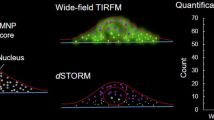
Abstract
A comprehensive understanding of interactions between nanoparticles (NPs) and biological components is critical to the clinical application of NPs and nanomedicine. Here we provide a step-by-step correlative imaging approach to investigate plasmonic NPs of different aggregation states at the single-cell level. Traceable spherical nucleic acids (SNAs) are fabricated by decorating 50-nm spherical gold NPs with fluorophore-labeled DNA, serving as dually emissive (fluorescent and plasmonic) NPs. The in situ correlative imaging with dark-field microscopy (DFM) and fluorescence microscopy (FM) reveals intracellular distribution of SNAs, whereas DFM combined with scanning electron microscopy (SEM) allows semi-quantification of SNA clustering states in solution. The imaging data are analyzed by ImageJ and a colorimetry-based algorithm written in Python. The clustering states of SNAs in a single cell can be efficiently distinguished within 20 s. This method can be readily installed to monitor real-time endocytosis and cellular distribution of plasmonic NPs of different aggregation states and to quantitatively image targets of interest (e.g., specific DNA, messenger RNA, peptides or proteins) in living cells. The entire procedure can be completed in 3–5 d and requires standard DFM, FM and SEM imaging and data analysis skills and equipment.








Similar content being viewed by others
Data availability
The data that support the findings of this study are provided in the article and its Supplementary Information files. Additional data are available from the corresponding author upon reasonable request. The primary data for Figs. 5c and 8b are provided as Source Data files with this protocol, and the primary data for Supplementary Figs. 1, 5 and 7b are provided as Supplementary Data 1, 2 and 3, respectively.
Code availability
The Python scripts for image analysis and aggregation classification of SNAs are available as Supplementary Software 1 of this protocol.
References
Cui, Z. et al. Nanodiamond autophagy inhibitor allosterically improves the arsenical-based therapy of solid tumors. Nat. Commun. 9, 4347 (2018).
Jiang, D. W. et al. DNA origami nanostructures can exhibit preferential renal uptake and alleviate acute kidney injury. Nat. Biomed. Eng. 2, 865–877 (2018).
Shi, B. et al. Theranostic nanoplatform with hydrogen sulfide activatable NIR responsiveness for imaging-guided on-demand drug release. Angew. Chem. Int. Ed. Engl. 58, 16826–16830 (2019).
Tang, W. et al. Emerging blood-brain-barrier-crossing nanotechnology for brain cancer theranostics. Chem. Soc. Rev. 48, 2967–3014 (2019).
Yu, G. C. et al. A discrete organoplatinum(II) metallacage as a multimodality theranostic platform for cancer photochemotherapy. Nat. Commun. 9, 4335 (2018).
Yamankurt, G. et al. Exploration of the nanomedicine-design space with high-throughput screening and machine learning. Nat. Biomed. Eng. 3, 318–327 (2019).
Ioannidis, J. P. A., Kim, B. Y. S. & Trounson, A. How to design preclinical studies in nanomedicine and cell therapy to maximize the prospects of clinical translation. Nat. Biomed. Eng. 2, 797–809 (2018).
van der Meel, R. et al. Smart cancer nanomedicine. Nat. Nanotechnol. 14, 1007–1017 (2019).
Chinen, A. B., Guan, C. X. M., Ko, C. H. & Mirkin, C. A. The impact of protein corona formation on the macrophage cellular uptake and biodistribution of spherical nucleic acids. Small 13, 1603847 (2017).
Li, H. et al. Molecular spherical nucleic acids. Proc. Natl Acad. Sci. USA 115, 4340–4344 (2018).
Rees, P., Wills, J. W., Brown, M. R., Barnes, C. M. & Summers, H. D. The origin of heterogeneous nanoparticle uptake by cells. Nat. Commun. 10, 2341 (2019).
Hinde, E. et al. Pair correlation rnicroscopy reveals the role of nanoparticle shape in intracellular transport and site of drug release. Nat. Nanotechnol. 12, 81–89 (2017).
Huang, Z. N., Cole, L. E., Callmann, C. E., Wang, S. & Mirkin, C. A. Sequence multiplicity within spherical nucleic acids. ACS Nano 14, 1084–1092 (2020).
Liu, M. et al. Real-time visualization of clustering and intracellular transport of gold nanoparticles by correlative imaging. Nat. Commun. 8, 15646 (2017).
Behzadi, S. et al. Cellular uptake of nanoparticles: journey inside the cell. Chem. Soc. Rev. 46, 4218–4244 (2017).
Xie, X. et al. Unraveling cell-type-specific targeted delivery of membrane-camouflaged nanoparticles with plasmonic imaging. Nano Lett. 20, 5228–5235 (2020).
Shen, J. et al. Fractal nanoplasmonic labels for supermultiplex imaging in single cells. J. Am. Chem. Soc. 141, 11938–11946 (2019).
Borkowska, M. et al. Targeted crystallization of mixed-charge nanoparticles in lysosomes induces selective death of cancer cells. Nat. Nanotechnol. 15, 331–341 (2020).
Wu, X. C. A., Choi, C. H. J., Zhang, C., Hao, L. L. & Mirkin, C. A. Intracellular fate of spherical nucleic acid nanoparticle conjugates. J. Am. Chem. Soc. 136, 7726–7733 (2014).
Narayan, S. P. et al. The sequence-specific cellular uptake of spherical nucleic acid nanoparticle conjugates. Small 11, 4173–4182 (2015).
Skakuj, K. et al. Conjugation chemistry-dependent T-cell activation with spherical nucleic acids. J. Am. Chem. Soc. 140, 1227–1230 (2018).
Sita, T. L. et al. Dual bioluminescence and near-infrared fluorescence monitoring to evaluate spherical nucleic acid nanoconjugate activity in vivo. Proc. Natl Acad. Sci. USA 114, 4129–4134 (2017).
Choi, C. H. J., Hao, L., Narayan, S. P., Auyeung, E. & Mirkin, C. A. Mechanism for the endocytosis of spherical nucleic acid nanoparticle conjugates. Proc. Natl Acad. Sci. USA 110, 7625–7630 (2013).
Li, K. et al. DNA-directed assembly of gold nanohalo for quantitative plasmonic imaging of single-particle catalysis. J. Am. Chem. Soc. 137, 4292–4295 (2015).
Liu, M. M. et al. Automated nanoplasmonic analysis of spherical nucleic acids clusters in single cells. Anal. Chem. 92, 1333–1339 (2020).
Jiang, Y. et al. Aptamer/AuNP biosensor for colorimetric profiling of exosomal proteins. Angew. Chem. Int. Ed. Engl. 56, 11916–11920 (2017).
Lee, K., Cui, Y., Lee, L. P. & Irudayaraj, J. Quantitative imaging of single mRNA splice variants in living cells. Nat. Nanotechnol. 9, 474–480 (2014).
Grigolato, F., Colonabo, C., Ferrari, R., Rezabkova, L. & Arosio, P. Mechanistic origin of the combined effect of surfaces and mechanical agitation on amyloid formation. ACS Nano 11, 11358–11367 (2017).
Dominguez-Medina, S. et al. Adsorption and unfolding of a single protein triggers nanoparticle aggregation. ACS Nano 10, 2103–2112 (2016).
Xu, F. et al. Lipid-mediated targeting with membrane-wrapped nanoparticles in the presence of corona formation. ACS Nano 10, 1189–1200 (2016).
Sugikawa, K., Kadota, T., Yasuhara, K. & Ikeda, A. Anisotropic self-assembly of citrate-coated gold nanoparticles on fluidic liposomes. Angew. Chem. 128, 4127–4131 (2016).
Yu, X. et al. Dressing up nanoparticles: a membrane wrap to induce formation of the virological synapse. ACS Nano 9, 4182–4192 (2015).
Choi, C. K. K. et al. Dopamine-mediated assembly of citrate-capped plasmonic nanoparticles into stable core-shell nanoworms for intracellular applications. ACS Nano 13, 5864–5884 (2019).
Sindhwani, S. et al. The entry of nanoparticles into solid tumours. Nat. Mater. 19, 566–575 (2020).
Cheng, X. J. et al. Light-triggered assembly of gold nanoparticles for photothermal therapy and photoacoustic imaging of tumors in vivo. Adv. Mater. 29, 1604894 (2017).
Dai, Q. et al. Quantifying the ligand-coated nanoparticle delivery to cancer cells in solid tumors. ACS Nano 12, 8423–8435 (2018).
Tan, S. F. et al. Real-time imaging of the formation of Au–Ag core-shell nanoparticles. J. Am. Chem. Soc. 138, 5190–5193 (2016).
Morimura, H. et al. Nano-analysis of DNA conformation changes induced by transcription factor complex binding using plasmonic nanodimers. ACS Nano 7, 10733–10740 (2013).
Visser, E. W. A., Horaacek, M. & Zijlistra, P. Plasmon rulers as a probe for real-time microsecond conformational dynamics of single molecules. Nano Lett. 18, 7927–7934 (2018).
Readman, C. et al. Anomalously large spectral shifts near the quantum tunnelling limit in plasmonic rulers with subatomic resolution. Nano Lett. 19, 2051–2058 (2019).
Chen, W., Zhang, S. P., Deng, Q. & Xu, H. X. Probing of sub-picometer vertical differential resolutions using cavity plasmons. Nat. Commun. 9, 801 (2018).
Syed, A. M. et al. Three-dimensional imaging of transparent tissues via metal nanoparticle labeling. J. Am. Chem. Soc. 139, 9961–9971 (2017).
Wang, K. et al. In situ detection and imaging of telomerase activity in cancer cell lines via disassembly of plasmonic core–satellites nanostructured probe. Anal. Chem. 89, 7262–7268 (2017).
Wu, D., Chen, Y., Hou, S., Fang, W. & Duan, H. Intracellular and cellular detection by SERS-active plasmonic nanostructures. Chembiochem 20, 2432–2441 (2019).
Nam, W. et al. Refractive-index-insensitive nanolaminated SERS substrates for label-free Raman profiling and classification of living cancer cells. Nano Lett 19, 7273–7281 (2019).
Zhang, Z. Q. et al. Quantitative evaluation of surface-enhanced Raman scattering nanoparticles for intracellular pH sensing at a single particle level. Anal. Chem. 91, 3254–3262 (2019).
Ando, J., Fujita, K., Smith, N. I. & Kawata, S. Dynamic SERS imaging of cellular transport pathways with endocytosed gold nanoparticles. Nano Lett. 11, 5344–5348 (2011).
Mayer, K. M. & Hafner, J. H. Localized surface plasmon resonance sensors. Chem. Rev. 111, 3828–3857 (2011).
Chen, Y., Xianyu, Y. & Jiang, X. Surface modification of gold nanoparticles with small molecules for biochemical analysis. Acc. Chem. Res. 50, 310–319 (2017).
Ye, S., Li, X., Wang, M. & Tang, B. Fluorescence and SERS imaging for the simultaneous absolute quantification of multiple miRNAs in living cells. Anal. Chem. 89, 5124–5130 (2017).
Dong, B. et al. Reversible self-assembly of nanoprobes in live cells for dynamic intracellular pH imaging. ACS Nano 13, 1421–1432 (2019).
Halo, T. L. et al. NanoFlares for the detection, isolation, and culture of live tumor cells from human blood. Proc. Natl Acad. Sci. USA 111, 17104–17109 (2014).
Wu, Y. et al. Gold nanoparticle loaded split-DNAzyme probe for amplified miRNA detection in living cells. Anal. Chem. 89, 8377–8383 (2017).
Li, D., Zhou, W., Yuan, R. & Xiang, Y. A DNA-fueled and catalytic molecule machine lights up trace under-expressed microRNAs in living cells. Anal. Chem. 89, 9934–9940 (2017).
Carnevale, K. J. F., Riskowski, R. A. & Strouse, G. F. A gold nanoparticle bio-optical transponder to dynamically monitor intracellular pH. ACS Nano 12, 5956–5968 (2018).
Yang, Y. et al. FRET nanoflares for intracellular mRNA detection: avoiding false positive signals and minimizing effects of system fluctuations. J. Am. Chem. Soc. 137, 8340–8343 (2015).
Bastús, N. G., Comenge, J. & Puntes, V. Kinetically controlled seeded growth synthesis of citrate-stabilized gold nanoparticles of up to 200 nm: size focusing versus Ostwald ripening. Langmuir 27, 11098–11105 (2011).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 9, 676–682 (2012).
Cutler, J. I., Auyeung, E. & Mirkin, C. A. Spherical nucleic acids. J. Am. Chem. Soc. 134, 1376–1391 (2012).
Zhang, X., Servos, M. R. & Liu, J. Instantaneous and quantitative functionalization of gold nanoparticles with thiolated DNA using a pH-assisted and surfactant-free route. J. Am. Chem. Soc. 134, 7266–7269 (2012).
Liu, B. & Liu, J. Freezing directed construction of bio/nano interfaces: reagentless conjugation, denser spherical nucleic acids, and better nanoflares. J. Am. Chem. Soc. 139, 9471–9474 (2017).
Lee, H. E. et al. Amino-acid- and peptide-directed synthesis of chiral plasmonic gold nanoparticles. Nature 556, 360–365 (2018).
Hakala, T. K. et al. Lasing in dark and bright modes of a finite-sized plasmonic lattice. Nat. Commun. 8, 13687 (2017).
Fang, W. et al. Quantizing single-molecule surface-enhanced Raman scattering with DNA origami metamolecules. Sci. Adv. 5, eaau4506 (2019).
Acknowledgements
We thank the National Natural Science Foundation of China (91953106, 21904041 and 21804088) and the China Postdoctoral Science Foundation (2019M661417) for support.
Author information
Authors and Affiliations
Contributions
Q.L. conceived the study. M.L., F.W., C.F. and Q.L. designed experiments. M.L. and F.W. performed experiments. X.Z. assisted with cellular culture and FM imaging. F.W. and X.M. assisted with writing the Python scripts. M.L., F.W., L.W., Y.T., C.F. and Q.L. analyzed data. M.L. and Q.L. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Protocols thanks Ramsey Majzoub, Bo Tang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol:
Liu, M. et al. Nat. Commun. 8, 15646 (2017): https://doi.org/10.1038/ncomms15646
Liu, M. et al. Anal. Chem. 92, 1333–1339 (2020): https://pubs.acs.org/doi/10.1021/acs.analchem.9b04500
Xie, X. et al. Nano Lett. 20, 5228–5235 (2020): https://doi.org/10.1021/acs.nanolett.0c01503
Supplementary information
Supplementary Information
Supplementary Figs. 1–7, Supplementary Tables 1 and 2, Supplementary Method 1 and Supplementary Note 1.
Supplementary Software 1
It contains five Python scripts, a .doc file giving a detailed protocol on how to run the five Python scripts and a file folder containing test data.
Supplementary Data 1
Statistical Source Data of Supplementary Fig 1.
Supplementary Data 2
Statistical source data of Supplementary Fig 5.
Supplementary Data 3
Statistical source data of Supplementary Fig 7.
Source data
Source Data Fig. 5
Statistical source data of Fig. 5c.
Source Data Fig. 8
Statistical source data of Fig. 8b.
Rights and permissions
About this article
Cite this article
Liu, M., Wang, F., Zhang, X. et al. Tracking endocytosis and intracellular distribution of spherical nucleic acids with correlative single-cell imaging. Nat Protoc 16, 383–404 (2021). https://doi.org/10.1038/s41596-020-00420-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-020-00420-1
- Springer Nature Limited
This article is cited by
-
Protein transfection via spherical nucleic acids
Nature Protocols (2022)