Abstract
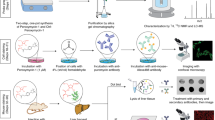
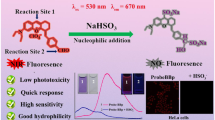
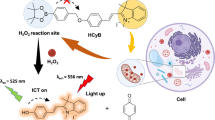
Hypochlorous acid (HOCl) is a critical member of the reactive oxygen species (ROS) produced by immune cells to fight infections. On the other hand, HOCl in homeostasis causes oxidative damage to biomolecules and is linked to many diseases, including inflammatory, neurodegenerative, and cardiovascular diseases. Herein, we detail a procedure for the preparation of a boron-dipyrromethene (BODIPY)-derived fluorescent probe for HOCl (BClO) and its application as an imaging reagent in living cells. BClO is synthesized in one pot through a four-step procedure that is nearly the same as that for conventional BODIPY dye preparation, except for the ratio of starting materials. BClO has an extremely rapid response (saturated within seconds) and is ultrasensitive to HOCl. The detection limit of BClO reaches the subnanomolar range, which is the highest HOCl sensitivity to date. Taking advantage of the ultrasensitive character of BClO, we have previously demonstrated its ability to detect endogenous HOCl generated by macrophages and shown that it can also be used to discriminate cancer cell lines (which show high HOCl production) from non-cancer cell lines (which show low HOCl production). The protocol requires ~2 d for probe synthesis and up to ~18 h for fluorescence imaging and flow cytometry assays.





Similar content being viewed by others
References
Murphy, M. P. et al. Unraveling the biological roles of reactive oxygen species. Cell Metab. 13, 361–366 (2011).
Finkel, T. Oxidant signals and oxidative stress. Curr. Opin. Cell Biol. 15, 247–254 (2003).
Dupré-Crochet, S., Erard, M. & Nüße, O. ROS production in phagocytes: why, when, and where? J. Leukoc. Biol. 94, 657–670 (2017).
Harrison, J. E. & Schultz, J. Studies on the chlorinating activity of myeloperoxidase. J. Biol. Chem. 251, 1371–1374 (1976).
Hampton, M. B., Kettle, A. J. & Winterbourn, C. C. Inside the neutrophil phagosome: oxidants, myeloperoxidase, and bacterial killing. Blood 92, 3007–3017 (1998).
Winterbourn, C. C., Hampton, M. B. & Kettle, A. J. Modeling the reactions of superoxide and myeloperoxidase in the neutrophil phagosome. J. Biol. Chem. 281, 39860–39869 (2006).
Steinbeck, M. J. et al. Myeloperoxidase and chlorinated peptides in osteoarthritis: potential biomarkers of the disease. J. Orthop. Res. 25, 1128–1135 (2007).
Pattison, D. I. & Davies, M. J. Reactions of myeloperoxidase-derived oxidants with biological substrates: gaining chemical insight into human inflammatory diseases. Curr. Med. Chem. 13, 3271–3290 (2006).
Andersen, J. K. Oxidative stress in neurodegeneration: cause or consequence? Nat. Rev. Neurosci. 10, S18–S25 (2004).
Nicholls, S. J. & Hazen, S. L. Myeloperoxidase and cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 25, 1102–1111 (2005).
Güngör, N., Knaapen, A. M. & van Schooten, F. J. Genotoxic effects of neutrophils and hypochlorous acid. Mutagenesis 25, 149–154 (2010).
London, S. J., Lehman, T. A. & Taylor, J. A. Myeloperoxidase genetic polymorphism and lung cancer risk. Cancer Res. 57, 5001–5003 (1997).
Soo, C. C., Haqquani, A. S. & Birnboim, H. C. Dose dependent effects of dietary alpha- and gamma-tocopherols on genetic instability in mouse mutatect tumors. J. Natl. Cancer Inst. 96, 796–800 (2004).
Zhu, H., Fan, J. & Peng, X. An “enhanced PET”-based fluorescent probe with ultrasensitivity for imaging basal and elesclomol-induced HClO in cancer cells. J. Am. Chem. Soc. 136, 12820–12823 (2014).
Zhang, R., Song, B. & Yuan, J. Bioanalytical methods for hypochlorous acid detection: recent advances and challenges. Trends Anal. Chem. 99, 1–33 (2018).
Klebanoff, S. J. Myeloperoxidase: friend and foe. J. Leukoc. Biol. 77, 598–625 (2005).
Winterbourn, C. C. Reconciling the chemistry and biology of reactive oxygen species. Nat. Chem. Biol. 4, 278–286 (2008).
Howard, J. K. & Smith, J. A. The oxidation of pyrrole. Chem. Asian J. 11, 155–167 (2016).
Boens, N. & Dehaen, W. Fluorescent indicators based on BODIPY. Chem. Soc. Rev. 41, 1130–1172 (2012).
Zhang, Y. et al. Recent progress in the development of fluorescent probes for the detection of hypochlorous acid. Sens. Actuators B 240, 18–36 (2017).
Chen, X. et al. Recent progress in the development of fluorescent, luminescent and colorimetric probes for detection of reactive oxygen and nitrogen species. Chem. Soc. Rev. 45, 2976–3016 (2016).
Zhang, Z., Fan, J. & Peng, X. Fluorescence completely separated ratiometric probe for HClO in lysosomes. Sens. Actuators B 246, 293–299 (2017).
Zhang, H. et al. Imaging lysosomal highly reactive oxygen species and lighting up cancer cells and tumors enabled by a Si-rhodamine-based near-infrared fluorescent probe. Biomaterials 133, 60–69 (2017).
Chen, X. & Yoon, J. Synthesis of a highly HOCl-selective fluorescent probe and its use for imaging HOCl in cells and organisms. Nat. Protoc. 11, 1219–1228 (2016).
Zhang, W. et al. Reversible two-photon fluorescent probe for imaging of hypochlorous acid in live cells and in vivo. Chem. Commun. 51, 10150–10153 (2015).
Yuan, L. et al. Development of targetable two-photon fluorescent probes to image hypochlorous acid in mitochondria and lysosome in live cell and inflamed mouse model. J. Am. Chem. Soc. 137, 5930–5938 (2015).
Xu, Q. & Yoon, J. A highly specific fluorescent probe for hypochlorous acid and its application in imaging microbe-induced HOCl production. J. Am. Chem. Soc. 135, 9944–9949 (2013).
Sun, Z.-N. & Yang, D. A highly specific BODIPY-based fluorescent probe for the detection of hypochlorous acid. Org. Lett. 10, 2171–2174 (2008).
Kenmoku, S. & Nagano, T. Development of a highly specific rhodamine-based fluorescence probe for hypochlorous acid and its application to real-time imaging of phagocytosis. J. Am. Chem. Soc. 129, 7317–7318 (2007).
Shepherd, J. & Libby, P. A fluorescent probe for the detection of myeloperoxidase activity in atherosclerosis-associated macrophages. Chem. Biol. 14, 1221–1231 (2007).
Toyokuni, S. et al. Persistent oxidative stress in cancer. FEBS Lett. 358, 1–3 (1995).
Hileman, E. O. et al. Intrinsic oxidative stress in cancer cells: a biochemical basis for therapeutic selectivity. Cancer Chemother. Pharmacol. 53, 209–219 (2004).
Pelicano, H., Carney, D. & Huang, P. ROS stress in cancer cells and therapeutic implications. Drug Resist. Updat. 7, 97–110 (2004).
Szatrowski, T. P. & Nathan, C. F. Production of large amounts of hydrogen peroxide by human tumor cells. Cancer Res. 51, 794–798 (1991).
Behrend, L. et al. Reactive oxygen species in oncogenic transformation. Biochem. Soc. Trans. 31, 1441–1444 (2003).
Kirshner, J. R. et al. Elesclomol induces cancer cell apoptosis through oxidative stress. J. Mol. Cancer Ther. 7, 2319–2327 (2008).
Acknowledgements
This work was financially supported by the National Science Foundation of China (21576037, 21406028, 21703025, and 21421005) and the NSFC-Liaoning United Fund (U1608222).
Author information
Authors and Affiliations
Contributions
H.Z. and Z.Z. contributed equally to this work; they designed and performed experiments, analyzed data, and wrote the paper; S.L. and J.D. designed and performed experiments; J.F. and X.P. discussed the results and commented on the manuscript at all stages.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key reference using this protocol
1. Zhu, H. et al. J. Am. Chem. Soc. 136, 12820–12823 (2014): https://doi.org/10.1021/ja505988g
Integrated supplementary information
Supplementary Figure 1 Absorption spectra of BClO upon NaOCl titration.
NaOCl concentrations vary ranging from 0 to 6 µM. The treatment of HOCl elicits tiny changes in the absorbance of BClO.
Supplementary Figure 2 Fluorescence spectra of BClO upon NaOCl titration.
NaOCl concentrations (from bottom to top): 0, 2, 4, 6, 8, 10, 12, 14, and 16 nM. Excitation at 480 nm.
Supplementary Figure 3 HOCl responsiveness of BClO at varied pH values.
(from left to right): 4.00, 4.53, 5.02, 5.52, 5.98, 6.51, 6.98, 7.40, 7.98, 8.51, and 9.02. Data represent mean values of fluorescence intensity ± standard derivation (n =3). Blue, red, and black dots represent data from 3 independent replicate experiments.
Supplementary Figure 4 Cell uptake of BClO in COS-7 and HeLa cells.
COS-7 (a) and HeLa (b) cells were incubated with BClO at 37 ºC, and fluorescence images were recorded at different time points: 0, 5, 10, and 20 min. After 20 min, BClO was completely activated by adding 12 µM of NaOCl. *: NaOCl (+). Scale bar is 200 µm. The intrinsic fluorescence of BClO in HeLa cells was higher than that in COS-7 cells, which is consistent with the result shown in Figure 4. After complete activation by NaOCl, BClO showed identical fluorescence in COS-7 and HeLa cells, suggesting its unbiased cell uptake in these cells.
Supplementary Figure 5 Fluorescence intensities extracted from ROIs in Supplementary Figure 4.
Data represent mean values of fluorescence intensity ± standard derivation (n=30).
Supplementary Figure 6 Flow cytometry analysis of BClO fluorescence in 6 types of living cells.
Three replicates were carried out. Representative results are shown. FSC: forward-scattered light; SSC: side-scattered light. Fluorescence signals were collected at 530 ± 30 nm upon excitation at 488 nm.
Supplementary Figure 7 Fluorescence imaging of endogenously produced HOCl in Raw 264.7 macrophages.
a) Raw 264.7 macrophages were stained with BClO only. b) Raw 264.7 macrophages were treated with LPS (1 µg/mL) for 24 h and PMA (1 µg/mL) for 1 h, followed by the incubation with BClO (1 µM) for 20 min.14.
Supplementary Figure 8 Time-dependent HOCl generation induced by elesclomol in live MCF-7 cells.
After the addition of elesclomol (2 µM) to MCF-7 cells that were prestained with BClO (1 µM), fluorescence images were recorded at different time points: a) 0 min; b) 30 min; c) 60 min; d) 90 min; e) 120 min; f) 180 min.14.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhu, H., Zhang, Z., Long, S. et al. Synthesis of an ultrasensitive BODIPY-derived fluorescent probe for detecting HOCl in live cells. Nat Protoc 13, 2348–2361 (2018). https://doi.org/10.1038/s41596-018-0041-6
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0041-6
- Springer Nature Limited
This article is cited by
-
Development of a Coumarin-derived Fluorescent Probe for Detection of HOCl and its Application in Cells and Zebrafish
Journal of Fluorescence (2024)
-
Activatable fluorescent probes for imaging and diagnosis of rheumatoid arthritis
Military Medical Research (2023)
-
In situ orderly self-assembly strategy affording NIR-II-J-aggregates for in vivo imaging and surgical navigation
Nature Communications (2023)
-
Preparation of near-infrared AIEgen-active fluorescent probes for mapping amyloid-β plaques in brain tissues and living mice
Nature Protocols (2023)
-
A fluorescent probe operating under weak acidic conditions for the visualization of HOCl in solid tumors in vivo
Science China Chemistry (2020)