Abstract
Diabrotica virgifera virgifera LeConte, the western corn rootworm (WCR) is one of the most destructive pests in the U.S. Corn Belt. Transgenic maize lines expressing various Cry toxins from Bacillus thuringiensis have been adopted as a management strategy. However, resistance to many Bt toxins has occurred. To investigate the mechanisms of Bt resistance we carried out RNA-seq using Illumina sequencing technology on resistant, eCry3.1Ab-selected and susceptible, unselected, whole WCR neonates which fed on seedling maize with and without eCry3.1Ab for 12 and 24 hours. In a parallel experiment RNA-seq experiments were conducted when only the midgut of neonate WCR was evaluated from the same treatments. After de novo transcriptome assembly we identified differentially expressed genes (DEGs). Results from the assemblies and annotation indicate that WCR neonates from the eCry3.1Ab-selected resistant colony expressed a small number of up and down-regulated genes following Bt intoxication. In contrast, unselected susceptible WCR neonates expressed a large number of up and down-regulated transcripts in response to intoxication. Annotation and pathway analysis of DEGs between susceptible and resistant whole WCR and their midgut tissue revealed genes associated with cell membrane, immune response, detoxification, and potential Bt receptors which are likely related to eCry3.1Ab resistance. This research provides a framework to study the toxicology of Bt toxins and mechanism of resistance in WCR, an economically important coleopteran pest species.
Similar content being viewed by others
Introduction
The Bt (Bacillus thuringiensis) Cry3 δ-endotoxin superfamily is known for its specificity to coleopteran species1,2. Transgenic maize hybrids expressing Cry3Bb1, mCry3A, eCry3.1Ab as well as Cry34/35Ab1 have been introduced to the market to manage western corn rootworm (WCR, Diabrotica virgifera virgifera LeConte). As a highly adaptive species, WCR has developed resistance to broadcast soil insecticides3, aerial spray insecticides for reducing adult number4,5, crop rotation6, and Cry3 Bt toxins. Laboratory selection experiments have developed WCR colonies resistant to Cry3Bb1, mCry3A, Cry34/35Ab1 and eCry3.1Ab by continuously rearing WCR on transgenic maize lines7,8,9,10. Resistance to Cry3 Bt toxins has become a practical issue since field resistant WCR populations have been reported in many locations11,12,13,14. Although eCry3.1Ab is the most recent Bt toxin15 to the market without reported control failure in the field, laboratory selection9 and cross-resistance experiments13 indicate that resistance to eCry3.1Ab is likely in the field due to the resistance to other Cry3 proteins16.
Three Cry toxins (Cry3Bb1, mCry3A and eCry3.1Ab) for WCR control are derived from the Cry3 superfamily. In lepidopteran systems, the consumption of Bt protoxin by larvae is followed by activation via cleavage by midgut proteases, binding to brush border receptors, toxin insertion into the epithelial cell membrane, pore formation by oligomerization, and finally, destruction of midgut epithelial cells17,18 followed by fatal sepsis19. Anything interfering with these processes (e.g. decreased activation, reduced receptor affinity, increased toxin degradation, or increased repair of midgut epithelial cells) could cause resistance20. However, the detailed interactions between Cry3 toxins and WCR is less known. Similar to Lepidoptera, reduced binding of mCry3A to the midgut epithelial cell membrane has been observed in resistant WCR colonies21. However, no Cry3 receptor has been validated in Coleopterans. Although potential receptors including cadherin-like protein22 and ATP-binding cassette (ABC) transporters23 are present in WCR, their roles in Bt resistance are questionable due to the lack of direct evidence of protein interactions.
Toxin dose is a very important component of what has been coined the ‘high-dose/refuge strategy’ to delay resistance14. In this theory the ‘high-dose’ refers to Bt toxins that cause very high mortality to pest populations. The initial frequency of alleles conferring resistance is low, fitness costs of resistance are present, mating between resistant and susceptible insects is random, and resistance to the Bt toxin is recessive24,25. For lepidopteran pests, for instance Heliothis virescens (Fabricius), the Cry1Ac is considered as a high-dose toxin, which is able to kill the insects with susceptible alleles. Only individuals with homozygous resistant alleles will survive, yielding a population with an extremely high resistance ratio to Bt toxin. The resistant genes could be identified by QTL mapping26 from a homozygous resistant population. In WCR, the situation is less optimal. All four WCR-targeting Bt toxins are considered as low to moderate dose27,28,29,30, where individuals heterozygous for resistance alleles may survive. Second, previous studies showed resistance to Bt in WCR may be caused by multiple genes23. The resistant genes selected in laboratory-selected resistant populations may not be sufficient to estimate the resistant alleles in field resistant populations due to founder effects. Third, the lack of a well-assembled, high-quality WCR genome sequence hampers the application of advanced genetic and molecular approaches to analyze resistant-related variation at the genomic level. RNA-seq followed by de novo assembly is an alternative for analyzing gene expression without a reference genome. In WCR, RNA-seq has been successfully used to study callus digestion31, interaction with corn defense chemicals32, adaptation to pesticides33, resistance to crop rotation34 and response to Cry34/35Ab135, and Cry3Bbb136.
To investigate the gene response to eCry3.1Ab intoxication and mechanisms of resistance, we sequenced the transcriptome of eCry3.1Ab-selected resistant and unselected, susceptible WCR feeding with maize root with and without eCry3.1Ab for 12 and 24 hours. Moreover, to further track down the tissue interactions with Bt toxin we sequenced the transcriptome of midgut from recovered neonates with the same treatment. The expression patterns of different populations under these conditions reveal how WCR respond to eCry3.1Ab and provide clues of resistance mechanisms.
Results and Discussion
Transcriptome assembly and annotation of WCR transcriptome
The transcriptome of whole larvae and midgut were separately de novo assembled. Reads from either whole larvae or midgut were individually pooled to increase the assembly coverage. Low quality and mitochondrial sequences were removed prior to assembly. Transcriptome assembly resulted in a whole larval transcriptome with 204,842 contigs from 57 Gb of reads and midgut transcriptome with 226,115 contigs from 137 Gb of reads. The two transcriptomes had comparable average sequence length (measured as N50, a weighted median statistic that more than half of the nucleotides of a transcriptome belong to the contigs of this size N50 or longer), GC content and length distribution (Table 1, Fig. 1). After removing duplicate contigs with more than 95% sequence similarity, we obtained 187,570 and 209,167 contigs respectively from larval and midgut transcriptome (Table 1). Herein, we refer to the two reduced redundancy sets of contigs as “unigene” sets. BUSCO37 was applied to evaluate the completeness of each unigene set. All of the transcriptome and unigene sets cover over 95% of insecta single-copy orthologs. Both unigene sets maintain the coverage and integrity while duplication is reduced, indicating the acceptable qualities for functional analysis (Table 2).
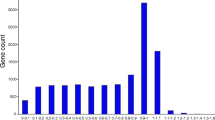
Transcriptomes and their unigene sets were aligned to the NCBI non-redundant protein database (NR) using BLASTX. Only 42% of larval and 38.2% of midgut unigenes had significant BLASTX results. The species distribution of BLASTX top hits indicated that a predominant number of unigenes could be annotated by the Coleopteran model species Tribolium castaneum (Herbst) (Table 3, Fig. 2). We also identified 372 and 400 unigenes from larval and midgut transcriptome, respectively, that were directly aligned to 13 known genes from Diabrotica species (Tables 3 and 4).
Differential expression analysis of WCR transcriptome
To identify the genes involved in eCry3.1Ab response, we analyzed the differentially expressed unigenes from whole larval and midgut transcriptomes between eCry3.1Ab-feeding and non-Bt isoline feeding WCR at both 12- and 24-hour time points. To understand the expression differences between eCry3.1Ab-selected and susceptible, control WCR from the same original population, we also compared the expression differences in whole larvae and midgut between two populations when being fed with the same kind of maize root. After the alignment and filtering out unigenes with extremely low expression levels, only 31,875 of larval and 22,954 of midgut transcriptome unigenes were proceeded to edgeR analysis38. The patterns of differentially expressed genes are shown in Fig. 3. Regardless of exposure time or tissue type, susceptible WCR differentially expressed a much larger number of genes in response to eCry3.1Ab intoxication. In contrast, eCry3.1-selected WCR had dramatically fewer differentially expressed genes (DEGs). The selected and unselected WCR shares many DEGs in both whole larvae and midgut transcriptome, while some unique DEGs were colony specific, especially in unselected colony (Fig. 4).
Transcriptional response of neonate WCR allowed feed 12 or 24 hrs on eCry3.1Ab or non-Bt isoline maize seedlings compared to resistant neonates feeding on isoline seedlings. R: eCry3.1Ab resistant WCR; S: susceptible WCR; Bt: eCry3.1Ab transgenic seedlings; Isoline: non-Bt seedlings; Differential expression pattern: (A) larval response at 12 hours. (B) Larval response at 24 hours. (C) Midgut response at 12 hours. (D) Midgut response at 24 hours (adjust p-value < 0.05, FDR test).
Differential expression pattern of resistant and susceptible WCR in whole larvae, or dissected midguts, when feeding on Cry3.1Ab transgenic maize seedlings vs. feeding on non-Bt isoline seedlings, for 12 or 24 hrs. In whole larvae (A,B,C and D) at 12 hrs feeding up-regulated (A), and down-regulated (B) contigs, showing overlapping expression. In whole larvae at 24 hrs feeding up-regulated (C), and down-regulated (D). In dissected midgut tissues (E,F,G and H) contigs up-regulated at 12 hr feeding (E), and down-regulated (F). At 24 hrs feeding contigs up-regulated (G) and down-regulated (H). Numbers in shown in parentheses are contigs with BLASTX annotation.
Albeit smaller in size, we identified DEGs between eCry3.1Ab-selected and unselected WCR, especially when both groups of neonates were fed on eCry3.1Ab maize root. The function of those DEGs and the pathways in which they were involved may reveal the physiological differences and the mechanism of eCry3.1Ab resistance in the selected population.
GO annotation and pathway analysis on eCry3.1Ab feeding WCR midgut
To further investigate the molecular and physiological adaptation to intoxication on eCry3.1Ab-selected WCR we applied gene ontology (GO) analysis to differentially expressed genes using Blast2GO. We compared the top 20 level-2 GO terms called from the unigene set from midgut DEGs between selected and unselected WCR feeding on eCry3.1Ab maize for 24 h. The GO terms were ranked based on number of unigenes of each GO term. The terms “metabolic process”, “catalytic activity” and “membrane” accounted for the majority of each of the three ontologies [Biological Process (BP), Molecular Function (MF) and Cellular Component (CC), respectively], suggesting the primary location and functions of eCry3.1Ab resistance. Other than that, the BP term “cellular process” and MF term “binding” also implied the binding and processing of toxin may have a role in eCry3.1Ab sensitivity.
We compared the distribution of level-2 GO terms between the DEG unigene set described above and midgut transcriptome unigene set (Table 5). The BP terms “Multiple-organism process”, “immune system process”, “detoxification” and “cell killing” and the CC terms “extracellular region” and “extracellular part” were among the most over represented GOs in DEG unigene sets, while the BP term “reproduction” and MF term “structural molecule activity” were the most under represented. However gene set enrichment analysis (GSEA) showed that no specific GO term was significantly enriched in the DEG unigene set.
We further predicted enzyme codes (EC) from GO terms for unigenes and used the EC to map differentially expressed unigenes to pathways from the Kyoto Encyclopedia of Genes and Genomes (KEGG) database39. The differentially expressed midgut unigenes were involved in 49 pathways, including purine metabolism (KEGG ID: 00230), glutathione metabolism (KEGG ID: 00480), fatty acid synthesis (KEGG ID: 00061), glycerophospholipid metabolism (KEGG ID: 00564) and drug metabolism (KEGG ID: 00983). The fatty acid synthesis pathway was also identified in whole larval differentially expressed unigenes. These results suggest that differences in eCry3.1Ab tolerances might arise from the alternation of genes related to detoxification, membrane functions and metabolism.
Expression of potential and novel eCry3.1Ab resistant genes
Research has revealed that cadherins26, ABC transporters40, and aminopeptidase N (APN)41 are Bt receptors in lepidopteran insects. Both cadherins and ABC transporters have been found in WCR22,23. Upon BLASTX, we found 102 unigenes with cadherin function, 209 unigenes with ABC transporter function, and 50 unigenes with APN function from the midgut unigene set. In whole larval unigene set, there were 78 unigenes with cadherin function, 191 unigenes with ABC transporter function, and 62 unigenes with APN function.
The midgut gene expression profile showed that cadherin was not significantly differentially expressed, while some ABC transporters (multidrug resistance-associated protein) and APNs were differentially expressed following Cry3.1Ab ingestion, especially in susceptible WCR (Table 6, Fig. 5). The expression level of one APN midgut unigene (comp127589_c1_seq. 1) was significantly higher in susceptible WCR midgut, when both selected and susceptible-WCR were given eCry3.1Ab for 24 hours. Two ABC transporter unigenes (comp121889_c0_seq. 1, comp126268_c0_seq. 1) showed the same pattern, but the increased expression in susceptible WCR was not significant. The protein structure of eCry3.1Ab toxin contains partial Cry1Ab sequence at its C-terminus15,42. In multiple lepidopteran species APNs serve as binding receptors of Cry1A toxins and mutations or reduced expression of APN result in resistance to Bt toxins41. Our results suggest that APN is a potential eCry3.1Ab target and the reduced expression level under intoxication might contribute to resistance in WCR.
Expression levels of 11 potential eCry3.1Ab resistance related genes in 8 WCR midgut treatment group (see Table 6). Expression level s are quantified by count per million read (cpm). The candidate genes are categorized into: 1: potential Bt receptors; 2: digestive proteases; 3: detoxification enzymes; 4: enzymes involve in drug metabolism pathway; 5: enzymes involve in membrane related pathways; 6: other candidates.
Proteases have been associated with Bt resistance either by increasing digestion of toxins43,44, or by decreasing the proteolytic activation of Bt pro-toxins45. We identified 112 unigenes with metalloprotease functions and 199 unigenes with cathepsin functions in the larval unigene set. In the midgut unigene set, there were 131 unigenes with metalloproteases functions and 234 unigenes with cathepsin functions. Expression levels of some proteases were regulated by Bt intoxication either in selected or susceptible WCR, while none of those digestive proteases was up regulated in selected WCR midgut compared to the susceptible one when both insects were fed with eCry3.1Ab maize.
Considering the expression patterns, GO annotation, and pathway analysis, we infer that at least two novel genes are likely involved in the resistance to eCry3.1Ab. Esterase has been reported involved in Cry1Ac resistance in Helicoverpa armigera46. In WCR, we observed a novel esterase (comp36305_c0_seq. 1) expressed in the midgut of selected colony was higher than unselected one after 24 hours of intoxication. The second gene is a dynein heavy chain-like protein (comp127369_c3_seq. 14). It was constitutively up regulated in selected WCR regardless of diet and time. Since dynein is a cytoskeletal motor protein involved in intracellular transportation and the movement of chromosomes, we propose that selected-WCR many have a stronger activity in either endocytosis or cell mitosis to remove the attached eCry3.1Ab molecules47, or to repair damaged epithelial cells48.
Conclusions
After a comprehensive analysis of an RNA-seq experiment with eCry3.1Ab-selected and susceptible, control WCR, the transcriptome, unigene sets and reads provided numerous resources for studying the interaction between Cry3 and this coleopteran species. This study is the first step approaching the delineation of Bt resistance mechanisms in WCR. We propose more than one potential mechanism of resistance to eCry3.1Ab – a dual action Bt toxin. With the recently published WCR genome sequence, future research will detect the genomic-wide genetic variations associated with Bt resistance. Studies to explore how Bt toxins affect gene alternative splicing and whether the alternatively sliced genes are related to Bt resistance in WCR. We are also developing cellular and molecular methods i.e. cultured cell expression, RNAi gene silencing, individual genotyping to further study the detailed mechanism of resistance to eCry3.1Ab as well as other Bt toxins. Continuous discoveries in this field will lead to improving strategies for insect resistant management and the developing of novel entomotoxins.
Material and Methods
Insects and bioassay
The eCry3.1Ab-selected resistant WCR colony was initially selected and reared on non-elite non-commercial eCry3.1Ab-expressing transgenic maize (event 5130) under laboratory conditions9. Both the selected and susceptible control colonies were developed from a single population and had been maintained on eCry3.1Ab-expressing transgenic maize (material ID 12MG00345) and its near-isoline (material ID 12MG001181), respectively, for more than 30 generations. For the current experimental design (Supplementary Table 1, column 1), both eCry3.1Ab-expressing and isoline maize seeds were surface sterilized and germinated in Petri dishes with moistened filter paper at 23 °C for 4–6 days without illumination. Approximately 30 neonates hatched within 24 hours were transferred to a Petri dish containing 3–4 seedlings of each line. After 12 or 24 hours feeding, the living first-instar larvae were recovered. In a separate identical experiment (Supplementary Table 1, column 2), the midgut was dissected from 30–40 recovered larvae of each Petri dish. Both whole larvae and midgut were flash frozen in liquid nitrogen and stored at −80 °C until processing. Both the whole larvae and midgut bioassays were replicated independently three times as full biological replications.
RNA extraction, library construction and sequencing
RNA was extracted using Trizol and purified by Direct-zol RNA Mini Prep kit (Zymo Research, Irving, CA). DNase treatment (ThermoFisher, Waltham, MA) was incorporated to remove genomic DNA contamination. RNA samples were checked for integrity using a fragment analyzer (University of Missouri DNA Core Laboratory). Strand-specific RNA-Seq libraries were prepared using the Illumina TruSeq HT Stranded Total RNA Library Prep Kit (Illumina, San Diego, CA), following the manufacturer’s instructions. For whole larvae and midgut respectively, 24 libraries (2 insect colonies × 2 corn lines × 2 times × 3 biological replications) were normalized, pooled and sequenced on two lanes of Illumina HiSeq2000 sequencer using 100-nucleotide pair-end protocol (Global Biologics LLC, Columbia, MO, USA).
De novo assembly of transcriptome
Adapters and low quality reads were trimmed using FastqMcf (version 1.04.803, https://github.com/ExpressionAnalysis/ea-utils/blob/wiki/FastqMcf.md) and Trimmomatic (version 0.36)49, respectively. To remove the mtDNA, the reads were aligned to the WCR mitochondrial genome50 using Bowtie 2 (version 4.7.7)51 with default settings. Paired reads concordantly aligned with no mismatch were considered as mitochondrial reads and discarded. These steps resulted in “clean reads” for assembly and differential expression analysis.
Trinity (version r2013-11-10)52 was used to de novo assemble larval and midgut transcriptomes using cleaned reads pooled from libraries of each sample type with default setting. The unigene set of each transcriptome was obtained by removing sequence with over 95% of similarity in Blast2GO53 (v4.1.9). BUSCO analyses (version v3 with Insecta odb9 dataset)37 were performed on transcriptome assemblies and unigene sets to evaluate their quality and completeness.
Differential expression analysis
Cleaned reads were aligned to the corresponding transcriptome unigene set using Bowtie 2 with default pair-end settings51. The output SAM files were converted to BAM format using SAMtools (version 0.1.20)54. The differential expression analysis was conducted in R (version 3.4.1)55. The read counts were called in R using the GenomicAlignment (version 1.6.3) and GenomicRanges (1.22.4) packages56. We counted only concordant alignment pairs while accepting multiple mapping reads due to the potential existence of isoforms in the unigene sets. The low expression unigenes were removed by applying filters with at least 2 count per million (CPM) over 3 samples. The differentially expressed contigs were assessed using the edgeR-robust algorithm of the edgeR package (version 3.12.1)38,57 with the trimmed mean of M-values (TMM) normalization method58,59. False discovery rate (FDR) was controlled at 0.05 by the edgeR package and was used to determine the significance of differentially expressed genes (DEGs).
Annotation and pathway analysis
Blast2GO was used for gene annotation, enrichment and pathway analysis. Transcriptomes were annotated by BLASTX against NCBI non-redundant (NR) database using an E-value cutoff of 1.0E-3. InterProScan was used to identify protein domains from 11 member databases60. Gene ontology terms (GO) were assigned based on the results of BLASTX and InterProScan. Gene set enrichment analysis (GSEA) was used to determine enriched GO terms by comparing the gene list versus the entire transcriptome. The enzyme codes were assigned to predicted enzymes function based on their GO terms. Those enzymes were mapped to Kyoto Encyclopedia of Gene and Genomes (KEGG) database for pathway analysis.
References
Perlak, F. J. et al. Genetically improved potatoes: protection from damage by Colorado potato beetles. Plant Molecular Biology 22, 313–321, https://doi.org/10.1007/bf00014938 (1993).
Lee, J.-H. et al. CryIIIA toxin gene expression in transgenic rice confers resistance to rice water weevil. Plant Cell, Tissue and Organ Culture (PCTOC) 115, 243–252, https://doi.org/10.1007/s11240-013-0356-5 (2013).
Ball, H. J. & Weekman, G. T. Insecticide resistance in the adult western corn rootworm in Nebraska. Journal of Economic Entomology 55, 439–441 (1962).
Meinke, L. J., Siegfried, B. D., Wright, R. J. & Chandler, L. D. Adult susceptibility of Nebraska western corn rootworm (Coleoptera: Chrysomelidae) populations to selected insecticides. Journal of Economic Entomology 91, 594–600 (1998).
Pereira, A. E. et al. Evidence of field-evolved resistance to bifenthrin in western corn rootworm (Diabrotica virgifera virgifera LeConte) populations in western Nebraska and Kansas. PLOS ONE 10, e0142299, https://doi.org/10.1371/journal.pone.0142299 (2015).
Levine, E., Spencer, J. L., Isard, S. A., Onstad, D. W. & Gray, M. E. Adaptation of the western corn rootworm to crop rotation: evolution of a new strain in response to a management practice. American Entomologist 48, 94–107 (2002).
Meihls, L. N. et al. Increased survival of western corn rootworm on transgenic corn within three generations of on-plant greenhouse selection. Proceedings of the National Academy of Sciences 105, 19177–19182, https://doi.org/10.1073/Pnas.0805565105 (2008).
Meihls, L. N., Higdon, M. L., Ellersieck, M. & Hibbard, B. E. Selection for resistance to mCry3A-expressing transgenic corn in western corn rootworm. Journal of Economic Entomology 104, 1045–1054 (2011).
Frank, D. L., Zukoff, A., Barry, J., Higdon, M. L. & Hibbard, B. E. Development of resistance to eCry3.1Ab-expressing transgenic maize in a laboratory-selected population of western corn rootworm (Coleoptera: Chrysomelidae). Journal of Economic Entomology 106, 2506–2513 (2013).
Lefko, S. A. et al. Characterizing laboratory colonies of western corn rootworm (Coleoptera: Chrysomelidae) selected for survival on maize containing event DAS-59122-7. Journal of Applied Entomology 132, 189–204, https://doi.org/10.1111/J.1439-0418.2008.01279.X (2008).
Gassmann, A. J., Petzold-Maxwell, J. L., Keweshan, R. S. & Dunbar, M. W. Field-evolved resistance to Bt maize by western corn rootworm. PLoS One 6, e22629, https://doi.org/10.1371/journal.pone.0022629 (2011).
Wangila, D. S., Gassmann, A. J., Petzold-Maxwell, J. L., French, B. W. & Meinke, L. J. Susceptibility of Nebraska western corn rootworm (Coleoptera: Chrysomelidae) populations to Bt corn events. Journal of Economic Entomology 108, 742–751, https://doi.org/10.1093/jee/tou063 (2015).
Zukoff, S. N. et al. Multiple assays indicate varying levels of cross resistance of Cry3Bb1-selected field populations of the western corn rootworm to mCry3A, eCry3.1Ab, and Cry34/35Ab1. Journal of Economic Entomology 109, 1387–1398 (2016).
Ludwick, D. C. et al. Minnesota field population of western corn rootworm (Coleoptera: Chrysomelidae) shows incomplete resistance to Cry34Ab1/Cry35Ab1 and Cry3Bb1. Journal of Applied Entomology 141, 28–40, https://doi.org/10.1111/jen.12377 (2017).
Walters, F. S., deFontes, C. M., Hart, H., Warren, G. W. & Chen, J. S. Lepidopteran-active variable-region sequence imparts coleopteran activity in eCry3.1Ab, an engineered Bacillus thuringiensis hybrid insecticidal protein. Applied and Environmental Microbiology 76, 3082–3088, https://doi.org/10.1128/aem.00155-10 (2010).
Ludwick, D. C. & Hibbard, B. C. Rootworm management: status of GM traits, insecticides and potential new tools. CAB Reviews 11, 048 (2016).
Bravo, A., Gill, S. S. & Soberón, M. Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 49, 423–435, https://doi.org/10.1016/j.toxicon.2006.11.022 (2007).
Knowles, B. H. Mechanism of action of Bacillus thuringiensis insecticidal δ-endotoxins. Advances in Insect Physiology 24, 275–308, https://doi.org/10.1016/S0065-2806(08)60085-5 (1994).
Caccia, S. et al. Midgut microbiota and host immunocompetence underlie Bacillus thuringiensis killing mechanism. Proceedings of the National Academy of Science USA 113, 9486–9491, https://doi.org/10.1073/pnas.1521741113 (2016).
Heckel, D. G. et al. The diversity of Bt resistance genes in species of Lepidoptera. Journal of Invertebrate Pathology 95, 192–197, https://doi.org/10.1016/j.jip.2007.03.008 (2007).
Zhao, J.-Z. et al. mCry3A-Selected western corn rootworm (Coleoptera: Chrysomelidae) colony exhibits high resistance and has reduced binding of mCry3A to midgut tissue. Journal of Economic Entomology 109, 1369–1377, https://doi.org/10.1093/jee/tow049 (2016).
Sayed, A. et al. A novel cadherin-like gene from western corn rootworm, Diabrotica virgifera virgifera (Coleoptera: Chrysomelidae), larval midgut tissue. Insect Molecular Biology 16, 591–600, https://doi.org/10.1111/j.1365-2583.2007.00755.x (2007).
Flagel, L. E. et al. Genetic markers for western corn rootworm resistance to Bt toxin. G3: Genes|Genomes|Genetics 5, 399–405, https://doi.org/10.1534/g3.114.016485 (2015).
Gould, F. Sustainability of transgenic insecticidal cultivars: integrating pest genetics and ecology. Annual Review of Entomology 43, 701–726, https://doi.org/10.1146/annurev.ento.43.1.701 (1998).
Tabashnik, B. E. & Gould, F. Delaying corn rootworm resistance to Bt corn. Journal of Economic Entomology 105, 767–776, https://doi.org/10.1603/EC12080 (2012).
Gahan, L. J., Gould, F. & Heckel, D. G. Identification of a gene associated with Bt resistance in Heliothis virescens. Science 293, 857–860 (2001).
Storer, N. P., Babcock, J. M. & Edwards, J. M. Field measures of western corn rootworm (Coleoptera: Chrysomelidae) mortality caused by Cry34/35Ab1 proteins expressed in maize event 59122 and implications for trait durability. Journal of Economic Entomology 99, 1381–1387, https://doi.org/10.1603/0022-0493-99.4.1381 (2006).
Hibbard, B. E., Meihls, L. N., Ellersieck, M. R. & Onstad, D. W. Density-dependent and density-independent mortality of the western corn rootworm: impact on dose calculations of rootworm-resistant Bt corn. Journal of Economic Entomology 103, 77–84, https://doi.org/10.1603/EC09277 (2010).
Hibbard, B. E. et al. Mortality of western corn rootworm larvae on MIR604 transgenic maize roots: field survivorship has no significant impact on survivorship of F1 progeny on MIR604. Journal of Economic Entomology 103, 2187–2196, https://doi.org/10.1603/EC10179 (2010).
Clark, T. L. et al. Mortality impact of MON863 transgenic maize roots on western corn rootworm larvae in the field. Journal of Applied Entomology 136, 721–729, https://doi.org/10.1111/j.1439-0418.2012.01709.x (2012).
Eyun, S.-i et al. Molecular evolution of glycoside hydrolase genes in the western corn rootworm (Diabrotica virgifera virgifera). PLoS ONE 9, e94052, https://doi.org/10.1371/journal.pone.0094052 (2014).
Miller, N. J. & Zhao, Z. Transcriptional responses of Diabrotica virgifera virgifera larvae to benzoxazinoids. Journal of Applied Entomology 139, 416–423, https://doi.org/10.1111/jen.12193 (2015).
Coates, B. S. et al. Quantitative trait locus mapping and functional genomics of an organophosphate resistance trait in the western corn rootworm, Diabrotica virgifera virgifera. Insect Molecular Biology 25, 1–15, https://doi.org/10.1111/imb.12194 (2016).
Chu, C.-C. et al. Patterns of differential gene expression in adult rotation-resistant and wild-type western corn rootworm digestive tracts. Evolutionary Applications 8, 692–704, https://doi.org/10.1111/eva.12278 (2015).
Wang, H. et al. Patterns of gene expression in western corn rootworm (Diabrotica virgifera virgifera) neonates, challenged with Cry34Ab1, Cry35Ab1 and Cry34/35Ab1, based on next-generation sequencing. Toxins 9, 124 (2017).
Rault, L. C. et al. Investigation of Cry3Bb1 resistance and intoxication in western corn rootworm by RNA sequencing. Journal of Applied Entomology in press (2018).
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212, https://doi.org/10.1093/bioinformatics/btv351 (2015).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140, https://doi.org/10.1093/bioinformatics/btp616 (2010).
Kanehisa, M. & Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28, 27–30 (2000).
Gahan, L. J., Pauchet, Y., Vogel, H. & Heckel, D. G. An ABC transporter mutation is correlated with insect resistance to Bacillus thuringiensis Cry1Ac toxin. PLOS Genetics 6, e1001248 (2010).
Bravo, A. et al. Oligomerization triggers binding of a Bacillus thuringiensis Cry1Ab pore-forming toxin to aminopeptidase N receptor leading to insertion into membrane microdomains. Biochimica et Biophysica Acta (BBA) - Biomembranes 1667, 38–46 (2004).
Walters, F. S., Stacy, C. M., Lee, M. K., Palekar, N. & Chen, J. S. An engineered chymotrypsin/cathepsin G site in domain I renders Bacillus thuringiensis Cry3A active against western corn rootworm larvae. Applied and environmental microbiology 74, 367–374 (2008).
Loseva, O. et al. Changes in protease activity and Cry3Aa toxin binding in the Colorado potato beetle: implications for insect resistance to Bacillus thuringiensis toxins. Insect Biochemistry and Molecular Biology 32, 567–577 (2002).
Rausell, C., Ochoa-Campuzano, C., Martínez-Ramírez, A. C., Bravo, A. & Real, M. D. A membrane associated metalloprotease cleaves Cry3Aa Bacillus thuringiensis toxin reducing pore formation in Colorado potato beetle brush border membrane vesicles. Biochimica et Biophysica Acta (BBA) - Biomembranes 1768, 2293–2299 (2007).
Wei, J. et al. Activation of Bt protoxin Cry1Ac in resistant and susceptible cotton bollworm. PLoS ONE 11, e0156560, https://doi.org/10.1371/journal.pone.0156560 (2016).
Gunning, R. V., Dang, H. T., Kemp, F. C., Nicholson, I. C. & Moores, G. D. New resistance mechanism in Helicoverpa armigera threatens transgenic crops expressing Bacillus thuringiensis Cry1Ac toxin. Applied and Environmental Microbiology 71, 2558–2563, https://doi.org/10.1128/aem.71.5.2558-2563.2005 (2005).
Bonfini, A., Liu, X. & Buchon, N. From pathogens to microbiota: how Drosophila intestinal stem cells react to gut microbes. Developmental & Comparative Immunology 64, 22–38, https://doi.org/10.1016/j.dci.2016.02.008 (2016).
Castagnola, A. & Jurat-Fuentes, J. L. Intestinal regeneration as an insect resistance mechanism to entompathogenic bacteria. Current Opinion in Insect Science 15, 104–110 (2016).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics, https://doi.org/10.1093/bioinformatics/btu170 (2014).
Coates, B. S. Assembly and annotation of full mitochondrial genomes for the corn rootworm species, Diabrotica virgifera virgifera and Diabrotica barberi (Insecta: Coleoptera: Chrysomelidae), using Next Generation Sequence data. Gene 542, 190–197, https://doi.org/10.1016/j.gene.2014.03.035 (2014).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nature Methods 9, 357–359, https://doi.org/10.1038/nmeth.1923 (2012).
Grabherr, M. G. et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nature Biotechnology 29, 644–652 (2011).
Conesa, A. et al. Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21, 3674–3676 (2005).
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079, https://doi.org/10.1093/bioinformatics/btp352 (2009).
Ihaka, R. & Gentleman, R. R: A language for data analysis and graphics. Journal of Computational and Graphical Statistics 5, 299–314, https://doi.org/10.1080/10618600.1996.10474713 (1996).
Lawrence, M. et al. Software for computing and annotating genomic ranges. PLoS Computational Biology 9, e1003118, https://doi.org/10.1371/journal.pcbi.1003118 (2013).
McCarthy, D. J., Chen, Y. & Smyth, G. K. Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variation. Nucleic Acids Research 40, 4288–4297 (2012).
Zhou, X., Lindsay, H. & Robinson, M. D. Robustly detecting differential expression in RNA sequencing data using observation weights. Nucleic Acids Research 42, e91–e91, https://doi.org/10.1093/nar/gku310 (2014).
Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biology 11, R25, https://doi.org/10.1186/gb-2010-11-3-r25 (2010).
Quevillon, E. et al. InterProScan: protein domains identifier. Nucleic Acids Research 33, W116–W120, https://doi.org/10.1093/nar/gki442 (2005).
Acknowledgements
Julie Barry (USDA-ARS Plant Genetics Research Unit) maintained and provided WCR colonies. Michelle Gregory (USDA-ARS- BCIRL) assisted with the experiments. Syngenta Biotechnology provided the transgenic and near-isoline maize seeds. Colin Diesh, Deepak Unni and Justin Le Tourneau (Dr. Elsik’s lab, Division of Animal Sciences, University of Missouri) maintained the server and assisted with the bioinformatics software. Funding was provided by Syngenta Biotechnology through Cooperative Research and Development Award No. 58-3K95-4-1697 to USDA-ARS to BEH and KSS, and National Science Foundation Award No. 1615789 to TJ. Funding was also provided by the University of Missouri, Division of Plant Sciences and USDA-ARS for ZZ stipend.
Author information
Authors and Affiliations
Contributions
Z.Z. designed and performed the experiments, data analyses, and drafted the manuscript. L.N.M. co-wrote the initial funded proposal, and designed and helped to perform initial experiments. K.S.S. co-wrote the funded proposal, designed experiments, and assisted with data analysis, manuscript writing, and overseeing the project. B.E.H. co-wrote the initial proposal, designed experiments, and assisted in writing the manuscript. C.G.E. provided significant advice on bioinformatics approaches, and assisted with manuscript writing. T.J. provided significant guidance on the statistical analysis. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, Z., Meihls, L.N., Hibbard, B.E. et al. Differential gene expression in response to eCry3.1Ab ingestion in an unselected and eCry3.1Ab-selected western corn rootworm (Diabrotica virgifera virgifera LeConte) population. Sci Rep 9, 4896 (2019). https://doi.org/10.1038/s41598-019-41067-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-41067-7
- Springer Nature Limited
This article is cited by
-
A draft Diabrotica virgifera virgifera genome: insights into control and host plant adaption by a major maize pest insect
BMC Genomics (2023)
-
Up-regulation of apoptotic- and cell survival-related gene pathways following exposures of western corn rootworm to B. thuringiensis crystalline pesticidal proteins in transgenic maize roots
BMC Genomics (2021)
-
Functional validation of DvABCB1 as a receptor of Cry3 toxins in western corn rootworm, Diabrotica virgifera virgifera
Scientific Reports (2020)