Abstract
CD4+ T cells are central to various immune responses, but the molecular programs that drive and maintain CD4+ T cell immunity are not entirely clear. Here we identify a stem-like program that governs the CD4+ T cell response in transplantation models. Single-cell-transcriptomic analysis revealed that naive alloantigen-specific CD4+ T cells develop into TCF1hi effector precursor (TEP) cells and TCF1−CXCR6+ effectors in transplant recipients. The TCF1−CXCR6+CD4+ effectors lose proliferation capacity and do not reject allografts upon adoptive transfer into secondary hosts. By contrast, the TCF1hiCD4+ TEP cells have dual features of self-renewal and effector differentiation potential, and allograft rejection depends on continuous replenishment of TCF1−CXCR6+ effectors from TCF1hiCD4+ TEP cells. Mechanistically, TCF1 sustains the CD4+ TEP cell population, whereas the transcription factor IRF4 and the glycolytic enzyme LDHA govern the effector differentiation potential of CD4+ TEP cells. Deletion of IRF4 or LDHA in T cells induces transplant acceptance. These findings unravel a stem-like program that controls the self-renewal capacity and effector differentiation potential of CD4+ TEP cells and have implications for T cell-related immunotherapies.






Similar content being viewed by others
Data availability
The scRNA-seq data have been deposited in the Gene Expression Omnibus under accession no. GSE221337. Raw Illumina sequencing reads of scRNA-seq datasets were aligned to reference mouse genome mm10 (Ensembl 93), http://jul2018.archive.ensembl.org/Mus_musculus/Info/Index. All other data supporting the findings of this study are available within the article and supplementary information. Source data are provided with this paper.
Code availability
ScRNA-seq data analyses were conducted using publicly available codes and open-source software packages. No new algorithms were developed for this study.
References
Pawlak, M., Ho, A. W. & Kuchroo, V. K. Cytokines and transcription factors in the differentiation of CD4(+) T helper cell subsets and induction of tissue inflammation and autoimmunity. Curr. Opin. Immunol. 67, 57–67 (2020).
Ruterbusch, M., Pruner, K. B., Shehata, L. & Pepper, M. In vivo CD4(+) T cell differentiation and function: revisiting the Th1/Th2 paradigm. Annu. Rev. Immunol. 38, 705–725 (2020).
Smith-Garvin, J. E., Koretzky, G. A. & Jordan, M. S. T cell activation. Annu. Rev. Immunol. 27, 591–619 (2009).
Schnell, A. et al. Stem-like intestinal Th17 cells give rise to pathogenic effector T cells during autoimmunity. Cell 184, 6281–6298 (2021).
Xia, Y. et al. BCL6-dependent TCF-1(+) progenitor cells maintain effector and helper CD4(+) T cell responses to persistent antigen. Immunity 55, 1200–1215 (2022).
Zhou, X. et al. Differentiation and persistence of memory CD8(+) T cells depend on T cell factor 1. Immunity 33, 229–240 (2010).
Goldman, N. et al. Intrinsically disordered domain of transcription factor TCF-1 is required for T cell developmental fidelity. Nat. Immunol. https://doi.org/10.1038/s41590-023-01599-7 (2023).
Choi, Y. S. et al. LEF-1 and TCF-1 orchestrate T(FH) differentiation by regulating differentiation circuits upstream of the transcriptional repressor Bcl6. Nat. Immunol. 16, 980–990 (2015).
Im, S. J. et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature 537, 417–421 (2016).
Utzschneider, D. T. et al. T cell factor 1-expressing memory-like CD8(+) T cells sustain the immune response to chronic viral infections. Immunity 45, 415–427 (2016).
Wu, T. et al. The TCF1-Bcl6 axis counteracts type I interferon to repress exhaustion and maintain T cell stemness. Sci. Immunol. 1, eaai8593 (2016).
Gray, S. M., Amezquita, R. A., Guan, T., Kleinstein, S. H. & Kaech, S. M. Polycomb repressive complex 2-mediated chromatin repression guides effector CD8(+) T cell terminal differentiation and loss of multipotency. Immunity 46, 596–608 (2017).
Man, K. et al. The transcription factor IRF4 is essential for TCR affinity-mediated metabolic programming and clonal expansion of T cells. Nat. Immunol. 14, 1155–116 (2013).
Bollig, N. et al. Transcription factor IRF4 determines germinal center formation through follicular T-helper cell differentiation. Proc. Natl Acad. Sci. USA 109, 8664–8669 (2012).
Brustle, A. et al. The development of inflammatory T(H)-17 cells requires interferon-regulatory factor 4. Nat. Immunol. 8, 958–966 (2007).
Staudt, V. et al. Interferon-regulatory factor 4 is essential for the developmental program of T helper 9 cells. Immunity 33, 192–202 (2010).
Yao, S. et al. Interferon regulatory factor 4 sustains CD8(+) T cell expansion and effector differentiation. Immunity 39, 833–845 (2013).
Honma, K. et al. Interferon regulatory factor 4 differentially regulates the production of Th2 cytokines in naive vs. effector/memory CD4+ T cells. Proc. Natl Acad. Sci. USA 105, 15890–15895 (2008).
Spiljar, M. & Kuchroo, V. K. Metabolic regulation and function of T helper cells in neuroinflammation. Semin. Immunopathol. 44, 581–598 (2022).
Wang, R. & Green, D. R. Metabolic checkpoints in activated T cells. Nat. Immunol. 13, 907–915 (2012).
Claps, G. et al. The multiple roles of LDH in cancer. Nat. Rev. Clin. Oncol. 19, 749–762 (2022).
Peng, M. et al. Aerobic glycolysis promotes T helper 1 cell differentiation through an epigenetic mechanism. Science 354, 481–484 (2016).
Xu, K. et al. Glycolytic ATP fuels phosphoinositide 3-kinase signaling to support effector T helper 17 cell responses. Immunity 54, 976–987 (2021).
Xu, K. et al. Glycolysis fuels phosphoinositide 3-kinase signaling to bolster T cell immunity. Science 371, 405–410 (2021).
Angelin, A. et al. Foxp3 reprograms T cell metabolism to function in low-glucose, high-lactate environments. Cell Metab. 25, 1282–1293 (2017).
Wagner, A. et al. Metabolic modeling of single Th17 cells reveals regulators of autoimmunity. Cell 184, 4168–4185 (2021).
Bolton, E. M., Gracie, J. A., Briggs, J. D., Kampinga, J. & Bradley, J. A. Cellular requirements for renal allograft rejection in the athymic nude rat. J. Exp. Med. 169, 1931–1946 (1989).
Krieger, N. R., Yin, D. P. & Fathman, C. G. CD4+ but not CD8+ cells are essential for allorejection. J. Exp. Med. 184, 2013–2018 (1996).
Wu, J. et al. Ablation of transcription factor IRF4 promotes transplant acceptance by driving allogenic CD4(+) T cell dysfunction. Immunity 47, 1114–1128 (2017).
Liu, Z., Fan, H. & Jiang, S. CD4(+) T-cell subsets in transplantation. Immunol. Rev. 252, 183–191 (2013).
Conlon, T. M. et al. Germinal center alloantibody responses are mediated exclusively by indirect-pathway CD4 T follicular helper cells. J. Immunol. 188, 2643–2652 (2012).
Wang, G. et al. IRF4 ablation in B cells abrogates allogeneic B cell responses and prevents chronic transplant rejection. J. Heart Lung Transpl. 40, 1122–1132 (2021).
Miyahara, Y. et al. Anti-TCRβ mAb induces long-term allograft survival by reducing antigen-reactive T cells and sparing regulatory T cells. Am. J. Transplant. 12, 1409–1418 (2012).
Chen, S. et al. Epigenetically modifying the Foxp3 locus for generation of stable antigen-specific Tregs as cellular therapeutics. Am. J. Transplant. 20, 2366–2379 (2020).
Zhang, H. D. et al. Ablation of interferon regulatory factor 4 in T cells induces ‘memory’ of transplant tolerance that is irreversible by immune checkpoint blockade. Am. J. Transplant. 19, 884–893 (2019).
Zou, D. et al. T cell exhaustion is associated with antigen abundance and promotes transplant acceptance. Am. J. Transplant. 20, 2540–2550 (2020).
Grubin, C. E., Kovats, S., deRoos, P. & Rudensky, A. Y. Deficient positive selection of CD4 T cells in mice displaying altered repertoires of MHC class II-bound self-peptides. Immunity 7, 197–208 (1997).
Gearty, S. V. et al. An autoimmune stem-like CD8 T cell population drives type 1 diabetes. Nature 602, 156–161 (2022).
Di Pilato, M. et al. CXCR6 positions cytotoxic T cells to receive critical survival signals in the tumor microenvironment. Cell 184, 4512–4530 (2021).
Ng, S. S. et al. The NK cell granule protein NKG7 regulates cytotoxic granule exocytosis and inflammation. Nat. Immunol. 21, 1205–1218 (2020).
Kim, C. H. et al. Bonzo/CXCR6 expression defines type 1-polarized T-cell subsets with extralymphoid tissue homing potential. J. Clin. Invest. 107, 595–601 (2001).
Rengarajan, J. et al. Interferon regulatory factor 4 (IRF4) interacts with NFATc2 to modulate interleukin 4 gene expression. J. Exp. Med. 195, 1003–1012 (2002).
Li, P. et al. BATF-JUN is critical for IRF4-mediated transcription in T cells. Nature 490, 543–546 (2012).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Lee, Y. et al. Induction and molecular signature of pathogenic TH17 cells. Nat. Immunol. 13, 991–999 (2012).
Graef, P. et al. Serial transfer of single-cell-derived immunocompetence reveals stemness of CD8(+) central memory T cells. Immunity 41, 116–126 (2014).
Aronoff, L., Epelman, S. & Clemente-Casares, X. Isolation and identification of extravascular immune cells of the heart. J. Vis. Exp. 23, 58114 (2018).
Stuart, T. et al. Comprehensive integration of single-cell data. Cell 177, 1888–1902 (2019).
Acknowledgements
This study was supported by internal funding from Houston Methodist Research Institute (to W.C. and S.G.Y.) and the US National Institutes of Health grants (R01 AI132492 to W.C. and R01 AI129906 to X.C.L.). The authors thank the staff at the Single Cell Genomics Core at Baylor College of Medicine (partially supported by the National Institutes of Health shared instrument grants (S10OD023469 and S10OD025240), P30EY002520 and CPRIT grant RP200504), the Biostatistics and Bioinformatics shared resources at Houston Methodist Neal Cancer Center and the Houston Methodist Flow Cytometry Core Facility for excellent services. Schematic diagrams of experimental design were created with BioRender.com.
Author information
Authors and Affiliations
Contributions
D.Z., X.X., X.C.L. and W.C. designed the study and wrote the manuscript. D.Z., Z.Y. and W.C. performed core experimental work and data analysis. Z.Y., L.W. and S.T.C.W. performed computational analyses. S.G.Y., G.W., Y.G., S.L., X.Z., N.M.G., L.J.M., A.O.G. and R.M.G. performed supporting experimental work and data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks Vijay Kuchroo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: N. Bernard, in collaboration with the Nature Immunology team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Single-cell transcriptome analysis of alloreactive CD4+ T cells (related to Fig. 1).
a–e, CD45.2+ WT B6 mice were adoptively transferred with CD45.1+ TEa CD4+ T cells, followed by BALB/c heart transplantation 1 d later. The transferred TEa cells from spleens and heart allografts were obtained at 7 days post-transplantation for scRNA-seq. a, Percentage heart allograft survival after transplantation. n = 5 mice. b, The gating strategy for sorting the transferred TEa cells from recipients. c, Violin plots show the expression distributions of indicated T cell marker genes, stem-like genes, and effector genes in each of the TEa cell clusters. d, Feature plots show the normalized gene expression of stem-like TEP cell and effector cell markers projected onto the uniform manifold approximation and projection (UMAP). e, Feature plots show the normalized gene expression of Th1, Th2, Th17, and Treg cell markers, projected onto the UMAP. In both d and e, gene expression level represented by color gradient ranging from gray (low expression) to purple (high expression).
Extended Data Fig. 2 Phenotypic characterization of the adoptively transferred TEa CD4+ T cells in transplant recipients (related to Figs. 2 and 3).
a,b, WT B6 and B6.Tcf7GFP mice were transplanted with BALB/c hearts. a, Experimental scheme. b, Percentage heart allografts in WT B6 and B6.Tcf7GFP mice (n = 6 mice per group). c–f, B6.Rag1–/– mice were adoptively transferred with CD45.1+ Tcf7GFP TEa cells and transplanted with BALB/c skins. c, Experimental scheme. d, Percentage skin allograft survival. n = 7 mice. e, TCF1.GFP expression of TEa cells in blood at indicated days after transplantation. Flow plots are gated on TEa cells. The line graph shows percentages of TCF1.GFP− cells in TEa cells. Data are mean ± SD (n = 8 mice). f, TCF1.GFP expression of TEa cells in indicated tissues at 14 days after transplantation. Flow plots are gated on TEa cells. The bar graph shows percentages of TCF1.GFP+ cells in TEa cells. Data are mean ± SD (n = 4 mice), and results are representative of two independent experiments. g, CD45.1+ Tcf7GFP TEa cells were adoptively transferred into Rag1−/− mice receiving BALB/c skin transplantation. At 14 days post-transplantation, TCF1hi and TCF1− TEa cells were isolated from the secondary lymphoid organs and transferred into new Rag1–/– hosts that received BALB/c skin transplantation. In these new hosts, representative flow plots show TEa cell frequencies among CD45+ cells in indicated organs 14 days post-transplantation (related to Fig. 3e). In a and c, experimental schemes were created with BioRender.com. P values are from two-tailed unpaired Student’s t-test (f) or log-rank test (b).
Extended Data Fig. 3 CD4+ TEP cells self-renew and replenish the effector cell pool.
a–c, CD45.1+ Tcf7GFP TEa cells were adoptively transferred into Rag1−/− recipients receiving BALB/c skin transplantation. Ly108hiCXCR6− TEP TEa and Ly108loCXCR6+ effector TEa cells were isolated from the spleens and lymph nodes at 14 days post-transplantation, and adoptively transferred into new Rag1−/− recipients receiving BALB/c skin transplantation. The phenotypic changes of TEP TEa and effector TEa cells in new recipients were analyzed at 14 days post-transplantation. a, Experimental scheme. b, Representative flow plots (related to Fig. 3j) show percentage TEa cells within CD45+ cells in new recipients that were transferred with Ly108hiCXCR6− or Ly108loCXCR6+ TEa cells. Flow plots are gated on CD45+ cells. c, Representative flow plots (related to Fig. 3k) show Ly108 and CXCR6 expression of TEa cells in new recipients that were transferred with Ly108hiCXCR6− TEP TEa cells. Flow plots are gated on TEa cells. d,e, Cxcr6−/− and WT B6 mice were transplanted with BALB/c hearts. d, Experimental scheme. e, Percentage heart allograft survival after transplantation. n = 6 mice for the WT group, and n = 5 mice for the Cxcr6−/− group. In a and d, experimental schemes were created with BioRender.com. The P value is from log-rank test (e).
Extended Data Fig. 4 TCF1 sustains stem-like CD4+ TEP cells (related to Fig. 4).
a–d, CD45.1+ Tcf7−/− TEa and CD45.2+ WT TEa cells were mixed in a 1:1 ratio and adoptively co-transferred into Rag1−/− mice, followed by BALB/c skin transplantation. TEa cells in recipients were analyzed at 14 days post-transplantation. a, Experimental scheme. b, Percentage Tcf7−/− TEa and WT TEa cells among total TEa cells in spleens and grafts. n = 4 mice. c, IFN-γ production by Tcf7−/− TEa and WT TEa cells in spleens and dLNs. n = 4 mice. d, Percentage Ly108hiCXCR6– cells within Tcf7−/− TEa or WT TEa cells in the indicated tissues. n = 4 mice. Flow plots in b‒d are gated on TEa cells. e, Rag1−/− mice were adoptively transferred with WT TEa cells transduced with mCherry alone (Ctrl-mCherry) or TCF1-mCherry, followed by BALB/c skin transplantation. Representative flow plots show Ly108 and CXCR6 expression of mCherry+ TEa cells in dLN at 14 days post-transplantation. Flow plots are gated on mCherry+ TEa cells. The bar graph shows percentage Ly108hiCXCR6+ cells in mCherry+ TEa cells. n = 4 mice for the Ctrl-mCherry group, and n = 3 mice for the TCF1-mCherry group. f,g, Tcf7fl/flCd4-Cre and Tcf7fl/fl control mice were transplanted with BALB/c hearts. f, Experimental scheme. g, Percentage heart allograft survival after transplantation. n = 6 mice per group. In a and f, experimental schemes were created with BioRender.com. In b‒e, data are presented as mean ± SD, and results are representative of two independent experiments. P values are from two-tailed unpaired Student’s t-test (b‒e) or log-rank test (g).
Extended Data Fig. 5 IRF4 governs the effector differentiation potential of stem-like CD4+ TEP cells (related to Fig. 4).
a–f, Rag1−/− mice were adoptively co-transferred with CD45.2+ Irf4−/− TEa and CD45.1+ WT TEa cells (in a 1:1 ratio), followed by BALB/c skin transplantation. a, Experimental scheme. Created with BioRender.com. b, Percentage Irf4−/− TEa and WT TEa cells within total TEa cells in blood at indicated days post-transplantation. Flow plots are gated on TEa cells. n = 4 mice. c, Representative flow plots show Ly108 and CXCR6 expression of TEa cells in dLN at 14 days post-transplantation. The bar graph shows percentage Ly108hiCXCR6– cells in Irf4−/− TEa and WT TEa cells. n = 3 mice. d, Percentage of TCF1+ cells within Irf4−/− TEa and WT TEa cells in the dLN at 14 days post-transplantation. n = 3 mice. e, Percentage of IFN-γ+ cells among Irf4−/− TEa and WT TEa cells in the dLN at 14 days post-transplantation. n = 3 mice. f, Percentage of granzyme B+ cells among Irf4−/− TEa and WT TEa cells in the dLN at 14 days post-transplantation. n = 3 mice. g, Rag1−/− mice were adoptively transferred with Irf4−/− TEa cells transduced with GFP alone (Ctrl–GFP) or IRF4–GFP, followed by BALB/c skin transplantation. Histogram and bar graph show CXCR6 expression of GFP+ TEa cells in the dLN at 14 days post-transplantation. n = 3 mice per group. In b‒g, data are mean ± SD. P values are from two-tailed unpaired Student’s t-test (b‒g).
Extended Data Fig. 6 T-bet is dispensable for the generation of Ly108loCXCR6+ effectors.
a–b, Rag1−/− mice were adoptively co-transferred with CD45.1+ Tbx21−/− TEa and CD45.2+ WT TEa cells (in a 1:1 ratio), followed by BALB/c skin transplantation. The transferred TEa cells were analyzed at 14 days post-transplantation. a, Percentage Tbx21−/− TEa and WT TEa cells within total TEa cells in indicated tissues. Flow plots are gated on TEa cells. n = 4 mice. b, Percentage Ly108loCXCR6+ cells in Tbx21−/− TEa and WT TEa cells. Flow plots are gated on WT TEa or Tbx21−/− TEa cells. n = 4 mice. c,d, Rag1−/− mice were adoptively transferred with Tbx21−/− TEa or WT TEa cells, followed by BALB/c skin transplantation. c, Experimental scheme. Created with BioRender.com. d, Percentage allograft survival. n = 5 mice per group. In a,b, data are mean ± SD, and results are representative of two independent experiments. P values are from two-tailed unpaired Student’s t-test (a,b) or log-rank test (d).
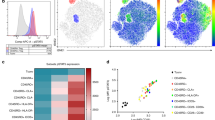
Extended Data Fig. 7 Identification of metabolic reactions that are significantly differentially active in stem-like TEP or effector CD4+ T cells.
a‒c, Compass algorithm was applied to analyze the metabolic states of alloantigen-specific TEa cells, based on scRNA-seq data. a, Differential activity of 1,497 reactions (colored dots) in 79 metabolic subsystems when compared between stem-like TEP TEa and effector TEa cells. Cohen′s d was used to determine the effect sizes. b, Volcano plots illustrate the detailed P values and effect sizes for metabolic reactions in 5 indicated subsystems when compared between stem-like TEP TEa and effector TEa cells. c, Spearman correlation of Compass scores with the expression of stem-like genes or effector genes. Rows are metabolic reactions selected from 8 subsystems listed in b and Fig. 5a–c. In b, data were analyzed by two-sided Wilcoxon rank-sum tests with Benjamini–Hochberg correction for multiple comparisons.
Extended Data Fig. 8 Enhanced glycolytic metabolism in CD4+ effector T cells.
a, Gene set enrichment analysis (GSEA) of scRNA-seq data identifies 801 pathways that are differentially expressed in TEa cell clusters. The bubble plot illustrates 48 representative pathways in each TEa cell cluster. Node sizes are proportional to NES scores from GSEA. Red color indicates upregulation in one cluster when compared to all other clusters. Blue color indicates downregulation in one cluster when compared to all other clusters. Color intensities correspond to FDR adjusted P values from GSEA. b, Schematic depicting the critical enzymes that catalyze sequential reactions in glycolysis. Adapted from ‘Glycolysis and Glycolytic Enzymes’, by BioRender.com (2023). Retrieved from https://app.biorender.com/biorender-templates. Genes significantly upregulated in effector (cluster 1) versus TEP (clusters 0 and 2) TEa cells were marked in bold red font with an asterisk. c, Violin plots show the expression distributions of indicated genes (encoding glycolytic enzymes) in each TEa cell clusters. In a, data were analyzed by weighted Kolmogorov-Smirnov tests, and false discovery rates (FDR) were calculated. In c, data were analyzed by two-sided Wilcoxon rank-sum tests with Bonferroni correction for multiple comparisons.
Extended Data Fig. 9 scRNA-seq identifies LDHA as a crucial regulator of effector differentiation of TEP cells.
a–d, On day 7 post-heart transplantation, transferred WT TEa and Ldha−/− TEa cells were isolated from recipient spleens for scRNA-seq analysis. a, UMAP analysis of TEa cells, including both WT TEa and Ldha−/− TEa cells from recipient spleens. Distinct color schemes were used to identify and visually represent the eight clusters (0–7). b, Feature plots project normalized Ldha gene expression onto the UMAPs for WT TEa or Ldha−/− TEa cell population. c,d, The compass algorithm was used to assess the metabolic states of WT stem-like and Ldha−/− stem-like TEa cells. c, Differential activity of metabolic reactions (colored dots) in metabolic subsystems between WT stem-like and Ldha−/− stem-like TEa cells, with Cohen’s d determining the effect sizes. d, Volcano plots illustrate the detailed P values and effect sizes for metabolic reactions in the citric acid cycle, oxidative phosphorylation, and glutamate metabolism. In d, data were analyzed by two-sided Wilcoxon rank-sum tests with Benjamini–Hochberg correction for multiple comparisons.
Extended Data Fig. 10 LDHA governs the effector differentiation potential of CD4+ TEP cells.
a, The UMAP feature plot shows normalized Ldha expression. Cluster numbers (related to Fig. 1b) are indicated in the UMAP plot. b, Experimental scheme for Fig. 6a. c, Experimental scheme for Fig. 6b. d‒h, CD45.1+ WT TEa and CD45.2+ Ldha−/− TEa cells were mixed in a 1:1 ratio, labeled with CTV, and adoptively co-transferred into Rag1−/− mice 1 d before BALB/c skin transplantation. The transferred TEa cells were analyzed by flow cytometry on day 14 post-skin transplantation. d, Experimental scheme. e, Percentage of WT TEa and Ldha−/− TEa cells among total TEa cells in dLNs and grafts. Flow plots are gated on TEa cells. n = 7 mice. f,g, Representative flow plots and bar graphs show % IFNγ+CTV− (f; n = 7 mice) and T-bet+CTV− (g; n = 7 mice) TEa cells among the transferred WT or Ldha−/− TEa cells in dLNs. h, Bar graphs show % TCF1−CTV−, Ly108−CTV−, CXCR6+CTV−, IFNγ+CTV−, and T-bet+CTV− TEa cells among the transferred WT or Ldha−/− TEa cells in spleens. n = 7 mice. In b‒d, experimental schemes were created with BioRender.com. In e‒h, data are mean ± SD, and results are pooled from two independent experiments. P values are from two-tailed unpaired Student’s t-test (e‒h).
Supplementary information
Source data
Source Data Fig. 1
Statistical Source Data.
Source Data Fig. 2
Statistical Source Data.
Source Data Fig. 3
Statistical Source Data.
Source Data Fig. 4
Statistical Source Data.
Source Data Fig. 6
Statistical Source Data.
Source Data Extended Data Fig. 1
Statistical Source Data.
Source Data Extended Data Fig. 2
Statistical Source Data.
Source Data Extended Data Fig. 3
Statistical Source Data.
Source Data Extended Data Fig. 4
Statistical Source Data.
Source Data Extended Data Fig. 5
Statistical Source Data.
Source Data Extended Data Fig. 6
Statistical Source Data.
Source Data Extended Data Fig. 10
Statistical Source Data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zou, D., Yin, Z., Yi, S.G. et al. CD4+ T cell immunity is dependent on an intrinsic stem-like program. Nat Immunol 25, 66–76 (2024). https://doi.org/10.1038/s41590-023-01682-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-023-01682-z
- Springer Nature America, Inc.
This article is cited by
-
Aerobic glycolysis enables the effector differentiation potential of stem-like CD4+ T cells to combat cancer
Cellular & Molecular Immunology (2024)
-
Reciprocal regulation of T follicular helper cells and dendritic cells drives colitis development
Nature Immunology (2024)
-
Stem-like T cells are associated with the pathogenesis of ulcerative colitis in humans
Nature Immunology (2024)