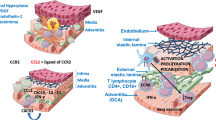
Abstract
Giant cell arteritis (GCA) and polymyalgia rheumatica (PMR) are closely related conditions characterized by systemic inflammation, a predominant IL-6 signature, an excellent response to glucocorticoids, a tendency to a chronic and relapsing course, and older age of the affected population. This Review highlights the emerging view that these diseases should be approached as linked conditions, unified under the term GCA–PMR spectrum disease (GPSD). In addition, GCA and PMR should be seen as non-monolithic conditions, with different risks of developing acute ischaemic complications and chronic vascular and tissue damage, different responses to available therapies and disparate relapse rates. A comprehensive stratification strategy for GPSD, guided by clinical findings, imaging and laboratory data, facilitates appropriate therapy and cost-effective use of health-economic resources. Patients presenting with predominant cranial symptoms and vascular involvement, who usually have a borderline elevation of inflammatory markers, are at an increased risk of sight loss in early disease but have fewer relapses in the long term, whereas the opposite is observed in patients with predominant large-vessel vasculitis. How the involvement of peripheral joint structures affects disease outcomes remains uncertain and understudied. In the future, all cases of new-onset GPSD should undergo early disease stratification, with their management adapted accordingly.
Key points
-
Giant cell arteritis (GCA) and polymyalgia rheumatica (PMR) are part of the same disease spectrum, which can be unified under the term GCA–PMR spectrum disease (GPSD).
-
Management of GPSD is characterized by a paradoxical contrast between short-term steroid responsiveness and long-term damage related to both the disease itself and its treatment.
-
Stratifying patients with GPSD according to clinical, imaging and laboratory features might help clinicians in personalizing the treatment to optimize the balance between effectiveness and safety.
-
A strong inflammatory activation, as indicated by high levels of classic markers but also of alternative biomarkers such as YKL-40 and osteopontin, predicts an increased risk of disease relapse.
-
Large-vessel involvement, peripheral arthritis and synovial hypertrophy in the shoulders are associated with a worse long-term outcome.
-
There is a need for prospective inception cohort studies of GPSD mapping of anatomical sites of inflammation and evolution during disease course to long-term critical outcomes.


Similar content being viewed by others
References
Buttgereit, F., Matteson, E. L. & Dejaco, C. Polymyalgia rheumatica and giant cell arteritis. J. Am. Med. Assoc. 324, 993–994 (2020).
van der Geest, K. S. M., Sandovici, M., Brouwer, E. & Mackie, S. L. Diagnostic accuracy of symptoms, physical signs, and laboratory tests for giant cell arteritis: a systematic review and meta-analysis. JAMA Intern. Med. 180, 1295–1304 (2020).
Salvarani, C., Cantini, F., Boiardi, L. & Hunder, G. G. Polymyalgia rheumatica and giant-cell arteritis. N. Engl. J. Med. 347, 261–271 (2002).
Dejaco, C. et al. Treat-to-target recommendations in giant cell arteritis and polymyalgia rheumatica. Ann. Rheum. Dis. https://doi.org/10.1136/ard-2022-223429 (2023).
Matteson, E. L., Buttgereit, F., Dejaco, C. & Dasgupta, B. Glucocorticoids for management of polymyalgia rheumatica and giant cell arteritis. Rheum. Dis. Clin. North. Am. 42, 75–90 (2016).
Hellmich, B. et al. 2018 Update of the EULAR recommendations for the management of large vessel vasculitis. Ann. Rheum. Dis. 79, 19–30 (2020).
Dejaco, C. et al. 2015 recommendations for the management of polymyalgia rheumatica: a European League Against Rheumatism/American College of Rheumatology collaborative initiative. Ann. Rheum. Dis. 74, 1799–1807 (2015).
Bond, M., Tomelleri, A., Buttgereit, F., Matteson, E. L. & Dejaco, C. Looking ahead: giant-cell arteritis in 10 years time. Ther. Adv. Musculoskelet. Dis. 14, 1759720X221096366 (2022).
Sebastian, A., Tomelleri, A. & Dasgupta, B. Current and innovative therapeutic strategies for the treatment of giant cell arteritis. Expert Opin. Orphan Drugs 9, 161–173 (2021).
Watts, R. A., Hatemi, G., Burns, J. C. & Mohammad, A. J. Global epidemiology of vasculitis. Nat. Rev. Rheumatol. 18, 22–34 (2022).
Li, K. J., Semenov, D., Turk, M. & Pope, J. A meta-analysis of the epidemiology of giant cell arteritis across time and space. Arthritis Res. Ther. 23, 82 (2021).
Mohammad, A. J., Nilsson, J. Å., Jacobsson, L. T. H., Merkel, P. A. & Turesson, C. Incidence and mortality rates of biopsy-proven giant cell arteritis in southern Sweden. Ann. Rheum. Dis. 74, 993–997 (2015).
Sharma A., Mohammad A., Turesson C. Incidence and prevalence of giant cell arteritis and polymyalgia rheumatica: a systematic literature review. Semin. Arthritis Rheum. 50, 1040–1048 (2020).
Hunder, G. G. The early history of giant cell arteritis and polymyalgia rheumatica: first descriptions to 1970. Mayo Clin. Proc. 81, 1071–1083 (2006).
Koster, M. J., Matteson, E. L. & Warrington, K. J. Large-vessel giant cell arteritis: diagnosis, monitoring and management. Rheumatology 57, ii32–ii42 (2018).
Dasgupta, B. et al. 2012 provisional classification criteria for polymyalgia rheumatica: a European League Against Rheumatism/American College of Rheumatology collaborative initiative. Ann. Rheum. Dis. 71, 484–492 (2012).
Kermani, T. A. & Warrington, K. J. Polymyalgia rheumatica. Lancet 381, 63–72 (2013).
Koster, M. J. et al. Giant cell arteritis and its mimics: a comparison of three patient cohorts. Semin. Arthritis Rheum. 50, 923–929 (2020).
van der Geest, K. S. M. et al. Comparison and validation of FDG-PET/CT scores for polymyalgia rheumatica. Rheumatology 61, 1072–1082 (2022).
Henckaerts, L., Gheysens, O., Vanderschueren, S., Goffin, K. & Blockmans, D. Use of 18F-fluorodeoxyglucose positron emission tomography in the diagnosis of polymyalgia rheumatica — a prospective study of 99 patients. Rheumatology 57, 1908–1916 (2018).
Schmidt, W. A. & Gromnica-Ihle, E. Incidence of temporal arteritis in patients with polymyalgia rheumatica: a prospective study using colour Doppler ultrasonography of the temporal arteries. Rheumatology 41, 46–52 (2002).
Alba, M. A. et al. Relapses in patients with giant cell arteritis: prevalence, characteristics, and associated clinical findings in a longitudinally followed cohort of 106 patients. Medicine 93, 194–201 (2014).
Hemmig, A. K. et al. Subclinical giant cell arteritis in new onset polymyalgia rheumatica: a systematic review and meta-analysis of individual patient data. Semin. Arthritis Rheum. 55, 152017 (2022).
Carmona, F. D. et al. A large-scale genetic analysis reveals a strong contribution of the HLA Class II region to giant cell arteritis susceptibility. Am. J. Hum. Genet. 96, 565–580 (2015).
Mackie, S. L. et al. Association of HLA-DRB1 amino acid residues with giant cell arteritis: genetic association study, meta-analysis and geo-epidemiological investigation. Arthritis Res. Ther. 17, 1–14 (2015).
Weyand, C. M., Hunder, N. N. H., Hicok, K. C., Hunder, G. G. & Goronzy, J. J. HLA-DRB1 alleles in polymyalgia rheumatica, giant cell arteritis, and rheumatoid arthritis. Arthritis Rheum. 37, 514–520 (1994).
Carmona, F. D., González-Gay, M. A. & Martín, J. Genetic component of giant cell arteritis. Rheumatology 53, 6–18 (2014).
González-Gay, M. A., Amoli, M. M., Garcia-Porrua, C. & Ollier, W. E. R. Genetic markers of disease susceptibility and severity in giant cell arteritis and polymyalgia rheumatica. Semin. Arthritis Rheum. 33, 38–48 (2003).
Ma-Krupa, W. et al. Activation of arterial wall dendritic cells and breakdown of self-tolerance in giant cell arteritis. J. Exp. Med. 199, 173–183 (2004).
Deng, J. et al. Toll-like receptors 4 and 5 induce distinct types of vasculitis. Circ. Res. 104, 488–495 (2009).
Pryshchep, O., Ma-Krupa, W., Younge, B. R., Goronzy, J. J. & Weyand, C. M. Vessel-specific Toll-like receptor profiles in human medium and large arteries. Circulation 118, 1276–1284 (2008).
Knab, K., Chambers, D. & Krönke, G. Synovial macrophage and fibroblast heterogeneity in joint homeostasis and inflammation. Front. Med. 9, 930 (2022).
Akbar, M. et al. Single cell and spatial transcriptomics in human tendon disease indicate dysregulated immune homeostasis. Ann. Rheum. Dis. 80, 1494–1497 (2021).
Mackie, S. L., Owen, C. E., Buchanan, R. R. C. & McGonagle, D. A shared basis for overlapping immunopathologies in giant cell arteritis and polymyalgia rheumatica. Lancet Rheumatol. 3, e826–e829 (2021).
van Sleen, Y. et al. Leukocyte dynamics reveal a persistent myeloid dominance in giant cell arteritis and polymyalgia rheumatica. Front. Immunol. 10, 1981 (2019).
Samson, M. et al. Th1 and Th17 lymphocytes expressing CD161 are implicated in giant cell arteritis and polymyalgia rheumatica pathogenesis. Arthritis Rheum. 64, 3788–3798 (2012).
Samson, M. et al. Involvement and prognosis value of CD8+ T cells in giant cell arteritis. J. Autoimmun. 72, 73–83 (2016).
Reitsema, R. D. et al. Contribution of pathogenic T helper 1 and 17 cells to bursitis and tenosynovitis in polymyalgia rheumatica. Front. Immunol. 13, 1–11 (2022).
Terrier, B. et al. Interleukin-21 modulates Th1 and Th17 responses in giant cell arteritis. Arthritis Rheum. 64, 2001–2011 (2012).
Deng, J., Younge, B. R., Olshen, R. A., Goronzy, J. J. & Weyand, C. M. Th17 and Th1 T-cell responses in giant cell arteritis. Circulation 121, 906–915 (2010).
Van der Geest, K. S. M. et al. Serum markers associated with disease activity in giant cell arteritis and polymyalgia rheumatica. Rheumatology 54, 1397–1402 (2015).
Dasgupta, B. & Panayi, G. S. Interleukin-6 in serum of patients with polymyalgia rheumatica and giant cell arteritis. Rheumatology 29, 456–458 (1990).
Stone, J. H. et al. Trial of tocilizumab in giant-cell arteritis. N. Engl. J. Med. 377, 317–328 (2017).
Bonelli, M. et al. Tocilizumab in patients with new onset polymyalgia rheumatica (PMR-SPARE): a phase 2/3 randomised controlled trial. Ann. Rheum. Dis. 81, 838–844 (2022).
Devauchelle-Pensec, V. et al. Effect of tocilizumab on disease activity in patients with active polymyalgia rheumatica receiving glucocorticoid therapy: a randomized clinical trial. J. Am. Med. Assoc. 328, 1053–1062 (2022).
Jiemy, W. F. et al. Distinct macrophage phenotypes skewed by local granulocyte macrophage colony-stimulating factor (GM-CSF) and macrophage colony-stimulating factor (M-CSF) are associated with tissue destruction and intimal hyperplasia in giant cell arteritis. Clin. Transl. Immunol. 9, e1164 (2020).
van Sleen, Y. et al. A distinct macrophage subset mediating tissue destruction and neovascularization in giant cell arteritis: implication of the YKL-40/interleukin-13 receptor α2 axis. Arthritis Rheumatol. 73, 2327–2337 (2021).
Meliconi, R. et al. Leukocyte infiltration in synovial tissue from the shoulder of patients with polymyalgia rheumatica. Quantitative analysis and influence of corticosteroid treatment. Arthritis Rheum. 39, 1199–1207 (1996).
Jiemy, W. F. et al. Expression of interleukin-6 in synovial tissue of patients with polymyalgia rheumatica. Ann. Rheum. Dis. 82, 440–442 (2022).
Corbera-Bellalta, M. et al. Blocking GM-CSF receptor α with mavrilimumab reduces infiltrating cells, pro-inflammatory markers and neoangiogenesis in ex vivo cultured arteries from patients with giant cell arteritis. Ann. Rheum. Dis. 81, 524–536 (2022).
Jiemy, W. F. et al. Proinflammatory monocytes and macrophages in synovial fluid and bursal tissue of patients with polymyalgia rheumatica: potent producers of IL-6 and GM-CSF. Ann. Rheum. Dis. 81, 8–9 (2022).
Szekanecz, Z. et al. Autoinflammation and autoimmunity across rheumatic and musculoskeletal diseases. Nat. Rev. Rheumatol. 17, 585–595 (2021).
Maz, M. et al. 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Giant Cell Arteritis and Takayasu Arteritis. Arthritis Care Res. 73, 1071–1087 (2021).
Langford, C. A. et al. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of giant cell arteritis. Arthritis Rheumatol. 69, 837–845 (2017).
Cid, M. C. et al. Efficacy and safety of mavrilimumab in giant cell arteritis: a phase 2, randomised, double-blind, placebo-controlled trial. Ann. Rheum. Dis. 81, 653–661 (2022).
Venhoff, N. et al. Efficacy and safety of secukinumab in patients with giant cell arteritis: study protocol for a randomized, parallel group, double-blind, placebo-controlled phase II trial. Trials 22, 543 (2021).
Hoffman, G. S. et al. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum. 46, 1309–1318 (2002).
Spiera, R. F. et al. A prospective, double-blind, randomized, placebo-controlled trial of methotrexate in the treatment of giant cell arteritis (GCA). Clin. Exp. Rheumatol. 19, 495–501 (2001).
Mahr, A. D. et al. Adjunctive methotrexate for treatment of giant cell arteritis: an individual patient data meta-analysis. Arthritis Rheum. 56, 2789–2797 (2007).
Tomelleri, A. et al. Long-term efficacy and safety of leflunomide in large-vessel giant-cell arteritis: a single centre ten-year experience. J. Clin. Rheumatol. 28, e297–e300 (2022).
Hočevar, A., Ješe, R., Rotar, Ž. & Tomšič, M. Does leflunomide have a role in giant cell arteritis? An open-label study. Clin. Rheumatol. 38, 291–296 (2019).
Das, S. et al. Efficacy of leflunomide as a steroid-sparing agent in treatment of Indian giant cell arteritis patients: a 2-year follow-up study. Int. J. Rheum. Dis. 25, 650–658 (2022).
Coath, F. et al. Giant cell arteritis: new concepts, treatments and the unmet need that remains. Rheumatology 58, 1123–1125 (2019).
Higashida-Konishi, M. et al. Giant cell arteritis successfully treated with subcutaneous tocilizumab monotherapy. Rheumatol. Int. 43, 545–549 (2023).
Saito, S. et al. Tocilizumab monotherapy for large vessel vasculitis: results of 104-week treatment of a prospective, single-centre, open study. Rheumatology 59, 1617–1621 (2020).
Jogimahanti, A. V., Kini, A. T., Irwin, L. E. & Lee, A. G. The cost-effectiveness of tocilizumab (Actemra) therapy in giant cell arteritis. J. Neuroophthalmol. 41, 342–350 (2021).
Stone, J. H. et al. Long-term effect of tocilizumab in patients with giant cell arteritis: open-label extension phase of the giant cell arteritis actemra (GiACTA) trial. Lancet Rheumatol. 3, e328–e336 (2021).
Tomelleri, A. et al. Effectiveness of a two-year tapered course of tocilizumab in patients with giant cell arteritis: a single-centre prospective study. Semin. Arthritis Rheum. 59, 152174 (2023).
Tomelleri, A. et al. Disease stratification in giant cell arteritis to reduce relapses and prevent long-term vascular damage. Lancet Rheumatol. 3, e886–e895 (2021).
Caporali, R. et al. Prednisone plus methotrexate for polymyalgia rheumatica: a randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 141, 493–500 (2004).
Van Der Veen, M. J., Dinant, H. J., Van Booma-Frankfort, C., Van Albada-Kuipers, G. A. & Bijlsma, J. W. J. Can methotrexate be used as a steroid sparing agent in the treatment of polymyalgia rheumatica and giant cell arteritis? Ann. Rheum. Dis. 55, 218 (1996).
Nazarinia, A. M., Moghimi, J. & Toussi, J. Efficacy of methotrexate in patients with polymyalgia rheumatica. Koomesh 14, 265–270 (2013).
Ferraccioli, G., Salaffi, F., De Vita, S., Casatta, L. & Bartoli, E. Methotrexate in polymyalgia rheumatica: preliminary results of an open, randomized study. J. Rheumatol. 23, 624–628 (1996).
Adizie, T., Christidis, D., Dharmapaliah, C., Borg, F. & Dasgupta, B. Efficacy and tolerability of leflunomide in difficult-to-treat polymyalgia rheumatica and giant cell arteritis: a case series. Int. J. Clin. Pract. 66, 906–909 (2012).
Spiera, R. et al. Sarilumab in patients with relapsing polymyalgia rheumatica: a phase 3, multicenter, randomized, double blind, placebo controlled trial (SAPHYR) [abstract]. Arthritis Rheumatol. 74, 1676 (2022).
Marsman, D. E., den Broeder, N., van den Hoogen, F. H. J., den Broeder, A. A. & van der Maas, A. Efficacy of rituximab in patients with polymyalgia rheumatica: a double-blind, randomised, placebo-controlled, proof-of-concept trial. Lancet Rheumatol. 3, e758–e766 (2021).
Dejaco, C., Duftner, C., Buttgereit, F., Matteson, E. L. & Dasgupta, B. The spectrum of giant cell arteritis and polymyalgia rheumatica: revisiting the concept of the disease. Rheumatology 56, 506–515 (2017).
Schmidt, W. A. Ultrasound in the diagnosis and management of giant cell arteritis. Rheumatology 57, ii22–ii31 (2018).
Biousse, V. & Newman, N. J. Ischemic optic neuropathies. N. Engl. J. Med. 372, 2428–2436 (2015).
Salvarani, C. et al. Risk factors for visual loss in an Italian population-based cohort of patients with giant cell arteritis. Arthritis Care Res. 53, 293–297 (2005).
Cid, M. C. et al. Association between strong inflammatory response and low risk of developing visual loss and other cranial ischemic complications in giant cell (temporal) arteritis. Arthritis Rheum. 41, 26–32 (1998).
van der Geest, K. S. M. et al. Ultrasonographic Halo Score in giant cell arteritis: association with intimal hyperplasia and ischaemic sight loss. Rheumatology 60, 4361–4366 (2021).
Makkuni, D. et al. Is intimal hyperplasia a marker of neuro-ophthalmic complications of giant cell arteritis? Rheumatology 47, 488–490 (2008).
Van Der Geest, K. S. M. M. et al. Novel ultrasonographic Halo Score for giant cell arteritis: assessment of diagnostic accuracy and association with ocular ischaemia. Ann. Rheum. Dis. 79, 393–399 (2019).
Vodopivec, I. & Rizzo, J. F. Ophthalmic manifestations of giant cell arteritis. Rheumatology 57, ii63–ii72 (2018).
Hayreh, S. S. Giant cell arteritis: its ophthalmic manifestations. Indian J. Ophthalmol. 69, 227 (2021).
Patil, P. et al. Fast track pathway reduces sight loss in giant cell arteritis: results of a longitudinal observational cohort study. Clin. Exp. Rheumatol. 33, S-103–S-106 (2015).
Gonzalez-Gay, M. A. et al. Strokes at time of disease diagnosis in a series of 287 patients with biopsy-proven giant cell arteritis. Medicine 88, 227–235 (2009).
De Boysson, H. et al. Giant cell arteritis-related stroke: a retrospective multicenter case-control study. J. Rheumatol. 44, 297–303 (2017).
Kermani, T. A. & Dasgupta, B. Current and emerging therapies in large-vessel vasculitis. Rheumatology 57, 1513–1524 (2018).
Hamrin, B., Jonsson, N. & Landberg, T. Involvement of large vessels in polymyalgia arteritica. Lancet 285, 1193–1196 (1965).
Espitia, O. et al. Comparison of idiopathic (isolated) aortitis and giant cell arteritis-related aortitis. A French retrospective multicenter study of 117 patients. Autoimmun. Rev. 15, 571–576 (2016).
Almeida-Morais, L. et al. Acute upper limb ischemia, a rare presentation of giant cell arteritis. Rev. Port. Cardiol. 35, 237.e1–237.e4 (2016).
Dejaco, C., Duftner, C., Dasgupta, B., Matteson, E. L. & Schirmer, M. Polymyalgia rheumatica and giant cell arteritis: management of two diseases of the elderly. Aging Health 7, 633–645 (2011).
Blockmans, D. et al. Repetitive 18F-fluorodeoxyglucose positron emission tomography in giant cell arteritis: a prospective study of 35 patients. Arthritis Rheum. 55, 131–137 (2006).
Muratore, F. et al. Large-vessel giant cell arteritis: a cohort study. Rheumatology 54, 463–470 (2015).
Monti, S. et al. Systematic literature review informing the 2018 update of the EULAR recommendation for the management of large vessel vasculitis: focus on giant cell arteritis. RMD Open 5, e001003 (2019).
Nesher, G., Nesher, R., Mates, M., Sonnenblick, M. & Breuer, G. S. Giant cell arteritis: intensity of the initial systemic inflammatory response and the course of the disease. Clin. Exp. Rheumatol. 26 (3 Supp 49), S30–S34 (2008).
Hutchings, A. et al. Clinical outcomes, quality of life, and diagnostic uncertainty in the first year of polymyalgia rheumatica. Arthritis Care Res. 57, 803–809 (2007).
Ramon, A. et al. The frequency of occult solid malignancy in patients with polymyalgia rheumatica-like symptoms. Ther. Adv. Musculoskelet. Dis. 13, 1759720X20984275 (2021).
Lavado-Pérez, C. et al. 18F-FDG PET/CT for the detection of large vessel vasculitis in patients with polymyalgia rheumatica. Rev. Esp. Med. Nucl. Imagen Mol. 34, 275–281 (2015).
Prieto-Peña, D. et al. Predictors of positive 18F-FDG PET/CT-scan for large vessel vasculitis in patients with persistent polymyalgia rheumatica. Semin. Arthritis Rheum. 48, 720–727 (2019).
Noval Menéndez, J., Serrano, M. T. C., Nuño Mateo, F. J. & Morís de la Tassa, J. Distal musculo-skeletal manifestations in rheumatic polymyalgia. Rev. Clin. Esp. 202, 385–387 (2002).
Pease, C. T. et al. Polymyalgia rheumatica can be distinguished from late onset rheumatoid arthritis at baseline: results of a 5-yr prospective study. Rheumatology 48, 123–127 (2009).
Cimmino, M. A., Parodi, M., Zampogna, G., Barbieri, F. & Garlaschi, G. Polymyalgia rheumatica is associated with extensor tendon tenosynovitis but not with synovitis of the hands: a magnetic resonance imaging study. Rheumatology 50, 494–499 (2011).
Karmacharya, P. et al. RS3PE revisited: a systematic review and meta-analysis of 331 cases. Clin. Exp. Rheumatol. 34, 404–415 (2016).
Owen, C. E. et al. Fusion of positron emission tomography/computed tomography with magnetic resonance imaging reveals hamstring peritendonitis in polymyalgia rheumatica. Rheumatology 57, 345–353 (2018).
Cimmino, M. A. et al. High frequency of capsular knee involvement in polymyalgia rheumatica/giant cell arteritis patients studied by positron emission tomography. Rheumatology 52, 1865–1872 (2013).
Salvarani, C. et al. Acute-phase reactants and the risk of relapse/recurrence in polymyalgia rheumatica: a prospective followup study. Arthritis Care Res. 53, 33–38 (2005).
Kimura, M. et al. Clinical characteristics of patients with remitting seronegative symmetrical synovitis with pitting edema compared to patients with pure polymyalgia rheumatica. J. Rheumatol. 39, 148–153 (2012).
Caporali, R. et al. Presenting features of polymyalgia rheumatica (PMR) and rheumatoid arthritis with PMR-like onset: a prospective study. Ann. Rheum. Dis. 60, 1021–1024 (2001).
Marzo-Ortega, H. et al. Evidence for a different anatomic basis for joint disease localization in polymyalgia rheumatica in comparison with rheumatoid arthritis. Arthritis Rheum. 56, 3496–3501 (2007).
Salvarani, C. et al. Distal musculoskeletal manifestations in polymyalgia rheumatica: a prospective follow-up study. Arthritis Rheum. 41, 1221–1226 (1998).
Hernández-Rodríguez, J. et al. A strong initial systemic inflammatory response is associated with higher corticosteroid requirements and longer duration of therapy in patients with giant-cell arteritis. Arthritis Rheum. 47, 29–35 (2002).
Burja, B. et al. Utility of serological biomarkers for giant cell arteritis in a large cohort of treatment-naïve patients. Clin. Rheumatol. 38, 317–329 (2019).
Hocevar, A. et al. Do early diagnosis and glucocorticoid treatment decrease the risk of permanent visual loss and early relapses in giant cell arteritis. Medicine 95, e3210 (2016).
Restuccia, G. et al. Long-term remission in biopsy proven giant cell arteritis: a retrospective cohort study. J. Autoimmun. 77, 39–44 (2017).
Bellan, M. et al. Role of positron emission tomography in the assessment of disease burden and risk of relapse in patients affected by giant cell arteritis. Clin. Rheumatol. 39, 1277–1281 (2020).
Martinez-Lado, L. et al. Relapses and recurrences in giant cell arteritis: a population-based study of patients with biopsy-proven disease from Northwestern Spain. Medicine 90, 186–193 (2011).
Prieto-González, S. et al. Serum osteopontin: a biomarker of disease activity and predictor of relapsing course in patients with giant cell arteritis. Potential clinical usefulness in tocilizumab-treated patients. RMD Open. 3, e000570 (2017).
Van Sleen, Y. et al. Markers of angiogenesis and macrophage products for predicting disease course and monitoring vascular inflammation in giant cell arteritis. Rheumatology 58, 1383–1392 (2019).
Nadkarni, S. et al. Investigational analysis reveals a potential role for neutrophils in giant-cell arteritis disease progression. Circ. Res. 114, 242–248 (2014).
Espígol-Frigolé, G. et al. Increased IL-17A expression in temporal artery lesions is a predictor of sustained response to glucocorticoid treatment in patients with giant-cell arteritis. Ann. Rheum. Dis. 72, 1481–1487 (2013).
Cid, M. C. et al. Association between increased CCL2 (MCP-1) expression in lesions and persistence of disease activity in giant-cell arteritis. Rheumatology 45, 1356–1363 (2006).
Hernández-Rodríguez, J. et al. Tissue production of pro-inflammatory cytokines (IL-1β, TNFα and IL-6) correlates with the intensity of the systemic inflammatory response and with corticosteroid requirements in giant-cell arteritis. Rheumatology 43, 294–301 (2004).
van der Geest, K. S. M. et al. What is the current evidence for disease subsets in giant cell arteritis? Arthritis Rheumatol. 70, 1366–1376 (2018).
Esen, I. et al. Plasma pyruvate kinase M2 as a marker of vascular inflammation in giant cell arteritis. Rheumatology 61, 3060–3070 (2022).
Muratore, F. et al. Aortic dilatation in patients with large vessel vasculitis: a longitudinal case control study using PET/CT. Semin. Arthritis Rheum. 48, 1074–1082 (2019).
Bilici Salman, R. et al. Diagnostic utility of serum biomarkers in large vessel vasculitis and their correlation with positron emission tomography. Mod. Rheumatol. 32, 938–945 (2022).
Rodriguez-Pla, A. et al. Evaluation of potential serum biomarkers of disease activity in diverse forms of vasculitis. J. Rheumatol. 47, 1001 (2020).
Nishimoto, N. et al. Mechanisms and pathologic significances in increase in serum interleukin-6 (IL-6) and soluble IL-6 receptor after administration of an anti-IL-6 receptor antibody, tocilizumab, in patients with rheumatoid arthritis and Castleman disease. Blood 112, 3959–3964 (2008).
Berger, C. T., Rebholz-Chaves, B., Recher, M., Manigold, T. & Daikeler, T. Serial IL-6 measurements in patients with tocilizumab-treated large-vessel vasculitis detect infections and may predict early relapses. Ann. Rheum. Dis. 78, 1012–1014 (2019).
Cantini, F. et al. Erythrocyte sedimentation rate and C-reactive protein in the evaluation of disease activity and severity in polymyalgia rheumatica: a prospective follow-up study. Semin. Arthritis Rheum. 30, 17–24 (2000).
Myklebust, G. & Gran, J. T. Prednisolone maintenance dose in relation to starting dose in the treatment of polymyalgia rheumatica and temporal arteritis. A prospective two-year study in 273 patients. Scand. J. Rheumatol. 30, 260–267 (2001).
Van Sleen, Y. et al. High angiopoietin-2 levels associate with arterial inflammation and long-term glucocorticoid requirement in polymyalgia rheumatica. Rheumatology 59, 176–184 (2020).
Owen, C. E. et al. Neutrophil to lymphocyte ratio predicts glucocorticoid resistance in polymyalgia rheumatica. Int. J. Rheum. Dis. 24, 56–62 (2021).
van Sleen, Y. et al. Angiopoietin-2/-1 ratios and MMP-3 levels as an early warning sign for the presence of giant cell arteritis in patients with polymyalgia rheumatica. Arthritis Res. Ther. 24, 65 (2022).
van der Geest, K. S. M. et al. Diagnostic value of 18FFDG-PET/CT in polymyalgia rheumatica: a systematic review and meta-analysis. Eur. J. Nucl. Med. Mol. Imaging 48, 1876–1889 (2021).
Dejaco, C. et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann. Rheum. Dis. 77, 636–643 (2018).
Sugihara, T. et al. Associated factors of poor treatment outcomes in patients with giant cell arteritis: clinical implication of large vessel lesions. Arthritis Res. Ther. 22, 1–12 (2020).
Blockmans, D. et al. Repetitive 18-fluorodeoxyglucose positron emission tomography in isolated polymyalgia rheumatica: a prospective study in 35 patients. Rheumatology 46, 672–677 (2007).
Czihal, M. et al. Impact of cranial and axillary/subclavian artery involvement by color duplex sonography on response to treatment in giant cell arteritis. J. Vasc. Surg. 61, 1285–1291 (2015).
De Boysson, H. et al. 18F-fluorodeoxyglucose positron emission tomography and the risk of subsequent aortic complications in giant-cell arteritis. Medicine 95, e3851 (2016).
de Boysson, H. et al. Different patterns and specific outcomes of large-vessel involvements in giant cell arteritis. J. Autoimmun. 103, 102283 (2019).
de Boysson, H. et al. Large-vessel involvement and aortic dilation in giant-cell arteritis. A multicenter study of 549 patients. Autoimmun. Rev. 17, 391–398 (2018).
Sammel, A. M. et al. Cranial and large vessel activity on positron emission tomography scan at diagnosis and 6 months in giant cell arteritis. Int. J. Rheum. Dis. 23, 582–588 (2020).
Grayson, P. C. et al. 18F-fluorodeoxyglucose–positron emission tomography as an imaging biomarker in a prospective, longitudinal cohort of patients with large vessel vasculitis. Arthritis Rheumatol. 70, 439–449 (2018).
Meller, J. et al. Early diagnosis and follow-up of aortitis with [18F]FDG PET and MRI. Eur. J. Nucl. Med. Mol. Imaging 30, 730–736 (2003).
Sebastian, A. et al. Efficacy and safety of tocilizumab in giant cell arteritis: a single centre NHS experience using imaging (ultrasound and PET-CT) as a diagnostic and monitoring tool. RMD Open 6, e001417 (2020).
Camellino, D., Duftner, C. & Dejaco, C. New insights into the role of imaging in polymyalgia rheumatica. Rheumatology 60, 1016–1033 (2021).
Salvarani, C. et al. Cervical interspinous bursitis in active polymyalgia rheumatica. Ann. Rheum. Dis. 67, 758–761 (2008).
Rehak, Z. et al. PET/CT imaging in polymyalgia rheumatica: praepubic 18F-FDG uptake correlates with pectineus and adductor longus muscles enthesitis and with tenosynovitis. Radiol. Oncol. 51, 8–14 (2017).
Laporte, J.-P. et al. Localized myofascial inflammation revealed by magnetic resonance imaging in recent-onset polymyalgia rheumatica and effect of tocilizumab therapy. J. Rheumatol. 46, 1619–1626 (2019).
Huwart, A. et al. Ultrasonography and magnetic resonance imaging changes in patients with polymyalgia rheumatica treated by tocilizumab. Arthritis Res. Ther. 20, 11 (2018).
Miceli, M. C. et al. Baseline shoulder ultrasonography is not a predictive marker of response to glucocorticoids in patients with polymyalgia rheumatica: a 12-month follow-up study. J. Rheumatol. 44, 241–247 (2017).
Ayano, M. et al. Shoulder ultrasound and serum lactate dehydrogenase predict inadequate response to glucocorticoid treatment in patients with polymyalgia rheumatica. Rheumatol. Int. 40, 1101–1109 (2020).
Macchioni, P. L., Catanoso, M. G., Pipitone, N., Boiardi, L. & Salvarani, C. Longitudinal examination with shoulder ultrasound of patients with polymyalgia rheumatica. Rheumatology 48, 1566–1569 (2009).
Nakamura, H. et al. Gadolinium-enhanced magnetic resonance imaging in shoulders contributes accurate diagnosis and predicting recurrence to patients with polymyalgia rheumatica. Clin. Exp. Rheumatol. 39, 84–90 (2021).
Mackie, S. L. et al. Whole-body MRI of patients with polymyalgia rheumatica identifies a distinct subset with complete patient-reported response to glucocorticoids. Ann. Rheum. Dis. 74, 2188–2192 (2015).
Giraud, N., Prati, C., Wendling, D. & Verhoeven, F. Prognostic value of 18F-fluorodeoxyglucose PET-CT score at baseline on the therapeutic response to prednisone in patients with polymyalgia rheumatica. Joint Bone Spine 88, 2020–2021 (2021).
van der Geest, K. S. M. et al. Positron emission tomography imaging in vasculitis. Cardiol. Clin. 41, 251–265 (2023).
Bley, T. A. et al. Comparison of duplex sonography and high-resolution magnetic resonance imaging in the diagnosis of giant cell (temporal) arteritis. Arthritis Rheum. 58, 2574–2578 (2008).
Chrysidis, S., Lage-Hansen, P. R., Svendsen, N. & Diamantopoulos, A. P. The fast-track outpatient clinic significantly decreases hospitalisation rates among polymyalgia rheumatica patients. BMC Rheumatol. 5, 37 (2021).
Nguyen, J. T. et al. Economic and objective burden of caregiving on informal caregivers of patients with systemic vasculitis. Musculoskelet. Care 17, 282–287 (2019).
De Smit, E., Palmer, A. J. & Hewitt, A. W. Projected worldwide disease burden from giant cell arteritis by 2050. J. Rheumatol. 42, 119–125 (2015).
Diamantopoulos, A. P., Haugeberg, G., Lindland, A. & Myklebust, G. The fast-track ultrasound clinic for early diagnosis of giant cell arteritis significantly reduces permanent visual impairment: towards a more effective strategy to improve clinical outcome in giant cell arteritis? Rheumatology 55, 66–70 (2016).
Sarnes, E. et al. Incidence and US costs of corticosteroid-associated adverse events: a systematic literature review. Clin. Ther. 33, 1413–1432 (2011).
Robson, J. C. et al. The relative risk of aortic aneurysm in patients with giant cell arteritis compared with the general population of the UK. Ann. Rheum. Dis. 74, 129–135 (2015).
Stone, W. M. & Fankhauser, G. T. Inflammatory aneurysms treated with EVAR. Semin. Vasc. Surg. 25, 227–231 (2012).
Broder, M. S. et al. Corticosteroid-related adverse events in patients with giant cell arteritis: a claims-based analysis. Semin. Arthritis Rheum. 46, 246–252 (2016).
Petri, H., Nevitt, A., Sarsour, K., Napalkov, P. & Collinson, N. Incidence of giant cell arteritis and characteristics of patients: data-driven analysis of comorbidities. Arthritis Care Res. 67, 390–395 (2015).
Manson, S. C., Brown, R. E., Cerulli, A. & Vidaurre, C. F. The cumulative burden of oral corticosteroid side effects and the economic implications of steroid use. Respir. Med. 103, 975–994 (2009).
Van Staa, T. P., Leufkens, H. G. M., Abenhaim, L., Zhang, B. & Cooper, C. Oral corticosteroids and fracture risk: relationship to daily and cumulative doses. Rheumatology 39, 1383–1389 (2000).
Kanis, J. A., Stevenson, M., McCloskey, E. V., Davis, S. & Lloyd-Jones, M. Glucocorticoid-induced osteoporosis: a systematic review and cost-utility analysis. Health Technol. Assess. 11, 1–231 (2007).
Sebastian, A. et al. Halo score (temporal artery, its branches and axillary artery) as a diagnostic, prognostic and disease monitoring tool for Giant Cell Arteritis (GCA). BMC Rheumatol. 4, 35 (2020).
Author information
Authors and Affiliations
Contributions
B.D., A.T., K.S.M.v.d.G., M.A.K., A.S., F.C., D.B. and Y.v.S researched data for the article. B.D., A.T., K.S.M.v.d.G., M.A.K., A.S., F.C. and C.D. contributed substantially to discussion of the content. B.D., A.T., K.S.M.v.d.G., M.A.K., A.S., F.C. and Y.v.S. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
K.S.M.v.d.G. declares that he has received grants from the Mandema Stipend and the FOREUM Foundation for Research in Rheumatology and personal fees from Roche and AbbVie, outside the submitted work. B.D. declares that he has received consulting fees from Chugai, Roche and Sanofi and sponsorship grants for international meetings and workshops with AbbVie, GlaxoSmithKline, Roche and Sanofi. C.D. declares that has he has received consulting and/or speaker’s fees from AbbVie, Eli Lilly, Galapagos, Janssen, Novartis, Pfizer, Roche and Sanofi, all unrelated to this manuscript. All other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Rheumatology thanks C. Owen; C. Marvisi, who co-reviewed with C. Salvarani; C. Salvarani; and W. Schmidt for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tomelleri, A., van der Geest, K.S.M., Khurshid, M.A. et al. Disease stratification in GCA and PMR: state of the art and future perspectives. Nat Rev Rheumatol 19, 446–459 (2023). https://doi.org/10.1038/s41584-023-00976-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-023-00976-8
- Springer Nature Limited