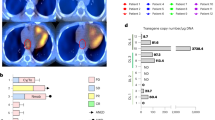
Abstract
Vα24-invariant natural killer T (NKT) cells have shown potent anti-tumor properties in murine tumor models and have been linked to favorable outcomes in patients with cancer. However, low numbers of these cells in humans have hindered their clinical applications. Here we report interim results from all three patients enrolled on dose level 1 in a phase 1 dose-escalation trial of autologous NKT cells engineered to co-express a GD2-specific chimeric antigen receptor (CAR) with interleukin-15 in children with relapsed or resistant neuroblastoma (NCT03294954). Primary and secondary objectives were to assess safety and anti-tumor responses, respectively, with immune response evaluation as an additional objective. We ex vivo expanded highly pure NKT cells (mean ± s.d., 94.7 ± 3.8%) and treated patients with 3 × 106 CAR-NKT cells per square meter of body surface area after lymphodepleting conditioning with cyclophosphamide/fludarabine (Cy/Flu). Cy/Flu conditioning was the probable cause for grade 3–4 hematologic adverse events, as they occurred before CAR-NKT cell infusion, and no dose-limiting toxicities were observed. CAR-NKT cells expanded in vivo, localized to tumors and, in one patient, induced an objective response with regression of bone metastatic lesions. These initial results suggest that CAR-NKT cells can be expanded to clinical scale and safely applied to treat patients with cancer.


Similar content being viewed by others
Data availability
All requests for raw and analyzed data and materials will be promptly reviewed by the Baylor College of Medicine Licensing Group to verify whether the request is subject to any intellectual property or confidentiality obligations. Patient-related data not included in the paper were generated as part of the clinical trial and might be subject to patient confidentiality restrictions. Any data and materials that can be shared will be released via a material transfer agreement. All raw data for single-cell sequencing have been deposited in the Gene Expression Omnibus with the accession code GSE154037. Source data are provided with this paper.
References
Godfrey, D. I., MacDonald, H. R., Kronenberg, M., Smyth, M. J. & Van Kaer, L. NKT cells: what’s in a name? Nat. Rev. Immunol. 4, 231–237 (2004).
Savage, A. K. et al. The transcription factor PLZF directs the effector program of the NKT cell lineage. Immunity 29, 391–403 (2008).
Metelitsa, L. S. Anti-tumor potential of type-I NKT cells against CD1d-positive and CD1d-negative tumors in humans. Clin. Immunol. 140, 119–129 (2011).
Metelitsa, L. S. et al. Natural killer T cells infiltrate neuroblastomas expressing the chemokine CCL2. J. Exp. Med. 199, 1213–1221 (2004).
Song, L. et al. Valpha24-invariant NKT cells mediate antitumor activity via killing of tumor-associated macrophages. J. Clin. Invest. 119, 1524–1536 (2009).
Tachibana, T. et al. Increased intratumor Valpha24-positive natural killer T cells: a prognostic factor for primary colorectal carcinomas. Clin. Cancer Res. 11, 7322–7327 (2005).
Cortesi, F. et al. Bimodal CD40/Fas-dependent crosstalk between iNKT cells and tumor-associated macrophages impairs prostate cancer progression. Cell Rep. 22, 3006–3020 (2018).
Noy, R. & Pollard, J. W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49–61 (2014).
Chang, D. H. et al. Sustained expansion of NKT cells and antigen-specific T cells after injection of alpha-galactosyl-ceramide loaded mature dendritic cells in cancer patients. J. Exp. Med. 201, 1503–1517 (2005).
Motohashi, S., Okamoto, Y., Yoshino, I. & Nakayama, T. Anti-tumor immune responses induced by iNKT cell-based immunotherapy for lung cancer and head and neck cancer. Clin. Immunol. 140, 167–176 (2011).
Exley, M. A. et al. Adoptive transfer of invariant NKT cells as immunotherapy for advanced melanoma: a phase I clinical trial. Clin. Cancer Res. 23, 3510–3519 (2017).
Heczey, A. et al. Invariant NKT cells with chimeric antigen receptor provide a novel platform for safe and effective cancer immunotherapy. Blood 124, 2824–2833 (2014).
Rotolo, A. et al. Enhanced anti-lymphoma activity of CAR19-iNKT cells underpinned by dual CD19 and CD1d targeting. Cancer Cell 34, 596–610 (2018).
Tahir, S. M. et al. Loss of IFN-gamma production by invariant NK T cells in advanced cancer. J. Immunol. 167, 4046–4050 (2001).
Tian, G. et al. CD62L+ NKT cells have prolonged persistence and antitumor activity in vivo. J. Clin. Invest. 126, 2341–2355 (2016).
Schulz, G. et al. Detection of ganglioside GD2 in tumor tissues and sera of neuroblastoma patients. Cancer Res. 44, 5914–5920 (1984).
Heczey, A. et al. CAR T cells administered in combination with lymphodepletion and PD-1 inhibition to patients with neuroblastoma. Mol. Ther. 25, 2214–2224 (2017).
Liu, D. et al. IL-15 protects NKT cells from inhibition by tumor-associated macrophages and enhances antimetastatic activity. J. Clin. Invest. 122, 2221–2233 (2012).
Xu, X., et al. NKT cells co-expressing a GD2-specific chimeric antigen receptor and IL-15 show enhanced in vivo persistence and antitumor activity against neuroblastoma. Clin. Cancer Res. 25, 7126–7138 (2019).
Cohn, S. L. et al. The international neuroblastoma risk group (INRG) classification system: an INRG task force report. J. Clin. Oncol. 27, 289–297 (2009).
Ghassemi, S. et al. Reducing ex vivo culture improves the antileukemic activity of chimeric antigen receptor (CAR) T cells. Cancer Immunol. Res. 6, 1100–1109 (2018).
Gattinoni, L. et al. Removal of homeostatic cytokine sinks by lymphodepletion enhances the efficacy of adoptively transferred tumor-specific CD8+ T cells. J. Exp. Med. 202, 907–912 (2005).
Park, J. R. et al. Revisions to the international neuroblastoma response criteria: a consensus statement from the National Cancer Institute Clinical Trials Planning Meeting. J. Clin. Oncol. 35, 2580–2587 (2017).
Decarolis, B. et al. Iodine-123 metaiodobenzylguanidine scintigraphy scoring allows prediction of outcome in patients with stage 4 neuroblastoma: results of the Cologne Interscore Comparison Study. J. Clin. Oncol. 31, 944–951 (2013).
Ngai, H. et al. IL-21 selectively protects CD62L+ NKT cells and enhances their effector functions for adoptive immunotherapy. J. Immunol. 201, 2141–2153 (2018).
Stoeckius, M. et al. Cell hashing with barcoded antibodies enables multiplexing and doublet detection for single cell genomics. Genome Biol. 19, 224 (2018).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Hafemeister, C. & Satija, R. Normalization and variance stabilization of single-cell RNA-seq data using regularized negative binomial regression. Genome Biol. 20, 296 (2019).
Butler, A., Hoffman, P., Smibert, P., Papalexi, E. & Satija, R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36, 411–420 (2018).
Guo, X. et al. Global characterization of T cells in non-small-cell lung cancer by single-cell sequencing. Nat. Med. 24, 978–985 (2018).
Zhang, L. et al. Lineage tracking reveals dynamic relationships of T cells in colorectal cancer. Nature 564, 268–272 (2018).
Yost, K. E. et al. Clonal replacement of tumor-specific T cells following PD-1 blockade. Nat. Med. 25, 1251–1259 (2019).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Acknowledgements
The authors are grateful to M. Brenner, H. Heslop and personnel from the cGMP facility at the Center for Cell and Gene Therapy for manufacturing CAR-NKT cells; A. Sher, K. Kukreja and P. Srivaths; staff of the Flow Cytometry Core Laboratory of the Texas Children’s Cancer and Hematology Center; P. Saha for excellent technical assistance; and E. Chomsky, E. Kiner and H. Sharim for help with scRNA-seq. This work was supported by grants and contracts from Alex’s Lemonade Stand Foundation for Childhood Cancer, Kuur Therapeutics (to L.S.M. and A.H.), the American Cancer Society (to A.H.), Cookies for Kids’ Cancer Foundation (to L.S.M.) and the Cancer Prevention and Research Institute of Texas (Baylor College of Medicine Comprehensive Cancer Center Training Program, RP160283, to H.N.).
Author information
Authors and Affiliations
Contributions
A.H. and L.S.M. designed the clinical trial. A.H. wrote the clinical protocol and was the principal investigator of the study. G.D., L.S.M., A.H. and B.S. developed and tested the CAR construct selected for the trial. S.R. wrote CAR-NKT cell manufacturing SOPs. S.R., K.L. and M.L. performed CAR-NKT cell manufacturing. A.N.C., H.N. and N.G. performed flow cytometry, O.D. performed qPCR, A.N.C. performed Luminex assay and B.L. performed Taqman RT–PCR. C.N.C.-F. performed bioenergetics analysis. X.X. performed the serial tumor challenge assay. T.R.-S. and A.M. performed scRNA-seq and analyzed data. The manuscript was written by A.H., A.N.C. and L.S.M. and edited by E.J.D.P. All authors discussed and interpreted the results.
Corresponding authors
Ethics declarations
Competing interests
A.H., A.N.C., G.D. and L.S.M are co-inventors on pending patent applications that relate to the use of NKT cells in cancer immunotherapy and have been licensed by Baylor College of Medicine to Kuur Therapeutics for commercial development. Kuur Therapeutics provided research support for this project (to L.S.M.) via a sponsored research agreement with Baylor College of Medicine. A.M., S.R., K.L., M.L., N.G., O.D., B.L., T.R.-S., C.N.C.-F., X.X., H.N., E.J.D.P. and B.S. declare no competing financial interests.
Additional information
Peer review information Saheli Sadanand was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Clinical trial design and enrollment.
a, Trial design: Cyclophosphamide/fludarabine (Cy/Flu) chemotherapy is given on days -4, -3, and -2 (purple arrows). CAR-NKTs are infused on day 0 (green arrow). Tumor biopsy is performed at week 2 (blue arrow) and response evaluation with imaging at week 4 (range 4-6; orange arrow). Black arrows indicate clinic visits and peripheral blood analysis. b, Enrollment CONSORT flow diagram.
Extended Data Fig. 2 Patient characteristics.
Indicated variables shown for the three patients enrolled on dose level 1. INSS: International Neuroblastoma Staging System; **Cy: cyclophosphamide, Flu: fludarabine. $: Dose: CAR-NKT cells / m2. Response criteria determined by revised International Neuroblastoma Response Criteria; partial response = elimination of at least 50% of bone metastatic lesions. DOD: Died of disease. AWD: Alive with disease.
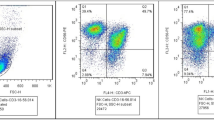
Extended Data Fig. 3 Characteristics of GD2-CAR-NKT cell products.
Patient cells were manufactured in the cGMP facility of the Center for Cell and Gene Therapy at Texas Children’s Hospital and evaluated using multi-color flow cytometry. a, NKT frequency (CD3+iNKT+) in initial apheresis product for the three patients. b, NKT purity and CAR expression at the end of manufacturing prior to cryopreservation. Shown are results from one of two independent experiments that produced similar results.
Extended Data Fig. 4 Adverse events.
Patients were monitored for adverse events from the beginning of lymphodepletion (day -4) until day 28 (four weeks post-infusion). Adverse events were described using Common Terminology Criteria for Adverse Events version 4.
Extended Data Fig. 5 Peripheral blood analysis pre- and post-infusion.
Peripheral blood samples were collected at indicated timepoints and a) the frequency of total white blood cells (WBCs), absolute lymphocyte counts (ALC), and absolute monocyte counts (AMC) were quantified in a standard clinical laboratory at the Texas Children’s Cancer Center. b, Frequency of myeloid and lymphoid cell subsets were quantified by flow cytometry staining. Absolute numbers of these subsets in peripheral blood were determined using flow cytometry data and AMC or ALC, respectively. Live PBMCs were gated and cell subsets were defined as shown in Supplementary Fig. 5. For patient 3, analyses of Treg and monocyte subsets were not performed at the week 4 timepoint.
Extended Data Fig. 6 Detection of CAR-NKT cells in treated patients pre- and post-infusion.
Flow cytometry staining showing iNKT+Vβ11+ cells after gating on CD3+ cells and corresponding GD2-CAR expression in patients at indicated timepoints. T-cells served as a negative control for CAR staining. N.D.: not defined; indicates that assay was not performed at given timepoint.
Extended Data Fig. 7 Expression of central memory and WNT pathway genes in pre-infusion patient products.
a, Gene expression in pre-infusion products from patients 1-3 for TNFRSF4 (encodes OX40), SELL (encodes CD62L), and genes related to Wnt-mediated signal transduction (https://www.gsea-msigdb.org/gsea/msigdb/cards/WNT_SIGNALING.html) overlaid on the UMAP 2D projection. b, Cryopreserved patient products were thawed and rested overnight prior to evaluating surface expression of OX40 (CD134) and CD62L (bottom) by flow cytometry using the corresponding fluorochrome-conjugated isotype controls (top). c, LEF1 expression in relation to CD62L was analyzed using intracellular flow cytometry (bottom) with the corresponding PE-conjugated isotype controls (top). Results are from one of two independent experiments with similar results.
Extended Data Fig. 8 Expression of metabolic genes and bioenergetic analysis of pre-infusion patient products.
a, Gene expression in pre-infusion products from patients 1-3 related to glycolysis (https://www.gsea-msigdb.org/gsea/msigdb/cards/KEGG_GLYCOLYSIS_GLUCONEOGENESIS) and oxidative phosphorylation (https://www.gsea-msigdb.org/gsea/msigdb/cards/KEGG_OXIDATIVE_PHOSPHORYLATION) overlaid on the UMAP 2D projection. b, Extracellular acidification rates (ECAR) and c) oxygen consumption rates (OCR) measured at baseline and in response to glucose (Glc), oligomycin (Oligo), 2-deoxy-D-glucose (2-DG), FCCP, or rotenone/antimycin A (R/A); data shown as mean ± SEM. d, Glycolytic and e) mitochondrial parameters derived from assay in b) and c) respectively; data shown as mean ± SD; P values by two-way ANOVA with Tukey’s correction for multiple comparisons (d,e). Shown are results from two independent experiments with 3 - 6 replicates per experiment.
Extended Data Fig. 9 Serial tumor challenge assay.
a, Patient GD2-CAR NKTs and NB cell line CHLA-255 were co-cultured in a 24-well plate. Five days later, cells were harvested for quantification by trypan-blue exclusion and FACS analysis. NKT cells were then re-plated at a 1:1 effector-to-target ratio with fresh tumor cells in fresh cell culture medium to start the next round with five rounds total. b, Representative flow cytometry analysis after round 4. Summary of c) tumor cell cytotoxicity and d) GD2-CAR NKT proliferation at the end of each five-day cycle. Data shown as mean ± SD from three technical replicates; P values by two-way ANOVA with Tukey’s correction for multiple comparisons (c and d).
Extended Data Fig. 10 Peripheral blood cytokine levels.
Peripheral blood was collected at indicated timepoints from the three patients and cytokine levels were quantified in triplicate by Luminex® assay. a, Cytokine expression is shown as log10 of pg/mL. b, IL7 and c) IL15 levels for the three patients at indicated timepoints. d, Peak IL15 concentration. Data shown as mean ± SD from three technical replicates (b-d); P values by unpaired two-tailed t-test (d).
Supplementary information
Supplementary Information
Supplementary Tables 1, 2 and 4; Supplementary Figs. 1–5; and Clinical Protocol.
Supplementary Table 3
Supplementary Table 3
Source data
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Source Data Extended Data Fig. 10
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Heczey, A., Courtney, A.N., Montalbano, A. et al. Anti-GD2 CAR-NKT cells in patients with relapsed or refractory neuroblastoma: an interim analysis. Nat Med 26, 1686–1690 (2020). https://doi.org/10.1038/s41591-020-1074-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-020-1074-2
- Springer Nature America, Inc.
This article is cited by
-
IL-12 reprograms CAR-expressing natural killer T cells to long-lived Th1-polarized cells with potent antitumor activity
Nature Communications (2024)
-
Targeting cytokine and chemokine signaling pathways for cancer therapy
Signal Transduction and Targeted Therapy (2024)
-
CAR-NKT Cells in Asthma: Use of NKT as a Promising Cell for CAR Therapy
Clinical Reviews in Allergy & Immunology (2024)
-
Generation of allogeneic CAR-NKT cells from hematopoietic stem and progenitor cells using a clinically guided culture method
Nature Biotechnology (2024)
-
Metastasis pattern and prognosis in children with neuroblastoma
World Journal of Surgical Oncology (2023)