Abstract
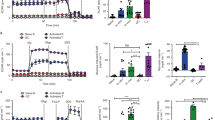
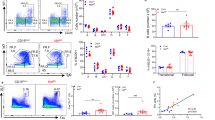
Following infection or vaccination, activated B cells at extrafollicular sites or within germinal centers (GCs) undergo vigorous clonal proliferation. Proliferating lymphocytes have been shown to undertake lactate dehydrogenase A (LDHA)-dependent aerobic glycolysis; however, the specific role of this metabolic pathway in a B cell transitioning from a naïve to a highly proliferative, activated state remains poorly defined. Here, we deleted LDHA in a stage-specific and cell-specific manner. We find that ablation of LDHA in a naïve B cell did not profoundly affect its ability to undergo a bacterial lipopolysaccharide-induced extrafollicular B cell response. On the other hand, LDHA-deleted naïve B cells had a severe defect in their capacities to form GCs and mount GC-dependent antibody responses. In addition, loss of LDHA in T cells severely compromised B cell-dependent immune responses. Strikingly, when LDHA was deleted in activated, as opposed to naïve, B cells, there were only minimal effects on the GC reaction and in the generation of high-affinity antibodies. These findings strongly suggest that naïve and activated B cells have distinct metabolic requirements that are further regulated by niche and cellular interactions.







Similar content being viewed by others
Data availability
All sequencing data generated in this study are deposited at the Gene Expression Omnibus under the accession code GSE232660. All custom pipelines developed for analysis of scRNA-seq data and Jh4 mutation data are available at https://github.com/ryashka/LDHA. Source data are provided with this paper.
References
Elsner, R. A. & Shlomchik, M. J. Germinal center and extrafollicular B cell responses in vaccination, immunity, and autoimmunity. Immunity 53, 1136–1150 (2020).
Mesin, L., Ersching, J. & Victora, G. D. Germinal center B cell dynamics. Immunity 45, 471–482 (2016).
Roco, J. A. et al. Class-switch recombination occurs infrequently in germinal centers. Immunity 51, 337–350 (2019).
Chang, C. H. et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell 153, 1239–1251 (2013).
Lunt, S. Y. & Vander Heiden, M. G. Aerobic glycolysis: meeting the metabolic requirements of cell proliferation. Annu. Rev. Cell Dev. Biol. 27, 441–464 (2011).
O’Neill, L. A., Kishton, R. J. & Rathmell, J. A guide to immunometabolism for immunologists. Nat. Rev. Immunol. 16, 553–565 (2016).
Warburg, O., Gawehn, K. & Geissler, A. W. Metabolism of leukocytes. Z. Naturforsch. B 13B, 515–516 (1958).
Peng, M. et al. Aerobic glycolysis promotes T helper 1 cell differentiation through an epigenetic mechanism. Science 354, 481–484 (2016).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Xu, K. et al. Glycolysis fuels phosphoinositide 3-kinase signaling to bolster T cell immunity. Science 371, 405–410 (2021).
Wu, L. et al. Niche-selective inhibition of pathogenic Th17 cells by targeting metabolic redundancy. Cell 182, 641–654 (2020).
Xu, K. et al. Glycolytic ATP fuels phosphoinositide 3-kinase signaling to support effector T helper 17 cell responses. Immunity 54, 976–987 (2021).
Crotty, S. T follicular helper cell differentiation, function, and roles in disease. Immunity 41, 529–542 (2014).
Akkaya, M. & Pierce, S. K. From zero to sixty and back to zero again: the metabolic life of B cells. Curr. Opin. Immunol. 57, 1–7 (2019).
Caro-Maldonado, A. et al. Metabolic reprogramming is required for antibody production that is suppressed in anergic but exaggerated in chronically BAFF-exposed B cells. J. Immunol. 192, 3626–3636 (2014).
Doughty, C. A. et al. Antigen receptor-mediated changes in glucose metabolism in B lymphocytes: role of phosphatidylinositol 3-kinase signaling in the glycolytic control of growth. Blood 107, 4458–4465 (2006).
Woodland, R. T. et al. Multiple signaling pathways promote B lymphocyte stimulator dependent B cell growth and survival. Blood 111, 750–760 (2008).
Jellusova, J. et al. Gsk3 is a metabolic checkpoint regulator in B cells. Nat. Immun. 18, 303–312 (2017).
Cho, S. H. et al. Germinal centre hypoxia and regulation of antibody qualities by a hypoxia response system. Nature 537, 234–238 (2016).
Weisel, F. J. et al. Germinal center B cells selectively oxidize fatty acids for energy while conducting minimal glycolysis. Nat. Immunol. 21, 331–342 (2020).
Haniuda, K., Fukao, S. & Kitamura, D. Metabolic reprogramming induces germinal center B cell differentiation through Bcl6 locus remodeling. Cell Rep. 33, 108333 (2020).
Chen, D. et al. Coupled analysis of transcriptome and BCR mutations reveals role of OXPHOS in affinity maturation. Nat. Immunol. 22, 904–913 (2021).
Waters, L. R., Ahsan, F. M., Wolf, D. M., Shirihai, O. & Teitell, M. A. Initial B cell activation induces metabolic reprogramming and mitochondrial remodeling. iScience 5, 99–109 (2018).
Price, M. J., Patterson, D. G., Scharer, C. D. & Boss, J. M. Progressive upregulation of oxidative metabolism facilitates plasmablast differentiation to a T-independent antigen. Cell Rep. 23, 3152–3159 (2018).
Hosios, A. M. & Vander Heiden, M. G. The redox requirements of proliferating mammalian cells. J. Biol. Chem. 293, 7490–7498 (2018).
Kwon, K. et al. Instructive role of the transcription factor E2A in early B lymphopoiesis and germinal center B cell development. Immunity 28, 751–762 (2008).
Chaudhuri, J. & Alt, F. W. Class-switch recombination: interplay of transcription, DNA deamination and DNA repair. Nat. Rev. Immunol. 4, 541–552 (2004).
Zdralevic, M. et al. Double genetic disruption of lactate dehydrogenases A and B is required to ablate the ‘Warburg effect’ restricting tumor growth to oxidative metabolism. J. Biol. Chem. 293, 15947–15961 (2018).
Allman, D. et al. Resolution of three nonproliferative immature splenic B cell subsets reveals multiple selection points during peripheral B cell maturation. J. Immunol. 167, 6834–6840 (2001).
Peng, S. L. Signaling in B cells via Toll-like receptors. Curr. Opin. Immunol. 17, 230–236 (2005).
Barwick, B. G., Scharer, C. D., Bally, A. P. R. & Boss, J. M. Plasma cell differentiation is coupled to division-dependent DNA hypomethylation and gene regulation. Nat. Immunol. 17, 1216–1225 (2016).
Tangye, S. G., Bryant, V. L., Cuss, A. K. & Good, K. L. BAFF, APRIL and human B cell disorders. Semin. Immunol. 18, 305–317 (2006).
Boothby, M. & Rickert, R. C. Metabolic regulation of the immune humoral response. Immunity 46, 743–755 (2017).
Reboldi, A. & Cyster, J. G. Peyer’s patches: organizing B-cell responses at the intestinal frontier. Immunol. Rev. 271, 230–245 (2016).
Jacob, J., Kelsoe, G., Rajewsky, K. & Weiss, U. Intraclonal generation of antibody mutants in germinal centres. Nature 354, 389–392 (1991).
Reimer, D. et al. Early CCR6 expression on B cells modulates germinal centre kinetics and efficient antibody responses. Immunol. Cell Biol. 95, 33–41 (2017).
Schwickert, T. A. et al. A dynamic T cell-limited checkpoint regulates affinity-dependent B cell entry into the germinal center. J. Exp. Med. 208, 1243–1252 (2011).
Jacobsen, J. T. et al. Expression of Foxp3 by T follicular helper cells in end-stage germinal centers. Science 373, eabe5146 (2021).
Ganeshan, K. & Chawla, A. Metabolic regulation of immune responses. Annu. Rev. Immunol. 32, 609–634 (2014).
Robbiani, D. F. et al. AID is required for the chromosomal breaks in c-myc that lead to c-myc/IgH translocations. Cell 135, 1028–1038 (2008).
Jolly, C. J., Klix, N. & Neuberger, M. S. Rapid methods for the analysis of immunoglobulin gene hypermutation: application to transgenic and gene targeted mice. Nucleic Acids Res. 25, 1913–1919 (1997).
Sheppard, S. et al. Lactate dehydrogenase A-dependent aerobic glycolysis promotes natural killer cell anti-viral and anti-tumor function. Cell Rep. 35, 109210 (2021).
DeFranco, A. L., Raveche, E. S. & Paul, W. E. Separate control of B lymphocyte early activation and proliferation in response to anti-IgM antibodies. J. Immunol. 135, 87–94 (1985).
Dominguez-Sola, D. et al. The proto-oncogene MYC is required for selection in the germinal center and cyclic reentry. Nat. Immunol. 13, 1083–1091 (2012).
Schuhmacher, M. et al. Control of cell growth by c-Myc in the absence of cell division. Curr. Biol. 9, 1255–1258 (1999).
Kwak, K., Akkaya, M. & Pierce, S. K. B cell signaling in context. Nat. Immunol. 20, 963–969 (2019).
Zhang, D. et al. Metabolic regulation of gene expression by histone lactylation. Nature 574, 575–580 (2019).
Fagone, P. et al. Phospholipid biosynthesis program underlying membrane expansion during B-lymphocyte differentiation. J. Biol. Chem. 282, 7591–7605 (2007).
Goldfinger, M. et al. De novo ceramide synthesis is required for N-linked glycosylation in plasma cells. J. Immunol. 182, 7038–7047 (2009).
Kirk, S. J., Cliff, J. M., Thomas, J. A. & Ward, T. H. Biogenesis of secretory organelles during B cell differentiation. J. Leukoc. Biol. 87, 245–255 (2010).
Wiest, D. L. et al. Membrane biogenesis during B cell differentiation: most endoplasmic reticulum proteins are expressed coordinately. J. Cell Biol. 110, 1501–1511 (1990).
Dufort, F.J. et al. Glucose-dependent de Novo Lipogenesis in B Lymphocytes. J. Bio. Chem. 289, 7011–7024 (2014).
Lam, W. Y. et al. Mitochondrial pyruvate import promotes long-term survival of antibody-secreting plasma cells. Immunity 45, 60–73 (2016).
Muschen, M. Metabolic gatekeepers to safeguard against autoimmunity and oncogenic B cell transformation. Nat. Rev. Immunol. 19, 337–348 (2019).
Lee, P. P. et al. A critical role for Dnmt1 and DNA methylation in T cell development, function, and survival. Immunity 15, 763–774 (2001).
Sage, P. T. & Sharpe, A. H. In vitro assay to sensitively measure TFR suppressive capacity and TFH stimulation of B cell responses. Methods Mol. Biol. 1291, 151–160 (2015).
Yewdell, W. T. et al. A Hyper-IgM syndrome mutation in activation-induced cytidine deaminase disrupts G-Quadruplex binding and genome-wide chromatin localization. Immunity 53, 952–970 (2020).
Yewdell, W. T. et al. Temporal dynamics of persistent germinal centers and memory B cell differentiation following respiratory virus infection. Cell Rep. 37, 109961 (2021).
Acknowledgements
R.S. was supported by the National Cancer Institute (U54CA137788); J.C. was supported by grants from the National Institutes of Health (R01AI072194 and R01AI124186) and from the National Cancer Institute (U54CA137788 and P30CA008748), the Starr Cancer Research Foundation, the Ludwig Center for Cancer Immunotherapy, MSKCC Functional Genomics and the Geoffrey Beene Cancer Center. M.O.L. was supported by a grant from the National Institutes of Health R01 (AI 102888). This work was supported in part by a grant from the Tri-Institutional Metabolomics Training Program (R25 AI140472) to J.R.C. J.R.C. is also supported by the Donald B. and Catherine C. Marron Cancer Metabolism Center. We thank A. Bravo for help with maintenance of the mouse colony. We thank all members of the laboratory of J.C. for helpful discussions and feedback. We acknowledge the use of the MSKCC Single-cell Analysis Innovation Lab for the generation of the scRNA-seq dataset and the Integrated Genomic Operation Core and the Molecular Cytology Core Facility at MSKCC supported by a Core Grant (P30CA008748).
Author information
Authors and Affiliations
Contributions
R.S. and J.C. conceptualized the work, analyzed the data and wrote the manuscript; R.M.S., P.C., K.C.F., Y.K., M.C., W.-F.Y., W.H., Z.-M.W. and S.V. collected, analyzed and validated data; R.M.S. and R.C. analyzed and interpreted scRNA-seq data; R.M.S. and K.C.F. conceptualized and carried out sequencing for the SHM assay; W.A. genotyped mice and made general reagents for experiments; M.O.L. generated conditional Ldha mice; J.R.C. analyzed data on B cell metabolism.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Immunology thanks Ulf Klein and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: L. A. Dempsey, in collaboration with the Nature Immunology team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Confirmation of Ldha deletion in naïve B cells.
a, PCR analysis of genomic DNA from naïve splenic B cells. Two Ldhafl/flCd23Cre and two Ldha+/+Cd23Cre control mice were analyzed for the presence of the deleted (null), floxed and wild type Ldha alleles. Data is representative of 3 independent experiments. b, Naïve splenic B cells harvested from mice of the indicated genotypes (n = 2 for Ldha+/+Cd23Cre, n = 3 for Ldhafl/flCd23Cre) or splenic B cells activated with LPS + IL-4 for 48 h (n = 3 for each group) were examined for expression of Ldha by qPCR. Heart tissue harvested from Ldha+/+Cd23Cre mice (n = 1) was used as a control. c, Naïve splenic B cells were activated ex vivo with LPS + IL-4 for 72 h and whole cell protein extracts were analyzed by immunoblotting using antibodies against LDHA or α-tubulin (control). Data is representative of three independent experiments. d, Naïve and activated B cells were analyzed for expression of Ldhb by qPCR. n = 3 for each group, except n = 2 for naive Ldha+/+Cd23Cre B cells and n = 1 for heart tissue. For panels b and d, data represents mean ± s.d.
Extended Data Fig. 2 LDHA governs aerobic glycolysis in B cells.
a, Schematic representation of mito stress assays performed in a Seahorse analyzer. The mito stress assay measures the oxygen flux of cells cultured in glucose-sufficient media supplemented with glutamine and sodium pyruvate. During this assay, the oxygen consumption rate (OCR) is first measured at baseline and after the sequential addition of oligomycin (which inhibits ATP synthase and reduces OCR), FCCP (an uncoupling agent that disrupts the mitochondrial membrane potential without inhibiting the electron transport chain and allows oxygen consumption to reach a maximum), and a mixture of rotenone and antimycin A (AA) (which shuts down mitochondrial respiration). b, Representative graph of OCR of naïve B cells at baseline and following the indicated perturbations. c, Quantification of OCR and of spare respiratory capacity (SRC) of naïve splenic B cells from Ldhafl/flCd23Cre and Ldha+/+Cd23Cre (control) mice. SRC was calculated as the difference of FCCP-stimulated maximum OCR and basal OCR. d, e, Naïve splenic B cells were activated with LPS + IL4, anti-CD40 + IL4 or anti-CD40 + IL4+anti-IgM for 48 h and OCR and SRC was quantified. p values were calculated using unpaired, two-tailed t-tests. For panels b–e, n = 6 for each indicated genotype and representative of three independent experiments. Data points and bars represent mean ± s.e.m.
Extended Data Fig. 3 Cellular homeostasis in LDHA-deficient activated B cells.
a, Uptake of the fluorescent glucose analog 2-NBDG. Naïve B cells (n = 5 for each genotype) were activated ex vivo with LPS + IL4 for 48 h, incubated with 11 mM of 2-NBDG for 15 min, and 2-NBDG uptake was quantified by flow cytometry. Data is representative of two independent experiments. b, Splenic B cells from Ldhafl/flCd23Cre (n = 3) and Ldha+/+Cd23Cre (control, n = 3) mice were activated ex vivo as indicated for 48 h and intracellular ATP level was determined. Data is representative of two independent experiments. c, d, Splenic B cells from Ldhafl/flCd23Cre and Ldha+/+Cd23Cre (control) mice were labeled with Cell Trace Violet (CTV) dye, activated ex vivo for 48 h with LPS + IL4, and CTV dilution was determined by flow cytometry as a measure of cell proliferation. Data shown is representative of three independent experiments. d, Splenic B cells (1 × 106) were harvested from mice of the indicated genotypes (n = 7 for each group), stimulated with LPS + IL-4 and cell numbers were quantified at the indicated time points. e, Splenic B cells (1 × 106) harvested from mice of the indicated genotypes (n = 4 for each group) were stimulated with LPS + TGFβ + anti-IgD ex vivo and cell numbers were determined at 96 h post-activation. Data is representative of two independent experiments. Bars represent mean ± s.e.m. *p ≤ 0.05, ****p ≤ 0.0001 by unpaired, two-tailed t-test.
Extended Data Fig. 4 B cell subsets at homeostasis.
a, Representative flow plot of splenic transitional T1 (B220+CD93+CD23−IgMhi), T2 (B220+CD93+CD23+IgMhi) and T3 (B220+CD93+CD23+IgMlow) B cells. Live singlets from the spleen were first gated for B220+CD93+ cells and then gated for surface expression of IgM and CD23 as indicated. b, Quantification of T1, T2, and T3 B cells in the spleens of mice of indicated genotypes. c, Representative flow plots of B1a (B220lowCD19+CD5+) B cells in the peritoneum of mice. Live singlets from the peritoneal cavity were first gated for B220lowCD19+cells and then gated for surface expression of CD52. d, Quantification of B1a cells in the peritoneum of mice of the indicated genotypes. e, Representative flow plots of marginal zone (MZ) B cells and follicular zone (FO) B cell subsets in the spleen of mice of the indicated genotypes. Live singlets from the spleen were first gated for mature B220+CD93−B cells and then analyzed for surface expression of CD21 and CD23. f,g, Frequency and absolute number of FO B cells. h, i, Frequency and absolute number of MZ B cells. j, Frequency of CD4+ and CD8+ T cells (among live singlets) in peripheral blood and k, in the spleen of mice of the indicated genotypes. Each datapoint represents a single mouse. Bars represent mean. *p ≤ 0.05, **p ≤ 0.01, p-values were calculated using unpaired, two-tailed t-test. For panels b, d, f–k, n = 5 of each genotype and data is representative of two independent experiments.
Extended Data Fig. 5 Confirmation of Ldha deletion in CD138+ plasmablasts, marginal zone (MZ) and follicular (FO) B cells; IgM ELISA of culture supernatants of ex vivo FO and MZ B cells.
a, Gating strategy for the analysis of B220lo CD138+ cells in the spleens of mice challenged with LPS. b, Representative PCR showing the genomic deletion of the floxed Ldha exon in CD138+ plasmablasts sorted from the spleens of Ldhafl/flCd23Cre and control mice at d5 after LPS challenge. Tail DNA was used as a control to detect both the null and floxed alleles. Image of ethidium bromide-stained gel shown is representative of three independent experiments. c, Purified FO or MZ B cells of the indicated genotypes (n = 3) were cultured ex vivo for 72 h with LPS + IL4 or LPS respectively and IgM concentration in the culture supernatants was determined by ELISA. Bars represent mean ± s.e.m. d, Representative PCR depicting genomic deletion of Ldha from sorted FO and MZ B cells cultured with LPS. Image of ethidium bromide-stained gel shown is representative of three independent experiments.
Extended Data Fig. 6 Gating strategy for analysis of GC B cells following NP-CGG immunization, quantification of GC density and gating strategy for sorting of GL7+ and GL7− B cells.
a, Gating strategy for analysis of GC B cells and associated populations in the spleens of NP-CGG immunized mice. b, Mice were immunized intraperitoneally with NP-CGG, boosted at d10 and the GC density in the spleen sections from Ldha+/+ Cd23Cre (grey bar) and Ldhafl/fl Cd23Cre (red bar) mice was quantified at d14 post-immunization. Each data point represents one independent experiment in which data from the spleen sections of 3 mice per genotype were pooled. Data presented is mean ± s.e.m. c, Detection of pre-GC B cells. Wild type mice were immunized intraperitoneally with NP-CGG or with PBS and the frequency of GL7+ cells quantified at d4 post-immunization. d, Gating strategy for the sorting of GL7+ and GL7− cells from the spleens of NP-CGG immunized mice at d4 post-immunization. Sorted GL7+ and GL7− cells were mixed 3:1 and subjected to scRNA sequencing analysis. Flow cytometry of the post-sort cells is shown.
Extended Data Fig. 7 LDHA in T cells regulates GC B cell responses.
Ldhafl/fl and Ldhafl/flCd4Cre mice were immunized intraperitoneally with NP-CGG, boosted at d10, and analyzed at d14. a, Immunofluorescence staining of frozen spleen sections harvested at d14. Image is representative of n = 3 mice per genotype. Scale bar represents 50 μm. b–e, Splenic B cells (5 × 105) from wild type mice were co-cultured with TFH cells (3 × 105) purified from Ldhafl/flCd4Cre (n = 2 pooled samples) or Ldhafl/fl (n = 2 pooled samples) control mice. The cells were analyzed at d6 post initiation of culture. b, c, Number of B and TFH cells at d6. d, e, Antibody titers of IgM and IgG1 in culture supernatant. *p ≤ 0.05, by unpaired, two-tailed t-test. Data represents two biological samples, each pooled from 4 mice per genotype. Bars represent either mean ± s.e.m (panels b, c, and e) or mean ± s.d. (panel d).
Extended Data Fig. 8 LDHA in T cells is dispensable for LPS-dependent extrafollicular responses.
The spleen of Ldhafl/flCd4Cre (n = 8) and Ldhafl/fl (control, n = 7) mice were analyzed at d5 after LPS administration. a, Staining of Ki67 in activated (GL7+) and unactivated (GL7−) B cells in mice of indicated genotypes. b, mTORC1 activity was quantified from the frequency of phosphorylated S6 (p-S6) positive cells in activated (GL7+) B cells. c, Frequency of CD4+ and CD8+ T cells among live singlets. d, Ki67 staining of CD4+ and CD8+ T cells. e, Quantification of naïve (CD62LhiCD44lo), central memory (CD62LhiCD44hi) and effector (CD62LloCD44hi) CD4+ and CD8+ T cells at d5 post-LPS challenge. Data is representative of three independent experiments. Bars represent mean ± s.e.m.
Extended Data Fig. 9 Ldhafl/flAicdaCre/+ mice can mount a robust GC response.
Ldhafl/flAicdaCre/+ and Ldha+/+AicdaCre/+ (control) mice were immunized intraperitoneally with NP-CGG, boosted at d10, and analyzed at d14. a, Viability of GC B cells as measured by cells negative for Zombie Red viability dye. b, Cell size of viable GC B cells as measured by quantifying the MFI of forward scatter (FSA). c, Mitochondrial mass was quantified by measuring the MFI of MitoTracker™ Green FM Dye among the GC and non-GC B cells. d, Frequency of dysfunctional mitochondria was measured by quantifying the fraction of live GC and non-GC B cells negative for MitoTracker™ Red CMXRos. e, PCR of genomic DNA of GC and non-GC B cells sorted from spleens of mice of the indicated genotypes. f, Immunoblotting of whole cell extracts prepared from GC and non-GC B cells sorted from the Peyer’s patches and spleens of mice of the indicated genotypes to detect LDHA and α-tubulin (control) proteins. g, Expression of Ldha and Ldhb in sorted GC B cells. Bars represent mean. Each datapoint represents a single mouse (n = 3). The box plot depicts the first quartile, third quartile, and the median of the dataset. Values that fall within 1.5 times the interquartile range above the third quartile and below the first quartile are represented by the whiskers. The gating strategy for the sorting of GC B cells is the same as that in Extended Data Fig. 6a. For panels a and b, n = 8 for each genotype; data is representative of two independent experiments. For panels c and d, n = 5 for each genotype and data is representative of two independent experiments. For panels e, and f, data is representative of 2 independent experiments with 3 to 5 mice per experiment.
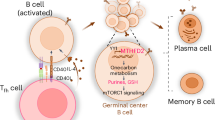
Extended Data Fig. 10 Working model depicting metabolic requirements in naïve and in activated B cells.
Proposed model suggesting different metabolic constraints in B cells committed to either a T-dependent or a T-independent pathway for an optimal humoral response. LDHA-mediated glycolysis is essential for the early activation of B cells during a T-dependent GC response but is largely dispensable once the activated B cells are fully engaged in a GC reaction ('>' sign symbolizes 'greater than'). On the other hand, a T-independent extrafollicular response is not reliant on LDHA-mediated glycolysis.
Supplementary information
Supplementary Information
Supplementary Table 1: Antibody catalog numbers
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Unmodified gel Extended Data Fig. 1c
Immunoblot
Unmodified gel Extended Data Fig. 9f
Immunoblot
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, R., Smolkin, R.M., Chowdhury, P. et al. Distinct metabolic requirements regulate B cell activation and germinal center responses. Nat Immunol 24, 1358–1369 (2023). https://doi.org/10.1038/s41590-023-01540-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-023-01540-y
- Springer Nature America, Inc.
This article is cited by
-
TCL1A-expressing B cells are critical for tertiary lymphoid structure formation and the prognosis of oral squamous cell carcinoma
Journal of Translational Medicine (2024)
-
Metabolic determinants of germinal center B cell formation and responses
Nature Chemical Biology (2024)
-
Single-B cell analysis correlates high-lactate secretion with stress and increased apoptosis
Scientific Reports (2024)
-
B cells ferment sugar before they (affinity) mature
Nature Immunology (2023)