Abstract
Most sensory information destined for the neocortex is relayed through the thalamus, where considerable transformation occurs1,2. One means of transformation involves interactions between excitatory thalamocortical neurons that carry data to the cortex and inhibitory neurons of the thalamic reticular nucleus (TRN) that regulate the flow of those data3,4,5,6. Although the importance of the TRN has long been recognised7,8,9, understanding of its cell types, their organization and their functional properties has lagged behind that of the thalamocortical systems they control. Here we address this by investigating the somatosensory and visual circuits of the TRN in mice. In the somatosensory TRN we observed two groups of genetically defined neurons that are topographically segregated and physiologically distinct, and that connect reciprocally with independent thalamocortical nuclei through dynamically divergent synapses. Calbindin-expressing cells—located in the central core—connect with the ventral posterior nucleus, the primary somatosensory thalamocortical relay. By contrast, somatostatin-expressing cells—which reside along the surrounding edges of the TRN—synapse with the posterior medial thalamic nucleus, a higher-order structure that carries both top-down and bottom-up information10,11,12. The two TRN cell groups process their inputs in pathway-specific ways. Synapses from the ventral posterior nucleus to central TRN cells transmit rapid excitatory currents that depress deeply during repetitive activity, driving phasic spike output. Synapses from the posterior medial thalamic nucleus to edge TRN cells evoke slower, less depressing excitatory currents that drive more persistent spiking. Differences in the intrinsic physiology of TRN cell types, including state-dependent bursting, contribute to these output dynamics. The processing specializations of these two somatosensory TRN subcircuits therefore appear to be tuned to the signals they carry—a primary central subcircuit tuned to discrete sensory events, and a higher-order edge subcircuit tuned to temporally distributed signals integrated from multiple sources. The structure and function of visual TRN subcircuits closely resemble those of the somatosensory TRN. These results provide insights into how subnetworks of TRN neurons may differentially process distinct classes of thalamic information.
Similar content being viewed by others
Main
We first investigated the cellular composition of the TRN, characterizing the expression of parvalbumin (PV), calbindin (CB) and somatostatin (SOM)—three markers that are useful in differentiating functionally distinct neural types in the neocortex and elsewhere13. Brain sections spanning the somatosensory sector of the TRN4,7,8 were prepared from SOM-Cre mice crossed with Cre-dependent tdTomato (tdT) reporters, then stained immunohistochemically for CB and PV (Fig. 1a).
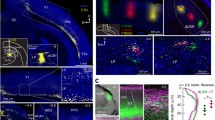
a, Confocal images of a 40-μm section, centred on the somatosensory TRN, from a SOM-Cre × tdTomato mouse. Immunohistochemical (IHC) staining was performed for PV and CB. SOM-Cre cells were genetically labelled (SOM-Cre × tdT). The outlines of the TRN are based on PV labelling. Scale bar, 100 μm. b, Confocal images of an 18-μm section from a wild-type mouse, showing fluorescence in situ hybridization experiments for the visualization of Pvalb, Sst and Calb1 mRNA (encoding PV, SOM and CB). Scale bar, 100 μm. c, Left, proportion of cells expressing PV, SOM–tdT or CB in the somatosensory TRN (outlined by the bracket in the leftmost image of a). Middle, proportions of cells expressing combinations of markers (n = 1,075 cells, 3 sections, 3 mice). Right, proportions of each marker in three zones of the somatosensory TRN. A total of 187 cells were in the medial zone (medial 20%), 746 in the central zone (central 60%) and 142 were in the lateral zone (lateral 20%). The leftmost images of a and b show the zones marked by dashed lines. Fractional SOM– tdT cell densities were higher in the edge zones than in the central zone, whereas CB cell densities were higher in the central zone (all P < 0.001, χ2, Yates’ correction). d, Left and middle, as for c but for Pvalb, Sst and Calb1 mRNA (n = 593 cells, 6 sections, 6 mice). Right, 98 cells were in the medial zone, 412 in the central zone and 83 in the lateral zone. Again, Sst-expressing and Calb1-expressing cells were differentially distributed across zones (all P < 0.001, χ2, Yates’ correction). e, Confocal image from a P23 SOM-Cre mouse. AAV9-DIO–GFP was injected into the TRN at P14 to assess SOM-Cre expression. GFP cells (pseudocoloured cyan) were largely absent in the central zone (replicated in 12 mice). Data are mean ± s.e.m. Scale bar, 200 μm.
Two neuron types in the somatosensory TRN
Nearly all somatosensory TRN cells expressed PV1,14, whereas only subsets expressed SOM–tdT (around 64%) or CB (around 48%) (Fig. 1c, Extended Data Fig. 1). The distributions of SOM–tdT cells and CB cells across the somatosensory TRN were complementary. The highest densities of SOM–tdT cells were near the medial and lateral edges of the sector27, whereas CB cells were concentrated near the centre and were almost absent along the edges (Fig. 1a, Extended Data Fig. 1). Quantitative comparisons between the medial 20%, lateral 20% and central 60% of the somatosensory TRN confirmed that the proportions of cells expressing SOM–tdT were higher in the edge zones than in the central zone (P < 0.001, χ2 test), whereas the reverse was true for CB (P < 0.001) (Fig. 1c).
To further investigate the organization of neurons across the TRN, we used in situ hybridization to assay expression of mRNA encoding SOM (Sst), CB (Calb1) and PV (Pvalb). The highest densities of SOM cells were again found in the medial and lateral edge zones, CB cells were clustered centrally, and nearly all cells were PV-positive (Fig. 1b, d, Extended Data Fig. 2). Notably, the segregation between edge SOM cells and central CB cells was more salient in the mRNA assay, mainly due to decreased proportions of SOM cells in the central zone (only 15.3% of central cells expressed Sst mRNA, whereas 54.7% expressed SOM–tdT; Fig. 1a–d).
The near absence of SOM-encoding mRNA in the central zone suggests that most neurons located there may not actually express SOM protein in mature mice, and that expression of tdT in the central cells of SOM-Cre × tdT mice could result from genetic recombination early in development and persistent tdT production thereafter15. To test for SOM expression in mature mice we initially attempted immunohistochemistry; however, we were unable to find SOM antibodies that were adequate for the TRN (not shown). As an alternative, we assayed Cre expression in mature SOM-Cre mice by injecting an adeno-associated virus driving Cre-dependent GFP into the TRN. Cre expression in the somatosensory TRN of these mice was almost entirely restricted to the edge zones27, consistent with the pattern of Sst mRNA expression (Fig. 1b, d, e, Extended Data Fig. 3). Together, these results indicate that the somatosensory TRN is composed of neurochemically distinct cell types segregated into separate zones: a core central zone composed mostly of CB-expressing neurons, flanked by edge zones of SOM-expressing neurons.
Primary and higher-order TRN subcircuits
The primary ventral posterior (VP) and higher-order posterior medial (POM) thalamocortical nuclei transmit distinct information to different targets in the neocortex and send collaterals to the TRN; the latter leads to both open-loop and closed-loop thalamic inhibition4,5,6,8. Clarifying the organization of these circuits, including how primary and higher-order thalamocortical nuclei synapse with subtypes of TRN neurons16, is essential to understanding thalamic information processing7. To this end, we selectively expressed ChR2–eYFP in the VP or the POM, then characterized their inputs to the TRN (Fig. 2). Notably, their projections were found to segregate topographically, in close alignment with the observed patterns of TRN cell types. VP axons terminated in the CB-rich central zone of the somatosensory TRN, whereas POM axons terminated along the SOM-dense medial and lateral edges (Fig. 2a, b, Extended Data Fig. 4a, c).
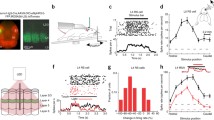
a, Representative confocal images illustrating the distinct projections from the POM and the VP to the TRN. AAV2-ChR2–eYFP was injected into the POM (left) or the VP (right) and projections to the TRN were characterized. Scale bars: 500 μm (main images); 100 μm (expanded images). b, Group data showing average thalamic projections to the TRN. Viral anterograde tracing as in a. The POM targeted the edges of the somatosensory TRN (n = 9 mice), and the VP targeted the central zone (n = 8 mice). Boundaries between the central and edge zones are drawn at 20% and 80% of the medial–lateral distance across the TRN. Right, average SOM–tdT and CB immunohistochemical profiles (n = 5 mice) aligned to the same reference TRN as the anterograde projection maps. Scale bar, 200 μm. c, Synaptic responses to POM input from cells across the TRN, testing how synaptic strength relates to anterograde fluorescence and topographical location. Left, image of live TRN (outlined) with ChR2–eYFP projections from the POM. Circles show the locations of recorded cells. Right, EPSCs evoked in TRN cells by optical activation of POM axons (−84 mV). The colours match the cells in the left image and the colours in d. Scale bar, 100 μm. d, Normalized fluorescence (black line) and synaptic charge (coloured dots) for cells in c, as a function of their medial–lateral position in the TRN. e, Group relationship between the location of the TRN soma and the evoked synaptic response. Somas close to the TRN edges responded strongly to input from the POM and those in the centre did not (for e, f, n = 21 cells, 5 slices, 5 mice; each preparation has a unique symbol). f, Synaptic responses of TRN cells to POM input (normalized charge) correlated with fluorescence from POM axons surrounding the cell (r = 0.75, P < 0.0001, two-tailed Pearson’s correlation). g, h, Same as c, d, except activating VP input. Scale bar, 100 μm. i, Group data showing that somas near the TRN centre respond more strongly to VP input than those near the edges (for i, j, n = 31 cells, 6 slices, 4 mice). j, Synaptic responses of TRN cells to VP input correlated with fluorescence from VP axons surrounding the cell (r = 0.68, P < 0.0001, two-tailed Pearson’s correlation).
Given this stark anatomical segregation of projections, it seemed likely that central and edge TRN cells would be selectively targeted by synapses from the VP and the POM, respectively. However, the dendrites of TRN neurons might extend into adjacent zones, leading to functional crosstalk among the circuits1,8,17. To address this, we mapped excitatory synaptic strengths of POM and VP inputs to cells located across the mediolateral axis of the somatosensory TRN. Consistent with the anatomy, synaptic responses to POM inputs were much stronger for TRN edge cells than for central cells, whereas VP inputs evoked the strongest responses in central cells (Fig. 2c–e, g–i, Extended Data Fig. 4b, d). Moreover, synaptic strengths for TRN cells correlated with fluorescence intensities of the afferent terminals near their soma (Fig. 2d, f, h, j). Together, our findings show that primary and higher-order somatosensory thalamic inputs to the TRN are topographically segregated and align with the neurochemical pattern of TRN cell types: the VP projects strongly to CB-expressing central cells, and the POM to SOM-expressing edge cells.
TRN subcircuits are functionally distinct
Primary and higher-order thalamocortical nuclei convey qualitatively different types of information10,12,18,19,20,21, and we asked whether VP and POM communication with the TRN might involve parallel differences in synaptic mechanisms. Thus, we compared dynamic features of the glutamatergic synaptic currents (kinetics and short-term synaptic depression) evoked by photostimulating the VP and POM inputs to the TRN. VP synaptic currents in central TRN cells were brief and depressed deeply during repetitive activation. Conversely, POM currents in TRN edge cells were longer-lasting and more stable, depressing significantly less (Fig. 3a–c, Extended Data Figs. 5, 6).
a, Representative locations of TRN cells targeted by the VP (black dot) and by the POM (red dots). b, POM-evoked EPSCs of edge cells are more prolonged than VP-evoked EPSCs of central cells. Left, normalized optogenetically evoked example EPSCs for each cell type (stimulus 1 of c; membrane potential −84 mV). Right, POM-evoked edge cell responses have greater fractional area in later stages of the EPSCs (P < 0.0001, unpaired two-tailed t-test; central: 48 cells, 14 mice; edge: 21 cells, 14 mice). c, Short-term synaptic depression. Left, representative EPSCs from a central cell (VP inputs) and an edge cell (POM inputs), normalized to response peaks (around 2 nA) (10 Hz LED trains). Right, group comparison: short-term depression was greater for central cells than for edge cells (P < 0.0001, ANOVA stimulus 2–10; all P < 0.005 from stimulus 5–10, two-tailed Bonferroni’s t-test; same cells as b). d, Left, intrinsic bursting of example central and edge cells. Offset bursts were triggered by injecting negative current to reach approximately −95 mV, followed by abrupt current removal. Right, number of spikes per burst as a function of position in the TRN (open circles: individual cells; filled circles: zone averages; 34 central cells, 11 mice; 22 medial cells, 10 mice; 16 lateral cells, 10 mice). Edge cells (red) discharged fewer spikes per burst than central cells (black) (unpaired two-tailed t-test, P < 0.0001). Data are mean ± s.e.m.
To understand how these dynamically distinct inputs to TRN neurons might be integrated postsynaptically, we assessed the intrinsic physiological characteristics of these neurons. Central and edge neurons differed in terms of a range of passive and active membrane properties17. Edge cells had higher resistances, lower capacitances and smaller somata than central cells. Action potential kinetics, afterpotentials and threshold currents also differed between the two neuron types (Extended Data Fig. 7, Supplementary Information 1). One notable intrinsic distinction between the TRN cell types, which could powerfully influence responses to synaptic input during certain behavioural states1,2,5,6,22,23,24,25,26, was the much greater tendency of central cells to fire spikes in high-frequency bursts (Fig. 3d). The bursting differences were consistent with stronger T-type (low-threshold) calcium currents in central cells17,27,28,29,30. Thus, nearly all central cells fired ‘offset bursts’ after release from hyperpolarizing stimuli (around 10 spikes per burst). By contrast, under matched stimulus conditions, most edge cells either failed to burst or fired weak bursts (around 2 spikes per burst; Fig. 3d, Supplementary Information 1). Neither cell type exhibited bursting when excited from a more depolarized steady-state (around −74 mV; Extended Data Fig. 7).
We next examined how the observed pathway-specific synaptic and intrinsic properties combine to control the spiking responses of TRN cells to their excitatory thalamic inputs. The brief and depressing synaptic inputs from the VP to central TRN cells, together with the propensity of central cells to burst, suggest that they might respond phasically—initially strong but quickly decreasing with repeated activation. By contrast, the kinetically slow and more stable inputs from the POM to the less bursty edge cells predict initially weaker—but more sustained—spiking.
First we generated simulated synaptic currents that matched the excitatory postsynaptic currents (EPSCs) previously recorded from central and edge TRN cells in response to their respective inputs. We then characterized TRN spike responses elicited by intracellular injection of these currents, delivered while the TRN cells were at their resting potentials (around −84 mV). As predicted, central cells responded to simulated VP inputs with initial bursts that sharply depressed. By contrast, the spiking responses of edge cells to simulated POM inputs were initially much weaker but persisted more during repetitive activation (Fig. 4a, b).
a, Simulated synaptic currents, modelled as average recorded EPSCs evoked by activation of VP or POM axons (as in Fig. 3, Methods), were applied to central and edge TRN cells. Left, response of a central cell (membrane potential, Vm) to simulated VP synaptic currents (Iinj). The first stimulus evoked a 12-spike burst, but responses depressed to 0 spikes by stimulus 8. Right, response of an edge cell to simulated POM synaptic currents. The first stimulus evoked 3 spikes, and spiking persisted during repetitive stimulation. b, Mean spike outputs of central cells to simulated VP currents, and edge cells to simulated POM currents (that is, to their native synaptic inputs). TRN cells were held in burst mode (−84 mV). Central cell responses were initially strong and depressed sharply, whereas edge responses were more stable (spike counts from stimulus 1 to stimulus 10 declined by 97% for central cells and 63% for edge cells; P < 0.0001, unpaired two-tailed t-test). For b–d: 13 central cells, 11 edge cells, 8 mice. c, Same as b except cells were held at −74 mV to reduce intrinsic bursting. Central responses became more edge-like, yet significant differences remained (spike counts from stimulus 1 to stimulus 10 declined by 77% for central and 33% for edge cells; P < 0.0003, unpaired two-tailed t-test). d, Spike responses of central cells and edge cells (−74 mV) to identical simulated synaptic currents (average of VP and POM EPSCs). Central and edge cell responses did not differ significantly (P = 0.19, unpaired two-tailed t-test). Data are mean ± s.e.m.
We then asked whether differences in intrinsic bursting (Fig. 3d) contribute to these marked differences in synaptically evoked spiking. To address this, we depolarized the TRN cells to −74 mV to partially inactivate T-type calcium channels and reduce intrinsic bursting, and then recharacterized their responses. The responses of central cells became far less phasic and were more persistent, whereas edge cells were hardly affected. Initial spiking was reduced by 41% in central cells but by only 23% in edge cells, and responses to later stimuli in the trains were enhanced more for central cells (Fig. 4c). These results indicate that intrinsic bursting in central TRN cells has a powerful role in their responses to excitatory inputs when that input arrives during relatively hyperpolarized states (for example, during sleep or periods of strong inhibition). The far smaller effects of polarization on edge cells indicates less influence of T-type calcium bursting and, importantly, weaker modulation by the types of membrane potential shifts that are thought to occur during behavioural state transitions1,6,22,23,24,25,26,31.
Finally, we considered whether the dynamic features of VP synaptic inputs to central cells (which are faster and more depressing than those to edge cells; Fig. 3a–c) also contributed to their phasic spike outputs. For this we generated simulated synaptic currents that were the average, in terms of EPSC kinetics and short-term depression, of the VP → central cell and POM → edge cell synaptic currents. We then tested the effects on evoked spiking, with steady-state potentials set to −74 mV to minimize bursting. Notably, responses of the two cell types were almost identical when triggered by the averaged synaptic input; central responses became more sustained and edge responses slightly more phasic (Fig. 4c, d). Together, these results indicate that central and edge cells differentially transform their native excitatory thalamic inputs into distinct spiking outputs through differences both in the dynamics of their synaptic inputs and in their intrinsic burstiness.
TRN inhibitory outputs are subcircuit-specific
To better understand the consequences of the distinct output from the TRN subcircuits, we examined their projections and the inhibitory feedback they produced. We found that the two TRN cell types predominantly inhibited the thalamocortical nuclei that drive them. That is, CB-expressing TRN cells projected to and inhibited neurons of the ventral posterior medial nucleus (VPM), whereas SOM-expressing edge cells bypassed the VPM and instead inhibited the POM (Extended Data Fig. 8). Thus, the primary and higher-order segregation of somatosensory reticulo-thalamic subcircuits seems to be largely reciprocal8,16,32.
Visual and somatosensory TRN have similar subcircuits
To test whether other sensory systems might share the salient structural and functional organization that we have described for the somatosensory TRN, we examined the visual TRN and its associated thalamocortical nuclei—the primary dorsal lateral geniculate and the higher-order lateral posterior nucleus (pulvinar). We found that the organization of the visual TRN, in terms of its synaptic input patterns and the intrinsic physiological properties of its neurons, was very similar to that of the somatosensory system (Extended Data Fig. 9). This suggests that a primary central core, flanked by higher-order edge neurons, might be a widespread TRN motif.
Discussion
Our results imply that sensory regions of the TRN have two discrete subcircuits that are distinguished by their structure and function (Extended Data Fig. 10). Structurally, two types of TRN neurons are segregated into central and edge zones and receive inputs from different thalamocortical nuclei. Functionally, the subcircuits have distinct dynamics, determined by the intrinsic physiology of their respective neurons and the properties of their excitatory thalamic synapses. The subcircuits seem to be tuned to temporal characteristics of the signals they process—transient sensory signals in the primary systems and more temporally distributed signals in the higher-order systems10,11,18,21.
A longstanding hypothesis suggests that the TRN serves as a gatekeeper of information flow, permitting distinct thalamocortical circuits to regulate one another and enabling functions such as selective attention1,5,9,33. It has been proposed that inhibitory crosstalk between thalamic circuits32,34 may underlie such regulation8,35,36. However, the sharp and reciprocal segregation of the subcircuits we observed here suggests that intrathalamic crosstalk might have a minor role. Instead, cross-system regulation in the thalamus could be mediated by other means—including descending cortical control of reticulo-thalamic subcircuits2,37,38,39, the nature of which is only beginning to emerge40.
The distinct kinetics of the parallel TRN subsystems suggests that arriving signals will be filtered through circuit-specific temporal tuning mechanisms. For example, it is generally thought that TRN neurons undergo shifts from bursting to tonic spiking mode as animals transition between behavioural states of quiescence and aroused wakefulness1,22,24,26, owing to activity-dependent changes in membrane potentials and neuromodulatory tone2,6,23,25,28,41. Our results suggest that such state transitions alter the spike mode in primary (central) TRN cells much more strongly than in higher-order (edge) cells27,31. This—in addition to the other kinetic differences between subcircuits that we describe here—are likely to have profound implications for the thalamic processing of afferent signals5, both ascending and descending38.
Previous studies of several species and sensory systems have suggested separate TRN laminae for primary and higher-order connections1,7,8, generally with just a single higher-order layer16,32,42,43. By contrast, in the somatosensory TRN of the mouse, we observed a primary core of CB cells surrounded by a higher-order shell-like zone of SOM cells. In at least one respect, this organization is reminiscent of the visual TRN of the galago, a primate; both systems have SOM-expressing zones that receive inputs from higher-order thalamus, and non-SOM zones receiving primary inputs43. The association between SOM neurons of the TRN and higher-order processing may therefore be a conserved feature of these circuits44. This finding, together with the association between CB expression and first-order processing, are of particular interest because they enable powerful new strategies for investigating the behavioural and perceptual functions of these distinct TRN circuits45.
Methods
Data reporting
No statistical methods were used to predetermine sample size. The experiments were not randomized and the investigators were not blinded to allocation during experiments and outcome assessment.
Mice
All procedures were approved by, and complied with all ethical regulations of, the Brown University Institutional Animal Care and Use Committee. The following mouse lines were used: SOM-IRES-Cre (The Jackson Laboratory, 013044), PV-Cre (The Jackson Laboratory, 008069), Calb1-IRES-Cre-D (‘CB-Cre’; The Jackson Laboratory, 028532), Vglut2-IRES-Cre (The Jackson Laboratory, 016963), GPR26-Cre (STOCK Tg(Gpr26-cre)KO250Gsat/Mmucd, MMRRC), Ai14 (The Jackson Laboratory, 007908), ICR (Charles River, CD-1[ICR], Strain Code 022). To fluorescently target SOM or PV cells in TRN, we bred the respective homozygous Cre mice (SOM-IRES-Cre, PV-Cre) with homozygous Cre-dependent tdTomato reporter mice (Ai14). In some experiments we instead injected viruses carrying Cre-dependent GFP genes into Cre mice (specified in the descriptions of the individual experiments, below). Of the 124 mice used in this study, 91 had C57 genetic backgrounds, 10 had ICR genetic backgrounds, and 23 had mixed C57/ICR backgrounds. Mice were maintained on a 12 h:12 h light/dark cycle, group-housed, and provided food and water ad libitum.
Immunohistochemistry
Mice from postnatal day (P)22–26 were deeply anaesthetized with Beuthanasia-D and intracardially perfused with 0.1 M phosphate buffer (PB) followed by 4% paraformaldehyde (PFA; in PB). Brains were post-fixed overnight at 4 °C in the same fixative and then transferred to a 30% sucrose/0.1 M PB solution until sectioning (4 °C, 2–3 days). Brain sections (40-μm thick) were cut on a freezing microtome at a somatosensory thalamocortical plane (35° tilt from coronal)46 designed to contain the somatosensory thalamus, TRN, barrel cortex and many of their interconnections. Next the sections were immunostained. In brief, they were washed 5 times in 0.1 M phosphate buffer containing 0.15 M NaCl, pH 7.4 (PBS) (5 min per wash), pre-incubated for 2 h at room temperature with a blocking solution (10% normal goat serum, 2% Triton X-100, 0.1% Tween 20 in 0.1 M PB), then incubated with primary antibodies for 5 days at 4 °C. After the primary incubation, sections were washed 8 times in PBS (5 min per wash), pre-incubated for 2 h in blocking solution, incubated with a secondary antibody solution for 3 days at 4 °C, then washed 8 times in PBS and 3 times in PB (5 min per wash). Sections were mounted and coverslipped using Prolong Gold or Prolong Gold with DAPI (Molecular Probes P36930 or P36931). Primary antibodies were: mouse monoclonal anti-parvalbumin (1:1,000, Swant clone 235, Lot 10-11F), rabbit polyclonal anti-calbindin D-28k (1:1,000, Swant clone CB-38a, Lot 9.03), mouse monoclonal anti-calbindin D-28k (1:1,000, Swant clone 300, Lot 07(F)), mouse monoclonal anti-NeuN (1:1,000, Millipore MAB377, Clone A60, Lot 2549411). Secondary antibodies were goat anti-mouse IgG (H+L) cross-adsorbed secondary antibody Pacific Blue (1:250, Molecular Probes P31582), goat anti-rabbit IgG (H+L) cross-adsorbed secondary antibody Alexa Fluor 647 (1:500, Molecular Probes A21244), goat anti-mouse IgG (H+L) cross-adsorbed secondary antibody Alexa Fluor 488 (1:250, Molecular Probes A11001), goat anti-rabbit IgG (H+L) highly cross-adsorbed secondary antibody Alexa Fluor 488 (1:250, Molecular Probes A11034). To test the specificity of the antibodies, no-primary and no-secondary controls were conducted for each. In addition, for the calbindin antibodies, we performed assays in which the antibodies were pre-adsorbed with calbindin protein (3.33 μg/1 μl of antibody, Swant recombinant rat Calbindin D-28) before tissue incubation. There was no clear labelling under any of the control conditions, providing support for the effectiveness and specificity of the antibodies. Confocal image stacks were taken on a Zeiss LSM 800, 20× objective, 0.26 μm pixel diameter.
For analysis of the immunohistochemical material, we selected sections spanning the somatosensory sector of TRN (approximately −1.7 mm to −1.1 mm from bregma), generally focusing on the anterior–posterior centre of that range (that is, −1.4 mm from bregma). The CellCounter ImageJ plugin was used to count cells positive for the tested markers. For accurate alignment across subjects and sections, small differences in TRN shapes were minimized by warping them to an average reference TRN image using the bUnwarpJ plugin in ImageJ. At least 12 landmark points were applied around the outer boundaries of each TRN (evenly spaced, 100 μm between points) for alignment. The TRN boundaries were identified using either the SOM-Cre × tdTomato or PV-Cre × tdTomato channel. Resulting corrections were applied to all channels.
Fluorescence in situ hybridization
Mice (P21–P27) were deeply anaesthetized with isofluorane and decapitated. The brains were removed while submerged in 4 °C saline solution and fresh-frozen on liquid nitrogen. Frozen brains were sectioned (18-μm thick) at the somatosensory thalamocortical plane (described in the above section ‘Immunohistochemistry’) using a cryostat (Leica), adhered to SuperFrost Plus slides (VWR, 48311-703), and refrozen (−80 °C) until used. Samples were fixed (4% PFA), processed as instructed in the ACD RNAScope Multiplex Fluorescent v2 Assay, and then immediately incubated with DAPI and coverslipped with ProLong Gold (Molecular Probes P36930). Probes for PV (Mm-Pvalb-C3, 421931-C3), SOM (Mm-Sst, 404631), CB (Mm-Calb1-C2, 428431-C2), and tdTomato (tdTomato-C3, 317041-C3) were purchased from Advanced Cell Diagnostics. Probes were visualized using the TSA Plus Fluorescein (1:1,000, Perkin Elmer NEL741E001KT), TSA Plus Cyanine 3 (1:1,000, Perkin Elmer NEL744E001KT), and TSA Plus Cyanine 5 (1:1,000, Perkin Elmer NEL745E001KT) evaluation kits. Confocal image stacks were taken on a Zeiss LSM 800, 20× objective, 0.26 μm pixel diameter.
One section was selected from each of six mice for the fluorescence in situ hybridization (FISH) analysis. These sections were centred on the somatosensory sector of the TRN (approximately 1.4 mm posterior to bregma). Four of the six mice were C57 wild-types. The remaining two mice were PV-Cre × Ai14 mice that expressed tdTomato in PV-Cre cells (the tdTomato-C3 probe, 317041-C3, was used as a proxy for PV for these two mice). Images were quantified using ImageJ. Regions of interest (ROIs) were manually drawn for each cell on the basis of the fluorescence signal of all three channels (SOM, CB and either PV or tdTomato). For quantification, the mean fluorescence intensity for a cell’s ROI was expressed as a percentage of the intensity range across the TRN of the section: 100 × ((mean intensity for the cell’s ROI)/(maximum pixel intensity in the TRN − minimum pixel intensity in the TRN)). Cells were considered positive for an RNA marker if this normalized expression for the marker was greater than 7.5%, which best matched qualitative visual assessment of expression thresholds.
Probes for PV (Mm-Pvalb-C4, 421931-C4), Cre (CRE-C3, 312281-C3), and SOM (Mm-Sst, 404631) were used to assess correspondence between Cre and Sst mRNA expression in the SOM-Cre mouse line (4 SOM-Cre × ICR mice).
Cell count analysis
For both immunohistochemical and FISH analysis, cells were counted in a region centred on somatosensory TRN extending 300 μm along the dorsal–ventral axis of the nucleus (dorsal–ventral boundaries indicated by the brackets in Fig. 1a, b, or the boxes in Extended Data Figs. 1b, 2a). This region consistently received axonal projections from the S1 cortex (data not shown) and the somatosensory thalamus (VP and POM; Fig. 2, Extended Data Fig. 4). Central/edge boundaries were drawn at 20% and 80% of the medial–lateral distance across the TRN.
Stereotactic injection procedure
Mice were anaesthetized with a mixture of Ketaset and Dexdormitor diluted in sterile saline (Ketaset, 70 mg/kg; Dexdormitor, 0.25 mg/kg; intraperitoneally). Once deeply anaesthetized, mice were placed into a stereotactic frame, and a craniotomy was made over the VP, POM, dorsal lateral geniculate nucleus of the thalamus (dLGN), lateral posterior nucleus of the thalamus (LP) or TRN. Virus solution was then pressure-ejected into the brain via a glass micropipette attached to a Picospritzer pressure system at a maximum rate of around 0.05 μl/min (10–30 min total injection times). After injection, the pipette was left in place for around 10 min before being slowly withdrawn from the brain. After surgery, mice were given Antisedan (2.5 mg/kg) to reverse the effects of Dexdormitor, and they were allowed to recover on a heating pad for around 1 h before being returned to their home cage. Experiments were usually performed about 10 days after the virus injections to allow for sufficient GFP or opsin expression (mean, 10.4 ± 0.3; range, 8–19 days).
Adeno-associated viruses (AAVs) and lentiviruses were acquired from the University of North Carolina, Addgene or the University of Pennsylvania Vector Cores and used at the following titers: (1) rAAV2/hSyn-ChR2(H134R)-eYFP-WPREpA (titre = ~3.93 × 1012 vg/ml), (2) pLenti-Synapsin-hChR2(H134R)-eYFP-WPRE (titre = ~2.53 × 1010 vg/ml), (3) AAV2.Syn.DIO.hChR2(H134R)-eYFP.WP.hGH (titre = ~2.32 × 1013 vg/ml), (4) rAAV2/hSyn-ChR2(H134R)-mCherry-WPREpA (titre = ~2.2 × 1012 vg/ml), (5) AAV9.Syn.DIO.EGFP.WPRE.hGH (titre = ~6.25 × 1012 vg/ml), (6) rAAV2/Syn-Flex-ChrimsonR-TdT (titre = ~3.8 × 1012 vg/ml).
For experiments testing projections from specific thalamocortical nuclei to the TRN (or from subtypes of TRN cells to thalamocortical nuclei), relatively small volumes (0.16–0.33 μl) of either AAV2 or lentivirus (described above) were stereotactically injected into individual presynaptic nuclei using the following mouse strains and coordinates. To test VP projections to the TRN (Figs. 2, 3, Extended Data Figs. 4–6), virus was injected into SOM-Cre × Ai14 (n = 15), ICR (n = 4), Vglut2-Cre × ICR (n = 5), or PV-Cre × Ai14 (n = 1) mice between P12 and P16 (mean 13.1 ± 0.2 days). Average coordinates from bregma for VP were 1.99 mm lateral, −0.74 mm posterior, 3.08 mm depth. For POM projections to TRN (Figs. 2–3, Extended Data Figs. 4–6), virus was injected into SOM-Cre × Ai14 (n = 8), ICR (n = 6), PV-Cre × Ai14 (n = 2), or GPR26-Cre × ICR (n = 2) mice between P11 and P14 (mean 12.0 ± 0.3). Coordinates for POM were 1.36 mm lateral, −1.17 mm posterior, 2.86 depth. For dLGN projections to TRN (Extended Data Fig. 9), virus was injected into SOM-Cre × Ai14 (n = 2), PV-Cre × Ai14 (n = 2) or GPR26-Cre × ICR (n = 1) mice between P14–P19 (mean 16.0 ± 0.8). Coordinates for dLGN were 2.38 mm lateral, −1.45 mm posterior, 2.35 depth. For LP projections to TRN (Extended Data Fig. 9), virus was injected into SOM-Cre × Ai14 (n = 6), PV-Cre × Ai14 (n = 6) or CB-Cre × Ai14 (n = 1) mice between P14–P17 (mean 15 ± 0.3). Coordinates for LP were 1.63 mm lateral, −1.36 mm posterior, 2.3 mm depth. For tests of the inhibitory outputs of TRN cell subtypes to VP and POM neurons (Extended Data Fig. 8), virus was injected into SOM-Cre × ICR (n = 5), CB-Cre × Ai14 (n = 1) or CB-Cre × ICR (n = 2) mice between P12 and P15 (mean 13.7 ± 0.3). Coordinates for these TRN injections were 2.2 mm lateral, −0.53 mm posterior, 3.0 mm depth.
For assessments of the positions of TRN cell subtypes (Fig. 1e, Extended Data Figs. 1, 3), relatively large volumes (1.3–2 μl) of AAV9 (described above) were injected across the entire dorsal–ventral extent of the TRN in SOM-Cre × Ai14 (n = 10), SOM-Cre × ICR (n = 2), PV-Cre × Ai14 (n = 4) or ICR (n = 2) mice between P14 and P25 (mean 16.3 ± 0.9). Average coordinates for these TRN injections were 2.2 mm lateral; −0.53 mm posterior, with continuous outflow of virus solution from 2.2 mm to 4.4 mm depth.
Slice preparation
Brain slices were prepared from P22–P34 mice of either sex as previously described40. Mice were deeply anaesthetized with isofluorane and decapitated. The brains were removed while submerged in cold (4 °C) oxygenated slicing solution containing (in mM): 3.0 KCl, 1.25 NaH2PO4, 10.0 MgSO4, 0.5 CaCl2, 26.0 NaHCO3, 10.0 glucose and 234.0 sucrose. Brains were then mounted, using a cyanoacrylate adhesive, onto the stage of a vibrating tissue slicer (Leica VT1000 or VT1200S) and somatosensory thalamocortical brain slices (300 μm thick, 35° tilt from coronal46) containing VP, POM, TRN, S1 and portions of dLGN and LP were obtained. Slices were incubated for around 1 min in the cold sucrose-based slicing solution, then transferred for 20 min to a holding chamber containing warm (32 °C) oxygenated (5% CO2, 95% O2) artificial cerebrospinal fluid (ACSF) solution. Finally, the slices were allowed to equilibrate in ACSF for 60 min at room temperature before imaging or recording. The ACSF solution contained (in mM): 126.0 NaCl, 3.0 KCl, 1.25 NaH2PO4, 1.0 MgSO4, 1.2 CaCl2, 26.0 NaHCO3, and 10.0 glucose.
Live imaging
Live sections (300 μm) centred on the somatosensory sector of TRN (−1.4 mm from bregma) were imaged using Nikon or Zeiss upright microscopes with 2.5–5× objectives and Andor Zyla sCMOS cameras. Both epifluorescent and transmitted light (bright-field) images were obtained to characterize the topographical positions of the TRN cell types and their connections with thalamic relay nuclei.
To generate the group maps showing the average VP and POM projections to the TRN (Fig. 2b), the TRN of each live slice was first outlined using either tdTomato expression driven by SOM-Cre (n = 6 for POM, n = 4 for VP) or bright-field images (n = 3 for POM, n = 4 for VPM). Those outlines were then used to warp the slice images to a common reference TRN (with the bUnwarpJ plugin in ImageJ—described for the immunohistochemical analysis above), allowing precise alignment across mice for averaging. The central/edge boundaries were drawn at 20% and 80% of the medial–lateral distance across the TRN.
Whole-cell recording procedure
Brain slices (300 μm) were placed in a submersion-type recording chamber maintained at 32 ± 1 °C and continuously superfused with oxygenated ACSF (above). Neurons were visualized for recording using DIC-IR optics with 40× water immersion objectives. Patch pipettes had tip resistances of 3–6 MΩ when filled with a potassium-based internal recording solution containing (in mM): 130.0 K-gluconate, 4.0 KCl, 2.0 NaCl, 10 HEPES, 0.2 EGTA, 4.0 ATP-Mg, and 0.3 GTP-Tris, 14.0 phosphocreatine-K (pH 7.25, ~290 mOsm). During all recordings, pipette capacitances were neutralized. Series resistances (~12–32 MΩ) were compensated online (100% for current–clamp, 60–70% for voltage-clamp). Pharmacological agents (stated in the figure legends when used) were diluted in ACSF just before use and applied though the bathing solution. Voltages reported here were corrected for a 14 mV liquid junction potential. The reversal potential for GABAA receptor-mediated responses in thalamic relay cells was −91 mV.
Measurements of intrinsic physiological properties
Resting membrane potentials were measured within 2 min of break-in. Steady-state potentials were adjusted to −74 mV or −84 mV with intracellular current to test physiological properties in tonic or burst mode, respectively. Input resistances (Rin) and membrane time constants (τm) were calculated from voltage responses (~3 mV deflections) to small negative current injections (3–50 pA, 600–1,000 ms). For τm, the voltage responses were fitted with a single exponential to the initial 50 ms of the response, omitting the first ms. Rin values were measured using Ohms law and input capacitances Cin were calculated as τm/Rin.
Threshold (rheobase) currents were measured as the minimum injected currents required to discharge an action potential (AP; determined using 1 s duration currents, 5 pA step increments). All other AP and after-AP properties were measured from the first AP discharged at the threshold current, but APs were only analysed if discharged in the initial 200 ms). AP voltage thresholds were measured, and verified by visual inspection, as the potential at which the rate of rise became greater than 10 V/s. AP amplitudes and after-AP properties were measured relative to the threshold potential. AP widths were measured at half of the AP amplitude (threshold voltage to peak). Fast afterhyperpolarizations (fast AHPs) were measured as the most hyperpolarized potential immediately succeeding the AP. In tonic mode, afterdepolarizations (ADPs) were measured as the most depolarized potential within 20 ms of the AP, and slow AHPs (sAHPs) were measured as the most hyperpolarized potential within 100 ms after the ADP. In burst mode, the AHP following the burst was measured as the minimum potential within 150 ms following the burst. ADP and sAHP measurements were not considered if a second AP (or tonic APs after a burst) confounded the measurements.
Repetitive spiking properties in tonic and burst mode were measured using positive current steps (25–200 pA, 25 pA increments, 1 s duration). Spike frequency adaptation was quantified as an adaptation ratio (frequency of the last 2 APs divided by the frequency of the first 2 APs) averaged across all sweeps in which the frequency of the last 2 APs was 20–60 Hz.
To elicit offset bursts (Fig. 3, Extended Data Fig. 9, Supplementary Information 1), the steady-state potential was adjusted to −74 mV with intracellular current, then 1 s duration negative currents were injected. Offset bursts were measured from trials in which negative current (20–300 pA, 20–25 pA test increments) led to a voltage of approximately −94 mV at the end of the current step.
Photostimulation
Synaptic physiology experiments were performed on inputs to TRN cells from excitatory thalamic relay neurons (Figs. 2, 3), and on inhibitory outputs of TRN cells to thalamic relay neurons. In both cases, ChR2 was optically excited using white light-emitting diodes (LEDs) (Mightex LCS-5500-03-22) controlled by Mightex LED controllers (SLCAA02-US or BLS-1000-2). The light was collimated and reflected through a 40× water immersion objective, resulting in a spot diameter of ~400 μm and a maximum LED power at the focal plane of 29.2 mW. The stimuli, delivered as 10 Hz trains of 1 ms flashes, were typically directed at ChR2-expressing presynaptic terminals by centring the light spot over the recorded postsynaptic cells (the postsynaptic cells did not express ChR2).
In a subset of experiments (Extended Data Fig. 6a–c), within-cell comparisons were made between TRN cell responses to stimulation directed at opsin-expressing presynaptic axons/terminals (from VP or POM) and stimulation directed further upstream, at or near the VP or POM cell bodies of origin. These experiments tested whether short-term synaptic plasticity differed when optogenetically stimulating soma/proximal axons of presynaptic cells versus their terminal boutons, as has been shown for some pathways47.
Simulated synaptic current injections
Simulated EPSC waveforms for thalamic (VP and POM) inputs to the TRN were generated from averaged measurements of optogenetically evoked synaptic responses (recorded in voltage clamp at −84 mV) from central and edge TRN cells (as in Fig. 3). These simulated synaptic currents for VP and POM inputs were matched for total synaptic charge across the 10 Hz trains (49.5 pC) and injected into the central and edge TRN cells to test features of integration in each cell type (described in the text and in Fig. 4).
Data analyses
Analyses of electrophysiological data were performed using CED Signal 6, Molecular Devices Clampfit 10, MATLAB and Microsoft Excel. Analyses of anatomical data were performed using ImageJ (plugins used: CellCounter, ROIManager, bUnwarpJ) and Microsoft Excel.
Statistical analyses
Statistical comparisons were performed using GraphPad Prism7 or SigmaPlot. Statistical tests used are indicated in the main text and figure legends. For representation of group data, centre values are means and error bars show s.e.m. Statistical significance was defined as P < 0.05, unless otherwise noted.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this paper.
Data availability
All data are available from the corresponding author upon reasonable request.
Change history
27 August 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41586-020-2680-3
References
Jones, E. G. The Thalamus (Cambridge Univ. Press, 2007).
Sherman, S. M. & Guillery, R. W. Functional organization of thalamocortical relays. J. Neurophysiol. 76, 1367–1395 (1996).
Hirsch, J. A., Wang, X., Sommer, F. T. & Martinez, L. M. How inhibitory circuits in the thalamus serve vision. Annu. Rev. Neurosci. 38, 309–329 (2015).
Crabtree, J. W. Functional diversity of thalamic reticular subnetworks. Front. Syst. Neurosci. 12, 41 (2018).
Halassa, M. M. & Acsády, L. Thalamic inhibition: diverse sources, diverse scales. Trends Neurosci. 39, 680–693 (2016).
Fogerson, P. M. & Huguenard, J. R. Tapping the brakes: cellular and synaptic mechanisms that regulate thalamic oscillations. Neuron 92, 687–704 (2016).
Guillery, R. W., Feig, S. L. & Lozsádi, D. A. Paying attention to the thalamic reticular nucleus. Trends Neurosci. 21, 28–32 (1998).
Pinault, D. The thalamic reticular nucleus: structure, function and concept. Brain Res. Rev. 46, 1–31 (2004).
Crick, F. Function of the thalamic reticular complex: the searchlight hypothesis. Proc. Natl Acad. Sci. USA 81, 4586–4590 (1984).
Diamond, M. E., von Heimendahl, M., Knutsen, P. M., Kleinfeld, D. & Ahissar, E. ‘Where’ and ‘what’ in the whisker sensorimotor system. Nat. Rev. Neurosci. 9, 601–612 (2008).
Mease, R. A., Sumser, A., Sakmann, B. & Groh, A. Cortical dependence of whisker responses in posterior medial thalamus in vivo. Cereb. Cortex 26, 3534–3543 (2016).
Sherman, S. M. Thalamus plays a central role in ongoing cortical functioning. Nat. Neurosci. 19, 533–541 (2016).
Fishell, G. & Rudy, B. Mechanisms of inhibition within the telencephalon: “where the wild things are”. Annu. Rev. Neurosci. 34, 535–567 (2011).
Hou, G., Smith, A. G. & Zhang, Z. W. Lack of intrinsic GABAergic connections in the thalamic reticular nucleus of the mouse. J. Neurosci. 36, 7246–7252 (2016).
Hu, H., Cavendish, J. Z. & Agmon, A. Not all that glitters is gold: off-target recombination in the somatostatin-IRES-Cre mouse line labels a subset of fast-spiking interneurons. Front. Neural Circuits 7, 195 (2013).
Lam, Y. W. & Sherman, S. M. Functional organization of the thalamic input to the thalamic reticular nucleus. J. Neurosci. 31, 6791–6799 (2011).
Lee, S. H., Govindaiah, G. & Cox, C. L. Heterogeneity of firing properties among rat thalamic reticular nucleus neurons. J. Physiol. (Lond.) 582, 195–208 (2007).
Diamond, M. E., Armstrong-James, M. & Ebner, F. F. Somatic sensory responses in the rostral sector of the posterior group (POm) and in the ventral posterior medial nucleus (VPM) of the rat thalamus. J. Comp. Neurol. 318, 462–476 (1992).
Roth, M. M. et al. Thalamic nuclei convey diverse contextual information to layer 1 of visual cortex. Nat. Neurosci. 19, 299–307 (2016).
Audette, N. J., Bernhard, S. M., Ray, A., Stewart, L. T. & Barth, A. L. Rapid plasticity of higher-order thalamocortical inputs during sensory learning. Neuron 103, 277–291.e4 (2019).
Zhang, W. & Bruno, R. M. High-order thalamic inputs to primary somatosensory cortex are stronger and longer lasting than cortical inputs. eLife 8, e44158 (2019).
Steriade, M., Domich, L. & Oakson, G. Reticularis thalami neurons revisited: activity changes during shifts in states of vigilance. J. Neurosci. 6, 68–81 (1986).
McCormick, D. A. & Bal, T. Sleep and arousal: thalamocortical mechanisms. Annu. Rev. Neurosci. 20, 185–215 (1997).
Marlinski, V. & Beloozerova, I. N. Burst firing of neurons in the thalamic reticular nucleus during locomotion. J. Neurophysiol. 112, 181–192 (2014).
Whitmire, C. J., Waiblinger, C., Schwarz, C. & Stanley, G. B. Information coding through adaptive gating of synchronized thalamic bursting. Cell Rep. 14, 795–807 (2016).
Llinás, R. R. & Steriade, M. Bursting of thalamic neurons and states of vigilance. J. Neurophysiol. 95, 3297–3308 (2006).
Clemente-Perez, A. et al. Distinct thalamic reticular cell types differentially modulate normal and pathological cortical rhythms. Cell Rep. 19, 2130–2142 (2017).
Destexhe, A., Contreras, D., Steriade, M., Sejnowski, T. J. & Huguenard, J. R. In vivo, in vitro, and computational analysis of dendritic calcium currents in thalamic reticular neurons. J. Neurosci. 16, 169–185 (1996).
Llinás, R. & Jahnsen, H. Electrophysiology of mammalian thalamic neurones in vitro. Nature 297, 406–408 (1982).
Lambert, R. C., Bessaïh, T., Crunelli, V. & Leresche, N. The many faces of T-type calcium channels. Pflugers Arch. 466, 415–423 (2014).
Kimura, A. & Imbe, H. Anatomically structured burst spiking of thalamic reticular nucleus cells: implications for distinct modulations of sensory processing in lemniscal and non-lemniscal thalamocortical loop circuitries. Eur. J. Neurosci. 41, 1276–1293 (2015).
Pinault, D., Bourassa, J. & Deschênes, M. The axonal arborization of single thalamic reticular neurons in the somatosensory thalamus of the rat. Eur. J. Neurosci. 7, 31–40 (1995).
McAlonan, K., Cavanaugh, J. & Wurtz, R. H. Guarding the gateway to cortex with attention in visual thalamus. Nature 456, 391–394 (2008).
Lee, S. C., Patrick, S. L., Richardson, K. A. & Connors, B. W. Two functionally distinct networks of gap junction-coupled inhibitory neurons in the thalamic reticular nucleus. J. Neurosci. 34, 13170–13182 (2014).
Crabtree, J. W. & Isaac, J. T. New intrathalamic pathways allowing modality-related and cross-modality switching in the dorsal thalamus. J. Neurosci. 22, 8754–8761 (2002).
Kimura, A. & Imbe, H. Robust subthreshold cross-modal modulation of auditory response by cutaneous electrical stimulation in first- and higher-order auditory thalamic nuclei. Neuroscience 372, 161–180 (2018).
Briggs, F. & Usrey, W. M. Emerging views of corticothalamic function. Curr. Opin. Neurobiol. 18, 403–407 (2008).
Crandall, S. R., Cruikshank, S. J. & Connors, B. W. A corticothalamic switch: controlling the thalamus with dynamic synapses. Neuron 86, 768–782 (2015).
Wimmer, R. D. et al. Thalamic control of sensory selection in divided attention. Nature 526, 705–709 (2015).
Nakajima, M., Schmitt, L. I. & Halassa, M. M. Prefrontal cortex regulates sensory filtering through a basal ganglia-to-thalamus pathway. Neuron 103, 445–458.e10 (2019).
Beierlein, M. Synaptic mechanisms underlying cholinergic control of thalamic reticular nucleus neurons. J. Physiol. (Lond.) 592, 4137–4145 (2014).
Conley, M., Kupersmith, A. C. & Diamond, I. T. The organization of projections from subdivisions of the auditory cortex and thalamus to the auditory sector of the thalamic reticular nucleus in galago. Eur. J. Neurosci. 3, 1089–1103 (1991).
Conley, M., Schmechel, D. E. & Diamond, I. T. Differential distribution of somatostatin-like immunoreactivity in the visual sector of the thalamic reticular nucleus in galago. Eur. J. Neurosci. 3, 237–242 (1991).
Graybiel, A. M. & Elde, R. P. Somatostatin-like immunoreactivity characterizes neurons of the nucleus reticularis thalami in the cat and monkey. J. Neurosci. 3, 1308–1321 (1983).
Daigle, T. L. et al. A suite of transgenic driver and reporter mouse lines with enhanced brain-cell-type targeting and functionality. Cell 174, 465–480. e22 (2018).
Agmon, A. & Connors, B. W. Thalamocortical responses of mouse somatosensory (barrel) cortex in vitro. Neuroscience 41, 365–379 (1991).
Jackman, S. L., Beneduce, B. M., Drew, I. R. & Regehr, W. G. Achieving high-frequency optical control of synaptic transmission. J. Neurosci. 34, 7704–7714 (2014).
Acknowledgements
We thank Z. Fu, G. Feng and their associates for interactions surrounding this project. We also thank S. Crandall, O. Ahmed, M. Zervas, D. Lipscombe, C. Chen, G. Manzano, S. Baskoylu, F. Pouille, B. Theyel and F. S. Susi for discussions. This work was supported by grants R01 NS100016, P20 GM103645, NSF 1738633, NSFGRFP 1058262 and NSF 1632738.
Author information
Authors and Affiliations
Contributions
R.I.M.-G., B.V., B.W.C. and S.J.C. designed the experiments. R.I.M.-G., B.V., J.B.Z., S.L.P., T.R.S. and S.J.C. conducted the experiments. R.I.M.-G., B.V., J.B.Z. and S.J.C. analysed the results. R.I.M.-G., B.V., B.W.C. and S.J.C. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks Karel Svoboda and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Somatostatin cells form a shell around a calbindin-rich central core of the somatosensory TRN; nearly all neurons in the somatosensory TRN express parvalbumin.
a, Epifluorescence images showing serial sections of live tissue expressing AAV9-DIO-GFP (9 days of expression) through the TRN of two P23 SOM-Cre mice. Anterior-posterior (AP) positions of the sections relative to bregma are indicated. Notice how the SOM cells form a shell-like structure surrounding the central core of the somatosensory TRN (centred ~ −1.4 mm from bregma) (experiment replicated 12 times). b, Top, dot plots showing the locations of the genetically or immunohistochemically labelled TRN cell types for sections at different AP distances from bregma. Bottom, proportion of cells expressing PV, CB, SOM-Cre × tdT, and combinations of the markers. Quantification was restricted to the boxed regions shown in the top panels (cell counts: left, 947; middle, 1,075; right, 843; 3 sections from 3 mice). The proportion of CB cells was highest in the middle section of the somatosensory TRN (−1.4 mm), whereas the proportion of SOM-Cre × tdT cells was lowest in the middle section and higher in the more anterior and posterior sections (P < 0.001, χ2 test with Yates’ correction). Data are mean ± s.e.m. c, Left, confocal images of the somatosensory sector of TRN in a PV-Cre × tdT mouse (red) stained for NeuN (green). Right, quantification of expression. In PV-Cre × tdT mice, 98.09% of TRN neurons were double positive for tdT and NeuN, 0.73% were positive only for tdT, and 1.18% were positive only for NeuN (1,218 neurons, 2 sections, 1 mouse). d, Same as c but tissue was stained immunohistochemically for parvalbumin. In PV-Cre × tdT mice, 98.66% of TRN neurons were double-positive for tdT and PV, 0.34% were positive only for tdT and 0.34% were positive only for PV (1,177 neurons, 2 sections, 1 mouse). e, DIC-IR (left) and tdT fluorescence (right) images of TRN in a live slice from a PV-Cre × tdT mouse. Asterisk indicates a soma that did not express tdT. During such live imaging, tdT-negative somata were relatively rare in TRN (>80% of somata visualized in DIC-IR expressed tdT; n = 5 slices, 3 mice; right panel). f, All tdT-negative cells targeted for recording (n = 8/8) had physiological properties of glia. Left, TRN glial cells had hyperpolarized resting potentials (typically less than −85 mV) and no action potentials even during large depolarizations.
Extended Data Fig. 2 Cells expressing Sst and Calb1 mRNA are topographically segregated in the somatosensory TRN.
a, Left, confocal image overlay of expression of Sst and Calb1 mRNA (encoding SOM and CB) through the TRN of a PV-Cre × tdT mouse. In the somatosensory TRN (boxed region), Sst expressing cells are preferentially localized in the medial and lateral edge zones, whereas Calb1 cells are generally in the central zone. Insets show mRNA expression patterns of cells in the lateral edge (top row), central (middle row), and medial edge (bottom row) zones. Although most cells predominantly expressed either Sst (red) or Calb1 (green) mRNA, some (about 10%) expressed both (arrow). b, Proportion of cells expressing various combinations of markers for each zone (for b–e, 593 cells, 6 sections, 6 mice; average of 98.9 ± 8.5 cells per section). 99.1 ± 0.2% of cells expressed PV. Most cells in the medial and lateral edge zones expressed only Sst and Pvalb, but not Calb1 (about 64%). By contrast, most central cells expressed only Calb1 and Pvalb, but not Sst (about 76%). c, Normalized Sst (left) or Calb1 (right) mRNA expression as a function of medial–lateral position in the the TRN (0 = most medial, 1 = most lateral). Cells expressing Sst but not Calb1 were concentrated near the edges (red) (mean Sst expression for edge vs central: 0.325 ± 0.020 versus 0.052 ± 0.006, P < 0.0001, unpaired two-tailed t-test). Cells in the centre generally had high levels of Calb1 but not Sst (mean Calb1 expression for central vs edge: 0.326 ± 0.012 versus 0.086 ± 0.012, P < 0.0001, unpaired two-tailed t-test). Around 10% of somatosensory TRN cells expressed both Sst and Calb1 (yellow) and clustered in the lateral half of the TRN, with their focus being near the boundary between the central and lateral zones (average normalized medial–lateral position 0.71 ± 0.02). Compared with the cells expressing Sst or Calb1 ‘alone’, the dual Sst/Calb1 cells had lower expression of both Sst (Sst/Calb1 cells: 0.25 ± 0.02 vs Sst only: 0.43 ± 0.02, P < 0.0001, unpaired two-tailed t-test) and Calb1 mRNA (Sst/Calb1 cells: 0.23 ± 0.15 vs Calb1 only: 0.39 ± 0.01, P < 0.0001, unpaired two-tailed t-test). Together this suggests they may represent a transitional population. About 9% of somatosensory TRN cells expressed PV only (grey) and were dispersed fairly evenly across the sector (mean medial-lateral position: 0.38 ± 0.03). d, Relative expression of Sst versus Calb1 (within-cell) as a function of medial-lateral position in the TRN (a value of 1.0 on the y-axis index indicates Sst expression only, a value of 0 indicates Calb1 only, and 0.5 indicates equal expression values for Sst and Calb1). Most cells (80%) had very high (>0.9) or very low (<0.1) relative expression scores, indicating most cells have strong biases towards a single marker (10:1 ratios or greater). Consistent with Fig. 1 and a–c, Sst-biased cells tended to cluster towards the TRN edges, whereas Calb1-biased were located centrally (edge versus central preference scores: 0.76 ± 0.03 versus 0.14 ± 0.01, P < 0.0001, paired two-tailed t-test). A moving average (11-point) is superimposed to illustrate the spatial trends in expression bias. Interestingly, even the dual-expressing Sst/Calb1 cells (yellow) tended to have a topographical preference according to relative Sst vs Calb1 expression: dual-expressing edge cells tended to have higher Sst:Calb1 preference scores than dual-expressing central cells (dual edge versus central cells: 0.58 ± 0.04 versus 0.47 ± 0.04, P = 0.06, unpaired two-tailed t-test). e, Calb1 expression as a function of Sst expression for cells in the medial, central, lateral, and combined zones. P values shown in the panels are the results of two-tailed paired (within-cell) t-tests comparing Calb1 and Sst expression. Data are mean ± s.e.m.
Extended Data Fig. 3 In the somatosensory TRN of postnatal SOM-Cre mice, both Cre expression and viral Cre-dependent protein expression are restricted mainly to SOM cells along the edges.
a, Epifluorescence images of virally induced, SOM-Cre-dependent GFP expression patterns in TRN (live 300-μm-thick slices from 7 mice). Virus carrying a Cre-dependent GFP gene (AAV9-DIO-GFP) was injected throughout the TRNs of SOM-Cre × tdTomato or SOM-Cre × ICR mice. This drove GFP expression (green) in SOM cells that expressed Cre at or after the time of the virus injection (age at injection P17.1 ± 1.18 days; age at imaging 26.8 ± 1.19). Mouse 1: images of GFP alone and overlay of GFP and genetically labelled tdTomato cells. Mice 2–7: GFP alone. Within the somatosensory TRN, expression of SOM-Cre × GFP was restricted to the edges of the nucleus and excluded from the central zone after early development. Experiment replicated 12 times. b, Same as in a except using PV-Cre mice (age at virus injection P14; age at imaging P23). Nearly all of the TRN cells expressed both genetically labelled PV-Cre × tdTomato and virally induced PV-Cre × GFP. In addition to demonstrating ubiquitous postnatal PV-Cre expression across the TRN, these results show that the virus has no obvious tropisms for different topographic regions of the TRN. Experiment replicated 4 times. c, Confocal images of Pvalb, Sst, and Cre mRNA expression through the TRN of a P27 SOM-Cre heterozygous mouse (18 μm thick section). The overall patterns of Sst and Cre mRNA labelling were nearly identical (experiment replicated in 5 sections from 4 mice). d, Sst and Cre mRNA were generally expressed in the same neurons (total counts: 167 cells, 5 sections, 4 mice; measured in the somatosensory sector of the TRN). Of the Cre-expressing cells, 88.3 ± 0.1% expressed Sst mRNA. Of the Sst-expressing cells, 96.1 ± 0.01% expressed Cre mRNA. e, AAV9-DIO-GFP injections into the TRNs of postnatal SOM-Cre mice led to GFP expression patterns that were very similar to Sst mRNA patterns. Left, DIC and epifluorescence images from a live 300-μm-thick section expressing AAV9-DIO-GFP (9 days after virus injection) in a SOM-Cre heterozygous mouse. This experiment was replicated 12 times (see a, Fig. 1g and Extended Data Fig. 1a for additional examples). f, Same as e but virus was injected into a wild-type mouse (ICR strain; no Cre expression) (tested in n = 2 mice). Matching display settings were used for the slices in c and d. The lack of fluorescent signal indicates that the AAV-DIO-GFP virus used here drives GFP expression only in Cre-expressing cells. Data are mean ± s.e.m.
Extended Data Fig. 4 VP afferents target the CB-dense centre of the somatosensory TRN, whereas POM afferents target the SOM-dense medial and lateral edges.
a, Four representative examples showing the POM injection sites and anterograde projections to TRN in live 300-μm slices. Red: SOM-Cre × tdT. Green: ChR2–eYFP. Boundaries of the thalamic nuclei (TRN outlined by dashed lines) were determined from SOM-Cre × tdT fluorescence and by bright-field images (not shown). Injection sites were located in POM nuclei, seen here as the large, bright eYFP spots medial to TRN. The smaller bands of eYFP along the medial and lateral edges of the somatosensory TRN are axons and terminals from the labelled POM cells. In addition to the TRN edges, POM also targeted layer 5a (L5a) and L1 of barrel cortex (but not L4; data not shown). Top left image shows the injection site for Fig. 2c, d. Experiment replicated in 18 mice. b, Left, schematic showing recording configuration (also applies to the experiments of Fig. 2c–f). Right, additional representative examples of synaptic responses of cells across the medial–lateral axis of somatosensory TRN evoked by POM stimulation. TRN cells near the edges of the somatosensory TRN received strong input from the POM whereas cells located in the centre of the TRN did not. Experiments replicated in 5 slices, 5 mice. c, Similar to a, but for VP afferents. VP afferents clearly targeted the central zone of TRN. VP also projected to L4 of the barrel cortex but not to L5a or L1 (not shown). Top left image shows the injection site for Fig. 2g, h. Experiments were replicated in 25 mice. d, Similar to b, but for VP. Left, schematic showing recording configuration (also applies to the experiments of Fig. 2g–j). Right, cells in the centre of the somatosensory TRN received stronger input from the VP than did cells located near the edges of the TRN. The experiment was replicated in 6 slices, 4 mice.
Extended Data Fig. 5 Comparison of EPSC kinetics and short-term synaptic depression based on topography or reporter labelling.
a, Schematic showing approximate locations of TRN somata targeted for each subcircuit. EPSCs in medial and lateral edge cells were examined separately here, using the dataset from Fig. 3a–c. V-clamp at −84 mV. b, Kinetics of the EPSCs. Left, optically evoked EPSCs of example cells from each group (stimulus 1 of c). EPSCs evoked by POM inputs to medial and lateral edge cells were broader, with greater fractional areas in the later portions of the responses than EPSCs evoked by VP inputs to central cells. Right, kinetic differences between responses are shown for two methods of quantification. The first is the ratio of the EPSC area during the last 90 ms of the response (10–100 ms from stimulus onset) to the full EPSC area (0–100 ms from stimulus onset; left graph). This ratio, which describes the fractional area of the late portion of the response, varied among cells groups (ANOVA, P < 0.0001). Central values were significantly lower than medial and lateral edge values (two-tailed Bonferroni’s t-tests, P < 0.002 and P < 0.002, respectively), whereas the two edge groups were not different from one another (two-tailed Bonferroni’s t-test, P = 0.99). As a second method of quantifying response kinetics we calculated the ratio of the full EPSC area (0–100 ms from stimulus onset) to the peak EPSC amplitude (right graph, as in Fig. 3b). This ratio also varied as a function of cell group (ANOVA, P < 0.0005). Again, central values were significantly lower than medial and lateral edge values (two-tailed Bonferroni’s t-tests, P < 0.002 and P < 0.03, respectively), whereas the two edge groups were not different from one another (two-tailed Bonferroni’s t-test, P = 0.99). The half-widths of EPSCs were not significantly different between any of the groups (all P > 0.05 for two-tailed Bonferroni’s t-tests; data not shown). For b, c, central cells: 48 cells from 14 mice; medial cells: 13 cells from 11 mice, lateral cells: 8 cells from 7 mice. c, Left, representative traces illustrating the short-term synaptic depression of optically evoked EPSCs across groups (10 Hz trains of LED stimuli: arrows). Right, average EPSC areas measured during the first 10 ms of the responses. Depression of VP-evoked responses in central cells was stronger than depression of POM-evoked responses in medial or lateral cells (two-tailed Bonferroni’s t-tests; central versus lateral and central versus medial, both P < 0.0001), whereas depression in the two edge cell groups did not differ significantly (two-tailed Bonferroni’s t-test, P = 0.99). d, Left, schematic showing experimental design. Recordings were made in slices from SOM-Cre × tdT (Ai14) mice. ChR2–eYFP was virally expressed in VP cells. We optically stimulated the VP cells/axons and recorded evoked synaptic responses from closely spaced tdT-positive and tdT-negative neurons in the central zone of the TRN. We ensured that both cells had similar eYFP terminal labelling surrounding them; usually the cells were separated by less than 50 μm. The goal was to test whether there were response differences between central TRN cells that were SOM-Cre × tdT-positive versus SOM-Cre × tdT-negative. Neurons were tdT-positive if they expressed Cre recombinase at any stage of their lives; in SOM-Cre mice, TRN central zone neurons seemed to express Cre only early in development (see text, Fig. 1b, e, Extended Data Figs. 1a, 2, 3). Middle and right, average optically evoked synaptic responses (1 ms LED pulses, 10 Hz trains) for tdT-positive and tdT-negative cell groups for stimuli delivered at 2× (middle) and 16× (right) the threshold for evoking a clear EPSC. Threshold intensity was not significantly different between tdT-positive and tdT-negative cells. The average LED power used at 2× threshold was 2.13 mW. Response magnitudes, short-term EPSC depression, and kinetics (inset traces) did not differ between tdT-positive and tdT-negative cells at 2× threshold (n = 19 pairs of cells) or at 16× threshold (n = 12 pairs of cells) (ANOVA, P = 0.98 and P = 0.61, respectively; all P > 0.05 for cell-type comparisons on individual stimuli in the trains, two-tailed Bonferroni’s t- tests). Thus, VP synaptic inputs seem to be no different for central TRN cells that express SOM-Cre during early development than for those that are SOM-Cre negative continuously. Data are mean ± s.e.m.
Extended Data Fig. 6 Differences in short-term depression between the two somatosensory thalamoreticular pathways do not depend on axon terminal stimulation, stimulus strength, or the temporal components of the responses measured.
Some studies have shown that synaptic release probability and short-term plasticity can be abnormal when optogenetic light stimuli are directed at opsin-expressing presynaptic terminals47. We addressed this issue in the experiments described in a–c. a, VP synapses onto central TRN cells undergo greater short-term depression than POM synapses onto edge cells when optically stimulating the thalamocortical (TC) terminals within TRN. Left, schematic showing recording configuration when optically stimulating over the terminals of the ChR2-expressing TC cells. For the experiments of a–c, GABAA receptors and T-type calcium channels were blocked with picrotoxin (50 μM) and TTA-P2 (1 μM), respectively. Right, average short-term synaptic depression. Light intensities were chosen to evoke around 1.5–3 nA peak responses. EPSC areas were measured over the first 10 ms of each response. There was greater short-term depression for the VP → central cell pathway (n = 20 cells, 5 mice) than the POM → edge cell pathway (n = 17 cells, 8 mice) (P < 0.0001, ANOVA stimulus 2–10; all P < 0.005 from stimulus 5–10, two-tailed Bonferroni’s t-test). b, VP inputs to central cells remain more depressing than POM inputs to edge cells when stimulating within the TC nuclei. Left, schematic showing recording configuration when optically stimulating over cell bodies and proximal axons of the ChR2-expressing TC cells. Same cells as a. Right, average short-term synaptic depression was greater in central than edge cells (P < 0.0001, ANOVA stimulus 2–10; all P < 0.05 from stimulus 5, 7–10, two-tailed Bonferroni’s t-test). This pattern of short-term depression in central cells (VP inputs) and edge cells (POM inputs) did not differ from the previously observed pattern in the same group of cells when stimulating over the TC terminals (a) or in other experiments of the study (d, e, Fig. 3, Extended Data Fig. 5). c, Optical control indicating that light scattering and spread to the TC terminals did not trigger the responses observed when stimulating within TC nuclei (that is, the responses in b). Left, schematic showing recording configuration. The LED was directed to ventrally located ‘control spots’, away from the ChR2-expressing TC pathway but equally distant from the TC terminals as the effective stimulus spots within the TC nuclei. Therefore, if synaptic responses were observed, they would be triggered by light spreading to the ChR2-expressing TC terminals within the TRN. The same cells were recorded as in a, b. Right, EPSC areas when stimulating over the TC nuclei versus the control spots using matching LED parameters (intensities and spot sizes). No clear responses were evoked when stimulating over the control spots, suggesting that light spread to the terminals did not drive the responses when stimulating within the TC nuclei (all P < 0.0001; two-tailed paired t-tests). d, Schematic showing recording configuration for e, f when optically stimulating VP inputs to central cells and POM inputs to edge cells. ChR2-expressing VP and POM inputs were photostimulated at their terminals (1 ms LED pulse, 10 Hz stimuli) and EPSCs were recorded in central and edge TRN cells, respectively. e, Differences in short-term depression of central and edge cells does not change with varying input strength. Maximum response amplitudes tended to be larger for central than for edge cells. Here we compared short-term depression of synaptic responses in central and edge cells, varying stimulus intensities for the central cells so that their responses were either weaker (top) or stronger (bottom) than those of edge cells (mean central EPSCs: 1.3 ± 0.2 nA and 5.5 ± 0.8 nA, respectively; mean edge EPSCs 2.1 ± 0.3 nA). There were no major differences in patterns of short-term synaptic depression for the weaker versus stronger responses. In both cases, there was more depression for the central cells than the edge cells (P < 0.0001, ANOVA stimulus 2–10; all P < 0.05 from stimulus 5–10, two-tailed Bonferoni’s t-test; central weak responses: n = 45 cells, 11 mice; central strong responses: n = 32 cells, 9 mice; edge cells: n = 21 cells, 14 mice). Central responses were acquired at approximately 2× the response threshold for the top row (average LED power 1.92 mW) and approximately 16× response threshold for the bottom row (average LED power 12.5 mW). f, Short-term synaptic depression is similar whether EPSCs are measured only during the initial fast components of the responses (the first 10 ms) or across longer durations (100 ms). Top, normalized EPSC area dynamics for the initial 10 ms of the responses (same plot shown on Fig. 3c). The EPSC amplitudes were similar for central and edge cells to stimulus 1 (mean central EPSCs: 2.0 ± 0.2 nA; mean edge EPSCs 2.1 ± 0.3 nA, unpaired two-tailed t-test, P = 0.93). There was greater short-term depression for the central cells than for the edge cells (P < 0.0001, ANOVA stimulus 2–10; all P < 0.005 from stimulus 5-10, two-tailed Bonferroni’s t-test; central cells: 48 cells, 14 mice; edge: 21 cells, 14 mice). Bottom, normalized area dynamics for the full responses (0–100 ms). Again, there was greater short-term depression for the central cells than for the edge cells (P < 0.0001, ANOVA stimulus 2–10; P < 0.05 for stimulus 8–10, two-tailed Bonferroni’s t-test). Thus, the overall short-term synaptic depression patterns are similar for early and later components of the EPSCs. Data are mean ± s.e.m.
Extended Data Fig. 7 Intrinsic cellular properties vary according to medial–lateral position in the TRN.
a–c, Inset, voltage responses to hyperpolarizing current steps for representative central and edge cells. Throughout this figure, central cells are coloured black and edge cells are red. Note the higher input resistance for the edge cell (316 vs 152 MΩ). Plots show passive membrane properties for central and edge cells as a function of soma position (far medial edge = position 0, far lateral edge = position 1; steady-state Vm ≈ −84 mV). Open circles indicate individual cell measurements. Filled circles are averages for the cell groups: medial edge, central and lateral edge. d, Central cells have larger somas than edge cells. ROIs outlining complete somas were hand-drawn with the aid of ROIManager (ImageJ) using the PV-IHC tissue from Fig. 1 (n = 575 cells; 3 sections; 3 mice). The mean soma area for central and edge were 110.1 μm2 vs 95.6 μm2, respectively (unpaired two-tailed t-test, P < 0.0001). Cell areas seem to change gradually with location. e, Representative examples of edge cell and central cell action potentials (APs). f–h, AP properties as a function of soma location. Half-width (f), AP maximum rate-of-rise (g), AP maximum rate-of-repolarization (h). Edge cells have wider APs due to slower repolarization slopes, suggesting differences in potassium conductances. i, Representative AP afterpotentials of each cell type. Edge cells generally have more hyperpolarized fast-afterhyperpolarizations (fast-AHPs), afterdepolarizations (ADPs), and slow-afterhyperpolarizations (slow-AHPs). j–l, Fast-AHP (j), ADP (k) and slow-AHP (l) as a function of somatic location (APs evoked from a steady state Vm ≈ −74 mV; values relative to AP threshold). m, Representative examples of repetitive tonic firing evoked by 75 pA current steps (from −74 mV baseline potentials) for central and edge cells. n, Spike frequency vs current intensity (FI) plots. Closed circles show the frequency for the first 2 spikes in each train. Open circles show the frequency for the last 2 spikes. The initial firing frequency is similar between edge and central cells at multiple current steps. However, edge cells fire at higher frequencies than central cells by the end of the current pulses, perhaps because edge cells have higher input resistances (for n, o, central cells n = 29, edge cells n = 40). o, Central cells have more spike frequency adaptation than edge cells (unpaired two-tailed t-test P < 0.0001). p–r, Same as m–o, except responses were evoked from steady-state potentials of −84 mV to elicit intrinsic bursts. Central cells showed more pronounced bursts than edge cells. On average, central cells required stronger currents to evoke spiking than edge cells when stimulated from −84 mV steady-state potentials (q) (see Supplementary Information 1 for more threshold information). However, the initial spike frequencies for central cells were much higher than those for edge cells due to their bursty nature (q). The frequencies by the ends of the trains were nearly identical for central and edge cells (q). Central cells had stronger spike frequency adaptation than edge cells (r), largely due to stronger initial bursting in central cells (unpaired two-tailed t-test, P < 0.0001; for q, r: central cells n = 31, edge cells n = 42). For the physiological measurements in this figure, central and edge cell recordings were interleaved within slices (drawn from at least 8 mice per measured property). Data are mean ± s.e.m. See Supplementary Information 1 for additional intrinsic physiological properties and statistical comparisons.
Extended Data Fig. 8 Inhibitory output from the TRN: SOM-expressing TRN edge cells project to the POM, whereas CB cells project to the VPM.
a, Fluorescence (left) and DIC (middle) images from a live 300-μm slice following ChR2–eYFP expression in CB-Cre-positive TRN cells. Fluorescent axons of these TRN CB cells target the VPM but not the POM. White and red circles show the locations of recorded VPM and POM neurons, respectively. Right, strong IPSCs were evoked in the VPM neurons (black) whereas almost no IPSCs were evoked in the POM neurons (red) by optical activation of CB-Cre TRN cells (each trace is the response for one cell, averaged over 10 sweeps; Vhold −34 mV). Arrows indicate onsets of LED flashes (1 ms duration). Glutamate receptors were blocked by applying APV (50 μM) and DNQX (20 μM). The experiment was replicated in 4 slices from 3 CB-Cre mice. b, Same as a except ChR2–eYFP expression was in SOM-Cre TRN edge cells. Notice both the strong axonal targeting and IPSCs in the POM, rather than in the VPM. The experiment was replicated in 6 slices from 5 SOM-Cre mice. c, Left, group plot comparing fluorescence projection intensities to the VPM and the POM from the two TRN cell types. Fluorescence was normalized to the maximum level across the thalamic relay nuclei, with zero values set to the minimum level in the slice (n = 6 slices from 5 SOM-Cre mice; n = 4 slices from 3 CB-Cre mice; dashed lines represent a second slice for one mouse). Projections of CB-Cre TRN cells were stronger in the VPM than in the POM, whereas projections of SOM-Cre TRN cells were stronger in the POM than in the VPM (all P < 0.0001, two-tailed Bonferroni’s t-tests). Right, strengths of IPSCs recorded in the VPM and the POM reflected their anatomical inputs from the TRN. IPSCs evoked by photostimulation of CB-Cre TRN central cells (black) were large in the VPM (n = 15, 3 mice) and very weak in the POM (n = 16, 3 mice) (two-tailed Bonferroni’s t- test, P < 0.0001). Conversely, IPSCs evoked by stimulation of SOM-Cre edge cells (red) were strong in the POM (n = 12 cells, 4 mice) and undetectable in the VPM (n = 15 cells, 4 mice) (two-tailed Bonferroni’s t- test, P < 0.0001). Open circles show response areas for individual cells. Closed circles show averages per mouse. Comparisons were made using matched LED power. Data are mean ± s.e.m.
Extended Data Fig. 9 Visual TRN and somatosensory TRN have similar centre and edge subcircuits.
a, Representative images showing dLGN and LP injection sites and anterograde projections to the TRN. dLGN projects to the central zone whereas LP projects to the edges of the visual sector of the TRN. This experiment was replicated in 5 mice with dLGN viral injections, and in 13 mice with LP viral injections. b, Two representative examples (left and middle panels) of synaptic responses from cells across the medial-lateral axis of visual TRN evoked by photostimulation of dLGN. Cells in the centre of visual TRN received strong input from dLGN, whereas inputs to cells located near the edges of TRN were very weak or undetectable (average of 10 sweeps, −84 mV). Right, relationship between soma location in TRN and synaptic response amplitude (n = 19 cells, 6 slice preparations, each preparation represented by a different symbol shape; means for the 3 zones are represented by filled symbols and connected by lines), showing that cells in the centre of visual TRN respond strongly to dLGN input and those in the medial and lateral edges have minimal dLGN inputs (as in Fig. 2, and Extended Data Fig. 4). c, Same as b except characterizing synaptic inputs to TRN from LP. Cells along the medial edge (and in some cases along the lateral edge) of visual TRN received strong input from LP. By contrast, cells located in the centre of visual TRN generally had weak or no responses to LP input (average of 10 sweeps, −84 mV; n = 49 cells,12 preps). d, Dynamic properties of visual thalamoreticular responses. To study short-term synaptic depression, photostimulation intensities were chosen to evoke peak responses of around 2.5 nA (average LED powers: 20.8 mW for LP, 6.6 mW for dLGN). EPSC areas were measured over the first 10 ms of each response. Short-term depression was more pronounced for the central cells than the edge cells (P < 0.0001, ANOVA, stimulus 2–10). Depression in the two edge cell groups did not differ (two-tailed Bonferroni’s t-test, all P > 0.99). Depression of dLGN-evoked responses in central cells was stronger than depression of LP-evoked responses in medial or lateral cells (two-tailed Bonferroni’s t-test; central vs medial, central vs lateral, both P < 0.0001; central group: 7 cells, 5 mice; medial group: 17 cells, 11 mice; lateral group: 7 cells, 7 mice). Analogous to subcircuits of the somatosensory TRN, dLGN-evoked EPSCs of central cells in the visual TRN were less prolonged than LP-evoked EPSCs of edge cells (not shown). For example, the ratio of the EPSC areas to the peak EPSC amplitudes (as in Fig. 3b) varied as a function of cell group (ANOVA, P < 0.009; central cells ratio = 2.74 ± 0.24; medial cells ratio 5.27 ± 0.49; lateral cells ratio 5.14 ± 0.60, same cells as for short-term depression analysis, above). The two edge groups did not differ (two-tailed Bonferroni’s t-test, P = 0.99). Central cell ratios were significantly lower than medial and lateral cell ratios (two-tailed Bonferroni’s t-test, P < 0.009, <0.05, respectively). e, Input resistances of medial edge (n = 16 cells, 14 mice), central (n = 14 cells, 13 mice), and lateral edge cells (n = 15 cells, 13 mice) varied as a function of cell group (P < 0.03, ANOVA). The two edge groups were not different from one another (two-tailed Bonferoni’s t-test, P = 0.99). Central cell input resistance was significantly different from medial, although not from lateral (two-tailed Bonferroni’s t-tests, P < 0.04 and P = 0.074, respectively). f, Central cells of visual TRN had more robust intrinsic bursting than those along the edges. Left, representative stimulus-offset bursts. Right, average number of spikes during bursts was 5.8 ± 0.76 for central cells (n = 19 cells, 15 mice), 0.8 ± 0.30 for medial cells (n = 19 cells, 14 mice), and 1.1 ± 0.50 for lateral cells (n = 14 cells, 12 mice) (P < 0.0001, ANOVA). The two edge groups were not different from one another (two-tailed Bonferoni’s t-test, P = 0.99), but the edge cells discharged far fewer spikes per burst than the central cells (two-tailed Bonferoni’s t-test, P < 0.0001 for both medial vs central cells and lateral vs central cells). For the experiments represented in this figure, the boundaries of the TRN were determined using either tdTomato expression driven by SOM-Cre or PV-Cre, IHC for PV, or brightfield images. Data are mean ± s.e.m.
Extended Data Fig. 10 Organizational and operational scheme for somatosensory circuits of the TRN.
a, Schematic illustrating the organization of the somatosensory sector of the TRN. Excitatory inputs from the primary VP thalamic nucleus innervate the CB-dense central zone of TRN (green cells), whereas higher-order POM inputs innervate the SOM-dense edges (red cells). The inhibitory projections of TRN cells to the VP and POM seem to reciprocate the patterns of their inputs (coloured axons in the schematic). The spatial organization of the neurochemically distinct TRN cell types, and the stark segregation of their primary and higher order connections, suggest a core-shell organization for somatosensory circuits of the TRN. b, Distinct responses of central and edge TRN cells to their thalamic inputs. The spiking responses of central and edge cells are determined by the dynamic synaptic properties of their respective thalamic inputs combined with the intrinsic physiological properties of the TRN cells themselves. The spiking of central cells evoked by VP input is phasic, with strong initial bursts that deeply depress with repetitive stimulation; this is consistent with a core circuit that processes transient sensory signals. By contrast, the spiking responses of edge cells to POM input are more sustained, with initially weaker but more persistent activation; this shell-like circuit is suited to processing temporally distributed higher-order signals.
Supplementary information
Supplementary Information
Supplementary Information Table 1 | Intrinsic physiological properties of central and edge TRN cells. Passive membrane properties, action potential (AP) kinetics, and repetitive firing properties are shown for central and edge neurons of somatosensory TRN (page 1) and visual TRN (page 2). Properties were measured in current-clamp mode, with steady-state Vm set to either -84 mV (“burst mode”, left) or -74 mV (“tonic mode”, right). “Offset bursts” are intrinsic bursts triggered by the offset of hyperpolarizing current pulses, as illustrated in Fig. 3d and Extended Data Fig. 9f. The numbers of cells are shown in parenthesis. T-tests were unpaired and two-tailed. Data are expressed as mean ± SEM.
Rights and permissions
About this article
Cite this article
Martinez-Garcia, R.I., Voelcker, B., Zaltsman, J.B. et al. Two dynamically distinct circuits drive inhibition in the sensory thalamus. Nature 583, 813–818 (2020). https://doi.org/10.1038/s41586-020-2512-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2512-5
- Springer Nature Limited
This article is cited by
-
Development of reciprocal connections between the dorsal lateral geniculate nucleus and the thalamic reticular nucleus
Neural Development (2024)
-
Convergent direct and indirect cortical streams shape avoidance decisions in mice via the midline thalamus
Nature Communications (2024)
-
Pallidal GABA B receptors: involvement in cortex beta dynamics and thalamic reticular nucleus activity
The Journal of Physiological Sciences (2023)
-
Thalamocortical loops as temporal demodulators across senses
Communications Biology (2023)
-
The endocannabinoid N-arachidonoyl dopamine is critical for hyperalgesia induced by chronic sleep disruption
Nature Communications (2023)