Abstract
The increased legal availability of cannabis has led to a common misconception that it is a safe natural remedy for, among others, pregnancy-related ailments such as morning sickness. Emerging clinical evidence, however, indicates that prenatal cannabis exposure (PCE) predisposes offspring to various neuropsychiatric disorders linked to aberrant dopaminergic function. Yet, our knowledge of how cannabis exposure affects the maturation of this neuromodulatory system remains limited. Here, we show that male, but not female, offspring of Δ9-tetrahydrocannabinol (THC)-exposed dams, a rat PCE model, exhibit extensive molecular and synaptic changes in dopaminergic neurons of the ventral tegmental area, including altered excitatory-to-inhibitory balance and switched polarity of long-term synaptic plasticity. The resulting hyperdopaminergic state leads to increased behavioral sensitivity to acute THC exposure during pre-adolescence. The neurosteroid pregnenolone, a US Food and Drug Administration (FDA) approved drug, rescues synaptic defects and normalizes dopaminergic activity and behavior in PCE offspring, thus suggesting a therapeutic approach for offspring exposed to cannabis during pregnancy.






Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings (Substance Abuse and Mental Health Services Administration, 2011).
Legal Topic Overviews: Possession of Cannabis for Personal Use (European Monitoring Centre for Drugs and Drug Addiction, 2012).
Alpar, A., Di Marzo, V. & Harkany, T. At the tip of an iceberg: prenatal marijuana and its possible relation to neuropsychiatric outcome in the offspring. Biol. Psychiatry 79, e33–e45 (2016).
Brown, Q. L. et al. Trends in marijuana use among pregnant and nonpregnant reproductive-aged women, 2002–2014. JAMA 317, 207–209 (2017).
Dickson, B. et al. Recommendations from cannabis dispensaries about first-trimester cannabis use. Obstet. Gynecol. 131, 1031–1038 (2018).
Volkow, N. D., Compton, W. M. & Wargo, E. M. The risks of marijuana use during Pregnancy. JAMA 317, 129–130 (2017).
Jansson, L. M., Jordan, C. J. & Velez, M. L. Perinatal marijuana use and the developing child. JAMA 320, 545–546 (2018).
Galve-Roperh, I. et al. Cannabinoid receptor signaling in progenitor/stem cell proliferation and differentiation. Prog. Lipid Res. 52, 633–650 (2013).
Maccarrone, M., Guzman, M., Mackie, K., Doherty, P. & Harkany, T. Programming of neural cells by (endo)cannabinoids: from physiological rules to emerging therapies. Nat. Rev. Neurosci. 15, 786–801 (2014).
Wu, C. S., Jew, C. P. & Lu, H. C. Lasting impacts of prenatal cannabis exposure and the role of endogenous cannabinoids in the developing brain. Future Neurol. 6, 459–480 (2011).
Morris, C. V., DiNieri, J. A., Szutorisz, H. & Hurd, Y. L. Molecular mechanisms of maternal cannabis and cigarette use on human neurodevelopment. Eur. J. Neurosci. 34, 1574–1583 (2011).
Huizink, A. C. Prenatal cannabis exposure and infant outcomes: overview of studies. Prog. Neuropsychopharmacol. Biol. Psychiatry 52, 45–52 (2014).
De Genna, N. M., Richardson, G. A., Goldschmidt, L., Day, N. L. & Cornelius, M. D. Prenatal exposures to tobacco and cannabis: associations with adult electronic cigarette use. Drug Alcohol Depend. 188, 209–215 (2018).
Fine, J. D. et al. Association of prenatal cannabis exposure with psychosis proneness among children in the adolescent brain cognitive development (ABCD) study. JAMA Psychiatry 7, 762–764 (2019).
Volkow, N. D., Fowler, J. S., Wang, G. J. & Swanson, J. M. Dopamine in drug abuse and addiction: results from imaging studies and treatment implications. Mol. Psychiatry 9, 557–569 (2004).
Grace, A. A. Dysregulation of the dopamine system in the pathophysiology of schizophrenia and depression. Nat. Rev. Neurosci. 17, 524–532 (2016).
Bloomfield, M. A., Ashok, A. H., Volkow, N. D. & Howes, O. D. The effects of Δ9-tetrahydrocannabinol on the dopamine system. Nature 539, 369–377 (2016).
Bourque, J., Afzali, M. H. & Conrod, P. J. Association of cannabis use with adolescent psychotic symptoms. JAMA Psychiatry 75, 864–866 (2018).
Di Forti, M. et al. The contribution of cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): a multicentre case–control study. Lancet Psychiatry 6, 427–436 (2019).
Buckholtz, J. W. et al. Mesolimbic dopamine reward system hypersensitivity in individuals with psychopathic traits. Nat. Neurosci. 13, 419–421 (2010).
Geschwind, D. H. & Flint, J. Genetics and genomics of psychiatric disease. Science 349, 1489–1494 (2015).
Insel, T. R. Rethinking schizophrenia. Nature 468, 187–193 (2010).
Heffernan, A. L. & Hare, D. J. Tracing environmental exposure from neurodevelopment to neurodegeneration. Trends Neurosci. 41, 496–501 (2018).
Richardson, K. A., Hester, A. K. & McLemore, G. L. Prenatal cannabis exposure—the “first hit” to the endocannabinoid system. Neurotoxicol. Teratol. 58, 5–14 (2016).
Young-Wolff, K. C. et al. Trends in self-reported and biochemically tested marijuana use among pregnant females in california from 2009–2016. JAMA 318, 2490–2491 (2017).
Wiley, J. L., O’Connell, M. M., Tokarz, M. E. & Wright, M. J. Jr. Pharmacological effects of acute and repeated administration of Δ9-tetrahydrocannabinol in adolescent and adult rats. J. Pharm. Exp. Ther. 320, 1097–1105 (2007).
Mehmedic, Z. et al. Potency trends of Δ9-THC and other cannabinoids in confiscated cannabis preparations from 1993 to 2008. J. Forensic Sci. 55, 1209–1217 (2010).
ElSohly, M. A. et al. Changes in cannabis potency over the last 2 decades (1995–2014): analysis of current data in the United States. Biol. Psychiatry 79, 613–619 (2016).
McGorry, P. D., Hickie, I. B., Yung, A. R., Pantelis, C. & Jackson, H. J. Clinical staging of psychiatric disorders: a heuristic framework for choosing earlier, safer and more effective interventions. Aust. NZ J. Psychiatry 40, 616–622 (2006).
Sherif, M., Radhakrishnan, R., D’Souza, D. C. & Ranganathan, M. Human laboratory studies on cannabinoids and psychosis. Biol. Psychiatry 79, 526–538 (2016).
Braff, D. L., Swerdlow, N. R. & Geyer, M. A. Gating and habituation deficits in the schizophrenia disorders. Clin. Neurosci. 3, 131–139 (1995).
Lammel, S., Ion, D. I., Roeper, J. & Malenka, R. C. Projection-specific modulation of dopamine neuron synapses by aversive and rewarding stimuli. Neuron 70, 855–862 (2011).
Malinow, R. & Tsien, R. W. Presynaptic enhancement shown by whole-cell recordings of long-term potentiation in hippocampal slices. Nature 346, 177–180 (1990).
Glebov, O. O. et al. Nanoscale structural plasticity of the active zone matrix modulates presynaptic function. Cell Rep. 18, 2715–2728 (2017).
Lovinger, D. M. in Pharmacology of Neurotransmitter Release Vol. 184 (eds Südhof, T.C. & Starke, K.) 435–477 (Springer, 2008).
Matyas, F. et al. Identification of the sites of 2-arachidonoylglycerol synthesis and action imply retrograde endocannabinoid signaling at both GABAergic and glutamatergic synapses in the ventral tegmental area. Neuropharmacology 54, 95–107 (2008).
Melis, M. et al. Enhanced endocannabinoid-mediated modulation of rostromedial tegmental nucleus drive onto dopamine neurons in Sardinian alcohol-preferring rats. J. Neurosci. 34, 12716–12724 (2014).
Bellone, C., Mameli, M. & Luscher, C. In utero exposure to cocaine delays postnatal synaptic maturation of glutamatergic transmission in the VTA. Nat. Neurosci. 14, 1439–1446 (2011).
Hausknecht, K. et al. Excitatory synaptic function and plasticity is persistently altered in ventral tegmental area dopamine neurons after prenatal ethanol exposure. Neuropsychopharmacology 40, 893–905 (2015).
Thomas, M. J., Malenka, R. C. & Bonci, A. Modulation of long-term depression by dopamine in the mesolimbic system. J. Neurosci. 20, 5581–5586 (2000).
Yamaguchi, T., Sheen, W. & Morales, M. Glutamatergic neurons are present in the rat ventral tegmental area. Eur. J. Neurosci. 25, 106–118 (2007).
Chieng, B., Azriel, Y., Mohammadi, S. & Christie, M. J. Distinct cellular properties of identified dopaminergic and GABAergic neurons in the mouse ventral tegmental area. J. Physiol. 589, 3775–3787 (2011).
Marin, O. Developmental timing and critical windows for the treatment of psychiatric disorders. Nat. Med. 22, 1229–1238 (2016).
Wong, P., Sze, Y., Chang, C. C., Lee, J. & Zhang, X. Pregnenolone sulfate normalizes schizophrenia-like behaviors in dopamine transporter knockout mice through the AKT/GSK3beta pathway. Transl Psychiatry 5, e528 (2015).
Vallee, M. et al. Pregnenolone can protect the brain from cannabis intoxication. Science 343, 94–98 (2014).
Wang, X., Dow-Edwards, D., Anderson, V., Minkoff, H. & Hurd, Y. L. In utero marijuana exposure associated with abnormal amygdala dopamine D2 gene expression in the human fetus. Biol. Psychiatry 56, 909–915 (2004).
DiNieri, J. A. et al. Maternal cannabis use alters ventral striatal dopamine D2 gene regulation in the offspring. Biol. Psychiatry 70, 763–769 (2011).
Kuepper, R. et al. Delta-9-tetrahydrocannabinol-induced dopamine release as a function of psychosis risk: 18F-fallypride positron emission tomography study. PLoS One 8, e70378 (2013).
McGrath, J. et al. Association between cannabis use and psychosis-related outcomes using sibling pair analysis in a cohort of young adults. Arch. Gen. Psychiatry 67, 440–447 (2010).
Compton, M. T. et al. Association of pre-onset cannabis, alcohol, and tobacco use with age at onset of prodrome and age at onset of psychosis in first-episode patients. Am. J. Psychiatry 166, 1251–1257 (2009).
Capone, F., Bonsignore, L. T. & Cirulli, F. Methods in the analysis of maternal behavior in the rodent. Curr. Protoc. Toxicol. Chapter 13, Unit13.19 (2005).
Frau, R. et al. Sleep deprivation disrupts prepulse inhibition of the startle reflex: reversal by antipsychotic drugs. Int. J. Neuropsychopharmacol. 11, 947–955 (2008).
Godar, S. C. et al. Maladaptive defensive behaviours in monoamine oxidase A-deficient mice. Int. J. Neuropsychopharmacol. 14, 1195–1207 (2011).
Frau, R. et al. The neurosteroidogenic enzyme 5α-reductase mediates psychotic-like complications of sleep deprivation. Neuropsychopharmacology 42, 2196–2205 (2017).
Bortolato, M., Godar, S. C., Davarian, S., Chen, K. & Shih, J. C. Behavioral disinhibition and reduced anxiety-like behaviors in monoamine oxidase B-deficient mice. Neuropsychopharmacology 34, 2746–2757 (2009).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates (Academic, 2007).
Devoto, P., Flore, G., Longu, G., Pira, L. & Gessa, G. L. Origin of extracellular dopamine from dopamine and noradrenaline neurons in the medial prefrontal and occipital cortex. Synapse 50, 200–205 (2003).
Melis, M., Camarini, R., Ungless, M. A. & Bonci, A. Long-lasting potentiation of GABAergic synapses in dopamine neurons after a single in vivo ethanol exposure. J. Neurosci. 22, 2074–2082 (2002).
Johnson, S.W. & North, R. A. Two types of neurone in the rat ventral tegmental area and their synaptic inputs. J. Physiol. 450, 455–468 (1992).
Ungless, M. A., Whistler, J. L., Malenka, R. C. & Bonci, A. Single cocaine exposure in vivo induces long-term potentiation in dopamine neurons. Nature 411, 583–587 (2001).
Barna, L. et al. Correlated confocal and super-resolution imaging by VividSTORM. Nat. Protoc. 11, 163–183 (2016).
Dani, A., Huang, B., Bergan, J., Dulac, C. & Zhuang, X. Superresolution imaging of chemical synapses in the brain. Neuron 68, 843–856 (2010).
Huang, B., Jones, S. A., Brandenburg, B. & Zhuang, X. Whole-cell 3D STORM reveals interactions between cellular structures with nanometer-scale resolution. Nat. Methods 5, 1047–1052 (2008).
Dudok, B. et al. Cell-specific STORM super-resolution imaging reveals nanoscale organization of cannabinoid signaling. Nat. Neurosci. 18, 75–86 (2015).
Melis, M. et al. PPARα regulates cholinergic-driven activity of midbrain dopamine neurons via a novel mechanism involving α7 nicotinic acetylcholine receptors. J. Neurosci. 33, 6203–6211 (2013).
Acknowledgements
The authors thank R. Tonini and O. J. Manzoni for discussions and comments on the manuscript, and G. Talani, M. Pignatelli, M. Tuveri, S. Aramo, G. Giua and B. Tuveri for their skillful assistance. The authors are also grateful to L. Barna for his help with STORM microscopy images, and thank Nikon Europe, Nikon Austria and Auro-Science Consulting for kindly providing microscopy support. The present study was supported by the University of Cagliari (RICCAR 2017 and 2018 to M.M.), the Region of Sardinia (RASSR32909 to M.M. and F72F16002850002 to R.F.), the Fondazione Banco di Sardegna (F71I17000200002 to R.F.), the European Molecular Biology Organization (ASTF 371-2016 to C.S.), the Fondazione Zardi Gori (to C.S.), the National Institutes of Health (R01DA022340 to J.F.C., R01NS099457 to I.K. and R01DA044925 to J.F.C., M.M. and I.K.), the Hungarian Academy of Sciences Momentum Program (LP-54/2013 to I.K.), and the National Research, Development and Innovation Office of Hungary (VKSZ-14-1-2015-0155 to I.K.). The project was also funded by the Ministry of National Economy for STORM super-resolution microscopy (VEKOP-2.3.3-15-2016-00013 to I.K.).
Author information
Authors and Affiliations
Contributions
R.F. and F.T. designed and performed the behavioral experiments and analyzed the data. S.F. and V.S. carried out behavioral observations. F.T. and V.S. prepared the figures. V.M. and C.I.P. carried out the confocal imaging and STORM experiments, the corresponding data analyses and prepared the figures. P.S. and P.D. carried out the cerebral microdialysis experiments and analyzed the data. M.C. and V.S. designed and performed the maternal observation experiments. C.S., V.S. and S.A. performed the chronic drug administration treatment. S.A. performed the DREADD experiments. J.F.C. designed the DREADD experiments and contributed to manuscript preparation. I.K. designed the confocal and STORM experiments, analyzed and supervised the imaging data and wrote the manuscript. M.M. conceived, designed and supervised the project, performed patch-clamp recordings, analyzed the data, prepared the figures and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Neuroscience thanks Camilla Bellone, Paul Kenny, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Effects of PCE on pregnancy outcome.
(a) Bar graph showing that the size of the litters of CTRL and PCE dams (nlitter= 37 and nlitter= 35, respectively) is similar (P=0.47, ns, t(70)=0.723; two-sided unpaired t-test). (b) Total maternal and (c) non maternal behavior displayed by CTRL and PCE dams (nlitter= 9 and nlitter= 11, respectively) towards the pups over a period of three weeks (P=0.84, ns, F(1,18)=0.043; two-way RM ANOVA). Data are represented as mean ± S.E.M. (d) Bar graph summarizing no differences in the weight of male pups prenatally treated with either vehicle (CTRL, npups= 38, nlitter= 17) or THC (PCE, npups= 34, nlitter=17) at postnatal day 25 (P=0.099, ns, t(70)=1.67; two-sided unpaired t-test). Data in (a,d) are represented as box-and-whiskers plot (including minima, maxima, and media values, and lower and upper quartiles) with single values.
Supplementary Figure 2 THC-induced deficits of PPI are age-dependent.
(a) Effect of THC challenge on PPI in CTRL and PCE young male adult (PND 60-70) offspring (P=0.64, ns, F(1,27)=2.5; two-way ANOVA; nrat= 8 per CTRL-VEH, CTRL-THC and PCE-VEH groups; nrat= 7 per PCE-THC group; nlitter= 6 per group). Data are represented as box-and-whiskers plot (including minima, maxima, and media values, and lower and upper quartiles) with single values. (b) Representative photograph of the probe location into the nucleus accumbens shell. The arrow indicates the tip of the probe. N= 7 experiments per group were repeated independently, with similar results obtained. (c) Schematic diagram of the cerebral area targeted by the microdialysis probe as indicated by the vertical line (nucleus accumbens shell, AP: +1.5, L: ±0.7, V: -7.0 from bregma) from Paxinos and Watson (2007). (d) Schematic showing the approximate localization of the probe for the experiments shown in Fig. 1c. NAcSh, nucleus accumbens shell; NAcC, nucleus accumbens core; CPu, caudate/putamen.
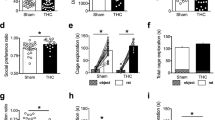
Supplementary Figure 3 PCE offspring behavioral phenotypes in male offspring.
(a) THC increases locomotion selectively in PCE offspring (**P=0.0024, interaction F(1,33)=7.851; CTRL-VEH and CTRL-THC: nrat= 8, PCE-VEH: nrat= 11, PCE-THC: nrat= 10). (b) THC prolongs total time spent (s) in center part of the arena (**P=0.005, interaction F(1,32)=6.03; CTRL-VEH and CTRL-THC: nrat= 8, PCE-VEH: nrat= 11, PCE-THC: nrat= 10). Confocal image of mCherry (c) and TH (d) channels of coronal brain sections, calibration bar: 1mm. N= 12 experiments were repeated independently, with similar results obtained. (e) PCE does not affect startle amplitude (P=0.77, ns, interaction F(1,59)=0.08; CTRL-VEH: nrat= 14; PCE-VEH and PCE-THC: nrat= 18 per group; CTRL-THC: nrat= 17). Startle amplitude values are represented as arbitrary units (AU). In the elevated plus maze, no differences were found in the (f) number of entries and (g) total duration of time spent by offspring in the closed, open arms and center position (number of entries in closed arms: P=0.10, ns, t(18)=1.72; duration: P=0.67, ns, t(18)=0.42; number of entries in open arms: P=0.93, ns, t(18)=0.08; duration: P=0.85, ns, t(18)=0.18; number of center transitions: P=0.33, ns, t(18)= 0.98; duration in center position: P=0.33, ns, t(18)= 0.99; two-tailed unpair t-test; nrat= 10 per group). All data are represented as box-and-whiskers plot (including minima, maxima, and media values, and lower and upper quartiles) with single values (each circle represents n= 1 rat). Unless otherwise indicated, data were analyzed with two-way ANOVA followed by Tukey’s multiple-comparisons test.
Supplementary Figure 4 PCE does not modify TH+ cell number in male offspring.
(a) Confocal image of anti-TH immunostaining in control rat brain. (b) Boxed region in a). (c) Confocal image of anti-TH immunostaining in PCE rat brain. (d) Boxed region in c). n= 12 images per animal were acquired independently, with similar results obtained. (e) Mean TH+ cell densities of the VTA ROIs showing no significant difference between treatment groups (P=0.665, ns; two-sided Mann-Whitney U-test). (f) Mean TH pixel intensities of the VTA ROI showing no significant difference between treatment groups (P=0.885, ns; two-sided Mann-Whitney U-test). Data in (e, f) are represented with floating bars (min to max, line at median) with single values (each circle represents n= 1 rat).
Supplementary Figure 5 Spike fidelity is enhanced in PCE male offspring.
(a and b) Representative traces of evoked action potentials (APs) in response to somatically injected current (top, in red the response elicited by 50 pA; bottom, in blue and red the elicited APs in response to 50 pA and 200 pA, respectively) by dopamine neurons from both CTRL (left) and PCE (right) offspring. Calibration bar: 100 ms, 50 mV. N= 20 and 21 experiments were repeated independently from PCE and CTRL groups, respectively, with similar results obtained. (c) PCE (ncells= 20, nrat= 10, nlitter= 6) reduced standard deviation of the latency (Jitter, ms) to the appearance of the first AP in response to somatically injected current (****P<0.0001, F(3,64)=27.16; PCE: P<0.0001, F(1,64)=44.79; two-way ANOVA) when compared to CTRL (ncells= 21, nrat =10, nlitter=6). Data are represented as box-and-whiskers plot (including minima, maxima, and media values, and lower and upper quartiles) with single values expressing average data for each animal. (d) The degree of spike fidelity in response to the current injected is revealed by the spike frequency of dopamine neurons plotted as a function of the jitter in PCE (ncells= 20, nrat= 10, nlitter= 6) and CTRL (ncells= 21, nrat= 10, nlitter= 6) offspring (*P=0.048, F(1,4)=7.844; linear regression). (e and f) Frequency distribution graphs show that PCE does not affect membrane capacitance (e) (P=0.52, ns; t(56)=0.63; PCE and CTRL: ncells= 71, nrat= 29, nlitter= 6) and inter-spike interval (ISI) (f) (P=0.45, ns; t(19)=0.768; PCE: ncells= 20, nrat= 11, nlitter= 6; CTRL: ncells= 21, nrat= 10, nlitter= 6). Data were analyzed with two-tailed unpaired t-test.
Supplementary Figure 6 Effects of THC on putative dopamine neurons of male offspring.
(a) Representative traces show spontaneous dopamine firing activity recorded from both CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 7, nrat= 7, nlitter= 5) offspring before (basal) and after bath application of THC (3μM, 5 min). Calibration bar: 40 s, 50 mV. (b) Dose-response curves for the effects of THC on spontaneous dopamine cell activity (****P<0.0001, F(3,44)=8.96; two-way ANOVA) recorded from CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 7, nrat= 7, nlitter= 5). Data are represented as mean ± S.E.M. (c) THC effect is prevented by the CB1 antagonist AM281 (0.5 μM) in both CTRL (ncells= 6, nrat= 6, nlitter= 2) and PCE (ncells= 6, nrat= 6, nlitter= 2) progenies (interaction THC x PCE: P<0.0001, F(2,30)=20.33; two-way ANOVA) (d) Representative traces showing no effect of THC on AP waveform: AP waveform before (purple and light blue) and after THC (grey and blue) from CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 7, nrat= 7, nlitter= 5) dopamine cells. No alterations in amplitude (P=0.36, ns, t11=0.94; two-sided unpaired t-test) and rise time (P=0.24, ns, t11=1.236; two-sided unpaired t-test) were detected between CTRL and PCE offspring. Calibration bar: 10pA, 10ms. (e) Representative traces of action potentials (APs) elicited in response to the maximum current injected (200 pA) by dopamine neurons of CTRL (ncells= 7, nrat= 7, nlitter= 2) and PCE (ncells= 7, nrat= 7, nlitter= 5) offspring before (basal) and after bath application of THC (3 μM, 5 min). Calibration bar: 40mV, 300 ms. N= 7 experiments per group were repeated independently, with similar results obtained (f) THC net effect (Δvalue= number of AP THC- number of AP basal) on spike frequency in response to somatically injected current in CTRL and PCE offspring (**P=0.002, F(9,88)=3.22; Two-way RM ANOVA followed by Tukey’s multiple comparisons test; CTRL: ncells= 7, nrat= 7, nlitter= 2 and PCE: ncells= 7, nrat= 7, nlitter= 5. (g) Effect of THC on the latency to first AP appearance in response to the smallest current injected (50 pA) in PCE cells (*P=0.02, t(11)= 2.1; two-tailed unpaired t test). Top, Representative traces of evoked APs in response to the minimum current injected (50 pA) by dopamine neurons before (light blue trace) and after THC application (blue trace). Calibration bar: 1s, 20 mV. CTRL: ncells= 7, nrat= 7, nlitter= 2 and PCE: ncells= 7, nrat= 7, nlitter= 5. (h) Percentage of cells eliciting APs at 50 pA in CTRL and PCE animals in this set of experiments is consistent with those presented in Fig. 2g. Unless otherwise indicated, for all panels, graphs show box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values expressing average data for each animal.
Supplementary Figure 7 Effects of PCE on excitatory to inhibitory balance and inhibitory properties of dopamine neurons in male offspring.
Insets show (a) representative traces of AMPA EPSCs (-70 mV) and GABAA IPSCs (0 mV), and (b) NMDA EPSCs (+40 mV) and GABAA IPSCs (-70 mV) recorded from CTRL and PCE offspring. Calibration bar: 20 ms, 20 pA. n= 6 experiments per group were repeated independently, with similar results obtained. (c) Quantification of the data showing how PCE affects the ratio between evoked excitatory and inhibitory synaptic currents, in particular the AMPA to GABAA PSC ratio (**P=0.008, t(10)=3.29, two-tailed unpaired t-test) and the NMDA to GABAA PSC ratio (*P=0.014, t(11)=2.93; two-tailed unpaired t-test) recorded from dopamine cells of CTRL (ncells=6, nrat=6, nlitter=3) and PCE (ncells=6, nrat=6, nlitter=4) offspring. (d) Representative traces of paired GABAA-mediated IPSCs. Calibration bar: 25 ms, 100 pA. n= 23 and 22 experiments were repeated independently from PCE and CTRL, respectively, with similar results obtained. (e) Graph shows that PCE increases (***P=0.0003, t(8)=4.087; two-tailed unpaired t-test) the paired-pulse ratio (IPSC2/IPSC1) of GABAA IPSCs recorded from PCE (ncells= 23, nrat= 12, nlitter= 10) with respect to CTRL (ncells= 22, nrat= 11, nlitter= 10) offspring. (f) Sample traces of mIPSCs recorded from CTRL and PCE cells. Calibration bar: 50 ms, 20 pA. n= 11 and 10 experiments were repeated independently from PCE and CTRL, respectively, with similar results obtained. (g) PCE decreases mIPSCs frequency (ncells= 11, nrat= 6, nlitter= 6) when compared to CTRL (ncells= 10, nrat= 5, nlitter= 4) offspring (****P<0.0001, t(9)=7.453; two-tailed unpaired t-test). (h) PCE does not affect amplitude of mIPSCs (P=0.49, ns, t(9)=0.712; two-tailed unpaired t-test) recorded from PCE (ncells= 11, nrat= 6, nlitter= 6) with respect to CTRL (ncells= 10, nrat= 5, nlitter= 4). For all panels, graphs show box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values expressing average data for each animal.
Supplementary Figure 8 Molecular and morphological parameters of VIAAT+ axon terminals in male offspring.
(a) Mean area of VIAAT- containing axon terminals determined by the active contour method in CTRL and PCE-treated animals showing no significant difference (P=0.665; two-sided Mann-Whitney U-test; nrat= 4 per group). (b) Cumulated mean pixel intensity of VIAAT- immunostaining in VIAAT+ axon terminals showing no significant difference between treatment groups (P=0.312; two-sided Mann-Whitney U-test; nrat= 4 per group). For (a) and (b) n= 26 images per PCE and n=31 images per CTRL group were acquired independently, with similar results obtained. (c) Mean of normalized bassoon NLP in active zones belonging to VIAAT+ terminals showing higher bassoon NLP in the PCE treatment group (*P=0.030; two-sided Mann-Whitney U-test; nrat= 4 per group, nlitter= 4 per group). (d) Mean area of active zones belonging to VIAAT+ axon terminals showing no difference between treatment groups (P=0.112; two-sided Mann-Whitney U-test; nrat= 4 per group, nlitter= 4 per group). Representative 3D reconstructions of a filled CTRL (e) and PCE (f) dopaminergic neuron (magenta) in the VTA with VIAAT+ inputs (cyan) impinging on them. Calibration bar: 0.5 mm. n= 7-8 images per animal were acquired independently, with similar results obtained. (g) Quantification of VIAAT input densities normalized to the surface of the biocytin objects per image (P=0.365; two sided Mann-Whitney U-test; ncells= 31 per CTRL group; ncells= 26 per PCE). Data in (a, b, c, d) are represented with floating bars (min to max, line at median) with single values (each circle represents n= 1 rat). Data in (g) are represented with box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values (each circle represents n= 1 cell). n= 26 images per PCE and n=31 images per CTRL group were acquired independently, with similar results obtained.
Supplementary Figure 9 Effect of PCE on morphological and synaptic properties of excitatory inputs on male VTA dopamine neurons.
Representative 3D reconstructions of a filled CTRL (a) and PCE (b) dopaminergic neuron (magenta) in the VTA with vGluT1+ inputs (cyan) impinging on them. Calibration bar: 0.5 mm. n= 8 images per animal were acquired independently, with similar results obtained. (c) Quantification of vGluT1 input densities normalized to the surface of the biocytin objects per image (**P=0.0004; two-tailed Mann-Whitney U-test). n= 26 images per PCE and n=31 images per CTRL group were acquired independently, with similar results obtained. (d) Representative traces of paired AMPA EPSCs recorded from putative dopamine neurons of pre-adolescent male offspring. Calibration bar: 25 ms, 100 pA. n= 26 and 30 experiments were repeated independently from PCE and CTRL slices, respectively, with similar results obtained. (e) PCE dopamine cells (ncells= 30, nrat= 17, nlitter= 15) exhibit an increased paired-pulse ratio (EPSC2/EPSC1) of AMPA EPSCs (****P<0.0001, t(30)=4.835; two-tailed unpaired t-test) with respect to CTRL (ncells= 26, nrat= 15, nlitter= 12) counterparts. (f) Sample traces of mEPSCs collected from CTRL and PCE dopamine cells. Calibration bar: 50 ms, 20 pA. n= 7 experiments per group were repeated independently, with similar results obtained. (g) Frequency of mEPSCs recorded from CTRL (ncells=7, nrat=5, nlitter=5) and PCE (ncells=7, nrat=5, nlitter=7) dopamine cells (P=0.88, t(8)=0.142; two-tailed unpaired t-test). (h) PCE increases the amplitude (**P=0.0028, t(8)=4.237; two-sided unpaired t-test) of mEPSCs (ncells=7, nrat=5, nlitter=7) with respect to CTRL (ncells=7, nrat=5, nlitter=5). For all panels, graphs show box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values expressing average data for each animal.
Supplementary Figure 10 Immunolabeling of TH is associated with electrophysiological properties of neurons in rat VTA horizontal slices.
Horizontal brain section immunolabeled for tyrosine hydroxylase (TH) (a) and containing a biocytin-filled cell (a,b) (calibration bar 0.5 mm). The arrows indicate a biocytin-filled cell. (c,d) Example traces demonstrating that putative dopamine neurons can be identified on the bases of the presence (c) of a large h-current (red trace; calibration bar: 200 pA, 20 ms) and the action potential width (d) in the lateral posterior VTA. Calibration bar: 10 pA, 10 ms. (e) TH immunolabeling, (f) byocitin labeling, (g) merged images. Calibration bar: 10 μm. (h,i) Example traces demonstrating that putative GABA neurons can be identified on the bases of the absence (h) of h-current (red trace; calibration bar 200 pA, 20 ms) and the action potential width (i) in the lateral posterior VTA. Calibration bar: 10 pA, 10 ms. (j) TH immunolabeling, (k) byocitin labeling, (l) merged images. Calibration bar: 10 μm. For panels a-g, n= 15 cells were acquired independently, with similar results obtained. (h-i) n= 18 experiments per group were performed independently, with similar results obtained. (j-l) n= 2 cells were acquired independently, with similar results obtained.
Supplementary Figure 11 Effect of PCE on synaptic properties of putative GABA neurons of lateral posterio VTA in male offspring.
(a) Representative traces (top) and min to max plots (down) summarizing the amplitude of evoked EPSCs (-70 mV) and IPSCs (0 mV) recorded from putative GABA neurons from CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 6, nrat= 6, nlitter= 4) offspring (P=0.29, ns, t(10)=1.109; two-tailed unpaired t-test). Calibration bar: 20 ms, 200 pA. (b) Quantification of the data summarizing that PCE GABA cells (ncells=6, nrat=6, nlitter=4) exhibit a decreased AMPA/NMDA ratio (**P=0.003, t(10)=3.81; two-tailed unpaired t-test) as compared to CTRL (ncells= 6, nrat= 6, nlitter= 3). Insets show representative traces of AMPA and NMDA EPSCs recorded from GABA cells held at +40 mV in slices from CTRL and PCE offspring. (c) PCE (ncells=6, nrat=6, nlitter=4) does not affect (P=0.74, ns, t(10)=0.34; two-tailed unpaired t-test) NMDA EPSC decay time kinetic (weighted tau, τ) with respect to CTRL (ncells= 6, nrat= 6, nlitter= 3). (d) Current-voltage relationship (I–V) curves of AMPA EPSCs recorded from GABAergic neurons do not differ (P=0.81, ns, F(2,30)=0.22; two-way RM ANOVA) in CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 6, nrat= 6, nlitter= 4) offspring. Data are represented as mean ± S.E.M. (e) Quantification of paired-pulse ratio (EPSC2/EPSC1) of AMPA EPSCs recorded from GABA cells in CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 6, nrat= 6, nlitter= 4) offspring (P=0.53, t(10)=0.65; two-tailed unpaired t-test). Insets show representative traces of paired AMPA EPSCs. Calibration bar: 50 ms, 100 pA. (f) Paired-pulse ratio (IPSC2/IPSC1) of GABAA IPSCs recorded from CTRL (ncells= 6, nrat= 6, nlitter= 3) and PCE (ncells= 6, nrat= 6, nlitter= 4) offspring do not differ (P=0.55, ns, t(10)=0.6147; two-tailed unpaired t-test). Insets show representative traces of paired GABAA IPSCs. Calibration bar: 50 ms, 100 pA. For all panels, graphs show box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values expressing average data for each animal.
Supplementary Figure 12 Restorative effects of systemic treatment with the FDA-approved pregnenolone on synaptic properties of VTA putative dopamine neurons in male PCE offspring.
(a) Pregnenolone (PREG, 6 mg kg-1, s.c.) restores excitatory to inhibitory balance (****P=0.0002, F(1,20)=20.62; two-way ANOVA) in dopamine neurons of PCE (VEH: ncells= 10, nrat= 8, nlitter= 5; PREG: ncells= 6, nrat= 4, nlitter= 3) without affecting CTRL (VEH: ncells=10, nrat=8, nlitter=5; PREG: ncells=6, nrat=4, nlitter=2) offspring. (b) Data summarize PREG effect on AMPA/NMDA ratio (P=0.0001, interaction F(1,18)=22.96; P=0.0008, PREG F(1,18)=15.96; two-way ANOVA) in CTRL (VEH: ncells= 9, nrat= 6, nlitter= 4; PREG: ncells=6, nrat=4, nlitter=2) and PCE (VEH: ncells= 10, nrat= 7, nlitter= 5; PREG: ncells= 6, nrat= 4, nlitter= 3) offspring. (c) Current-voltage relationship (I–V) curves of AMPA EPSCs recorded from dopamine neuron of CTRL (VEH: ncells= 9, nrat= 6, nlitter= 4; PREG: ncells= 6, nrat= 4, nlitter= 2) and PCE (VEH: ncells= 10, nrat= 7, nlitter= 5; PREG: ncells= 6, nrat= 4, nlitter= 3) offspring (P=0.09, interaction F(6,30)=2.041; two-way ANOVA). Data are represented as mean ± S.E.M. (d) Min to max plots showing that PREG normalizes (*P=0.033, interaction F(1,24)=5.08; PREG: P=0.016, F(1,24)=6.718; two-way ANOVA) NMDAR EPSC decay time kinetic (weighted tau, τ) in PCE (VEH: ncells= 18, nrat= 12, nlitter=6; PREG: ncells= 6, nrat= 4, nlitter= 3) animals when compared to CTRL (VEH: ncells= 18, nrat= 12, nlitter= 6; PREG: ncells= 6, nrat= 4, nlitter= 2). (e) PREG restores paired-pulse facilitation (EPSC2/EPSC1) of AMPA EPSCs (**P=0.0018, interaction F(1,24)=14.67; P=0.0038, PREG F(1,14)=5.207; two-way ANOVA) recorded from dopamine cells in PCE (VEH: ncells= 8, nrat= 5, nlitter= 2; PREG: ncells= 8, nrat= 4, nlitter= 2) when compared with CTRL offspring (VEH: ncells= 7, nrat= 5, nlitter= 2; PREG: ncells= 8, nrat= 4, nlitter= 2). (f) PREG corrects (**P=0.0049, interaction F(1,18)=10.28; P=0.009, PREG F(1,18)=8.549; two-way ANOVA) the paired-pulse facilitation (IPSC2/IPSC1) of GABAA IPSCs recorded from dopaminergic neuron of PCE (VEH: ncells= 8, nrat= 5, nlitter= 3; PREG: ncells= 4, nrat= 4, nlitter= 4) as per CTRL offspring (VEH: ncells= 7, nrat= 5, nlitter= 2; PREG: ncells= 6, nrat= 4, nlitter= 2). Unless otherwise indicated, for all panels, graphs show box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values expressing average data for each animal.
Supplementary Figure 13 The pharmacological inhibition of 3-beta HSD enzyme does not affect the protective effect of pregnenolone in male PCE-THC treated offspring.
Effect of subchronic treatment with pregnenolone (PREG 6 mg kg-1, s.c.), alone or in combination with trilostane (TRI, 15 mg kg-1, s.c.) on sensorimotor gating alterations induced by acute THC in CTRL and PCE progenies. PREG alone or in combination with TRI reversed PPI deficits in PCE offspring (*P<0.05 between PCE-VEH-VEH-THC and PCE-PREG-VEH-THC, ***P<0.001 between PCE-VEH-VEH-THC vs PCE-PREG-TRIL-THC, ****P<0.0001 between CTRL-VEH-VEH-THC and PCE-VEH-VEH-THC, interaction F(1,105)=5.815; CTRL groups: nrat= 54, PCE groups: nrat= 58). All data are represented as box-and-whisker plots (including minima, maxima, and median values, and lower and upper quartiles) with single values expressing average data for each animal. Data were analyzed with three-way ANOVA followed by Tukey’s multiple-comparisons test.
Supplementary information
Supplementary information
Supplementary Figs. 1–13 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Frau, R., Miczán, V., Traccis, F. et al. Prenatal THC exposure produces a hyperdopaminergic phenotype rescued by pregnenolone. Nat Neurosci 22, 1975–1985 (2019). https://doi.org/10.1038/s41593-019-0512-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-019-0512-2
- Springer Nature America, Inc.
This article is cited by
-
Prenatal Cannabis Use and Offspring Autism-Related Behaviors: Examining Maternal Stress as a Moderator in a Black American Cohort
Journal of Autism and Developmental Disorders (2024)
-
Prenatal Exposure to Cannabis: Effects on Childhood Obesity and Cardiometabolic Health
Current Obesity Reports (2024)
-
Cannabis for morning sickness: areas for intervention to decrease cannabis consumption during pregnancy
Journal of Cannabis Research (2023)
-
Characterization of cannabinoid plasma concentration, maternal health, and cytokine levels in a rat model of prenatal Cannabis smoke exposure
Scientific Reports (2023)
-
Prenatal THC exposure induces long-term, sex-dependent cognitive dysfunction associated with lipidomic and neuronal pathology in the prefrontal cortex-hippocampal network
Molecular Psychiatry (2023)