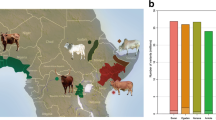
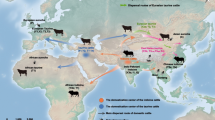
Abstract
Cattle pastoralism plays a central role in human livelihood in Africa. However, the genetic history of its success remains unknown. Here, through whole-genome sequence analysis of 172 indigenous African cattle from 16 breeds representative of the main cattle groups, we identify a major taurine × indicine cattle admixture event dated to circa 750–1,050 yr ago, which has shaped the genome of today’s cattle in the Horn of Africa. We identify 16 loci linked to African environmental adaptations across crossbred animals showing an excess of taurine or indicine ancestry. These include immune-, heat-tolerance- and reproduction-related genes. Moreover, we identify one highly divergent locus in African taurine cattle, which is putatively linked to trypanotolerance and present in crossbred cattle living in trypanosomosis-infested areas. Our findings indicate that a combination of past taurine and recent indicine admixture-derived genetic resources is at the root of the present success of African pastoralism.






Similar content being viewed by others
Data availability
The newly generated sequences for 114 African cattle and two African buffalo samples are available from the Sequence Read Archive (SRA) with the Bioproject accession number PRJNA574857. The publicly available sequences were downloaded from the SRA and China National GeneBank (CNGB) with the following project accession numbers; CNP0000189 (Achai, Bhagnari, Cholistani, Dajal, Dhanni, Gabrali, Hariana, Lohani, Red Sindhi, Sahiwal and Tharparkar), PRJNA318087 (Angus, Ankole, Jersey, Kenya Boran, Kenana, N’Dama and Ogaden), PRJNA514237 (Boskarin, Limia, Maremmana, Maronesa, Pajuna, Podolica and Sayaguesa), PRJNA324822 (Brahman), PRJNA343262 (Brahman, Gir, Hereford, Nelore and Simmental), PRJNA432125 (Brahman), PRJEB28185 (Eastern Finn and Western Finn), PRJNA210523 (Hanwoo), PRJNA379859 (Hariana, Sahiwal and Thaparkar), PRJNA210521 (Holstein), PRJNA386202 (Muturu) and PRJNA507259 (Nelore). The known variants file (ARS1.2PlusY_BQSR_v3.vcf.gz) for base quality score recalibration was provided by the 1000 Bull Genomes Project (http://www.1000bullgenomes.com/). The annotation of the candidate regions was based on the ARS-UCD1.2 Gene Transfer Format file (.gtf) from Ensembl release 99 (http://www.ensembl.org/). The PANTHER database (http://pantherdb.org/) was used for functional enrichment analysis of a candidate gene set.
References
Schneider, H. K. A model of African indigenous economy and society. Comp. Stud. Soc. Hist. 7, 37–55 (1964).
Di Lernia, S. et al. Inside the ‘African cattle complex’: animal burials in the Holocene central Sahara. PLoS ONE 8, e56879 (2013).
Mwai, O., Hanotte, O., Kwon, Y.-J. & Cho, S. African indigenous cattle: unique genetic resources in a rapidly changing world. Asian-Australas. J. Anim. Sci. 28, 911–921 (2015).
Roberts, C. & Gray, A. Studies on trypanosome-resistant cattle. II. The effect of trypanosomiasis on N’dama, Muturu and Zebu cattle. Trop. Anim. Health Prod. 5, 220–233 (1973).
Hanotte, O. et al. Geographic distribution and frequency of a taurine Bos taurus and an indicine Bos indicus Y specific allele amongst sub‐Saharan African cattle breeds. Mol. Ecol. 9, 387–396 (2000).
Hanotte, O. et al. African pastoralism: genetic imprints of origins and migrations. Science 296, 336–339 (2002).
Loftus, R. T., MacHugh, D. E., Bradley, D. G., Sharp, P. M. & Cunningham, P. Evidence for two independent domestications of cattle. Proc. Natl Acad. Sci. USA 91, 2757–2761 (1994).
MacHugh, D. E., Shriver, M. D., Loftus, R. T., Cunningham, P. & Bradley, D. G. Microsatellite DNA variation and the evolution, domestication and phylogeography of taurine and zebu cattle (Bos taurus and Bos indicus). Genetics 146, 1071–1086 (1997).
Achilli, A. et al. Mitochondrial genomes of extinct aurochs survive in domestic cattle. Curr. Biol. 18, R157–R158 (2008).
Bibi, F. A multi-calibrated mitochondrial phylogeny of extant Bovidae (Artiodactyla, Ruminantia) and the importance of the fossil record to systematics. BMC Evolut. Biol. 13, 166 (2013).
Gifford-Gonzalez, D. & Hanotte, O. Domesticating animals in Africa. in The Oxford Handbook of African Archaeology 491–506 (Oxford University Press, 2013).
Blench, R. & MacDonald, K. The Origins and Development of African Livestock: Archaeology, Genetics, Linguistics and Ethnography (Routledge, 2006).
Ajmone‐Marsan, P., Garcia, J. F. & Lenstra, J. A. On the origin of cattle: how aurochs became cattle and colonized the world. Evol. Anthropol. 19, 148–157 (2010).
Manning, K. The first herders of the West African Sahel: inter-site comparative analysis of zooarchaeological data from the lower Tilemsi Valley, Mali. in People and Animals in Holocene Africa. Recent Advances in Archaeozoology 75–85 (Africa Magna, 2011).
Hildebrand, E. A. & Grillo, K. M. Early herders and monumental sites in eastern Africa: dating and interpretation. Antiquity 86, 338–352 (2012).
Chritz, K. L. et al. Climate, ecology, and the spread of herding in eastern Africa. Quat. Sci. Rev. 204, 119–132 (2019).
Lesur, J., Hildebrand, E. A., Abawa, G. & Gutherz, X. The advent of herding in the Horn of Africa: new data from Ethiopia, Djibouti and Somaliland. Quat. Int. 343, 148–158 (2014).
Gifford-Gonzalez, D. & Hanotte, O. Domesticating animals in Africa: implications of genetic and archaeological findings. J. World Prehist. 24, 1–23 (2011).
Epstein, H. The Origin of the Domestic Animals of Africa (Africana Publishing Corporation, 1971).
Gifford-Gonzalez, D. Animal disease challenges to the emergence of pastoralism in sub-Saharan Africa. Afr. Archaeol. Rev. 17, 95–139 (2000).
Sadr, K. The archaeology of herding in southernmost Africa. in The Oxford Handbook of African Archaeology 645–655 (Oxford University Press, 2013).
Gifford-Gonzalez, D. ‘Animal disease challenges’ fifteen years later: the hypothesis in light of new data. Quat. Int. 436, 283–293 (2017).
Felius, M., Koolmees, P. A., Theunissen, B., European Cattle Genetic Diversity Consortium & Lenstra, J. A. On the breeds of cattle—historic and current classifications. Diversity 3, 660–692 (2011).
Freeman, A. et al. Admixture and diversity in West African cattle populations. Mol. Ecol. 13, 3477–3487 (2004).
Bradley, D. G., MacHugh, D. E., Cunningham, P. & Loftus, R. T. Mitochondrial diversity and the origins of African and European cattle. Proc. Natl Acad. Sci. USA 93, 5131–5135 (1996).
Bonfiglio, S. et al. Origin and spread of Bos taurus: new clues from mitochondrial genomes belonging to haplogroup T1. PLoS ONE 7, e38601 (2012).
Tarekegn, G. M. et al. Variations in mitochondrial cytochrome b region among Ethiopian indigenous cattle populations assert Bos taurus maternal origin and historical dynamics. Asian-Australas. J. Anim. Sci. 31, 1393 (2018).
Pérez‐Pardal, L. et al. Y‐specific microsatellites reveal an African subfamily in taurine (Bos taurus) cattle. Anim. Genet. 41, 232–241 (2010).
Mbole-Kariuki, M. N. et al. Genome-wide analysis reveals the ancient and recent admixture history of East African Shorthorn Zebu from Western Kenya. Heredity 113, 297 (2014).
Bahbahani, H. et al. Signatures of selection for environmental adaptation and zebu × taurine hybrid fitness in East African Shorthorn Zebu. Front. Genet. 8, 68 (2017).
Kim, J. et al. The genome landscape of indigenous African cattle. Genome Biol. 18, 34 (2017).
Verhoeven, K. J., Macel, M., Wolfe, L. M. & Biere, A. Population admixture, biological invasions and the balance between local adaptation and inbreeding depression. Proc. R. Soc. B Biol. Sci. 278, 2–8 (2010).
Hovick, S. M. & Whitney, K. D. Hybridisation is associated with increased fecundity and size in invasive taxa: meta‐analytic support for the hybridisation‐invasion hypothesis. Ecol. Lett. 17, 1464–1477 (2014).
Medugorac, I. et al. Whole-genome analysis of introgressive hybridization and characterization of the bovine legacy of Mongolian yaks. Nat. Genet. 49, 470 (2017).
Chen, N. et al. Whole-genome resequencing reveals world-wide ancestry and adaptive introgression events of domesticated cattle in East Asia. Nat. Commun. 9, 2337 (2018).
Wu, D.-D. et al. Pervasive introgression facilitated domestication and adaptation in the Bos species complex. Nat. Ecol. Evol. 2, 1139–1145 (2018).
Wu, C.-I. & Ting, C.-T. Genes and speciation. Nat. Rev. Genet. 5, 114 (2004).
Tijjani, A., Utsunomiya, Y. T., Ezekwe, A., Nash, O. & Hanotte, O. H. Genome sequence analysis reveals selection signatures in endangered trypano-tolerant West African Muturu cattle. Front. Genet. 10, 442 (2019).
Bahbahani, H. et al. Signatures of positive selection in African Butana and Kenana dairy zebu cattle. PLoS ONE 13, e0190446 (2018).
Rege, J., Ayalew, W., Getahun, E., Hanotte, O. & Dessie, T. DAGRIS (Domestic Animal Genetic Resources Information System) (International Livestock Research Institute, 2006).
Canavez, F. C. et al. Genome sequence and assembly of Bos indicus. J. Heredity 103, 342–348 (2012).
Browning, S. R. & Browning, B. L. Rapid and accurate haplotype phasing and missing-data inference for whole-genome association studies by use of localized haplotype clustering. Am. J. Hum. Genet. 81, 1084–1097 (2007).
Bahbahani, H., Afana, A. & Wragg, D. Genomic signatures of adaptive introgression and environmental adaptation in the Sheko cattle of southwest Ethiopia. PLoS ONE 13, e0202479 (2018).
Alexander, D. H., Novembre, J. & Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 19, 1655–1664 (2009).
Patterson, N. et al. Ancient admixture in human history. Genetics 192, 1065–1093 (2012).
Pickrell, J. K. et al. Ancient west Eurasian ancestry in southern and eastern Africa. Proc. Natl Acad. Sci. USA 111, 2632–2637 (2014).
Porter, V., Alderson, L., Hall, S. J. & Sponenberg, D. P. Mason’s World Encyclopedia of Livestock Breeds and Breeding (Cabi, 2016).
Rege, J. Zebu Cattle of Kenya: Uses, Performance, Farmer Preferences, Measures of Genetic Diversity and Options for Improved Use (ILRI 2001).
Park, S. D. et al. Genome sequencing of the extinct Eurasian wild aurochs, Bos primigenius, illuminates the phylogeography and evolution of cattle. Genome Biol. 16, 234 (2015).
Hellenthal, G. et al. A genetic atlas of human admixture history. Science 343, 747–751 (2014).
Dias-Alves, T., Mairal, J. & Blum, M. G. Loter: a software package to infer local ancestry for a wide range of species. Mol. Biol. Evol. 35, 2318–2326 (2018).
Morchikh, M. et al. HEXIM1 and NEAT1 long non-coding RNA form a multi-subunit complex that regulates DNA-mediated innate immune response. Mol. Cell 67, 387–399.e5 (2017).
Flach, H. et al. Mzb1 protein regulates calcium homeostasis, antibody secretion, and integrin activation in innate-like B cells. Immunity 33, 723–735 (2010).
Patel, S. & Jin, L. TMEM173 variants and potential importance to human biology and disease. Genes Immun. 20, 82 (2019).
Qiu, X.-B., Shao, Y.-M., Miao, S. & Wang, L. The diversity of the DnaJ/Hsp40 family, the crucial partners for Hsp70 chaperones. Cell. Mol. Life Sci. 63, 2560–2570 (2006).
Delbes, G., Yanagiya, A., Sonenberg, N. & Robaire, B. PABP interacting protein 2 (Paip2) is a major translational regulator involved in the maturation of male germ cells and male fertility. Biol. Reprod. 81, 167–167 (2009).
McReynolds, S. et al. Toward the identification of a subset of unexplained infertility: a sperm proteomic approach. Fertil. Steril. 102, 692–699 (2014).
Kuo, Y.-C. et al. SEPT12 orchestrates the formation of mammalian sperm annulus by organizing core octameric complexes with other SEPT proteins. J. Cell Sci. 128, 923–934 (2015).
Zhao, Y. et al. The NLRC4 inflammasome receptors for bacterial flagellin and type III secretion apparatus. Nature 477, 596 (2011).
Canna, S. W. et al. An activating NLRC4 inflammasome mutation causes autoinflammation with recurrent macrophage activation syndrome. Nat. Genet. 46, 1140 (2014).
Kitamura, A., Sasaki, Y., Abe, T., Kano, H. & Yasutomo, K. An inherited mutation in NLRC4 causes autoinflammation in human and mice. J. Exp. Med. 211, 2385–2396 (2014).
Wang, X. et al. The tick protein Sialostatin L2 binds to Annexin A2 and inhibits NLRC4-mediated inflammasome activation. Infect. Immun. 84, 1796–1805 (2016).
Rege, J., Aboagye, G. & Tawah, C. Shorthorn cattle of West and Central Africa. I. Origin, distribution, classification and population statistics. World Anim. Rev. 78, 2–13 (1994).
Yi, X. et al. Sequencing of 50 human exomes reveals adaptation to high altitude. Science 329, 75–78 (2010).
MacEachern, S., Hayes, B., McEwan, J. & Goddard, M. An examination of positive selection and changing effective population size in Angus and Holstein cattle populations (Bos taurus) using a high density SNP genotyping platform and the contribution of ancient polymorphism to genomic diversity in domestic cattle. BMC Genomics 10, 181 (2009).
Flori, L. et al. Adaptive admixture in the West African bovine hybrid zone: insight from the Borgou population. Mol. Ecol. 23, 3241–3257 (2014).
Newman, J. L. The Peopling of Africa: A Geographic Interpretation (Yale University Press, 1995).
Russell, J. M., Verschuren, D. & Eggermont, H. Spatial complexity of ‘Little Ice Age’ climate in East Africa: sedimentary records from two crater lake basins in western Uganda. Holocene 17, 183–193 (2007).
Phoofolo, P. Epidemics and revolutions: the rinderpest epidemic in late nineteenth-century Southern Africa. Past Present 138, 112–143 (1993).
Loh, P.-R. et al. Inferring admixture histories of human populations using linkage disequilibrium. Genetics 193, 1233–1254 (2013).
Boivin, N., Crowther, A., Prendergast, M. & Fuller, D. Q. Indian Ocean food globalisation and Africa. Afr. Archaeol. Rev. 31, 547–581 (2014).
Burrow, H. M. et al. Towards a new phenotype for tick resistance in beef and dairy cattle: a review. Anim. Prod. Sci. 59, 1401–1427 (2019).
Hansen, P. Physiological and cellular adaptations of zebu cattle to thermal stress. Anim. Reprod. Sci. 82, 349–360 (2004).
Mirkena, T. et al. Genetics of adaptation in domestic farm animals: a review. Livest. Sci. 132, 1–12 (2010).
Porto-Neto, L. R. et al. Genomic divergence of zebu and taurine cattle identified through high-density SNP genotyping. BMC Genomics 14, 876 (2013).
Bahbahani, H. et al. Signatures of positive selection in East African Shorthorn Zebu: a genome-wide single nucleotide polymorphism analysis. Sci. Rep. 5, 11729 (2015).
Kasarapu, P. et al. The Bos taurus–Bos indicus balance in fertility and milk related genes. PLoS ONE 12, e0181930 (2017).
Boone, M. & Deen, P. M. Physiology and pathophysiology of the vasopressin-regulated renal water reabsorption. Pflugers Arch. 456, 1005 (2008).
Sodhi, M. et al. Microsatellite analysis of genetic population structure of Zebu cattle (Bos indicus) breeds from North-Western region of India. Anim. Biotechnol. 22, 16–29 (2011).
Yang, Z. et al. ATG4B (Autophagin-1) phosphorylation modulates autophagy. J. Biol. Chem. 290, 26549–26561 (2015).
Ishikawa, H., Ma, Z. & Barber, G. N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461, 788–792 (2009).
Yamada, S. et al. Quantitative analysis of cytokine mRNA expression and protozoan DNA load in Theileria parva-infected cattle. J. Vet. Med. Sci. 71, 49–54 (2009).
McElroy, A. K. & Nichol, S. T. Rift Valley fever virus inhibits a pro-inflammatory response in experimentally infected human monocyte derived macrophages and a pro-inflammatory cytokine response may be associated with patient survival during natural infection. Virology 422, 6–12 (2012).
Smetko, A. et al. Trypanosomosis: potential driver of selection in African cattle. Front. Genet. 6, 137 (2015).
Murray, M., Trail, J., Davis, C. & Black, S. Genetic resistance to African trypanosomiasis. J. Infect. Dis. 149, 311–319 (1984).
Safran, M. et al. GeneCards version 3: the human gene integrator. Database 2010, 1–16 (2010).
Pomerantz, J. L., Denny, E. M. & Baltimore, D. CARD11 mediates factor‐specific activation of NF‐κB by the T cell receptor complex. EMBO J. 21, 5184–5194 (2002).
Hara, H. et al. The MAGUK family protein CARD11 is essential for lymphocyte activation. Immunity 18, 763–775 (2003).
Noyes, H. et al. Genetic and expression analysis of cattle identifies candidate genes in pathways responding to Trypanosoma congolense infection. Proc. Natl Acad. Sci. USA 108, 9304–9309 (2011).
Cecchi, G., Paone, M., Herrero, R. A., Vreysen, M. J. & Mattioli, R. C. Developing a continental atlas of the distribution and trypanosomal infection of tsetse flies (Glossina species). Parasit. Vectors 8, 284 (2015).
Lemecha et al. Response of four indigenous cattle breeds to natural tsetse and trypanosomosis challenge in the Ghibe valley of Ethiopia. Vet. Parasitol. 141, 165–176 (2006).
Naessens, J., Teale, A. & Sileghem, M. Identification of mechanisms of natural resistance to African trypanosomiasis in cattle. Vet. Immunol. Immunopathol. 87, 187–194 (2002).
Hanotte, O. et al. Mapping of quantitative trait loci controlling trypanotolerance in a cross of tolerant West African N’Dama and susceptible East African Boran cattle. Proc. Natl Acad. Sci. USA 100, 7443–7448 (2003).
Courtin, D. et al. Host genetics in African trypanosomiasis. Infect. Genet. Evol. 8, 229–238 (2008).
Ciccia, A. et al. Identification of FAAP24, a Fanconi anemia core complex protein that interacts with FANCM. Mol. Cell 25, 331–343 (2007).
Cohn, M. A. et al. A UAF1-containing multisubunit protein complex regulates the Fanconi anemia pathway. Mol. Cell 28, 786–797 (2007).
Kumar, L. et al. Leucine-rich repeat containing 8A (LRRC8A) is essential for T lymphocyte development and function. J. Exp. Med. 211, 929–942 (2014).
Ball, E. A. et al. IFNAR1 controls progression to cerebral malaria in children and CD8+ T cell brain pathology in Plasmodium berghei–infected mice. J. Immunol. 190, 5118–5127 (2013).
Makina, S. O. et al. Genome-wide scan for selection signatures in six cattle breeds in South Africa. Genet. Sel. Evol. 47, 92 (2015).
Gautier, M. et al. A whole genome Bayesian scan for adaptive genetic divergence in West African cattle. BMC Genomics 10, 550 (2009).
Kahle, D. & Wickham, H. ggmap: spatial visualization with ggplot2. R. J. 5, 144–161 (2013).
Danecek, P. et al. The variant call format and VCFtools. Bioinformatics 27, 2156–2158 (2011).
Lee, H.-J. et al. Deciphering the genetic blueprint behind Holstein milk proteins and production. Genome Biol. Evol. 6, 1366–1374 (2014).
Shin, D.-H. et al. Deleted copy number variation of Hanwoo and Holstein using next generation sequencing at the population level. BMC Genomics 15, 240 (2014).
Heaton, M. P. et al. Using diverse US beef cattle genomes to identify missense mutations in EPAS1, a gene associated with pulmonary hypertension. F1000Res. 5, 2003 (2016).
Taylor, J. F. et al. Lessons for livestock genomics from genome and transcriptome sequencing in cattle and other mammals. Genet. Sel. Evol. 48, 59 (2016).
Andrews, S. FastQC: a quality control tool for high throughput sequence data. Babraham Bioinformatics http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (2010).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
Cingolani, P. et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly 6, 80–92 (2012).
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Evanno, G., Regnaut, S. & Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol. Ecol. 14, 2611–2620 (2005).
Weir, B. S. & Cockerham, C. C. Estimating F‐statistics for the analysis of population structure. Evolution 38, 1358–1370 (1984).
Nguyen, L.-T., Schmidt, H. A., Von Haeseler, A. & Minh, B. Q. IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274 (2015).
Kalyaanamoorthy, S., Minh, B. Q., Wong, T. K., von Haeseler, A. & Jermiin, L. S. ModelFinder: fast model selection for accurate phylogenetic estimates. Nat. Methods 14, 587 (2017).
Felsenstein, J. Evolutionary trees from DNA sequences: a maximum likelihood approach. J. Mol. Evol. 17, 368–376 (1981).
Kousathanas, A. et al. Inferring heterozygosity from ancient and low coverage genomes. Genetics 205, 317–332 (2017).
Ma, L. et al. Cattle sex-specific recombination and genetic control from a large pedigree analysis. PLoS Genet. 11, e1005387 (2015).
Lawson, D. J., Hellenthal, G., Myers, S. & Falush, D. Inference of population structure using dense haplotype data. PLoS Genet. 8, e1002453 (2012).
Voight, B. F., Kudaravalli, S., Wen, X. & Pritchard, J. K. A map of recent positive selection in the human genome. PLoS Biol. 4, e72 (2006).
Maclean, C. A., Chue Hong, N. P. & Prendergast, J. G. hapbin: an efficient program for performing haplotype-based scans for positive selection in large genomic datasets. Mol. Biol. Evol. 32, 3027–3029 (2015).
Utsunomiya, Y. et al. Genomic clues of the evolutionary history of Bos indicus cattle. Anim. Genet. 50, 557–568 (2019).
Koufariotis, L. et al. Sequencing the mosaic genome of Brahman cattle identifies historic and recent introgression including polled. Sci. Rep. 8, 17761 (2018).
O’brien, A. M. P. et al. Low levels of taurine introgression in the current Brazilian Nelore and Gir indicine cattle populations. Genet. Sel. Evol. 47, 31 (2015).
Zerbino, D. R. et al. Ensembl 2018. Nucleic Acids Res. 46, D754–D761 (2017).
Mi, H., Muruganujan, A., Ebert, D., Huang, X. & Thomas, P. D. PANTHER version 14: more genomes, a new PANTHER GO-slim and improvements in enrichment analysis tools. Nucleic Acids Res. 47, D419–D426 (2019).
Croft, D. et al. The Reactome pathway knowledgebase. Nucleic Acids Res. 42, D472–D477 (2014).
Acknowledgements
This work was supported by a grant from the Next-Generation BioGreen 21 Program and Post-Genome Project (Project Nos. PJ01323701 and PJ01040601), Rural Development Administration, Republic of Korea. Sampling of cattle populations was supported by the CGIAR Livestock and Fish CRP (Uganda and Ethiopia), the University of Khartoum (Sudan) and the National Biotechnology Development Agency (NABDA) (Nigeria). The following institutions and their personnel provided help for the sampling of the African cattle: the ILRI Kapiti Ranch; the Ministry of Animal Resources, Fisheries and Range (Sudan); the Ol Pejeta Conservancy (Kenya); the Institute of Biodiversity (Ethiopia); and the Directors of Veterinary Services and the cattle keepers from Ethiopia, Kenya, Uganda and Sudan. The ILRI livestock genomics program is supported by the CGIAR Research Program on Livestock (CRP Livestock), which is supported by contributors to the CGIAR Trust Fund (http://www.cgiar.org/about-us/our-funders/). This research was funded in part by the Bill & Melinda Gates Foundation and with UK aid from the UK Foreign, Commonwealth and Development Office (Grant Agreement OPP1127286) under the auspices of the Centre for Tropical Livestock Genetics and Health (CTLGH), established jointly by the University of Edinburgh, SRUC (Scotland’s Rural College) and the International Livestock Research Institute. The findings and conclusions contained within are those of the authors and do not necessarily reflect positions or policies of the Bill & Melinda Gates Foundation or the UK Government. We thank the reviewers for their critical and constructive comments on the manuscript, and D. Gifford-Gonzalez (University of California, Santa Cruz, CA, USA) for a critical reading of the manuscript in light of the current knowledge on the archeology and history of African pastoralism.
Author information
Authors and Affiliations
Contributions
K.K. and O.H. devised the main conceptual ideas. O.H. and H.K. managed the project. D.L., S.C., S.J.O., H.-K.L., O.A.M., T.D., S.K., O.H. and H.K. conceived of and designed all of the described experiments. O.A.M., T.D., B.S., G.M.T. and A.T. contributed to sample collection and laboratory work. K.K., T.K., D.Y., J. Jang, S.S., S.L., J. Jung and H.J. analyzed the data. K.K., C.J., J.K. and O.H. drafted the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Improvement in genotype concordance after genotype refinement using BEAGLE as a function of depth coverage.
The y-axis shows the concordance between genotypes called from sequencing data compared to genotypes obtained using the BovineSNP50 Genotyping BeadChip.
Extended Data Fig. 2 Delta K of cluster number K in genetic clustering analysis using ADMIXTURE.
A subset of ~1.6 million SNPs (linkage disequilibrium (LD)-based pruning using PLINK v1.9 with ‘-indep-pairwise 50 10 0.1’ option) was used for K from 1 to 10. The delta K analysis suggests K = 2 as the most likely number of distinct genetic ancestries among the 10 Ks (delta K = 31.02).
Extended Data Fig. 3 Mean pairwise Fst values between cattle breeds represented by more than one animal.
Sheko is indicated as yellow.
Extended Data Fig. 4 Estimated heterozygosity of cattle breeds.
The lower and upper bounds of box correspond to the first and third quartiles (the 25th and 75th percentiles), respectively. The horizontal line in the box represents the median value. The upper and lower whisker extend from the bounds to the largest and lowest value no further than 1.5 * interquartile range (IQR), respectively. The number of biologically independent animals used in this analysis for each breed is as follows: Achai (2), Afar (9), Angus (10), Ankole (10), Arsi (10), Barka (9), Bhagnari (3), Boskarin (1), Brahman (20), Butana (20), Cholistani (2), Dajal (1), Dhanni (2), Eastern Finn (5), Ethiopian Boran (10), Fogera (9), Gabrali (2), Gir (4), Goffa (10), Hanwoo (23), Hariana (3), Hereford (18), Holstein (10), Horro (11), Jersey (10), Kenya Boran (10), Kenana (13), Limia (1), Lohani (1), Maremmana (3), Maronesa (1), Mursi (10), Muturu (10), N’Dama (13), Nelore (10), Ogaden (9), Pajuna (2), Poldolica (1), Red Sindhi (1), Sahiwal (2), Sayaguesa (2), Sheko (9), Simmental (11), Tharparkar(3) and Wetern Finn (5). Sheko is indicated as yellow.
Extended Data Fig. 5 Runs of homozygosity patterns of cattle breeds.
Sheko is indicated as yellow.
Extended Data Fig. 6 Weighted LD decay in the Kenya Boran breed before and after fitted with a double-pulse admixture model.
The red curve shows the exponential fit to the data. a, Weighted LD fitted by a single-pulse admixture model, when using EAT and Muturu as a reference population separately. b, Weighted LD fitted by a double-pulse admixture model, when using EAT and Muturu as a reference population separately.
Extended Data Fig. 7 Distribution of proportions of SNPs with |iHS | ≥ 2 and taurine ancestry in each 50-kb window.
a, Distribution of proportions of SNPs with |iHS | ≥ 2. b, Distribution of taurine ancestry. The windows with SNPs less than 10 were removed. Dashed lines indicate the highest 1% for a, and highest or lowest 0.5% in b.
Extended Data Fig. 8 Distribution of taurine ancestry in the candidate regions (the highest 1% for proportion of SNPs with |iHS | ≥ 2), and whole genome windows.
Dashed lines indicate mean (top 1% in iHS analysis: 26.14%, and whole genome: 32.49%).
Extended Data Fig. 9 Distribution of taurine ancestry according to quantiles of proportions of SNPs with |iHS | ≥ 2 in each 50-kb window.
The lower and upper bounds of box correspond to the first and third quartiles (the 25th and 75th percentiles), respectively. The horizontal line in the box represents the median value. The upper and lower whisker extend from the bounds to the largest and lowest value no further than 1.5 * interquartile range (IQR), respectively. Asterisk indicates the highest 1% with proportions of SNPs with |iHS | ≥ 2. n = 149 (African humped cattle) biologically independent animals were used in this analysis.
Extended Data Fig. 10 Distribution of average taurine ancestry generated by resampling random windows (same number of windows as the candidate) for 0.1 million times.
Asterisk indicates average taurine ancestry of the candidate windows from iHS analysis.
Supplementary information
Supplementary Information
Supplementary Note, Tables 1–13 and Figs. 1–3
Rights and permissions
About this article
Cite this article
Kim, K., Kwon, T., Dessie, T. et al. The mosaic genome of indigenous African cattle as a unique genetic resource for African pastoralism. Nat Genet 52, 1099–1110 (2020). https://doi.org/10.1038/s41588-020-0694-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41588-020-0694-2
- Springer Nature America, Inc.
This article is cited by
-
Whole-genome resequencing provides insights into the diversity and adaptation to desert environment in Xinjiang Mongolian cattle
BMC Genomics (2024)
-
Multimodal interactions in Stomoxys navigation reveal synergy between olfaction and vision
Scientific Reports (2024)
-
Exploring the genetic factors behind the discrepancy in resistance to bovine tuberculosis between African zebu cattle and European taurine cattle
Scientific Reports (2024)
-
Genomic Reference Resource for African Cattle: Genome Sequences and High-Density Array Variants
Scientific Data (2024)
-
Whole genome sequencing of simmental cattle for SNP and CNV discovery
BMC Genomics (2023)