Abstract
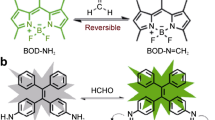
Formaldehyde (FA) is the simplest active carbonyl species that can be spontaneously produced in the body and plays important roles in human cognitive ability and spatial memory. However, excessive intake of FA may cause a series of diseases, including cancer, diabetes, heart and liver diseases and various neuropathies. Hence, the exploration of sensitive and fast detection methods for FA is crucial to understand and diagnose these diseases. Recently, fluorescent probes have been increasingly employed as powerful tools for detecting a broad range of different small molecules due to their high selectivity, rapid response, convenient operation and relatively non-invasive nature. Thus, we have developed two naphthalimide-based fluorescent probes for detecting FA in cells and in lysosomes. Compared with other FA fluorescent probes, these two probes have several advantages, including high sensitivity and selectivity, excellent two-photon properties and high signal-to-noise ratio. In this protocol, we provide detailed procedures for the synthesis of the two probes; characterization of their sensitivity, selectivity and stability in solution; and representative application procedures for detecting FA in living cells and mouse liver tissue slices. The protocol requires ~88 h to synthesize the probes, ~24 h to characterize the probes in solution and ~25 h to carry out the biological fluorescence imaging experiments in cells and liver tissue slices.









Similar content being viewed by others
Data availability
The data that support this study are available from the corresponding author on request.
References
Salthammer, T. Formaldehyde in the ambient atmosphere: from an indoor pollutant to an outdoor pollutant? Angew. Chem. Int. Ed. 52, 3320–3327 (2013).
Salthammer, T., Mentese, S. & Marutzky, R. Formaldehyde in the indoor environment. Chem. Rev. 110, 2536–2572 (2010).
World Health Organization. Guidelines for Drinking-Water Quality 2nd edn, Vol. 2: Health Criteria and Other Supporting Information (World Health Organization, 1993).
Bruemmer, K. J., Brewer, T. F. & Chang, C. J. Fluorescent probes for imaging formaldehyde in biological systems. Curr. Opin. Chem. Biol. 39, 17–23 (2017).
Kalasz, H. Biological role of formaldehyde, and cycles related to methylation, demethylation, and formaldehyde production. Mini. Rev. Med. Chem. 3, 175–192 (2003).
O’Sullivan, J. et al. Semicarbazide-sensitive amine oxidases: enzymes with quite a lot to do. Neurotoxicology 25, 303–315 (2004).
Ai, L. et al. Endogenous formaldehyde is a memory-related molecule in mice and humans. Commun. Biol. 2, 446 (2019); correction 2, 479 (2019).
Songur, A., Ozen, O. A. & Sarsilmaz, M. The toxic effects of formaldehyde on the nervous system. Rev. Environ. Contam. Toxicol. 203, 105–118 (2010).
Tulpule, K. & Dringen, R. Formaldehyde in brain: an overlooked player in neurodegeneration? J. Neurochem. 127, 7–21 (2013).
Jones, P. A. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 13, 484–492 (2012).
Tong, Z. Q. et al. Accumulated hippocampal formaldehyde induces age-dependent memory decline. Age 35, 583–596 (2013).
Baan, R., Grosse, Y. & Straif, K. A review of human carcinogens–part F: chemical agents and related occupations. Lancet Oncol. 10, 13–14 (2009).
Cao, D. et al. Coumarin-based small-molecule fluorescent chemosensors. Chem. Rev. 119, 10403–10519 (2019); correction 119, 11550–11550 (2019).
Chan, J., Dodani, S. C. & Chang, C. J. Reaction-based small-molecule fluorescent probes for chemoselective bioimaging. Nat. Chem. 4, 973–984 (2012).
Wu, D. et al. Fluorescent chemosensors: the past, present and future. Chem. Soc. Rev. 46, 7105–7123 (2017).
Brewer, T. F. & Chang, C. J. An aza-Cope reactivity-based fluorescent probe for imaging formaldehyde in living cells. J. Am. Chem. Soc. 137, 10886–10889 (2015).
Roth, A. et al. A reaction-based fluorescent probe for imaging of formaldehyde in living cells. J. Am. Chem. Soc. 137, 10890–10893 (2015).
Tang, Y. H. et al. Strategies for designing organic fluorescent probes for biological imaging of reactive carbonyl species. Chem. Soc. Rev. 48, 4036–4048 (2019).
Ohata, J., Bruemmer, K. J. & Chang, C. J. Activity-based sensing methods for monitoring the reactive carbon species carbon monoxide and formaldehyde in living systems. Acc. Chem. Res. 52, 2841–2848 (2019).
Xu, Z. Q. et al. Recent advances in formaldehyde-responsive fluorescent probes. Chin. Chem. Lett. 28, 1935–1942 (2017).
Heck, H. D., White, E. L. & Casanovaschmitz, M. Determination of formaldehyde in biological tissues by gas chromatography/mass spectrometry. J. Mass Spectrom. 9, 347–353 (1982).
Tang, Y. H. et al. Development of a two-photon fluorescent probe for imaging of endogenous formaldehyde in living tissues. Angew. Chem. Int. Ed. 55, 3356–3359 (2016).
Tang, Y. H. et al. Lysosome-targeted turn-on fluorescent probe for endogenous formaldehyde in living cells. Anal. Chem. 88, 9359–9363 (2016).
Kato, S., Burke, P. J. & Koch, T. H. Formaldehyde in human cancer cells: detection by preconcentration-chemical ionization mass spectrometry. Anal. Chem. 73, 2992–2997 (2001).
Lee, M. H. et al. Naphthalimide trifluoroacetyl acetonate: a hydrazine-selective chemodosimetric sensor. Chem. Sci. 4, 4121–4126 (2013).
Liu, X. L. et al. Ratiometric two-photon fluorescent probes for mitochondrial hydrogen sulfide in living cells. J. Org. Chem. 79, 9481–9489 (2014).
Yu, H., Xiao, Y. & Jin, L. A lysosome-targetable and two-photon fluorescent probe for monitoring endogenous and exogenous nitric oxide in living cells. J. Am. Chem. Soc. 134, 17486–17489 (2012).
Badii, F. & Howell, N. K. Elucidation of the effect of formaldehyde and lipids on frozen stored cod collagen by FT-Raman spectroscopy and differential scanning calorimetry. J. Agric. Food Chem. 51, 1440–1446 (2003).
Xie, Z. D. et al. A highly selective two-photon fluorogenic probe for formaldehyde and its bioimaging application in cells and zebrafish. Sens. Actuator B Chem. 241, 1050–1056 (2017).
Liu, W. et al. A reactivity-based [18 F]FDG probe for in vivo formaldehyde imaging using positron emission tomography. Chem. Sci. 7, 5503–5507 (2016).
Song, H., Rajendiran, S. & Kim, N. A tailor designed fluorescent ‘turn-on’ sensor of formaldehyde based on the BODIPY motif. Tetrahedron Lett. 53, 4913–4916 (2012).
Zhou, W. et al. HCHO-reactive molecule with dual-emission-enhancement property for quantitatively detecting HCHO in near 100% water solution. Sens. Actuator B Chem. 209, 664–669 (2015).
Achmann, S., Hämmerle, M. & Moos, R. Amperometric enzyme-based biosensor for direct detection of formaldehyde in the gas phase: dependence on electrolyte composition. Electroanalysis 20, 410–417 (2008).
Feng, L., Musto, C. J. & Suslick, K. S. A simple and highly sensitive colorimetric detection method for gaseous formaldehyde. J. Am. Chem. Soc. 132, 4046–4047 (2010).
Takeuchi, A. et al. Determination of formaldehyde in urine by headspace gas chromatography. Bull. Environ. Contam. Toxicol. 79, 1–4 (2007).
Beasley, R. K. et al. Sampling of formaldehyde in air with coated solid sorbent and determination by high performance liquid chromatography. Anal. Chem. 52, 1110–1114 (1980).
Xu, J. et al. A simple naphthalene-based fluorescent probe for high selective detection of formaldehyde in toffees and HeLa cells via aza-Cope reaction. Talanta 160, 645–652 (2016).
Li, J. et al. A two-photon fluorescent probe for bio-imaging of formaldehyde in living cells and tissues. Analyst 141, 3395–3402 (2016).
Bruemmer, K. J. et al. Chemiluminescent probes for activity-based sensing of formaldehyde released from folate degradation in living mice. Angew. Chem. Int. Ed. 57, 7508–7512 (2018).
Liu, C. X. et al. Nanomolar fluorescent quantitative detection of formaldehyde with a 8-hydroxyquinoline derivative in aqueous solution and electrospun nanofibers. Sens. Actuator B Chem. 219, 185–191 (2015).
He, L. W. et al. A ratiometric fluorescent formaldehyde probe for bioimaging application. Chem. Commun. 52, 4029–4032 (2016).
Brewer, T. F. et al. A 2-aza-Cope reactivity-based platform for ratiometric fluorescence imaging of formaldehyde in living cells. Chem. Sci. 8, 4073–4081 (2017).
Ma, Y. Y. et al. Development of a unique reversible fluorescent probe for tracking endogenous sulfur dioxide and formaldehyde fluctuation in vivo. Chem. Commun. 55, 11263–11266 (2019).
Ma, Y. Y. et al. Rational design of a reversible fluorescent probe for sensing sulfur dioxide/formaldehyde in living cells, zebrafish, and living mice. Anal. Chem. 91, 10723–10730 (2019).
Brendel, K. et al. Precision cut tissue slices in culture: a new tool in pharmacology. Proc. West. Pharmacol. Soc. 30, 291–293 (1987).
Gandolfi, A. J., Wijeweera, J. & Brendel, K. Use of precision-cut liver slices as an in vitro tool for evaluating liver function. Toxicol. Pathol. 24, 58–61 (1996).
Acknowledgements
This work was financially supported by NSFC (21472067, 21672083 and 21877048), the Taishan Scholar Foundation (TS201511041) and the startup fund of the University of Jinan (309-10004).
Author information
Authors and Affiliations
Contributions
W.L. directed the research and conceived the project with Y. T. The experiments were performed by Y. T. and Y. Z. All the authors analyzed the data and contributed to the manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks Zhiqian Tong and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Tang, Y. et al. Angew. Chem. Int. Ed. 55, 3356–3359 (2016): https://onlinelibrary.wiley.com/doi/10.1002/anie.201510373
Tang, Y. et al. Anal. Chem. 88, 9359–9363 (2016): https://pubs.acs.org/doi/10.1021/acs.analchem.6b02879
Supplementary information
Supplementary Information
Supplementary Figs. 1–15.
Rights and permissions
About this article
Cite this article
Tang, Y., Zhao, Y. & Lin, W. Preparation of robust fluorescent probes for tracking endogenous formaldehyde in living cells and mouse tissue slices. Nat Protoc 15, 3499–3526 (2020). https://doi.org/10.1038/s41596-020-0384-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-020-0384-7
- Springer Nature Limited
This article is cited by
-
Preparation of near-infrared AIEgen-active fluorescent probes for mapping amyloid-β plaques in brain tissues and living mice
Nature Protocols (2023)
-
Imaging in experimental models of diabetes
Acta Diabetologica (2022)
-
Recent Progress in Fluorescent Formaldehyde Detection Using Small Molecule Probes
Journal of Analysis and Testing (2022)