Abstract
Previous studies have shown that rabbit IgG antibodies against Schistosoma mansoni egg antigens (SmSEA) cross-react with allergens in natural rubber latex, peanuts and grass and tree pollens. Here we describe antigenic molecules that cross-react with rabbit anti-S. mansoni IgG antibodies in extracts of the house dust mite (HDM) Dermatophagoides farinae, the Australian cockroach (ACR) Periplaneta australasiae and in the venom of the honey bee Apis mellifera (HBV). Tandem mass spectrometry identified the cross-reactive allergens as Der f 15 in HDM, two homologues of the Periplaneta americana cockroach allergen Cr-PI/Per a 3 in ACR and two isoforms of the allergen Api m 1 (phospholipase A2: PLA2) in HBV. Cross-reactive rabbit anti-SmSEA IgG antibodies eluted from the three invertebrate allergens reacted with S. mansoni egg antigens and variably with schistosome cercarial and worm antigens. Treatment of the electroblotted allergens with sodium metaperiodate abrogated most of the cross-reactivity of the rabbit anti-SmSEA antibodies, suggesting it was due to cross-reactive carbohydrate determinants (CCDs). Furthermore, analyses of the allergens’ amino acid sequences indicated that they had potential for both N- and O-linked glycosylation. A potential role for the CCDs shared by the schistosome and invertebrates in inducing an allergy-protective effect, as proposed by the hygiene hypothesis, is discussed.
Similar content being viewed by others
Introduction
In recent decades there has been a steep rise in the occurrence of allergic diseases, particularly in countries with well-developed health systems1,2. The ‘hygiene hypothesis’ is often offered as an explanation, i.e., due to an increase in healthy living conditions and/or prevention of infection by vaccinations and drugs, populations have suffered less from parasitic and microbial infections3 and development of human immune systems is now different from those of pre-hygienic, infection-rife eras4. A consequence can be an untimely, pathological immune response to environmental and air-borne antigens responsible for asthma and allergies, and which ‘normal’ human immune responsiveness would have perceived as innocuous.
In helminth-endemic countries in particular an inverse correlation has been observed between infection with helminths, including schistosomes, and allergic sensitization5,6,7 for which situation the hygiene hypothesis has been evoked as an explanation8.
Immune responses that cause allergic reactions are so-called Th2-type responses9,10,11 and Th2 cell-driven immunity is also induced by helminth infections12,13,14. Th2-biased immune responsiveness is however downregulated during chronic helminth infection in human hosts and this in turn has a suppressive effect on allergic sensitization in such individuals15,16,17. How this occurs has not been fully elucidated, though one explanation invokes activation of regulatory T cells (Tregs) and production of IL-10 and TGF-β, anti-inflammatory cytokines which have a dampening effect on atopic diseases such as allergy and asthma18,19,20.
‘Blocking antibodies’ may provide an alternative explanation for the hygiene hypothesis. Helminth infections induce the production of large amounts of non-specific IgE21, which could saturate Fcε receptors on mast cells and basophils and ‘block’ the cross-linking of such receptors by allergen-induced specific IgE antibody. There is, however, evidence against this possibility22,23,24. Nevertheless, the results presented here are consistent with an alternative possible ‘blocking antibody’ explanation since many proteins of plants and invertebrates (including helminths) are glycoproteins, the glycan epitopes of which can induce IgE antibodies25,26,27, some of which are known to be antigenically cross-reactive: so called cross-reactive carbohydrate determinants (CCDs)28. Glycan epitopes can also induce production of IgG antibodies29. The possibility that anti-S. mansoni IgG antibodies that cross-react with allergens could therefore block IgE-mediated allergic reactions is discussed as an explanation for the hygiene hypothesis.
We have so far demonstrated antigenic cross-reactivity, most likely due to CCDs, between S. mansoni and identified allergens in several different plants30,31,32. Those studies have been extended here to explore cross-reactivity between S. mansoni and allergenic invertebrates: namely, the house dust mite33, the cockroach34 and honey bee venom35.
Material and Methods
Except when stated otherwise, all chemicals and reagents were of analytical grade and purchased from Sigma-Aldrich (now Merck), Poole, Dorset, England.
Ethical approval
Experiments using mice for production of S. mansoni antigens and rabbits for production of antisera were approved by the Animal Welfare and Ethical Review Board of the University of Nottingham in which these materials were produced and the work was carried out in accordance with the regulations set out in the UK Animals (Scientific Procedures) Act, 1986, (Project licence numbers PPL 40/3024 and 40/3595). Animals were euthanized using a lethal dose of pentobarbitone anaesthetic.
Preparation of soluble extracts of SmSEA
A Puerto-Rican isolate of S. mansoni was maintained by continuous passaging through the snail Biomphalaria glabrata and random-bred, CD1 strain mice.
S. mansoni soluble egg antigens (SmSEA) were extracted from eggs from the livers and intestines of infected mice harbouring adult worms as previously described36. The soluble extract was lyophilised and stored in 1 mg aliquots at −80 °C until use. It was used for immunoblotting at a concentration of 5 mg/ml protein. Extracts from S. mansoni worms and cercariae were prepared as described previously37.
Preparation of allergen extracts
Powder of the house dust mite Dermatophagoides farinae (HDM) was incorporated (50 mg/ml) in phosphate-buffered saline (PBS), pH 7.4. The suspension was agitated gently for 30 minutes and centrifuged at 10,000 × g for 10 minutes, all at room temperature in a microcentrifuge. The supernatant containing 4.4 mg/ml protein was collected and kept in 500 µl aliquots at −20 °C in the short term (2 weeks) and at −80 °C in the longer term and they were used within 4–8 weeks of preparation (i.e., solution or suspension in buffer), after which fresh extracts were prepared for any subsequent experiments. Adults of a local infestation of cockroaches, identified as Periplaneta australasiae (ACR), were macerated in a mortar and pestle, suspended in an equal volume of PBS, agitated gently for 30 minutes at room temperature, sonicated on ice with 3 × 10 second pulses at 5,000 Hz separated by 10 second breaks and the suspension, in a 50 ml centrifuge tube, was centrifuged for 10 minutes at 4000 × g at room temperature utilising a swing-bucket centrifuge rotor. The supernatant containing 20 mg/ml protein was stored similarly to that of HDM extract. For production of bee venom (HBV), worker honey bees were anaesthetised with carbon dioxide gas and decapitated. The sting and venom sac of ~100 bees were gently pulled from the abdomen and placed in 1 ml ice-cooled distilled water, macerated with a plastic pipette tip, dispensed in 500 μl aliquots and centrifuged at 10,000 × g for 10 minutes. The supernatant contained 2.5 mg/ml protein and was stored as for other extracts.
Aqueous extracts of rubber latex (Hevea brasiliensis) (10.5 mg/ml), peanut (Arachis hypogaea) (33.4 mg/ml), tomato (Lycopersicon esculatum) (18.8 mg/ml), avocado (Persea americana) (22 mg/ml) and kiwi fruit (Actinidia deliciosa) (9.4 mg/ml) were prepared as described previously30. Estimates of protein concentrations in SmSEA and other extracts were done using the adapted38 Bio-Rad DC protein assay method (Bio-Rad Laboratories, Watford, UK), with bovine serum albumin (BSA) as standard.
Preparation of rabbit antisera
Rabbits were laboratory-maintained under strict monitoring for health and hygiene and during the course of these studies there was no record of any occurrences of HDM, cockroach infestations or bees in the laboratories. Polyspecific antisera were raised against SmSEA by weekly immunization of New Zealand white rabbits as described previously39. Thus, a soluble extract of SmSEA prepared as described above containing approximately 5 mg/ml SEA was emulsified in an equal volume of complete Freund’s adjuvant and 1 ml of the emulsified homogenate was administered to rabbits in 0.1 ml quantities both intramuscularly (in both hind legs) and subcutaneously at multiple dorsal sites. The rabbits were serially bled via an ear vein weekly and injections were continued until a strong antibody response was obtained against the SEA immunogen in immunoelectrophoresis. Rabbits were terminally anaesthetised and exsanguinated by cardiac puncture and sera were collected and stored at −20 °C. Sera from 2 immunized rabbits, BR84 and 1025Z, were individually used here for experiments on the house dust mite and on the cockroach and honey bee venom, respectively.
Honey bee venom phospholipase A2 was purchased from Sigma-Aldrich (now Merck), UK, and a polyclonal rabbit antiserum raised by injecting 50 µg that had been dissolved in 1 ml PBS and emulsified with 1 ml Freund’s adjuvant in several subcutaneous sites. The injections were repeated weekly for 5 weeks, after which the rabbit was exsanguinated and the serum aliquoted and stored at −20 °C until required.
Sera from rabbits injected with Freund’s adjuvant alone, developed as scheduled above, were used as control normal rabbit sera (NRS).
One-dimensional sodium dodecyl polyacrylamide gel electrophoresis (SDS-PAGE) and western immunoblotting
SDS-PAGE methodology was adapted40 and performed as described41 using 12% or 8% polyacrylamide gels. SDS-PAGE gels were run using a Bio-Rad Mini Protean II electrophoresis system (Bio-Rad Laboratories, California, USA). Western immunoblotting was adapted and performed as described previously30,42. SDS-PAGE-resolved proteins were transferred to nitrocellulose membranes (NCM) and probed with rabbit antisera diluted 1:100 in Tris-buffered saline with 0.5% v/v Tween 20 (TBST) overnight at 4 °C as the primary antibody, followed by 2 hours at room temperature in a solution of horse radish peroxidise (HRP)-conjugated goat anti-rabbit IgG antibodies (Sigma Aldrich, now Merck, UK) diluted 1:1000 in TBST as the secondary antibody. The immunoblots were developed using 4-chloro-1-naphthol substrate (Sigma Aldrich, now Merck, UK) as described by the manufacturer. At least three replicates of SDS-PAGE gels and immunoblots have been done for cross-reactive experiments in the present work.
Staining and purification of electrophoresed proteins in SDS-PAGE
SDS-PAGE gels containing electrophoresed proteins were stained with Coomassie blue for protein visualization using SimplyBlue SafeStain (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. Partial purification of proteins was achieved by excision of the bands of interest (identified by matching with western blots displaying the immunoreactivities of interest) from a stained gel, followed by overnight elution in 10% SDS, 0.06 M Tris-HCl, pH 7.0, buffer at 37 °C30,31. Solutions containing eluted proteins were concentrated using Amicon ultra centrifugal filters, 3000 molecular weight cut-off (Millipore, Corrigtwohill, Co. Cork, Ireland) and re-electrophoresed in a second SDS-PAGE as recently described31,43. Purified, concentrated proteins were stored at −20 °C.
Purification of cross-reactive anti-SmSEA IgG antibodies by acid-elution
Anti-SmSEA antibodies that were cross-reactive with molecules from the allergen extracts were purified by acid-elution using an adapted method44 modified as described30 (Supplementary Fig. S1). Protein extracts were individually loaded into a single, wide lane in a SDS-PAGE gel and electro-transferred to a nitrocellulose membrane. The membrane was blocked and incubated with the primary rabbit antiserum as described above. 1 cm parallel longitudinal nitrocellulose paper strips were cut from each edge of the immunoblot, washed and incubated with horse-radish peroxidase-conjugated goat anti-rabbit IgG antibodies and chromogenically stained. The position of the target immune-complex in the undeveloped main part of the immunoblot was determined by realigning the two stained strips against the sides of the membrane. A horizontal strip containing the immune-complex of interest was then cut from the blot and the antibodies were eluted therefrom in 1 ml of 0.1 M glycine buffer, pH 2.8, gently agitated at room temperature for 10 minutes. The eluting buffer was collected and neutralised using 1 M Tris, pH 8.0, and stored at −20 °C. The strip was washed three times in PBS, each for 5 minutes with gentle agitation. The process of incubating the strip with primary antibody, washing and antibody-elution using low pH buffer was repeated up to 4 times.
Treatment of proteins electroblotted onto NCM with sodium metaperiodate
The technique was adapted30 and performed as described earlier45,46. Briefly, nitrocellulose membranes carrying electroblotted antigens were incubated for 1 hour in 10 mM sodium metaperiodate dissolved in 0.05 M sodium acetate buffer, pH 4.5, in the dark at room temperature. Controls were treated in 0.05 M sodium acetate buffer, pH 4.5 under similar conditions, but without the metaperiodate. The membranes were washed three times in TBST and the process for western immunoblotting was then continued with blocking, incubation with primary and secondary antibodies and development methods as described above.
Tandem mass spectrometry (TMS) analysis of protein samples and data analysis
Protein bands excised from Coomassie blue-stained gels were subjected to nanoflow Liquid Chromatography Electrospray Ionization TMS (nLC-ESI MS/MS)47 as described elsewhere48. The analysis was done by the BSRC Mass Spectrometry and Proteomics Facility, University of St Andrews, UK. The MS/MS data were analysed using the Mascot algorithm (Matrix Science), against the NCBInr database (August 2016). A protein was accepted as identified if it had two or more peptides with Mascot Ion Scores above the Identity Threshold (P < 0.05). TMS-identified proteins were further analysed using the BLAST Tool (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and the pairwise sequence alignment tool (http://www.ebi.ac.uk/Tools/psa/). The prediction of potential glycosylation sites on TMS-identified allergens was done using the CBS Software Prediction Servers NetNGlyc and NetOGlyc (http://www.cbs.dtu.dk/services/NetNGlyc/ & http://www.cbs.dtu.dk/services/NetOGlyc/).
Results
Resolution of HDM, ACR and HBV invertebrate extracts by SDS-PAGE and reactivity of rabbit anti-SmSEA IgG antibodies thereon
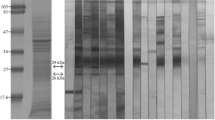
Figure 1 shows a composite of results of electrophoretic resolution of material from 3 allergenic invertebrates by one-dimensional SDS-PAGE, followed by staining with Coomassie blue or electroblotting on NCM and probing with a rabbit anti-SmSEA antiserum. Figure 1a comprises the results on the HDM extract electrophoresed in a 12% polyacrylamide gel. Lane 1 in the Coomassie blue-stained gel shows a protein band of molecular size >90 kDa which appears to be the sole reactant with IgG antibodies in rabbit BR84 anti-SmSEA antiserum in the immunoblot (Fig. 1a, lane 2). Normal rabbit serum gave no reactivity against HDM (Fig. 1a, lane 3). A sample of the >90 kDa band in the Coomassie-stained gel (indicated by a red arrow against lane 1, Fig. 1a) was submitted for TMS analysis.
Coomassie blue-stained SDS-PAGE and western immunoblots of crude invertebrate extracts, and purified proteins from house dust mite (D. farinae), Australian cockroach (P. australasiae) and honey bee (A. mellifera) venom, probed with anti-S. mansoni SEA antisera. (a) D. farinae extract (44 µg/lane): lane 1, Coomassie blue-stained gel; lanes 2,3, Western immunoblots; (bi) P. australasiae extract (200 µg/lane): lane 1, Coomassie blue-stained gel; lanes 2,3 immunoblots; (bii), P. australasiae purified proteins (10.6 µg/lane): Coomassie blue-stained purified P. australasiae protein at ~85 kDa (lane 4) and ~88 kDa (lane 5); immunoblot of purified cockroach proteins of ~85 kDa (lane 6) and ~88 kDa (lane 7). (c) Honey bee venom (25 µg/lane): lane 1, Coomassie blue-stained gel; lanes 2,3, Western immunoblots. 12% SDS PAGE gels were used in panels (a,c) and 8% SDS PAGE gels in panels (bi) and (bii). Blots were probed with anti-S. mansoni SEA antiserum BR84 on lane 2 in (a); anti-SmSEA antiserum 1025Z on lane 2 in (bi), on lanes 6 and 7 in (bii) and on lane 2 in (c), or normal rabbit serum on lanes 3 in (a), (bi) and (c). Cross-reactive protein bands at >90 kDa (a), ~85 kDa & ~88 kDa (bi and bii) and ~17 kDa & 19 kDa (c) that were selected for TMS are arrowed. M = protein molecular size marker. Supplementary Fig. S5 shows uncropped blot images for lane 2 (bi) and lanes 4–7 (bii).
Fig. 1bi, lane 1 shows the ACR extract in a Coomassie blue-stained 8% polyacrylamide gel. Lane 2 in Fig. 1bi is an immunoblot of the electrophoresed ACR probed with anti-SmSEA 1025Z IgG antibodies. At least four molecules between 46 kDa and 100 kDa were cross-reactive with the anti-SmSEA antibodies (Fig. 1bi, lane 2). No reactivity against ACR was observed with normal rabbit serum (Fig. 1bi, lane 3). Gel slices containing two molecules estimated to be 85 kDa to 88 kDa were excised from a replicate of the gel in lane 1 and the proteins were eluted from the respective gel slices by overnight incubation in buffer. The eluted proteins were re-electrophoresed for Coomassie blue-staining (Fig. 1bii, lanes 4 and 5). After immunoblotting the purified molecules each reacted with IgG antibodies in the rabbit anti-SmSEA 1025Z antiserum (Fig. 1bii, lanes 6 and 7) and samples of each excised from the acrylamide gels were therefore subjected to TMS analysis.
In HBV several proteins with molecular sizes ranging from <20 kDa to >100 kDa reacted with the rabbit anti-SmSEA 1025Z serum (Fig. 1c). Two of these molecules of ~17 kDa and ~19 kDa were purified and found to react with a rabbit antiserum raised against HBV PLA2 (Supplementary Fig. S2). These two molecules were taken forward for analysis by TMS.
Characterization of schistosome cross-reactive invertebrate molecules by tandem mass spectrometry (TMS)
Molecules in the invertebrate extracts that cross-reacted with anti-SmSEA antibodies were selected for further analysis by mass spectrometry, the selected proteins having been excised from well-washed Coomassie blue-stained gels. The results of TMS on the sole cross-reactive >90 kDa protein in the HDM extract (Fig. 1a) are summarized in Supplementary Table S1 and they indicate that a known HDM allergen, Der f 15 (gi: 5815436), was present. The TMS did not detect any peptides other than those from Der f 15.
The results of TMS analysis of two purified ACR proteins of estimated size ~85 kDa and ~88 kDa (Fig. 1bii, lanes 4 and 5) are given in Supplementary Tables S2 and S3 and they indicate both protein bands contained peptides present in the sequence of protein Cr-PI, allergen Per a 3 (gi: 284518363) from Periplaneta americana.
TMS analysis of the ~17 kDa and ~19 kDa HBV molecules (Fig. 1c) confirmed that both protein bands contained HBV phospholipase A2 (gi: 5627, GenBank: CAA34681.1), also known as the allergen Api m 1 (Supplementary Tables S4 and S5).
Reactivity and periodate-sensitivity of acid-eluted antibodies
Supplementary Fig. S1 illustrates how rabbit anti-SmSEA BR84 antibodies that were cross-reactive with the >90 kDa HDM molecule were purified by acid-elution from electroblotted HDM extract. This method was also used to purify anti-SmSEA IgG antibodies that cross-reacted with cockroach and bee venom antigens. Eluted antibodies were obtained at concentrations that ranged from 1 to 1.3 mg/ml. The acid-eluted antibodies were used to probe electroblots of extracts from HDM and from different stages of the S. mansoni life-cycle and the results are shown in Fig. 2a.
Western blot of D. farinae and S. mansoni extracts probed with rabbit BR84 IgG antibodies eluted from a >90 kDa D. farinae molecule. (a) Eluted antibodies were used to probe: 1, D. farinae extract (44 µg/lane); 2, SmSEA (20 µg/lane); 3, S. mansoni cercarial homogenate (20 µg/lane); 4, S. mansoni worm homogenate (20 µg/lane). (b) The same as (a), but treated with 10 mM Na-metaperiodate for one hour prior to application of primary antibody. M = protein molecular size marker.
Reactivity of the eluted antibodies against HDM was relatively weak, including against the >90 kDa molecule itself (Fig. 2a, lane 1). There was however intense reactivity against the constituents of SmSEA over a wide range of molecular size (Fig. 2a, lane 2), though only relatively low reactivity against S. mansoni cercarial or adult worm antigens (Fig. 2a, lanes 3 and 4). No reactivity was observed on any of the parasite extracts with a normal rabbit serum (Supplementary Fig. S3).
Figure 2b shows the result of treatment of a replicate NCM film carrying the same electroblotted antigens as in Fig. 2a, with 10 mM Na-metaperiodate for 1 hour before incubating the film with the anti-HDM eluted antibodies. There is no antibody reactivity against the HDM extract or the S. mansoni cercarial and worm extracts, and the intensity of reactivity against the SmSEA is much reduced. Supplementary Fig. S4 illustrates SDS-PAGE gels of resolved proteins in the three stages of S. mansoni life-cycle extracts used in blots in Fig. 2.
Results in Fig. 3a show the reactivity of BR84 antibodies that had been acid-eluted from the >90 kDa HDM against the HDM extract and against extracts from a variety of plants. The reactivity against HDM was relatively weak as in Fig. 2a, lane 1, but there was reactivity against a range of antigens in the different plant extracts, with relatively intense reactivity against peanut and avocado. Treatment of the electroblotted extracts with 10 mM Na-metaperiodate prior to incubation with the acid-eluted rabbit antibodies resulted in complete abrogation of the antibody cross-reactivity against all the extracts (Fig. 3b). No reactivity was observed on any of the plants with a normal rabbit serum (Supplementary Fig. S3).
Western blots of extracts of allergenic plants probed with rabbit BR84 anti-SmSEA antibodies eluted from a >90 kDa D. farinae molecule. (a) Eluted antibodies were used to probe: 1, D. farinae (44 µg/lane); 2, latex (157.5 µg/lane); 3, tomato (188 µg/lane); 4, peanut (167 µg/lane); 5, avocado (220 µg/lane); 6, kiwi fruit (94 µg/lane). (b) The same as (a), but after treatment with 10 mM Na-metaperiodate for one hour prior to application of primary antibody. M = protein molecular size marker.
Figure 4a shows the reactivity of 1025Z antibodies eluted from both the ~85 kDa and the ~88 kDa ACR molecules against a variety of antigens in extracts from different life cycle stages of S. mansoni. (Elution from both molecules together was justified since the TMS results indicated that both were orthologues of the cockroach allergen Per a 3). The eluted anti-85/88 kDa ACR antibodies reacted relatively intensely against those two molecules in the ACR extract (Fig. 4a, lane 1). Likewise, reactivity of anti-85/88 kDa ACR antibodies against SmSEA was also relatively intense (Fig. 4a, lane 2), as well as against S. mansoni worm and cercarial molecules (Fig. 4a, lanes 3 and 4 respectively), all of which showed more intense staining than that of the eluted anti-HDM >90 kDa antibodies in Figs. 2, 3. After Na-metaperiodate treatment neither the 85 kDa nor 88 kDa ACR molecules reacted with the acid-eluted anti-ACR 85/88 kDa antibodies (Fig. 4b, lane 1). Cross-reactivity of these acid-eluted antibodies against adult schistosome worm and cercarial antibodies was also almost completely abrogated (Fig. 4b, lanes 3 and 4). In periodate-treated SmSEA only a broad band between the 32 and 46 kDa markers remained reactive with these antibodies (Fig. 4b, lane 2).
Western immunoblots of P. australasiae and S. mansoni extracts probed with rabbit 1025Z anti-SmSEA IgG antibodies eluted from ~85 kDa and ~88 kDa P. australasiae molecules. (a) Eluted antibodies were used to probe: 1, P. australasiae extract (200 µg/lane); 2, S. mansoni SEA (20 µg/lane); 3, S. mansoni worm homogenate (20 µg/lane); 4, S. mansoni cercariae homogenate (20 µg/lane). (b) The same as (a) but after treatment with 10 mM Na-metaperiodate for one hour. M = protein molecular size marker. Supplementary Figs. S6 and S7 show uncropped blot images for lanes 1–4 of panels (a,b).
Figure 5 shows the results of probing different extracts with 1025Z antibodies eluted from the ~17 kDa and ~19 kDa HBV molecules. As with the antibodies eluted from the two ACR molecules, the two HBV molecules were not treated separately here because they were shown by TMS to be isoforms of PLA2/Api m 1. The eluted antibodies reacted against a variety of molecules in SmSEA and S. mansoni worm and cercarial antigens (Fig. 5a, lanes 1, 2 and 3 respectively) and against extracts of latex and peanut (Fig. 5a, lanes 4 and 5 respectively). In SmSEA the most intense reactivity was against a ~100 kDa molecule. Treatment of the NCM with 10 mM Na-metaperiodate prior to application of the acid-eluted primary antibodies resulted in abrogation of all the cross-reactivity observed in Fig. 5a, except for a residual faint smudge of reactivity between 32 kDa and 46 kDa in SmSEA.
Western immunoblots of extracts of S. mansoni and allergenic plants probed with rabbit 1025Z IgG antibodies eluted from ~17–19 kDa bee venom molecules. (a) Eluted antibodies were used to probe: 1, S. mansoni SEA (20 µg/lane); 2, S. mansoni worm homogenate (20 µg/lane); 3, S. mansoni cercariae homogenate (20 µg/lane); 4, latex (157.5 µg/lane); 5, peanut (167 µg/lane). (b) The same as (a) but after treatment with 10 mM Na- metaperiodate for an hour before incubation with primary antibody. M = protein molecular size marker. Supplementary Fig. S8 shows uncropped blot images for lanes 1–5 (a).
Identification of potential glycosylation sites in amino-acid sequences of invertebrate allergens
Bioinformatic analysis of the potential N-linked and O-linked glycosylation sites for the 3 invertebrate allergenic molecules are indicated in Supplementary Tables S6 to S10. HDM Der f 15 has three N-linked sites (Supplementary Table S6) and 54 O-linked sites (Supplementary Table S7). ACR Per a 3 has two N-linked sites (Supplementary Table S8) and no O-linked sites. HBV Api m 1 has one N-linked site (Supplementary Table S9) and three O-linked sites (Supplementary Table S10).
Discussion
Previous studies investigating antigenic cross-reactivity between S. mansoni and allergens from different organisms have demonstrated cross-reactivity between the helminth and the allergens Hev b 7 from natural rubber latex Hevea brasiliensis30, Ara h 1 from the peanut Arachis hypogaea31 and a total of five distinct allergenic molecules in plant pollens32. The aforementioned studies have now been extended to exploration of cross-reactive antigenic relationships between S. mansoni and extracts from three invertebrates which are important causes of allergic reactions, particularly asthma and hypersensitive skin reactions.
Hypersensitive pathological immune responses against the constituents of such invertebrates constitute serious world-wide health problems. Thus, as stated by Calderon et al.33, ‘The house dust mite (HDM) is a major perennial allergen source and a significant cause of allergic rhinitis and allergic asthma. Prevalence data for HDM allergen sensitization vary from 65 to 130 million persons in the general population worldwide to as many as 50% among asthmatic patients’. Likewise cockroaches, established as an important cause of allergies over 50 years ago49, represent one of the most common sources of indoor allergens worldwide, with 40–60% of patients with asthma in urban and inner-city areas possessing IgE antibodies to cockroach allergens34,50. Estimates of the prevalence of systemic (anaphylactic) reactions to hymenoptera stings in adult Americans range from 0.5% to 3.3%51 while European epidemiological studies report a prevalence of systemic reactions between 0.3% and 7.5%52.
Der f 15, a >90 kDa HDM molecule found here to be the sole reactant with anti-SmSEA antibodies, is a chitinase. Together with the chitinase-like Der f 18, Der f 15 is considered a minor allergen for humans53,54, but a major allergen for dogs55. Most dogs with HDM-associated atopic dermatitis had IgE antibodies to two proteins of apparent molecular weight 98 kDa and 109 kDa55. The two proteins had identical amino acid sequences which predicted a protein of 63.2 kDa55 and which TMS analysis for the >90 kDa HDM protein studied here gave as 63.8 kDa (Supplementary Table S1). Rabbit anti-SmSEA IgG antibodies here reacted against only one molecule between 93 kDa and 100 kDa, presumably the smaller of the two molecules identified as Der f 15 by McCall et al.55,. The 30–40 kDa difference between the size predicted by the 555 amino acid-long sequence and that indicated in their immunoblots was attributed to extensive glycosylation55. Our analysis of potential sites for glycosylation indicates that Der f 15 may be glycosylated via numerous O-linked sites, particularly in a threonine/proline-rich section of its sequence (amino acids 415–498) found towards the C-terminal end of the molecule and three N-linked sites, though only one N-linked site was predicted in the analysis by McCall et al.55.
Alignment of linear amino-acid sequences of Der f 15 with those of three SmSEA molecules which have also previously been implicated in antigenic cross-reactivity with other allergen molecules30,31,32 shows only 10–15% identity (data available on request). The reactivity of the eluted antibodies against the HDM extract and schistosome egg antigens was abrogated by periodate-treatment, except for some reactivity that remained in the SmSEA. Thus, the cross-reactivity of anti-SmSEA antibodies with Der f 15 seems likely to be due to recognition of glycan rather than peptide epitopes. Amid a wide-ranging smudge of cross-reactivity, three molecules of ~100 kDa, 30–35 kDa and 25–28 kDa in SEA reacted with antibodies eluted from Der f 15. These SEA molecules may be, respectively, the immunodominant S. mansoni egg antigens kappa-556, IPSE/alpha-157 and omega-158. All three SmSEA molecules are N-glycosylated and their respective patterns of glycosylation have been elucidated: kappa-559, IPSE/alpha-160 and omega-161. O-linked glycans have not been found on the above-mentioned SEA proteins, so some of the cross-reactivity of the rabbit anti-SmSEA antibodies with Der f 15 may be associated with glycosylation of the N-linked site(s) on the latter molecule. None of the other SmSEA molecules reacting with the acid-eluted anti-Der f 15 antibodies have been investigated, but O-linked glycans thereon62 may have induced antibodies cross-reactive with Der f 15. The notion that the cross-reactivity between Der f 15 and antigens in SmSEA is due to shared cross-reactive carbohydrate determinants (CCDs)25,28 is supported by the observation here that the anti-SmSEA antibodies eluted from Der f 15 reacted against not only SmSEA, but also against various plant extracts. Furthermore, all the reactivity against the plant extracts was abrogated completely by prior treatment of the material electroblotted onto the nitrocellulose film with Na-metaperiodate, which destroys carbohydrate determinants63,64.
For this work a local infestation of the Australian cockroach P. australasiae provided a convenient, cost-free source of cockroach-derived material. At least four molecules in the cockroach extract, mainly between 46 kDa and 100 kDa, were found to be cross-reactive with rabbit anti-SmSEA antibodies in the present work. TMS analysis of two of the SmSEA cross-reactive molecules of ~85 kDa and ~88 kDa indicated they both contained peptides with sequences identical to those found in P. americana protein Cr-PI, allergen Per a 3 (gi: 284518363)65,66. Periplaneta americana and Blatella germanica are deemed responsible for most allergic reactions to cockroach with twelve P. americana allergens known34 (http://www.allergen.org/). Unsurprisingly, at the time of writing a Cr-PI/Per a 3 homologue of P. australasiae is not amongst the 75 sequences from this cockroach species currently in the NCBI protein database, compared with a listing of >2250 P. americana proteins, including ~10 sequences for Cr-PI and/or Per a 3. Further discussion of the P. australasiae molecule studied here is therefore necessarily restricted to what is known about its P. americana orthologue.
Cr-PI/Per a 3 is an important human allergen as partially purified fractions of Cr-PI elicited skin reactions in 73% of patients who were sensitive to the crude P. americana extracts67. This qualifies Per a 3 as a major allergen68. Four isomers of Per a 3 are known with molecular sizes ranging from 46–79 kDa based on amino acid sequence65,66,69. The sizes of native forms of the allergen molecules in preparations of Cr-PI were estimated to be 72 kDa and 78 kDa67 while the molecular weights of mature Per a 3 calculated from amino acid sequences of two recombinant clones were given as 75.5 kDa and 79.3 kDa66. The former two values for the native P. americana molecules are lower than the values of 85 kDa and 88 kDa estimated for native forms of the two schistosome cross-reactive proteins in the P. australasiae extract studied here, which may be due to differences between the two cockroach species. The two isoforms of Per a 3 studied by Wu et al.66, were both found to have two sites for potential N-linked glycosylation, as was found by the analysis here. As with Der f 15 above, a low degree of amino acid sequence similarity found by pair-wise comparisons between Per a 3 and three immunodominant antigens of SmSEA: kappa-5, IPSE/alpha-1 and omega-1 (data available on request) suggests the antigenic cross-reactivity is unlikely to be due to shared peptide sequences. The identity of the S. mansoni worm and cercarial antigens that cross-reacted with the P. australasiae Per a 3 homologue is unknown, but all that cross-reactivity, apart from a broad band in SmSEA between 32 kDa and 46 kDa, disappeared as a result of periodate-treatment.
Comparison of the results from Der f 15 and Per a 3 indicate that the anti-SmSEA antibodies eluted from Der f 15 reacted strongly against SmSEA, but much less intensively against parasite cercarial and worm antigens and Der f 15 itself. In contrast, antibodies eluted from Per a 3 reacted well against molecules in all 3 schistosome extracts as well as the Per a 3 double band, this despite the fact that Der f 15 is potentially more heavily glycosylated than Per a 3. There is a large difference between the calculated amino-acid mass of Der f 15 (63.882 kDa) and its mass when resolved by electrophoresis (98 kDa), but this difference was much lower for Per a 3 which has a sequence-calculated mass of 82.3 kDa and of 85–88 kDa when derived by electrophoresis. In this study, analysis of the Der f 15 amino-acid sequence indicated the molecule has many more potential sites for O-linked glycosylation than Per a 3, which is predicted to have none, but otherwise little is known about these two molecules. The different reactivities of the anti-SmSEA antibodies eluted from Der f 15 with extracts of the three schistosome stages may be consistent with the previous findings of the presence of multifucosylated antennae in mature eggs and miracidia and its absence in worms62, indicating that this cross-reactivity may be due to α3-core fucose residues.
Twelve honey bee (Apis mellifera) allergens are currently listed (http://www.allergen.org/). Phospholipase A2 (PLA2) was identified as a major allergen early on70 and it constitutes 12% of the material in honey bee venom71. Three isoforms of PLA2 have been identified, of estimated molecular sizes 16 kDa, 18 kDa and 20 kDa: the larger two molecules are glycosylated while the 16 kDa is not72. The two isoforms of estimated size 17 kDa and 19 kDa observed here that are antigenically cross-reactive with anti-SmSEA antibodies may thus correspond with the two larger isoforms described by Altmann et al.72, N-glycosylated via just one asparagine residue situated at position 1373 and Asn 41 as described here (Supplementary Table S9). In the present study, anti-SmSEA antibodies eluted from electroblots of the 17/19 kDa isoforms of PLA2 reacted against a variety of schistosome antigens, but again nearly all this cross-reactivity did not survive periodate treatment. One ~100 kDa molecule in SmSEA was particularly reactive with the eluted anti-17/19 kDa antibodies, and this may be kappa-556. As with Der f 15 and Per a 3, pairwise comparison of the amino acid sequences indicates PLA2 and kappa-5 have little identity (~6%) or similarity (~12%) (data available on request), indicating that glycan epitopes are probably responsible for the antigenic cross-reactivity between the two molecules and anti-schistosome antibodies.
Glycan residues on both the larger-sized PLA2 variants were observed to contain fucose, mannose and N-acetylglucosamine, while the largest had, in addition, N-acetylgalactosamine72,74. Xylose and fucose are constituents of many plant and invertebrate glycoproteins27,75. Xylose is, however, generally considered absent in PLA274,76, though paradoxically a rabbit antiserum specific for an oligosaccharide-linked xylose residue was found to react with PLA277 and Altmann et al.72 found traces of this monosaccharide, which they considered an impurity, in their preparation of the 20 kDa isoform. The antigenic cross-reactivity between PLA2 and the putatively identified SmSEA antigen kappa-5 is therefore potentially interesting: kappa-5 has four potential N-glycosylation sites carrying triantennary glycans composed of a core region that is both difucosylated and xylosylated59, but the core α3-fucose residues may be primarily responsible for the cross-reactivity. The alpha 1,3 fucosylated glycan residue of PLA2/Api m 1 is cross-reactive with plant glycoproteins75, as also illustrated here by reactivity of anti-SmSEA antibodies eluted from PLA2 with a ~46 kDa band in a rubber latex extract and several proteins in peanut. The 46 kDa latex molecule could be Hev b 7, shown previously to be antigenically cross-reactive with S. mansoni30 and which might be responsible for reactivity to natural rubber latex in some patients with insect venom allergy78.
Antigenic reactivity of a band of 32–46 kDa in SmSEA survived periodate-treatment to some extent in the present study. The antigen responsible for this is likely IPSE/alpha-157, a molecule which binds to immunoglobulins in a non-immunological manner79. Apparent periodate-resistance of the antigenicity of IPSE/alpha-1 has been discussed elsewhere32. The supposition that the cross-reactivity between rabbit anti-SmSEA antibodies and the allergens is due to shared CCDs is supported by results (personal communication: R Hokke, 13 July 2018) showing that antibodies in the rabbit sera were highly reactive on a microarray of synthetic N-linked glycans with α3-fucose containing core structures, alone or in combination with β2-xylose (see80,81 for principles of the method). Core α3-fucosylation is characteristic of the N-linked glycans of IPSE/alpha-160 and omega-161, while the core of kappa-5 N-linked glycans is modified by both α3-fucose and β2-xylose59. A recent study81 has confirmed that core β-1,2-xylose- and α-1,3-fucose-specific antibody responses are associated with S. mansoni infections in rural environments.
The present study has used rabbit anti-S. mansoni IgG antibodies to demonstrate antigenic cross-reactivity between antigens and allergens from three invertebrates, with potential involvement of cross-reactive carbohydrate determinants (CCDs). The existence of antigenic cross-reactivity between plants, invertebrates and molluscs, attributable to CCDs, has been known for a long time77,82,83. This is, however, seemingly the first time the carbohydrate determinants on a helminth’s antigens have been shown to be cross-reactive with those of known invertebrate allergens. In the context of the present results, dogs subjected to allergen-specific immunotherapy for atopic dermatitis produced IgG antibodies (isotype not specified) against a 98 kDa D. farinae molecule, presumed to be Der f 1584 and specific immunotherapy with whole bee venom resulted in an increase in the ratio of anti-PLA2 IgG4:IgE in serum85. High IgG levels, including anti-CCD-specific IgG4, are a frequent outcome of successful specific immunotherapy for allergies86 and pollen immunotherapy29. High levels of IgG4 are also found in subjects with chronic helminth infections87,88 and a recent study in Uganda on the relationship between immune responses to S. mansoni and allergy found that: ‘total and allergen-specific IgG4/IgE ratios were mostly inversely associated with atopy, implying that the regulatory role of IgG4 against allergy might best be assessed relative to IgE'89. Thus, it could be envisaged from our results that anti-schistosome anti-CCD IgGs induced by schistosome antigens that share identical epitopes with invertebrate allergens may induce IgGs that elicit a blocking effect on allergenic IgE.
IgG4 antibodies are postulated to inhibit immediate hypersensitivity reactions by two possible mechanisms: sequestering antigen and thus preventing cross-linking of receptor-bound IgE, and/or inhibiting IgE-facilitated allergen presentation to antigen presenting B cells90. An alternative hypothesis involves co-aggregation of FcεRI to FcγRIIb, e.g. by immune complexes, which has been shown to block basophil and mast cell activation in vitro91,92,93,94. The effect has been confirmed in vivo using chimeric human-cat Fcγ-Fel d 1 allergen fusion proteins95.
Future experiments may seek to investigate potential blocking effects of cross-reactive anti-S. mansoni IgG on different allergen-induced IgE antibodies and their potential interference with early- and/or late-phase allergic reactions. A perhaps surprising statistic is that of the molecules in plants and invertebrates that so far happen to have been studied because they are cross-reactive with anti-SmSEA antibodies, all ten are known allergens; namely Hev b 7 in natural rubber latex30, Ara h 1 in peanut31, Bet v 1, Bet v 6.01 and a glutathione S-transferase in birch tree pollen32, the GST having been subsequently shown to be an allergen96, Phl p 1 and Phl p 5 in Timothy grass pollen32, and Der f 15, Per a 3 and Api m 1 in this study. The rabbit anti-SmSEA antibodies used in this series of studies however cross-reacted with more than one molecule in the extract of peanut31 and more than five plant pollen molecules32. If many of these are also found to be known allergens, CCD-dependent cross-reactivity between S. mansoni and allergens might be quite commonplace.
Data availability
The data generated and/or analysed during the current study are available from the corresponding author on reasonable request.
Change history
20 May 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Okada, H., Kuhn, C., Feillet, H. & Bach, J. F. The ‘hygiene hypothesis’ for autoimmune and allergic diseases: an update. Clin. Exp. Immunol. 160, 1–9 (2010).
Bach, J. F. The effect of infections on susceptibility to autoimmune and allergic diseases. New Eng. J. Med. 347, 911–920 (2002).
Rook, G. A., Raison, C. L. & Lowry, C. A. Microbial ‘old friends’, immunoregulation and socioeconomic status. Clin. Exp. Immunol. 177, 1–12 (2014).
Guarner, F. et al. Mechanisms of disease: the hygiene hypothesis revisited. Nature clinical practice. Gastr. Hepatol. 3, 275–284 (2006).
Rujeni, N., Taylor, D. W. & Mutapi, F. Human schistosome infection and allergic sensitisation. J. Parasitol. Res., https://doi.org/10.1155/2012/154743 (2012).
Araujo, M. I., Hoppe, B. S., Medeiros, M. Jr. & Carvalho, E. M. Schistosoma mansoni infection modulates the immune response against allergic and auto-immune diseases. Mem. Instit. Oswaldo Cruz 99, 27–32 (2004).
Smits, H. H., Everts, B., Hartgers, F. C. & Yazdanbakhsh, M. Chronic helminth infections protect against allergic diseases by active regulatory processes. Curr. Allergy Asthma Rep. 10, 3–12 (2010).
Rook, G. A. The hygiene hypothesis and the increasing prevalence of chronic inflammatory disorders. Trans. Roy. Soc. Trop. Med. Hyg. 101, 1072–1074 (2007).
Sokol, C. L., Barton, G. M., Farr, A. G. & Medzhitov, R. A mechanism for the initiation of allergen-induced T helper type 2 responses. Nature mmunol. 9, 310–318 (2008).
Oliphant, C. J., Barlow, J. L. & McKenzie, A. N. Insights into the initiation of type 2 immune responses. Immunology 134, 378–385 (2011).
Licona-Limon, P., Kim, L. K., Palm, N. W. & Flavell, R. A. TH2, allergy and group 2 innate lymphoid cells. Nature Immunol. 14, 536–542 (2013).
Anthony, R. M., Rutitzky, L. I., Urban, J. F. Jr., Stadecker, M. J. & Gause, W. C. Protective immune mechanisms in helminth infection. Nature Rev. Immunol. 7, 975–987 (2007).
Harris, N. & Gause, W. C. To B or not to B: B cells and the Th2-type immune response to helminths. Tr. Immunol. 32, 80–88 (2011).
Fitzsimmons, C. M., Falcone, F. H. & Dunne, D. W. Helminth Allergens, parasite-Specific IgE, and its protective role in human immunity. Front. Immunol. 5, 61 (2014).
Fallon, P. G. & Mangan, N. E. Suppression of T(H)2-type allergic reactions by helminth infection. Nat. Rev. Immunol. 7, 220–230 (2007).
Maizels, R. M. Parasitic helminth infections and the control of human allergic and autoimmune disorders. Clin. Microbiol. Infect. 22, 481–486 (2016).
Yazdanbakhsh, M. & Matricardi, P. M. Parasites and the hygiene hypothesis - Regulating the immune system? Clin. Rev. Allergy Immunol. 26, 15–23 (2004).
Wilson, M. S. et al. Suppression of allergic airway inflammation by helminth-induced regulatory T cells. J. Exp. Med. 202, 1199–1212 (2005).
Couper, K. N., Blount, D. G. & Riley, E. M. IL-10: the master regulator of immunity to infection. J. Immunology 180, 5771–5777 (2008).
Doetze, A. et al. Antigen-specific cellular hyporesponsiveness in a chronic human helminth infection is mediated by T(h)3/T(r)1-type cytokines IL-10 and transforming growth factor-beta but not by a T(h)1 to T(h)2 shift. Int. Immunol. 12, 623–630 (2000).
Jarrett, E. & Bazin, H. Elevation of total serum IgE in rats following helminth parasite infection. Nature 251, 613–614 (1974).
Jarrett, E., Mackenzie, S. & Bennich, H. Parasite-induced ‘nonspecific’ IgE does not protect against allergic reactions. Nature 283, 302–304 (1980).
Mitre, E., Norwood, S. & Nutman, T. B. Saturation of immunoglobulin E (IgE) binding sites by polyclonal IgE does not explain the protective effect of helminth infections against atopy. Inf. Immun. 73, 4106–4111 (2005).
Pritchard, D. I. et al. Basophil competence during hookworm (Necator americanus) infection. Am. J. Trop. Med. Hyg. 77, 860–865 (2007).
Altmann, F. The role of protein glycosylation in allergy. Int. Arch. Allergy Immunol. 142, 99–115 (2007).
Homann, A., Schramm, G. & Jappe, U. Glycans and glycan-specific IgE in clinical and molecular allergology: Sensitization, diagnostics, and clinical symptoms. J. Allergy Clin. Immunol. 140, 356–368 (2017).
van Ree, R. et al. Beta(1,2)-xylose and alpha(1,3)-fucose residues have a strong contribution in IgE binding to plant glycoallergens. J. Biol. Chem. 275, 11451–11458 (2000).
Aalberse, R. C. & van Ree, R. Crossreactive carbohydrate determinants. Clin. Rev. Alllergy Immunol. 15, 375–387 (1997).
van Ree, R. & Aalberse, R. C. Demonstration of carbohydrate-specific immunoglobulin G4 antibodies in sera of patients receiving grass pollen immunotherapy. Int. Arch. Allergy Immunol. 106, 146–148 (1995).
Doenhoff, J. M. et al. Cross-reactivity between Schistosoma mansoni antigens and the latex allergen Hev b 7: putative implication of cross-reactive carbohydrate Determinants (CCDs). PLoS One 11, e0159542, https://doi.org/10.1371/journal.pone.0159542 (2016).
Igetei, J. E., El-Faham, M., Liddell, S. & Doenhoff, M. J. Antigenic cross-reactivity between Schistosoma mansoni and peanut: a role for cross-reactive carbohydrate determinants (CCDs) and implications for the hygiene hypothesis. Immunology 150, 506–517 (2017).
Igetei, J. E., El-Faham, M., Liddell, S., Schramm, G. & Doenhoff, M. J. Antigenic cross-reactivity between Schistosoma mansoni and pollen allergens from the birch tree (Betula verrucosa) and Timothy grass (Phleum pratense): involvement of shared glycan epitopes and implications for the hygiene hypothesis. Int. J. Parasitol. 48, 345–357 (2018).
Calderon, M. A. et al. Respiratory allergy caused by house dust mites: What do we really know? J. Allergy Clin. Immunol. 136, 38–48 (2015).
Do, D. C., Zhao, Y. & Gao, P. Cockroach allergen exposure and risk of asthma. Allergy 71, 463–474 (2016).
Casale, T. B. & Burks, A. W. Clinical practice. Hymenoptera-sting hypersensitivity. New Eng. J. Med. 370, 1432–1439 (2014).
Doenhoff, M. J., Modha, J., Curtis, R. H. & Adeoye, G. O. Immunological identification of Schistosoma mansoni peptidases. Mol. Biochem. Parasitol. 31, 233–240 (1988).
Doenhoff, M. J. et al. Immunological control of hepatotoxicity and parasite egg excretion in Schistosoma mansoni infections: stage specificity of the reactivity of immune serum in T-cell deprived mice. Trans. Roy. Soc. Trop. Med. Hyg. 75, 41–53 (1981).
Lowry, O. H., Rosebrough, N. J., Farr, A. L. & Randall, R. J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951).
Dunne, D. W., Agnew, A. M., Modha, J. & Doenhoff, M. J. Schistosoma mansoni egg antigens: preparation of rabbit antisera with monospecific immunoprecipitating activity, and their use in antigen characterization. Parasite Immunol. 8, 575–586 (1986).
Laemmli, U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Studier, F. W. Analysis of bacteriophage T7 early RNAs and proteins on slab gels. J. Mol. Biol. 79, 237–248 (1973).
Towbin, H., Staehelin, T. & Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Nat. Acad. Sci. USA 76, 4350–4354 (1979).
Igetei, J. E., Liddell, S., El-Faham, M. & Doenhoff, M. J. Purification of a chymotrypsin-like enzyme present on adult Schistosoma mansoni worms from infected mice and its characterization as a host carboxylesterase. Parasitology 143, 646–657 (2016).
Rybicki, E. P. Affinity purification of specific antibodies from plant virus capsid protein immobilised on nitrocellulose. J. Phytopath. 116, 30–38 (1986).
Hamilton, J. V., Chiodini, P. L., Fallon, P. G. & Doenhoff, M. J. Periodate-sensitive immunological cross-reactivity between keyhole limpet haemocyanin (KLH) and serodiagnostic Schistosoma mansoni egg antigens. Parasitology 118, 83–89 (1999).
Eberl, M. et al. Antibodies to glycans dominate the host response to schistosome larvae and eggs: is their role protective or subversive? J. Infect. Dis. 183, 1238–1247 (2001).
Shevchenko, A., Wilm, M., Vorm, O. & Mann, M. Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal. Chem. 68, 850–858 (1996).
Kershaw, J. L., Botting, C. H., Brownlow, A. & Hall, A. J. Not just fat: investigating the proteome of cetacean blubber tissue. Conserv. Physiol. 6, coy003, https://doi.org/10.1093/conphys/coy003 (2018).
Bernton, H. S. & Brown, H. Insect Allergy–Preliminary Studies of the Cockroach. J. Allergy 35, 506–513 (1964).
Sohn, M. H. & Kim, K. E. The cockroach and allergic diseases. Allergy Asthma Imm. Res. 4, 264–269 (2012).
Moffitt, J. E. et al. Stinging insect hypersensitivity: a practice parameter update. J. Allergy Clin. Immunol. 114, 869–886 (2004).
Bilo, B. M. & Bonifazi, F. Epidemiology of insect-venom anaphylaxis. Curr. Opin. Allergy Clin. Immunol. 8, 330–337 (2008).
Thomas, W. R. Hierarchy and molecular properties of house dust mite allergens. Allergol. Int. Off. J. Jap. Soc. Allergol. 64, 304–311 (2015).
Hales, B. J. et al. Quantitation of IgE binding to the chitinase and chitinase-like house dust mite allergens Der p 15 and Der p 18 compared to the major and mid-range allergens. Int. Arch. Allergy Immunol. 160, 233–240 (2013).
McCall, C. et al. Characterization and cloning of a major high molecular weight house dust mite allergen (Der f 15) for dogs. Vet. Immunol. Immunopath. 78, 231–247 (2001).
Schramm, G. et al. Molecular characterisation of kappa-5, a major antigenic glycoprotein from Schistosoma mansoni eggs. Mol. Biochem. Parasitol. 166, 4–14 (2009).
Schramm, G. et al. IPSE/alpha-1: a major immunogenic component secreted from Schistosoma mansoni eggs. Mol. Biochem. Parasitol. 147, 9–19 (2006).
Fitzsimmons, C. M. et al. Molecular characterization of omega-1: a hepatotoxic ribonuclease from Schistosoma mansoni eggs. Mol. Biochem. Parasitol. 144, 123–127 (2005).
Meevissen, M. H. J. et al. Targeted glycoproteomic analysis reveals that kappa-5 is a major, uniquely glycosylated component of Schistosoma mansoni egg antigens. Mol. Cell. Prot. 10, https://doi.org/10.1074/mcp.M110.005710 (2011).
Wuhrer, M. et al. IPSE/alpha-1, a major secretory glycoprotein antigen from schistosome eggs, expresses the Lewis X motif on core-difucosylated N-glycans. FEBS J. 273, 2276–2292 (2006).
Meevissen, M. H. J. et al. Structural Characterization of Glycans on Omega-1, a major Schistosoma mansoni egg glycoprotein that drives Th2 responses. J. Prot. Res. 9, 2630–2642 (2010).
Smit, C. H. et al. Glycomic analysis of life stages of the human parasite Schistosoma mansoni reveals developmental expression profiles of functional and antigenic glycan motifs. Mol. Cell. Prot. 14, 1750–1769 (2015).
Bobbitt, J. M. Periodate oxidation of carbohydrates. Adv. Carb. Chem. 48, 1–41 (1956).
Woodward, M. P., Young, W. W. Jr. & Bloodgood, R. A. Detection of monoclonal antibodies specific for carbohydrate epitopes using periodate oxidation. J. Imm. Meth. 78, 143–153 (1985).
Wu, C. H., Lee, M. F., Wang, N. M. & Luo, S. F. Sequencing and immunochemical characterization of the American cockroach per a 3 (Cr-PI) isoallergenic variants. Mol. Imm. 34, 1–8 (1997).
Wu, C. H., Lee, M. F., Liao, S. C. & Luo, S. F. Sequencing analysis of cDNA clones encoding the American cockroach Cr-PI allergens. Homology with insect hemolymph proteins. J. Biol. Chem. 271, 17937–17943 (1996).
Wu, C. H. & Lan, J. L. Cockroach hypersensitivity: isolation and partial characterization of major allergens. J. Allergy Clin. Immunol. 82, 727–735 (1988).
Lowenstein, H. Quantitative immunoelectrophoretic methods as a tool for the analysis and isolation of allergens. Prog. Allergy 25, 1–62 (1978).
Pomes, A., Mueller, G. A., Randall, T. A., Chapman, M. D. & Arruda, L. K. New insights into cockroach allergens. Curr. Allergy Asthma Rep. 17, 25, https://doi.org/10.1007/s11882-017-0694-1 (2017).
Sobotka, A. K. et al. Allergy to insect stings. II. Phospholipase A: the major allergen in honeybee venom. J. Allergy Clin. Immunol. 57, 29–40 (1976).
Habermann, E. Bee and wasp venoms. Science 177, 314–322 (1972).
Altmann, F., Kubelka, V., Staudacher, E., Uhl, K. & Marz, L. Characterization of the isoforms of phospholipase-a(2) from honeybee venom. Insect Biochem. 21, 467–472 (1991).
Shipolini, R. A., Callewaert, G. L., Cottrell, R. C. & Vernon, C. A. The amino-acid sequence and carbohydrate content of phospholipase A2 from bee venom. Eur. J. Biochem. FEBS 48, 465–476 (1974).
Kubelka, V. et al. Primary structures of the N-linked carbohydrate chains from honeybee venom phospholipase A2. Eur. J. Biochem. FEBS 213, 1193–1204 (1993).
Prenner, C., Mach, L., Glossl, J. & Marz, L. The antigenicity of the carbohydrate moiety of an insect glycoprotein, honey-bee (Apis mellifera) venom phospholipase A2. The role of alpha 1,3-fucosylation of the asparagine-bound N-acetylglucosamine. Biochem. J. 284, 377–380 (1992).
Faye, L., Gomord, V., Fitchette-Laine, A. C. & Chrispeels, M. J. Affinity purification of antibodies specific for Asn-linked glycans containing alpha 1–>3 fucose or beta 1–>2 xylose. Anal. Biochem. 209, 104–108 (1993).
Faye, L. & Chrispeels, M. J. Common antigenic determinants in the glycoproteins of plants, mollusks and insects. Glycocon. J. 5, 245–256 (1988).
Mahler, V., Gutgesell, C., Valenta, R. & Fuchs, T. Natural rubber latex and hymenoptera venoms share immunoglobinE-epitopes accounting for cross-reactive carbohydrate determinants. Clin. Exp. Allergy 36, 1446–1456 (2006).
Meyer, N. H. et al. A crystallin fold in the interleukin-4-inducing principle of Schistosoma mansoni eggs (IPSE/alpha-1) mediates IgE binding for antigen-independent basophil activation. J. Biol. Cehm. ournal of biological chemistry 290, 22111–22126 (2015).
Amoah, A. S. et al. Identification of dominant anti-glycan IgE responses in school children by glycan microarray. J. Allergy Clin. Immunol. 141, 1130–1133 (2018).
Nkurunungi, G. et al. Microarray assessment of N-glycan-specific IgE and IgG profiles associated with Schistosoma mansoni infection in rural and urban Uganda. Scientific Rep. 9, 3522 (2019).
Aalberse, R. C., Koshte, V. & Clemens, J. G. Immunoglobulin E antibodies that crossreact with vegetable foods, pollen, and Hymenoptera venom. J. Allergy Clin. Immunol. 68, 356–364 (1981).
Wilson, I. B. Glycosylation of proteins in plants and invertebrates. Curr. Opin. Struct. Biol. 12, 569–577 (2002).
Hou, C. C., Griffin, C. E. & Hill, P. B. Dermatophagoides farinae-specific IgG responses in atopic dogs undergoing allergen-specific immunotherapy with aqueous vaccines. Vet. Dermat. 19, 215–220 (2008).
Akdis, C. A. et al. Epitope-specific T cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J. Clin. Invest. 98, 1676–1683 (1996).
Matsuoka, T., Shamji, M. H. & Durham, S. R. Allergen immunotherapy and tolerance. Allergol. Int. Off. J. Jap. Soc. Allergol. 62, 403–413 (2013).
Kurniawan, A. et al. Differential expression of IgE and IgG4 specific antibody responses in asymptomatic and chronic human filariasis. J. Immunol. 150, 3941–3950 (1993).
Ottesen, E. A., Skvaril, F., Tripathy, S. P., Poindexter, R. W. & Hussain, R. Prominence of IgG4 in the IgG antibody response to human filariasis. J. Immunol. 134, 2707–2712 (1985).
Nkurunungi, G. et al. Schistosoma mansoni-specific immune responses and allergy in Uganda. Parasite Immunol. 40, https://doi.org/10.1111/pim.12506 (2018).
James, L. K. & Till, S. J. Potential mechanisms for IgG4 inhibition of immediate hypersensitivity reactions. Curr. Allergy Asthma Rep. 16, 23 (2016).
Kepley, C. L. et al. Negative regulation of Fc epsilon RI signaling by Fc gamma RII costimulation in human blood basophils. J. Allergy Clin. Immunol. 106, 337–348 (2000).
Malbec, O. et al. Fc epsilon receptor I-associated lyn-dependent phosphorylation of Fc gamma receptor IIB during negative regulation of mast cell activation. J. Immunol. 160, 1647–1658 (1998).
Daeron, M. et al. The same tyrosine-based inhibition motif, in the intracytoplasmic domain of Fc gamma RIIB, regulates negatively BCR-, TCR-, and FcR-dependent cell activation. Immunity 3, 635–646 (1995).
Fong, D. C. et al. Selective in vivo recruitment of the phosphatidylinositol phosphatase SHIP by phosphorylated Fc gammaRIIB during negative regulation of IgE-dependent mouse mast cell activation. Immunol. Lett. 54, 83–91 (1996).
Terada, T., Zhang, K., Belperio, J., Londhe, V. & Saxon, A. A chimeric human-cat Fcgamma-Fel d1 fusion protein inhibits systemic, pulmonary, and cutaneous allergic reactivity to intratracheal challenge in mice sensitized to Fel d1, the major cat allergen. Clin. Immunol. 120, 45–56 (2006).
Deifl, S. et al. Glutathione-S-Transferase: A Minor Allergen in Birch Pollen due to Limited Release from Hydrated Pollen. PlOS One 9, e109075 (2014).
Acknowledgements
The authors are very grateful to David Richards, University of East Anglia, UK, for a valuable donation of honey bee venom, and to Cornelis H. (Ron) Hokke for preliminary analysis of reactivity of the rabbit anti-SmSEA antisera used in this study against a microarray of synthetic N-linked glycans. The current research was partly funded by awards of a University of Nottingham International Research Excellence scholarship 2011 to JEI, an Erasmus + scholarship of the European Union to SR and a grant from the Islamic Development Bank to FG.
Author information
Authors and Affiliations
Contributions
M.H.E.-F., F.G., J.E.I. and S.R. performed the experiments; M.J.D., F.H.F. and G.S. supervised; all authors contributed to drafting, reading and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Faham, M.H., Gai, F., Igetei, J.E. et al. Antigenic cross-reactivity between Schistosoma mansoni and allergenic invertebrates putatively due to shared glycanic epitopes. Sci Rep 10, 3350 (2020). https://doi.org/10.1038/s41598-020-59892-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-59892-6
- Springer Nature Limited
This article is cited by
-
Are humanized IgE reporter systems potential game changers in serological diagnosis of human parasitic infection?
Parasitology Research (2022)