary epithelial cells have shown that manipulation of the PEMT pathway affects intracellular LD size8. Although the cow’s diet can also affect the relative proportions of the major polar lipid classes42, the cows in the current study received the same diet and did not differ in their daily concentrate intake. Therefore, dietary changes are unlikely to be the reason for the observed trend towards higher relative PC abundance and the significantly increased PC/PE ratios in the SMFG group. Whatever the reason for the increased PC content in milk from SMFG cows, the effect of increased PC/PE ratios, and possibly PC species with higher unsaturation, could reduce the propensity of LDs to participate in LD fusion and could impact MFG size.
Our study provides first evidence for a potential role of ePE and confirms previously suggested roles for the proportion of major PLs, particularly the ratio of PC/PE in the size development of MFGs. Our study presents a comparison of the lipidomic profiles of the selected cows and we would like to put forward potential reasons for the observed differences. Firstly, the activity of metabolic pathways that utilise or produce DG could regulate MFG size development in the mammary gland. More specifically, we suggest that the variation in MFG size between the studied groups could be determined by the metabolic activity of the pathways for TG, PL and possibly SL synthesis. The possible relationship between these pathways and MFG size is illustrated in Fig. 1. Since the same enzymes are responsible for the synthesis of ether lipid species and their diacyl equivalents, the final concentration may depend on the relative abundance of their metabolic precursors. Indeed, alkylacylglycerol, the direct precursor for ePL synthesis, is predicted to stimulate ePL synthesis, while DG stimulates the synthesis of PE and PC43. Therefore, increased usage of DG for TG synthesis, possibly due to increased DGAT1 activity, could lead to reduced DG availability for PC and PE synthesis. Accordingly, MFG size has been related to the DGAT1 K232A polymorphism, with the KK genotype showing lower PL/TG ratios (larger MFG) compared to the AA genotype44. This is attributed to a higher activity of the DGAT1 K variant resulting in increased fat content45 as well as a changed FA composition46. The results from these studies align with the phenotypical differences seen between the SMFG and LMFG cows in our previous study, where we found less UFAs, but more C16 FAs in the MFG core of LMFG cows21. Accordingly, the K genetic variant is related to an increased content of C16:0 as well as SFAs and reduced contents of unsaturated C18 FAs and C14:046. However, in our study the average fat yield and fat content were similar between the studied groups, which is in contrast to the increased fat content which is usually characteristic for the K variant47. Under the assumption of increased DGAT1 activity in LMFG cows, this could lead to the reduced availability of DG for PL synthesis because it is primarily used for TG synthesis. Consequently, the limited availability of DG as a precursor for diacyl-PL synthesis could lead to a shift towards ePL synthesis in LMFG cows. Therefore, we suggest that ePL synthesis could represent a possible salvage pathway, when PL synthesis through the conventional pathways is limited, for example when DG is predominantly used for TG synthesis. The fact that plasmenyl-PE is the dominant type of plasmalogen found in most tissues and is the precursor for plasmenyl-PC synthesis explains why mainly ePE species were relatively more abundant in LMFG cows compared to SMFG cows.
Secondly, the lipidomic profile of the milk from the studied cows also revealed differences in some Cer and SM lipid species. Ceramides have been linked to insulin resistance in cows and humans48,49. In addition, Cer and particularly the balance between SM and Cer may also be involved in LD biogenesis and this potential relationship is illustrated in Fig. 1. Some evidence suggests that SL, PL and TG synthesis pathways are interconnected and have important effects on LD composition and size50. For example, increased SMS1 activity in hepatocytes has been shown to activate PC sensing mechanisms and increase de novo PC synthesis, channelling DGs towards PC synthesis and away from TG synthesis through the DGAT pathway51. Although not statistically significant, the ratio of SM/Cer showed a numerical increase in SMFG compared to LMFG cows (22%, P = 0.192). Whether or not an increased SM/Cer could potentially have contributed to the increased PC/PE ratios in SMFG cows will need to be elucidated in future studies.
Conclusion
The results obtained from PL and SL analyses indicated several differences in the lipidomic profiles between the studied groups and we have suggested possible explanations for the observed differences. Firstly, milk from SMFG cows contained more PC relative to PE compared to LMFG cows, a finding that is in accordance with some of the available literature. However, a novel finding of the current study was that the lipidomic profile of milk from the LMFG group was characterised by increased levels of plasmanyl- and plasmenyl-PE. This is the first time that a potential relationship between ether linked PE and MFG size has been suggested, and the underlying mechanisms leading to an increased relative abundance of these lipid species in LMFG cows remains unknown. Moreover, our findings support the hypothesis that in addition to the impact of major lipid classes, especially the relative proportions of the main polar lipid classes, minor lipid classes can also impact MFG size. Therefore, further studies should focus on the precise role of ePL in the mammary gland, their location within the mammary epithelial cell and their absolute abundance, along with the required concentration to exert their suggested biological functions. Overall, the results from the current study suggest a potential relationship between the milk lipidome and the characteristic MFG size of individual cows. Some of these relationships have been suggested for the first time and warrant further investigation. However, it should be noted that the impact of parity on the milk lipidome could not be entirely separated from the observed relationship between the milk lipidome and MFG size. Moreover, we cannot exclude that other metabolic differences between the studied animals, that were not measured in the current study, could have contributed to the individual variation in the milk lipidome of the selected cows.
Materials and methods
Cow selection and milk sampling
A total of 12 Holstein–Friesian cows were selected from the main herd for this study. The cows are part of the herd at the University of Melbourne Dookie Dairy in Victoria, Australia (latitude 36° 25′ 31.3″ S, longitude 145° 42′ 36.6″ E). The herd comprises approximately 150 cows, which are milked by an automatic milking system (Lely Astronaut; Lely, Maassluis, The Netherlands) and pasture fed and kept outside all year. The cows’ diet in the paddock is supplemented with hay and silage if needed and cereal grain-based concentrate is fed during milking. Of the 12 selected cows, six cows consistently produced milk with small average MFG size (< 3.5 µm, expressed as the volume weighted mean diameter D[4,3]), forming the SMFG group. The remaining six cows formed the LMFG size group, which produced milk with an average diameter of > 4.5 µm (Fig. 4).
Study design for the current study, including milk sampling and cow selection based on a pre-trial observation period. The top left panel shows the consistency in volume weighted mean diameter D4,3 of milk fat globules (MFG) from selected cows compared to the whole herd. Mean symbols (black triangles) and individual values (blue and orange circles for small MFG (SMFG) and large MFG (LMFG) cows, respectively) are shown for the 12 selected cows individually, where error bars represent 95% confidence intervals. Boxplots are shown for the SMFG and LMFG group compared to the data collected for the whole herd including the pre-trial period and the four sampling days of the current study. Asterisks denote outliers 1.5 times the interquartile range above or below the upper or lower quartile, respectively. Each selected cow was measured a minimum of three and a maximum of 9 times. The data presented in the top left panel of this figure is also presented as part of another study21 using milk samples from the same cows selected for the current study which were collected at the same time.
Milk from each selected cow was sampled twice on separate days approximately two weeks apart, resulting in a total of 24 samples from 12 cows. The cows were sampled in two cohorts as illustrated in Fig. 4. The first cohort of six cows, including three per study group, were sampled on two separate days in autumn (March 2016) and the second cohort on two days in winter (June 2016). Milk samples were collected during morning milking (0800 to 1000 h). For the lipidomic analysis, whole raw milk samples (30 mL in 5 ml aliquots) were collected and frozen within 1 min from the end of milking in a liquid nitrogen dry shipper and were subsequently kept at − 80 °C. The University of Melbourne Dookie Dairy is a commercially operating farm and milk samples were taken from the entire milk volume collected from the cows during routine milking.
One of the goals of this study was to select cows that consistently produce milk with average MFG sizes on the small or large end of the spectrum observed within a herd but have otherwise similar milk production traits and are in similar stages of lactation. Therefore, cows were surveyed over one year prior to this study and the selected animals were measured between three to nine times, including the two samples per cow taken for the current study. The MFG size measurements taken of the selected groups compared to the whole herd during the observation period was reported in our previous study21.The average MFG size from this pre-trial observation period for the whole herd and the selected groups are shown in Fig. 4. The animals selected for this study were also used for another study looking at the FA profile of the MFG core21, where milk samples were collected at the same time as the samples collected for the current work. Therefore, the descriptive data for milk production and animal parameters presented in Table 1 and MFG size measurements for the pre-trial observation period presented in Fig. 4 are identical to the data presented in our other study21.
The selected cows differed in MFG size and parity, while milk composition, number of milking sessions per day and concentrate intake were statistically similar between the groups (Table 1). Cows were further selected based on stage of lactation, which resulted in a range of 184–230 days in milk for the first cohort of cows and 57–135 days for the second cohort. Overall, the SMFG cows were, on average, 25 days later in their lactation cycle. Due to the year-round calving pattern in our herd, we decided to perform sampling in two separate seasons in order to obtain two groups which contain animals with the widest possible difference in average MFG size (Fig. 4). Because the goal was to capture the milk lipidomic differences underlying the individual variation in MFG size observed in our herd, season and days in milk were included in the statistical model as fixed effects.
MFG size measurements and proximate analysis
MFG size measurements and proximate analysis of freshly collected milk samples were performed the day after milk collection following storage in ice water overnight. Fat and protein concentrations were determined using a LactoScope FTIR 20 (Delta Instruments, Melbourne, Australia). MFG size was measured as previously described13 with a Mastersizer 2000 particle analyser (Malvern Instruments, Malvern, UK). Prior to particle size analysis milk was mixed 1:1 with 35 mM EDTA, pH 7.0 to dissociate casein micelles and the obscuration rate was kept between 12 to 15%. Refractive indices for milk fat and water were set to 1.46 and 1.33, respectively. Each sample was measured in duplicate and the volume-weighted mean diameter D4,3 was used as the measure for the average MFG size.
Lipidomic analysis and data curation
From 24 milk samples, 20 µL of each sample were used for lipid extraction using a modified Folch extraction protocol52. Briefly, 20 µL of milk samples were suspended in 400 µL ice cold chloroform:methanol (2:1, v/v) containing 10 mg/L of each internal standard. The internal standards were PC19:0/19:0, PE-d31, PG17:0/17:0 and TG-d5 19:0/12:0/19:0 (Product # 850,367, 8,609,040, 860,374, and 830,456, from Avanti Polar Lipids, Alabama, USA). Samples were vortexed and then mixed at 950 rpm for 30 min at 27˚C with a Thermomixer C (Eppendorf South Pacific Pty Ltd, Macquarie Park, Australia). Samples were centrifuged at 16,100 × g (Beckman Coulter Microfuge® 22R Refrigerated Microcentrifuge, Beckman Coulter Australia Pty Ltd, Sydney, Australia) for 10 min and the supernatant transferred to fresh LoBind Eppendorf tubes. Samples were completely dried in a vacuum concentrator, with the temperature maintained at 30–35 °C (Christ® RVC 2–33, Martin Christ Gefriertrocknungsanlagen GmbH, Osterode am Harz, Germany). The samples were reconstituted with methanol:water-saturated butanol (100 µL, 1:9, v/v). Pooled biological quality control samples (PBQC) were prepared by pooling aliquots of the extracts from each sample and ran after every five samples.
Extracted lipids were processed and detected by Metabolomics Australia (Bio21 Institute, Melbourne, VIC, Australia) as previously described27,28 using an Agilent 1,290 liquid chromatography (LC) system and Triple Quadrupole 6,490 mass spectrometer (MS, Agilent Technologies Australia, Mulgrave, Australia). Briefly, the lipid material (1 μL) was separated using a Zorbax Eclipse Plus C-18 column (100 mm × 2.1 mm × 1.8 µm, Agilent Technologies Australia, 0.4 mL/min) with a mobile phase solvent system of A: water/acetonitrile/isopropanol (50:30:20, v/v/v) and B: water/acetonitrile/isopropanol (1:9:90, v/v/v), both with 10 mM ammonium formate, using a flow rate of 400 µl/min. After equilibration with 10% B, the gradient elution used was 10%-45% B (2.7 min), 53% B (0.10 min), 65% B (0.20 min), 89% B (0.10 min), 92% B (1.9 min), 100% B (1.9 min), then 10% B (1.1 min), total run time 14 min. The column was preconditioned with 10% B for 30 min before a batch analysis. The lipids were detected in positive ionisation mode with dynamic scheduled multiple reaction monitoring. The MS parameters and MRM transitions of each lipid class, subclass and individual lipid species have been previously described27,28. Data processing was performed using Agilent’s Mass Hunter Quantitative Analysis (QQQ) software (Agilent Technologies Australia).
Lipid nomenclature
Lipid species were named according to the LIPID MAPS nomenclature described in Liebisch et al.53. Lipid species identified on a group level were named according to their lipid class followed by the total number of carbons and double bonds (e.g. PC 34:1). Where known, the individual FA composition is denoted in brackets separated by an underscore, e.g. PC (18:0_18:1). The underscore signifies that the stereospecific position number (sn1 or sn2) of the FA on the glycerol backbone is unknown. Furthermore, the O- or P- in the PC and PE lipid classes as well as the lysophospholid species designates the ether or vinyl-ether linkages in plasmanyl-and plasmenyl-species, e.g. PE (O-18:1_18:0).
Statistical analysis
Peak areas of each detected compound were normalised by dividing the respective peak by the aggregate median for all detected compounds (per sample). Ratios of PC/PE and SM/Cer were calculated by calculating the sum of peak areas of the respective lipid class and were not median normalised. The rationale behind calculating PC/PE and SM/Cer ratio was threefold: (1) calculating the ratio of these metabolites can reduce the bias of the normalisation technique (here median normalisation) and resemble the use of an internal standard, which can reduce the impact of biological variability on the data interpretation54; (2) calculating the ratio of two metabolites that are the substrate and the product of a biochemical reaction can provide an estimation of the rate of the reaction54 and, in our case, can provide an indication of the activity of the reaction between the selected groups; and (3) the PC/PE and SM/Cer ratios have been used as an indicator for membrane integrity39,55 and PC/PE is further used as an indicator for PEMT pathway activity in liver lipid metabolism39. The data was transformed using the natural logarithm and the mean of duplicate measurements for each cow were used for statistical analysis. Because sampling was performed across two seasons a two-way ANOVA was used for statistical analysis including the studied groups (LMFG and SMFG cows), days in milk and season as fixed effects using the ‘lm’ function combined with the ‘tidy’ function as part of the ‘broom’ package56 in R version 3.5.157. The procedure by Benjamini and Hochberg58 was used to correct false positives for multiple comparisons and only corrected P values are shown. Interactions between the fixed effects were not significant. The estimated difference between the means of the two studied groups based on data transformed using the natural logarithm, represents the fold change of SMFG cows compared to that of the reference group (LMFG cows). If multiplied by 100, the estimated difference between the groups, calculated by the linear model, can be interpreted as a percentage59. Although absolute abundance of the compounds was not measured, sums of the median-normalised peak areas of individual lipid species within a lipid class were calculated to estimate the difference in relative abundance of each lipid class. The results of this analysis were transformed using the natural logarithm and significance testing was performed using two-way ANOVA, as described for the individual lipid species, including the correction for multiple testing. Graphs were produced using the ‘ggplot2′ package60 in R.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Heid, H. W. & Keenan, T. W. Intracellular origin and secretion of milk fat globules. Eur. J. Cell Biol. 84, 245–258 (2005).
Spitsberg, V. L. Invited review: Bovine milk fat globule membrane as a potential nutraceutical. J. Dairy Sci. 88, 2289–2294 (2005).
Argov-Argaman, N. Symposium review: Milk fat globule size: Practical implications and metabolic regulation. J. Dairy Sci. 102, 2783–2795 (2019).
Michalski, M.-C. et al. Functionality of smaller vs control native milk fat globules in Emmental cheeses manufactured with adapted technologies. Food Res. Int. 40, 191–202 (2007).
Wilfling, F. et al. Triacylglycerol synthesis enzymes mediate lipid droplet growth by relocalizing from the ER to lipid droplets. Dev. Cell 24, 384–399 (2013).
Masedunskas, A., Chen, Y., Stussman, R., Weigert, R. & Mather, I. H. Kinetics of milk lipid droplet transport, growth, and secretion revealed by intravital imaging: Lipid droplet release is intermittently stimulated by oxytocin. Mol. Biol. Cell 28, 935–946 (2017).
Thiam, A. R., Farese, R. V. Jr. & Walther, T. C. The biophysics and cell biology of lipid droplets. Nat. Rev. Mol. Cell Biol. 14, 775–786 (2013).
Cohen, B.-C., Raz, C., Shamay, A. & Argov-Argaman, N. Lipid droplet fusion in mammary epithelial cells is regulated by phosphatidylethanolamine metabolism. J. Mammary Gland Biol. Neoplasia 22, 235–249 (2017).
Arisawa, K. et al. Saturated fatty acid in the phospholipid monolayer contributes to the formation of large lipid droplets. Biochem. Biophys. Res. Commun. 480, 641–647 (2016).
Dewettinck, K. et al. Nutritional and technological aspects of milk fat globule membrane material. Int. Dairy J. 18, 436–457 (2008).
Carroll, S. M. et al. Milk composition of Holstein, Jersey, and Brown Swiss cows in response to increasing levels of dietary fat. Anim. Feed Sci. Technol. 131, 451–473 (2006).
El-Zeini, H. M. Microstructure, rheological and geometrical properties of fat globules of milk from different animal species. Pol. J. Food Nutr. Sci. 56, 147–153 (2006).
Logan, A., Auldist, M., Greenwood, J. & Day, L. Natural variation of bovine milk fat globule size within a herd. J. Dairy Sci. 97, 4072–4082 (2014).
Couvreur, S., Hurtaud, C., Marnet, P. G., Faverdin, P. & Peyraud, J. L. Composition of milk fat from cows selected for milk fat globule size and offered either fresh pasture or a corn silage-based diet. J. Dairy Sci. 90, 392–403 (2007).
Michalski, M.-C., Briard, V. & Michel, F. Optical parameters of milk fat globules for laser light scattering measurements. Le Lait 81, 787–796 (2001).
Fleming, A. et al. Variation in fat globule size in bovine milk and its prediction using mid-infrared spectroscopy. J. Dairy Sci. 100, 1640–1649 (2017).
Wiking, L., Stagsted, J., Björck, L. & Nielsen, J. H. Milk fat globule size is affected by fat production in dairy cows. Int. Dairy J. 14, 909–913 (2004).
Couvreur, S. & Hurtaud, C. Relationships between milks differentiated on native milk fat globule characteristics and fat, protein and calcium compositions. Animal 11, 507–518 (2017).
Altenhofer, C. et al. Temporal variation of milk fat globule diameter, fat and cholesterol content and milk epithelial cell gene expression in dairy cows. Int. J. Dairy Technol. 68, 519–526 (2015).
Walter, L. et al. The effect of physiological state, milk production traits and environmental conditions on milk fat globule size in cow’s milk. J. Dairy Res. 86, 454–460 (2019).
Walter, L., Shrestha, P., Fry, R., Leury, B. & Logan, A. Lipid metabolic differences in cows producing small or large milk fat globules: Fatty acid origin and the role of membrane proteins. J. Dairy Sci. 103, 1920–1930 (2020).
Lopez, C. et al. Fat globules selected from whole milk according to their size: Different compositions and structure of the biomembrane, revealing sphingomyelin-rich domains. Food Chem. 125, 355–368 (2011).
Lu, J. et al. The protein and lipid composition of the membrane of milk fat globules depends on their size. J. Dairy Sci. 99, 4726–4738 (2016).
Mesilati-Stahy, R., Mida, K. & Argov-Argaman, N. Size-dependent lipid content of bovine milk fat globule and membrane phospholipids. J. Agric. Food Chem. 59, 7427–7435 (2011).
Li, Q. et al. Lipidomics profiling of goat milk, soymilk and bovine milk by UPLC-Q-exactive orbitrap mass spectrometry. Food Chem. 224, 302–309 (2017).
Liu, Z., Rochfort, S. & Cocks, B. Milk lipidomics: What we know and what we don’t. Prog. Lipid Res. 71, 70–85 (2018).
Huynh, K. et al. Lipidomic profiling of murine macrophages treated with fatty acids of varying chain length and saturation status. Metabolites 8, 29 (2018).
Huynh, K. et al. High-throughput plasma lipidomics: Detailed mapping of the associations with cardiometabolic risk factors. Cell Chem. Biol. 26, 71-84.e4 (2019).
Sokol, E., Ulven, T., Færgeman, N. J. & Ejsing, C. S. Comprehensive and quantitative profiling of lipid species in human milk, cow milk and a phospholipid-enriched milk formula by GC and MS/MSALL. Eur. J. Lipid Sci. Technol. 117, 751–759 (2015).
Glaser, P. E. Plasmenylethanolamine facilitates rapid membrane fusion: A stopped-flow kinetic investigation correlating the propensity of a major plasma membrane constituent to adopt an hii phase with its ability to promote membrane fusion. Biochemistry 33, 5805–5812 (1994).
Dean, J. M. & Lodhi, I. J. Structural and functional roles of ether lipids. Protein Cell 9, 196–206 (2018).
Wallner, S. & Schmitz, G. Plasmalogens the neglected regulatory and scavenging lipid species. Chem. Phys. Lipids 164, 573–589 (2011).
Nagan, N. & Zoeller, R. A. Plasmalogens: biosynthesis and functions. Prog. Lipid Res. 40, 199–229 (2001).
Rahmatyar, Z. & Wiking, L. Fatty acid composition and thermal behavior of small and large milk fat globules. Milchwissenschaft 67, 34–38 (2012).
Brites, P. et al. Alkyl-glycerol rescues plasmalogen levels and pathology of ether-phospholipid deficient mice. PLoS ONE 6, e28539 (2011).
Guo, Y. et al. Functional genomic screen reveals genes involved in lipid-droplet formation and utilization. Nature 453, 657–661 (2008).
Fauquant, C., Briard-Bion, V., Leconte, N., Guichardant, M. & Michalski, M.-C. Membrane phospholipids and sterols in microfiltered milk fat globules. Eur. J. Lipid Sci. Technol. 109, 1167–1173 (2007).
McManaman, J. L. Lipid transport in the lactating mammary gland. J. Mammary Gland Biol. Neoplasia 19, 35–42 (2014).
Li, Z. et al. The ratio of phosphatidylcholine to phosphatidylethanolamine influences membrane integrity and steatohepatitis. Cell Metab. 3, 321–331 (2006).
Hörl, G. et al. Sequential synthesis and methylation of phosphatidylethanolamine promote lipid droplet biosynthesis and stability in tissue culture and in vivo. J. Biol. Chem. 286, 17338–17350 (2011).
DeLong, C. J., Shen, Y.-J., Thomas, M. J. & Cui, Z. Molecular distinction of phosphatidylcholine synthesis between the CDP-choline pathway and phosphatidylethanolamine methylation pathway. J. Biol. Chem. 274, 29683–29688 (1999).
Lopez, C. et al. Phospholipid, sphingolipid, and fatty acid compositions of the milk fat globule membrane are modified by diet. J. Agric. Food Chem. 56, 5226–5236 (2008).
Honsho, M., Asaoku, S. & Fujiki, Y. Posttranslational regulation of fatty acyl-CoA reductase 1, far1, controls ether glycerophospholipid synthesis. J. Biol. Chem. 285, 8537–8542 (2010).
Argov-Argaman, N., Mida, K., Cohen, B.-C., Visker, M. & Hettinga, K. Milk fat content and DGAT1 genotype determine lipid composition of the milk fat globule membrane. PLoS ONE 8, e68707 (2013).
Grisart, B. et al. Positional candidate cloning of a QTL in dairy cattle: Identification of a missense mutation in the bovine DGAT1 gene with major effect on milk yield and composition. Genome Res. 12, 222–231 (2002).
Schennink, A. et al. DGAT1 underlies large genetic variation in milk-fat composition of dairy cows. Anim. Genet. 38, 467–473 (2007).
Grisart, B. et al. Genetic and functional confirmation of the causality of the DGAT1 K232A quantitative trait nucleotide in affecting milk yield and composition. Proc. Natl. Acad. Sci. 101, 2398–2403 (2004).
Kasumov, T. et al. Improved insulin sensitivity after exercise training is linked to reduced plasma C14:0 ceramide in obesity and type 2 diabetes. Obesity (Silver Spring) 23, 1414–1421 (2015).
Rico, J. E., Bandaru, V. V. R., Dorskind, J. M., Haughey, N. J. & McFadden, J. W. Plasma ceramides are elevated in overweight Holstein dairy cows experiencing greater lipolysis and insulin resistance during the transition from late pregnancy to early lactation. J. Dairy Sci. 98, 7757–7770 (2015).
Deevska, G. M. & Nikolova-Karakashian, M. N. The expanding role of sphingolipids in lipid droplet biogenesis. Biochim Biophys. Acta BBA Mol. Cell Biol. Lipids 1862, 1155–1165 (2017).
Deevska, G. M. et al. Novel interconnections in lipid metabolism revealed by overexpression of sphingomyelin synthase-1. J. Biol. Chem. 292, 5110–5122 (2017).
Folch, J., Lees, M. & Stanley, G. H. S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 226, 497–509 (1957).
Liebisch, G. et al. Shorthand notation for lipid structures derived from mass spectrometry. J. Lipid Res. 54, 1523–1530 (2013).
Petersen, A.-K. et al. On the hypothesis-free testing of metabolite ratios in genome-wide and metabolome-wide association studies. BMC Bioinform. 13, 120 (2012).
Pullmannová, P. et al. Effects of sphingomyelin/ceramide ratio on the permeability and microstructure of model stratum corneum lipid membranes. Biochim. Biophys. Acta BBA Biomembr. 1838, 2115–2126 (2014).
Robinson, D. & Hayes, A. broom: Convert statistical analysis objects into tidy tibbles (2018).
Core Team, R. R: A language and environment for statistical computing (R Foundation for Statistical Computing, Vienna, 2018).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B Methodol. 57, 289–300 (1995).
Cole, T. J. Sympercents: symmetric percentage differences on the 100 log(e) scale simplify the presentation of log transformed data. Stat. Med. 19, 3109–3125 (2000).
Wickham, H. ggplot2: Elegant graphics for data analysis (Springer, New York, 2016).
Fagone, P. & Jackowski, S. Membrane phospholipid synthesis and endoplasmic reticulum function. J. Lipid Res. 50, S311–S316 (2009).
Acknowledgements
We would like to thank Dr Sue Finch from the Melbourne Statistical Consulting platform for her assistance with the statistical analysis as well as Kenny Oluboyede and Ashley Gabler for their help with sample collection at the University of Melbourne Dairy.
Author information
Authors and Affiliations
Contributions
LW collected samples, performed the data analysis, statistical analysis and wrote the manuscript. VKN performed LC–MS/MS analysis, sample preparations and contributed to data analysis and interpretation. DT and VKN developed and validated the method. DT, VKN, LW, RF, AL and BL designed the study and reviewed the manuscript. AL, RF and BL supervised the study and acquired the funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Walter, L., Narayana, V.K., Fry, R. et al. Milk fat globule size development in the mammary epithelial cell: a potential role for ether phosphatidylethanolamine. Sci Rep 10, 12299 (2020). https://doi.org/10.1038/s41598-020-69036-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-69036-5
- Springer Nature Limited