Abstract
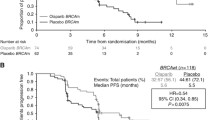
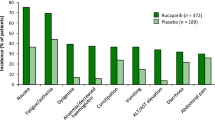
Poly(adenosine diphosphate-ribose) polymerase (PARP) inhibitors as maintenance therapy after first-line chemotherapy have improved progression-free survival in women with advanced ovarian cancer; however, not all PARP inhibitors can provide benefit for a biomarker-unselected population. Senaparib is a PARP inhibitor that demonstrated antitumor activity in patients with solid tumors, including ovarian cancer, in phase 1 studies. The multicenter, double-blind, phase 3 trial FLAMES randomized (2:1) 404 females with advanced ovarian cancer (International Federation of Gynecology and Obstetrics stage III–IV) and response to first-line platinum-based chemotherapy to senaparib 100 mg (n = 271) or placebo (n = 133) orally once daily for up to 2 years. The primary endpoint was progression-free survival assessed by blinded independent central review. At the prespecified interim analysis, the median progression-free survival was not reached with senaparib and was 13.6 months with placebo (hazard ratio 0.43, 95% confidence interval 0.32–0.58; P < 0.0001). The benefit with senaparib over placebo was consistent in the subgroups defined by BRCA1 and BRCA2 mutation or homologous recombination status. Grade ≥3 treatment-emergent adverse events occurred in 179 (66%) and 27 (20%) patients, respectively. Senaparib significantly improved progression-free survival versus placebo in patients with advanced ovarian cancer after response to first-line platinum-based chemotherapy, irrespective of BRCA1 and BRCA2 mutation status and with consistent benefits observed between homologous recombination subgroups, and was well tolerated. These results support senaparib as a maintenance treatment for patients with advanced ovarian cancer after a response to first-line chemotherapy. ClinicalTrials.gov identifier: NCT04169997.




Similar content being viewed by others
Data availability
The study protocol is published with this manuscript as Supplementary Information. All extracted data, including deidentified individual information, trial-level data (analysis datasets) and other information (for example, clinical study reports and informed consent form) are available from the corresponding author upon request, provided the FLAMES trial is not part of an ongoing or planned regulatory submission. The corresponding author will also accept requests for clinical trial data for unlicensed products and indications.
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
National Cancer Institute. Cancer stat facts: ovarian cancer. National Cancer Institute Surveillance, Epidemiology, and End Results Program 2013–2019 https://seer.cancer.gov/statfacts/html/ovary.html (2019).
National Comprehensive Cancer Network. Ovarian cancer including fallopian tube cancer and primary peritoneal cancer, version 2.2023. NCCN https://www.nccn.org/professionals/physician_gls/pdf/ovarian.pdf (2022).
Berek, J. S., Renz, M., Kehoe, S., Kumar, L. & Friedlander, M. Cancer of the ovary, fallopian tube, and peritoneum: 2021 update. Int. J. Gynaecol. Obstet. 155, 61–85 (2021).
González-Martín, A. et al. Newly diagnosed and relapsed epithelial ovarian cancer: ESMO clinical practice guideline for diagnosis, treatment and follow-up. Ann. Oncol. 34, 833–848 (2023).
DiSilvestro, P. & Alvarez Secord, A. Maintenance treatment of recurrent ovarian cancer: is it ready for prime time? Cancer Treat. Rev. 69, 53–65 (2018).
O’Malley, D. M., Krivak, T. C., Kabil, N., Munley, J. & Moore, K. N. PARP inhibitors in ovarian cancer: a review. Target. Oncol. 18, 471–503 (2023).
Huang, X.-Z. et al. Efficacy and prognostic factors for PARP inhibitors in patients with ovarian cancer. Front. Oncol. 10, 958 (2020).
Moore, K. et al. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N. Engl. J. Med. 379, 2495–2505 (2018).
Ray-Coquard, I. et al. Olaparib plus bevacizumab as first-line maintenance in ovarian cancer. N. Engl. J. Med. 381, 2416–2428 (2019).
González-Martín, A. et al. Niraparib in patients with newly diagnosed advanced ovarian cancer. N. Engl. J. Med. 381, 2391–2402 (2019).
Monk, B. J. et al. A randomized, phase III trial to evaluate rucaparib monotherapy as maintenance treatment in patients with newly diagnosed ovarian cancer (ATHENA–MONO/GOG-3020/ENGOT-ov45). J. Clin. Oncol. 40, 3952–3964 (2022).
ZEJULA (niraparib) prescribing information. GlaxoSmithKline https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Zejula_Capsules/pdf/ZEJULA-CAPSULES-PI-PIL.PDF (2022).
RUBRACA (rucaparib) prescribing information. Pharma and Schweiz GmbH https://www.rubraca.com/sites/default/files/RubracaUSPI.pdf (2023).
Coleman, R. L. et al. Veliparib with first-line chemotherapy and as maintenance therapy in ovarian cancer. N. Engl. J. Med. 381, 2403–2415 (2019).
Bell, D. et al. Integrated genomic analyses of ovarian carcinoma. Nature 474, 609–615 (2011).
Cai, S. X. et al. Abstract 1399: discovery and development of PARP inhibitor senaparib. Cancer Res. 82, 1399 (2022).
Cao, J. et al. Safety, tolerability, and pharmacokinetics of senaparib, a novel PARP1/2 inhibitor, in Chinese patients with advanced solid tumors: a phase I trial. Oncologist 28, e1259–e1267 (2023).
Gao, B. et al. A phase 1 dose-escalation study of the poly(ADP-ribose) polymerase inhibitor senaparib in Australian patients with advanced solid tumors. Cancer 129, 1041–1050 (2023).
Banerjee, S. et al. Maintenance olaparib for patients with newly diagnosed advanced ovarian cancer and a BRCA mutation (SOLO1/GOG 3004): 5-year follow-up of a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 22, 1721–1731 (2021).
Li, N. et al. Treatment with niraparib maintenance therapy in patients with newly diagnosed advanced ovarian cancer: a phase 3 randomized clinical trial. JAMA Oncol. 9, 1230–1237 (2023).
de Souza, P. et al. 574P updated results of phase I study of senaparib (IMP4297) in Australian patients with advanced solid tumours. Ann. Oncol. 31, S490 (2020).
Lynparza approved in China as 1st-line maintenance treatment with bevacizumab for HRD-positive advanced ovarian cancer. AstraZeneca https://www.astrazeneca.com/media-centre/press-releases/2022/lynparza-approved-in-china-for-ovarian-cancer.html (2022).
Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. National Cancer Institute https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf (2017).
Acknowledgements
This study was funded by IMPACT Therapeutics (Shanghai) Inc. The sponsor had a role in the study design, data collection, data analysis, data interpretation and writing and approval of this manuscript for publication. The authors wish to acknowledge and thank the study patients and their families, the investigators and the study teams at each of the participating centers, as well as S.X. Cai, Y. Tian, H. Xia, B. Li and Y. Zhang of IMPACT Therapeutics (Shanghai) Inc. and the entire research team of the FLAMES trial. Writing and editorial assistance was provided by J. Kolston and M. Yu at Parexel and was funded by IMPACT Therapeutics (Shanghai) Inc.
Author information
Authors and Affiliations
Consortia
Contributions
X.Wu and J.L. were the cochief investigators for this trial and are co-first authors of the manuscript. X.Wu, J.L., J.W., L.W. and Z.Lin contributed to the trial design and were involved in project administration and supervision. All authors contributed to patient accrual, trial conduct, treatment, safety assessments and data acquisition. All authors had full access to and verified the data in this study. All authors interpreted the data, reviewed the draft and final versions of the manuscript and provided final approval to submit this article for publication.
Corresponding author
Ethics declarations
Competing interests
C.X. and C.-Y.H. are employees of IMPACT Therapeutics (Shanghai) Inc., Shanghai, China. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Michael Friedlander and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ulrike Harjes, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1
Structures of senaparib, olaparib, niraparib, and rucaparib.
Extended Data Fig. 2 Chemotherapy-free interval, time to first subsequent therapy or death, and time to treatment discontinuation or death.
Kaplan–Meier estimates of chemotherapy-free interval (a), time to first subsequent therapy or death (b), and time to treatment discontinuation or death (c) in the ITT population. CFI, chemotherapy-free interval; CI, confidence interval; HR, hazard ratio; ITT, intention-to-treat; n/N, number with disease progression or death/total number evaluable; NR, not reached; TDT, time to treatment discontinuation or death; TFST, time to first subsequent therapy or death.
Supplementary information
Supplementary Information
Supplementary Methods and Study Protocol.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, X., Liu, J., Wang, J. et al. Senaparib as first-line maintenance therapy in advanced ovarian cancer: a randomized phase 3 trial. Nat Med 30, 1612–1621 (2024). https://doi.org/10.1038/s41591-024-03003-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-024-03003-9
- Springer Nature America, Inc.