Abstract
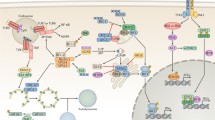
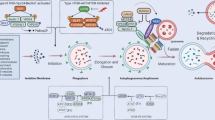
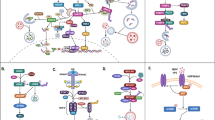
Autophagy is a constitutive cellular process of degradation required to maintain homeostasis and turn over spent organelles and aggregated proteins. For some viruses, the process can be antiviral, degrading viral proteins or virions themselves. For many other viruses, the induction of the autophagic process provides a benefit and promotes viral replication. In this Review, we survey the roles that the autophagic pathway plays in the replication of viruses. Most viruses that benefit from autophagic induction block autophagic degradation, which is a ‘bend, but don’t break’ strategy initiating but limiting a potentially antiviral response. In almost all cases, it is other effects of the redirected autophagic machinery that benefit these viruses. This rapid mechanism to generate small double-membraned vesicles can be usurped to shape membranes for viral genome replication and virion maturation. However, data suggest that autophagic maintenance of cellular homeostasis is crucial for the initiation of infection, as viruses have evolved to replicate in normal, healthy cells. Inhibition of autophagic degradation is important once infection has initiated. Although true degradative autophagy is probably a negative for most viruses, initiating nondegradative autophagic membranes benefits a wide variety of viruses.






Similar content being viewed by others
References
Yamaguchi, O., Murakawa, T., Nishida, K. & Otsu, K. Receptor-mediated mitophagy. J. Mol. Cell. Cardiol. 95, 50–56 (2016).
Parzych, K. R. & Klionsky, D. J. An overview of autophagy: morphology, mechanism, and regulation. Antioxid. Redox Signal. 20, 460–473 (2014).
Qin, Z.-H. Autophagy: Biology and Diseases: Basic Science (Springer Nature, 2019).
Bhutia, S. K. & Dhiman, R. Autophagy Processes and Mechanisms (Elsevier Science, 2023).
Rodolfo, C., Campello, S. & Cecconi, F. Mitophagy in neurodegenerative diseases. Neurochem. Int. 117, 156–166 (2018).
Cerri, S. & Blandini, F. Role of autophagy in Parkinson’s disease. Curr. Med. Chem. 26, 3702–3718 (2019).
Guo, F., Liu, X., Cai, H. & Le, W. Autophagy in neurodegenerative diseases: pathogenesis and therapy. Brain Pathol. 28, 3–13 (2018).
Henderson, P. & Stevens, C. The role of autophagy in Crohn’s disease. Cells 1, 492–519 (2012).
Ichimiya, T. et al. Autophagy and autophagy-related diseases: a review. Int. J. Mol. Sci. 21, 8974 (2020).
Yamamoto, H., Zhang, S. & Mizushima, N. Autophagy genes in biology and disease. Nat. Rev. Genet. 24, 382–400 (2023).
Puri, C., Vicinanza, M. & Rubinsztein, D. C. Phagophores evolve from recycling endosomes. Autophagy 14, 1475–1477 (2018).
Rubinsztein, D. C., Shpilka, T. & Elazar, Z. Mechanisms of autophagosome biogenesis. Curr. Biol. 22, R29–R34 (2012).
Johansen, T. & Lamark, T. Selective autophagy mediated by autophagic adapter proteins. Autophagy 7, 279–296 (2011).
Katsuragi, Y., Ichimura, Y. & Komatsu, M. p62/SQSTM1 functions as a signaling hub and an autophagy adaptor. FEBS J. 282, 4672–4678 (2015).
Lamark, T., Svenning, S. & Johansen, T. Regulation of selective autophagy: the p62/SQSTM1 paradigm. Essays Biochem. 61, 609–624 (2017).
Zhao, Y. G., Codogno, P. & Zhang, H. Machinery, regulation and pathophysiological implications of autophagosome maturation. Nat. Rev. Mol. Cell Biol. 22, 733–750 (2021).
Chen, Y. & Yu, L. Autophagic lysosome reformation. Exp. Cell Res. 319, 142–146 (2013).
Corona Velazquez, A. F. & Jackson, W. T. So many roads: the multi-faceted regulation of autophagy induction. Mol. Cell. Biol. 38, e00303-13 (2018).
Zachari, M. & Ganley, I. G. The mammalian ULK1 complex and autophagy initiation. Essays Biochem. 61, 585–596 (2017).
Wirth, M., Joachim, J. & Tooze, S. A. Autophagosome formation—the role of ULK1 and Beclin1–PI3KC3 complexes in setting the stage. Semin. Cancer Biol. 23, 301–309 (2013).
Menon, M. B. & Dhamija, S. Beclin 1 phosphorylation—at the center of autophagy regulation. Front. Cell Dev. Biol. 6, 137 (2018).
Maruyama, T. & Noda, N. N. Autophagy-regulating protease Atg4: structure, function, regulation and inhibition. J. Antibiot. 71, 72–78 (2017).
Tanida, I. Autophagy basics. Microbiol. Immunol. 55, 1–11 (2011).
Tanida, I., Ueno, T. & Kominami, E. LC3 conjugation system in mammalian autophagy. Int. J. Biochem. Cell Biol. 36, 2503–2518 (2004).
Nakatogawa, H. Two ubiquitin-like conjugation systems that mediate membrane formation during autophagy. Essays Biochem. 55, 39–50 (2013).
Lystad, A. H., Carlsson, S. R. & Simonsen, A. Toward the function of mammalian ATG12-ATG5-ATG16L1 complex in autophagy and related processes. Autophagy 15, 1485–1486 (2019).
Dooley, H. C. et al. WIPI2 links LC3 conjugation with PI3P, autophagosome formation, and pathogen clearance by recruiting Atg12–5-16L1. Mol. Cell 55, 238–252 (2014).
Sanchez-Wandelmer, J. & Reggiori, F. Amphisomes: out of the autophagosome shadow? EMBO J. 32, 3116–3118 (2013).
Shvarev, D. et al. Structure of the HOPS tethering complex, a lysosomal membrane fusion machinery. eLife 11, e80901 (2022).
Song, H., Orr, A. S., Lee, M., Harner, M. E. & Wickner, W. T. HOPS recognizes each SNARE, assembling ternary trans-complexes for rapid fusion upon engagement with the 4th SNARE. eLife 9, e53559 (2020).
McEwan, D. G. et al. PLEKHM1 regulates autophagosome-lysosome fusion through HOPS complex and LC3/GABARAP proteins. Mol. Cell 57, 39–54 (2015).
Ahmad, L., Mostowy, S. & Sancho-Shimizu, V. Autophagy-virus interplay: from cell biology to human disease. Front. Cell Dev. Biol. 6, 155 (2018).
Levine, B. Eating oneself and uninvited guests: autophagy-related pathways in cellular defense. Cell 120, 159–162 (2005).
Paludan, C. et al. Endogenous MHC class II processing of a viral nuclear antigen after autophagy. Science 307, 593–596 (2005).
Kirkegaard, K., Taylor, M. P. & Jackson, W. T. Cellular autophagy: surrender, avoidance and subversion by microorganisms. Nat. Rev. Microbiol. 2, 301–314 (2004).
Narendra, D. P. et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol. 8, e1000298 (2010).
Vives-Bauza, C. et al. PINK1-dependent recruitment of Parkin to mitochondria in mitophagy. Proc. Natl Acad. Sci. USA 107, 378–383 (2010).
Mochida, K. & Nakatogawa, H. ER-phagy: selective autophagy of the endoplasmic reticulum. EMBO Rep. 23, e55192 (2022).
Singh, R. et al. Autophagy regulates lipid metabolism. Nature 458, 1131–1135 (2009).
Ferrell, J. E. Jr & Ha, S. H. Ultrasensitivity part I: michaelian responses and zero-order ultrasensitivity. Trends Biochem. Sci. 39, 496–503 (2014).
Ferrell, J. E. Jr & Ha, S. H. Ultrasensitivity part II: multisite phosphorylation, stoichiometric inhibitors, and positive feedback. Trends Biochem. Sci. 39, 556–569 (2014).
Ferrell, J. E. Jr & Ha, S. H. Ultrasensitivity part III: cascades, bistable switches, and oscillators. Trends Biochem. Sci. 39, 612–618 (2014).
Ferrell, J. E. Jr Self-perpetuating states in signal transduction: positive feedback, double-negative feedback and bistability. Curr. Opin. Cell Biol. 14, 140–148 (2002).
Klionsky, D. J. et al. Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition). Autophagy 17, 1–382 (2021).
Schaaf, M. B. E., Keulers, T. G., Vooijs, M. A. & Rouschop, K. M. A. LC3/GABARAP family proteins: autophagy-(un)related functions. FASEB J. 30, 3961–3978 (2016).
Bampton, E. T. W., Goemans, C. G., Niranjan, D., Mizushima, N. & Tolkovsky, A. M. The dynamics of autophagy visualized in live cells: from autophagosome formation to fusion with endo/lysosomes. Autophagy 1, 23–36 (2005).
Dales, S., Eggers, H. J., Tamm, I. & Palade, G. E. Electron mcroscopic study of the formation of poliovirus. Virology 26, 379–389 (1965).
Smith, J. D. & de Harven, E. Herpes simplex virus and human cytomegalovirus replication in WI-38 cells. III. Cytochemical localization of lysosomal enzymes in infected cells. J. Virol. 26, 102–109 (1978).
Liang, X. H. et al. Protection against fatal Sindbis virus encephalitis by beclin, a novel Bcl-2-interacting protein. J. Virol. 72, 8586–8596 (1998).
Orvedahl, A. et al. HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe 1, 23–35 (2007).
Verpooten, D., Ma, Y., Hou, S., Yan, Z. & He, B. Control of TANK-binding kinase 1-mediated Signaling by the γ134.5 Protein of Herpes Simplex Virus 1. J. Biol. Chem. 284, 1097–1105 (2009).
Pilli, M. et al. TBK-1 promotes autophagy-mediated antimicrobial defense by controlling autophagosome maturation. Immunity 37, 223–234 (2012).
Alexander, D. E., Ward, S. L., Mizushima, N., Levine, B. & Leib, D. A. Analysis of the role of autophagy in replication of herpes simplex virus in cell culture. J. Virol. 81, 12128–12134 (2007).
Ames, J. et al. OPTN is a host intrinsic restriction factor against neuroinvasive HSV-1 infection. Nat. Commun. 12, 5401 (2021).
Orvedahl, A. et al. Autophagy protects against Sindbis virus infection of the central nervous system. Cell Host Microbe 7, 115–127 (2010).
Rubio, R. M. & Mohr, I. Inhibition of ULK1 and Beclin1 by an α-herpesvirus Akt-like Ser/Thr kinase limits autophagy to stimulate virus replication. Proc. Natl Acad. Sci. USA 116, 26941–26950 (2019).
Wang, R. C. et al. Akt-mediated regulation of autophagy and tumorigenesis through Beclin 1 phosphorylation. Science 338, 956–959 (2012).
Chaumorcel, M. et al. The human cytomegalovirus protein TRS1 inhibits autophagy via its interaction with Beclin 1. J. Virol. 86, 2571–2584 (2012).
Mouna, L. et al. Analysis of the role of autophagy inhibition by two complementary human cytomegalovirus BECN1/Beclin 1-binding proteins. Autophagy 12, 327–342 (2016).
Castro-Gonzalez, S. et al. HIV-1 Nef counteracts autophagy restriction by enhancing the association between BECN1 and its inhibitor BCL2 in a PRKN-dependent manner. Autophagy 17, 553–577 (2021).
Li, J. C. B. et al. HIV-1 trans-activator protein dysregulates IFN-γ signaling and contributes to the suppression of autophagy induction. AIDS 25, 15–25 (2011).
Campbell, G. R. & Spector, S. A. Hormonally active vitamin D3 (1α,25-Dihydroxycholecalciferol) triggers autophagy in human macrophages that inhibits HIV-1 infection. J. Biol. Chem. 286, 18890–18902 (2011).
Campbell, G. R. & Spector, S. A. Vitamin D inhibits human immunodeficiency virus type 1 and Mycobacterium tuberculosis infection in macrophages through the induction of autophagy. PLoS Pathog. 8, e1002689 (2012).
Campbell, G. R., Bruckman, R. S., Chu, Y.-L. & Spector, S. A. Autophagy induction by histone deacetylase inhibitors inhibits HIV type 1. J. Biol. Chem. 290, 5028–5040 (2015).
Hernaez, B. et al. A179L, a new viral Bcl2 homolog targeting Beclin 1 autophagy related protein. Curr. Mol. Med. 13, 305–316 (2013).
Zvereva, A. S. et al. Viral protein suppresses oxidative burst and salicylic acid-dependent autophagy and facilitates bacterial growth on virus-infected plants. N. Phytol. 211, 1020–1034 (2016).
Yang, M. et al. Barley stripe mosaic virus γb protein subverts autophagy to promote viral infection by disrupting the ATG7-ATG8 interaction. Plant. Cell 30, 1582–1595 (2018).
Corona, A. K., Saulsbery, H. M., Corona Velazquez, A. F. & Jackson, W. T. Enteroviruses remodel autophagic trafficking through regulation of host SNARE proteins to promote virus replication and cell exit. Cell Rep. 22, 3304–3314 (2018).
Mohamud, Y. et al. Enteroviral infection inhibits autophagic flux via disruption of the SNARE complex to enhance viral replication. Cell Rep. 22, 3292–3303 (2018).
Jassey, A. et al. Starvation after infection restricts enterovirus D68 replication. Autophagy 19, 112–125 (2022).
Ding, B. et al. Phosphoprotein of human parainfluenza virus type 3 blocks autophagosome-lysosome fusion to increase virus production. Cell Host Microbe 15, 564–577 (2014).
Lin, Y. et al. Hepatitis B virus is degraded by autophagosome-lysosome fusion mediated by Rab7 and related components. Protein Cell 10, 60–66 (2019).
Lin, Y. et al. Synaptosomal-associated protein 29 is required for the autophagic degradation of hepatitis B virus. FASEB J. 33, 6023–6034 (2019).
Zhong, L., Hu, J., Shu, W., Gao, B. & Xiong, S. Epigallocatechin-3-gallate opposes HBV-induced incomplete autophagy by enhancing lysosomal acidification, which is unfavorable for HBV replication. Cell Death Dis. 6, e1770 (2015).
Metz, P. et al. Dengue virus inhibition of autophagic flux and dependency of viral replication on proteasomal degradation of the autophagy receptor p62. J. Virol. 89, 8026–8041 (2015).
Miao, G. et al. ORF3a of the COVID-19 virus SARS-CoV-2 blocks HOPS complex-mediated assembly of the SNARE complex required for autolysosome formation. Dev. Cell 56, 427–442.e5 (2021).
Zhang, Y. et al. The SARS-CoV-2 protein ORF3a inhibits fusion of autophagosomes with lysosomes. Cell Discov. 7, 31 (2021).
Granato, M. et al. Epstein-barr virus blocks the autophagic flux and appropriates the autophagic machinery to enhance viral replication. J. Virol. 88, 12715–12726 (2014).
Gonnella, R. et al. BFRF1 protein is involved in EBV-mediated autophagy manipulation. Microbes Infect. 22, 585–591 (2020).
Gannagé, M. et al. Matrix protein 2 of influenza A virus blocks autophagosome fusion with lysosomes. Cell Host Microbe 6, 367–380 (2009).
Gladue, D. P. et al. Foot-and-mouth disease virus nonstructural protein 2C interacts with Beclin1, modulating virus replication. J. Virol. 86, 12080–12090 (2012).
Sir, D. et al. Induction of incomplete autophagic response by hepatitis C virus via the unfolded protein response. Hepatology 48, 1054–1061 (2008).
Huang, H. et al. Hepatitis C virus inhibits AKT-tuberous sclerosis complex (TSC), the mechanistic target of rapamycin (MTOR) pathway, through endoplasmic reticulum stress to induce autophagy. Autophagy 9, 175–195 (2013).
Ke, P.-Y. & Chen, S. S.-L. Activation of the unfolded protein response and autophagy after hepatitis C virus infection suppresses innate antiviral immunity in vitro. J. Clin. Invest. 121, 37–56 (2011).
Wang, L., Tian, Y. & Ou, J.-H. J. HCV induces the expression of Rubicon and UVRAG to temporally regulate the maturation of autophagosomes and viral replication. PLoS Pathog. 11, e1004764 (2015).
Aydin, Y. et al. Extracellular vesicle release promotes viral replication during persistent HCV infection. Cells 10, 984 (2021).
Jones-Jamtgaard, K. N., Wozniak, A. L., Koga, H., Ralston, R. & Weinman, S. A. Hepatitis C virus infection increases autophagosome stability by suppressing lysosomal fusion through an Arl8b-dependent mechanism. J. Biol. Chem. 294, 14257–14266 (2019).
Korolchuk, V. I. et al. Lysosomal positioning coordinates cellular nutrient responses. Nat. Cell Biol. 13, 453–460 (2011).
Kim, N. et al. Interferon-inducible protein SCOTIN interferes with HCV replication through the autolysosomal degradation of NS5A. Nat. Commun. 7, 10631 (2016).
Dales, S., Eggers, H. J., Tamm, I. & Palade, G. E. Electron microscopic study of the formation of poliovirus. Virology 26, 379–389 (1965).
Jackson, W. T. et al. Subversion of cellular autophagosomal machinery by RNA viruses. PLoS Biol. 3, e156 (2005).
Taylor, M. P. & Kirkegaard, K. Modification of cellular autophagy protein LC3 by poliovirus. J. Virol. 81, 12543–12553 (2007).
Corona Velazquez, A., Corona, A. K., Klein, K. A. & Jackson, W. T. Poliovirus induces autophagic signaling independent of the ULK1 complex. Autophagy 14, 1201–1213 (2018).
Bird, S. W., Maynard, N. D., Covert, M. W. & Kirkegaard, K. Nonlytic viral spread enhanced by autophagy components. Proc. Natl Acad. Sci. USA 111, 13081–13086 (2014).
Chen, Y.-H. et al. Phosphatidylserine vesicles enable efficient en bloc transmission of enteroviruses. Cell 160, 619–630 (2015).
Richards, A. L. & Jackson, W. T. Intracellular vesicle acidification promotes maturation of infectious poliovirus particles. PLoS Pathog. 8, e1003046 (2012).
Wen, W. et al. Selective autophagy receptor SQSTM1/ p62 inhibits Seneca Valley virus replication by targeting viral VP1 and VP3. Autophagy 17, 3763–3775 (2021).
Hou, L. et al. Seneca valley virus activates autophagy through the PERK and ATF6 UPR pathways. Virology 537, 254–263 (2019).
Klein, K. A. & Jackson, W. T. Human rhinovirus 2 induces the autophagic pathway and replicates more efficiently in autophagic cells. J. Virol. 85, 9651–9654 (2011).
Wong, J. et al. Autophagosome supports coxsackievirus B3 replication in host cells. J. Virol. 82, 9143–9153 (2008).
Alirezaei, M., Flynn, C. T., Wood, M. R. & Whitton, J. L. Pancreatic acinar cell-specific autophagy disruption reduces coxsackievirus replication and pathogenesis in vivo. Cell Host Microbe 11, 298–305 (2012).
Huang, S.-C., Chang, C.-L., Wang, P.-S., Tsai, Y. & Liu, H.-S. Enterovirus 71-induced autophagy detected in vitro and in vivo promotes viral replication. J. Med. Virol. 81, 1241–1252 (2009).
Fu, Y. et al. Enterovirus 71 induces autophagy by regulating has-miR-30a expression to promote viral replication. Antivir. Res. 124, 43–53 (2015).
Lee, Y.-R., Wang, P.-S., Wang, J.-R. & Liu, H.-S. Enterovirus 71-induced autophagy increases viral replication and pathogenesis in a suckling mouse model. J. Biomed. Sci. 21, 80 (2014).
Ferraris, P., Blanchard, E. & Roingeard, P. Ultrastructural and biochemical analyses of hepatitis C virus-associated host cell membranes. J. Gen. Virol. 91, 2230–2237 (2010).
Sir, D. et al. Replication of hepatitis C virus RNA on autophagosomal membranes. J. Biol. Chem. 287, 18036–18043 (2012).
Dreux, M., Gastaminza, P., Wieland, S. F. & Chisari, F. V. The autophagy machinery is required to initiate hepatitis C virus replication. Proc. Natl Acad. Sci. USA 106, 14046–14051 (2009).
Lee, Y.-R. et al. Autophagic machinery activated by dengue virus enhances virus replication. Virology 374, 240–248 (2008).
Panyasrivanit, M., Khakpoor, A., Wikan, N. & Smith, D. R. Co-localization of constituents of the dengue virus translation and replication machinery with amphisomes. J. Gen. Virol. 90, 448–456 (2009).
Khakpoor, A., Panyasrivanit, M., Wikan, N. & Smith, D. R. A role for autophagolysosomes in dengue virus 3 production in HepG2 cells. J. Gen. Virol. 90, 1093–1103 (2009).
Liang, Q. et al. Zika virus NS4A and NS4B proteins deregulate Akt-mTOR signaling in human fetal neural stem cells to inhibit neurogenesis and induce autophagy. Cell Stem Cell 19, 663–671 (2016).
Tiwari, S. K. et al. Zika virus depletes neural stem cells and evades selective autophagy by suppressing the Fanconi anemia protein FANCC. EMBO Rep. 21, e49183 (2020).
Cao, B., Parnell, L. A. & Diamond, M. S. Inhibition of autophagy limits vertical transmission of Zika virus in pregnant mice. J. Exp. Med. 214, 2303–2313 (2017).
Sumpter, R. Jr et al. Fanconi anemia proteins function in mitophagy and immunity. Cell 165, 867–881 (2016).
Orvedahl, A. et al. Image-based genome-wide siRNA screen identifies selective autophagy factors. Nature 480, 113–117 (2011).
Zhou, Z. et al. Autophagy is involved in influenza A virus replication. Autophagy 5, 321–328 (2009).
Wang, R. et al. Autophagy promotes replication of influenza A virus in vitro. J. Virol. 93, e01984-18 (2019).
Sir, D. et al. The early autophagic pathway is activated by hepatitis B virus and required for viral DNA replication. Proc. Natl Acad. Sci. USA 107, 4383–4388 (2010).
Tian, Y., Sir, D., Kuo, C.-F., Ann, D. K. & Ou, J.-H. J. Autophagy required for hepatitis B virus replication in transgenic mice. J. Virol. 85, 13453–13456 (2011).
Li, J. et al. Subversion of cellular autophagy machinery by hepatitis B virus for viral envelopment. J. Virol. 85, 6319–6333 (2011).
Grégoire, I. P. et al. IRGM is a common target of RNA viruses that subvert the autophagy network. PLoS Pathog. 7, e1002422 (2011).
Richetta, C. et al. Sustained autophagy contributes to measles virus infectivity. PLoS Pathog. 9, e1003599 (2013).
Nowag, H. et al. Macroautophagy proteins assist epstein barr virus production and get incorporated into the virus particles. EBioMedicine 1, 116–125 (2014).
Taisne, C. et al. Human cytomegalovirus hijacks the autophagic machinery and LC3 homologs in order to optimize cytoplasmic envelopment of mature infectious particles. Sci. Rep. 9, 4560 (2019).
Buckingham, E. M., Carpenter, J. E., Jackson, W. & Grose, C. Autophagy and the effects of its inhibition on varicella-zoster virus glycoprotein biosynthesis and infectivity. J. Virol. 88, 890–902 (2014).
Buckingham, E. M., Jarosinski, K. W., Jackson, W., Carpenter, J. E. & Grose, C. Exocytosis of varicella-zoster virus virions involves a convergence of endosomal and autophagy pathways. J. Virol. 90, 8673–8685 (2016).
Ma, J., Sun, Q., Mi, R. & Zhang, H. Avian influenza A virus H5N1 causes autophagy-mediated cell death through suppression of mTOR signaling. J. Genet. Genomics 38, 533–537 (2011).
Meng, C. et al. Newcastle disease virus triggers autophagy in U251 glioma cells to enhance virus replication. Arch. Virol. 157, 1011–1018 (2012).
Sun, Y. et al. Autophagy benefits the replication of Newcastle disease virus in chicken cells and tissues. J. Virol. 88, 525–537 (2014).
Meng, S. et al. Avian reovirus triggers autophagy in primary chicken fibroblast cells and Vero cells to promote virus production. Arch. Virol. 157, 661–668 (2012).
Berryman, S. et al. Foot-and-mouth disease virus induces autophagosomes during cell entry via a class III phosphatidylinositol 3-kinase-independent pathway. J. Virol. 86, 12940–12953 (2012).
McFarlane, S. et al. Early induction of autophagy in human fibroblasts after infection with human cytomegalovirus or herpes simplex virus 1. J. Virol. 85, 4212–4221 (2011).
Shelly, S., Lukinova, N., Bambina, S., Berman, A. & Cherry, S. Autophagy is an essential component of Drosophila immunity against vesicular stomatitis virus. Immunity 30, 588–598 (2009).
Sun, P. et al. Foot-and-mouth disease virus capsid protein VP2 activates the cellular EIF2S1-ATF4 pathway and induces autophagy via HSPB1. Autophagy 14, 336–346 (2018).
O’Donnell, V. et al. Foot-and-mouth disease virus utilizes an autophagic pathway during viral replication. Virology 410, 142–150 (2011).
Huang, Y.-P. et al. Autophagy is involved in assisting the replication of Bamboo mosaic virus in Nicotiana benthamiana. J. Exp. Bot. 70, 4657–4670 (2019).
Reggiori, F. et al. Coronaviruses Hijack the LC3-I-positive EDEMosomes, ER-derived vesicles exporting short-lived ERAD regulators, for replication. Cell Host Microbe 7, 500–508 (2010).
Noack, J., Bernasconi, R. & Molinari, M. How viruses hijack the ERAD tuning machinery. J. Virol. 88, 10272–10275 (2014).
Miller, K. et al. Coronavirus interactions with the cellular autophagy machinery. Autophagy 16, 2131–2139 (2020).
Fletcher, K. et al. The WD40 domain of ATG16L1 is required for its non-canonical role in lipidation of LC3 at single membranes. EMBO J. 37, e97840 (2018).
Durgan, J. et al. Non-canonical autophagy drives alternative ATG8 conjugation to phosphatidylserine. Mol. Cell 81, 2031–2040.e8 (2021).
Wang, Y. et al. Control of infection by LC3-associated phagocytosis, CASM, and detection of raised vacuolar pH by the V-ATPase-ATG16L1 axis. Sci. Adv. 8, eabn3298 (2022).
Suhy, D. A., Giddings, T. H. Jr & Kirkegaard, K. Remodeling the endoplasmic reticulum by poliovirus infection and by individual viral proteins: an autophagy-like origin for virus-induced vesicles. J. Virol. 74, 8953–8965 (2000).
Benard, G. & Rossignol, R. Ultrastructure of the mitochondrion and its bearing on function and bioenergetics. Antioxid. Redox Signal. 10, 1313–1342 (2008).
Bouchard, M. J. & Schneider, R. J. The enigmatic X gene of hepatitis B virus. J. Virol. 78, 12725–12734 (2004).
Kim, S.-J. et al. Hepatitis B virus disrupts mitochondrial dynamics: induces fission and mitophagy to attenuate apoptosis. PLoS Pathog. 9, e1003722 (2013).
Kim, S.-J., Syed, G. H. & Siddiqui, A. Hepatitis C virus induces the mitochondrial translocation of Parkin and subsequent mitophagy. PLoS Pathog. 9, e1003285 (2013).
Kim, S.-J. et al. Hepatitis C virus triggers mitochondrial fission and attenuates apoptosis to promote viral persistence. Proc. Natl Acad. Sci. USA 111, 6413–6418 (2014).
Hara, Y. et al. Hepatitis C virus core protein suppresses mitophagy by interacting with parkin in the context of mitochondrial depolarization. Am. J. Pathol. 184, 3026–3039 (2014).
Jassey, A. et al. Hepatitis C virus non-structural protein 5A (NS5A) disrupts mitochondrial dynamics and induces mitophagy. Cells 8, 290 (2019).
Afzal, M. S. et al. Regulation of core expression during the hepatitis C virus life cycle. J. Gen. Virol. 96, 311–321 (2015).
Ding, B. et al. The matrix protein of human parainfluenza virus type 3 induces mitophagy that suppresses interferon responses. Cell Host Microbe 21, 538–547.e4 (2017).
Vescovo, T. et al. Autophagy protects cells from HCV-induced defects in lipid metabolism. Gastroenterology 142, 644–653.e3 (2012).
Heaton, N. S. & Randall, G. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microbe 8, 422–432 (2010).
Farré, J.-C. & Subramani, S. Mechanistic insights into selective autophagy pathways: lessons from yeast. Nat. Rev. Mol. Cell Biol. 17, 537–552 (2016).
Lennemann, N. J. & Coyne, C. B. Dengue and Zika viruses subvert reticulophagy by NS2B3-mediated cleavage of FAM134B. Autophagy 13, 322–332 (2017).
Lipatova, Z. & Segev, N. A role for macro-ER-phagy in ER quality control. PLoS Genet. 11, e1005390 (2015).
Acknowledgements
The authors thank the members of the Jackson, Frieman and Coughlan labs for weekly discussion sessions and M. Patricia (2017) for inspiring the title. This work was funded by National Institutes of Health (NIH)/National Institute of Allergy and Infectious Diseases (NIAID) grants R01AI141359 and R01AI104928 to W.T.J. The authors apologize to any authors whose work was missed or given short mention because of space limitations.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Microbiology thanks Christian Münz; Rupert Beale, who co-reviewed with Carmen Figueras-Novoa; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jassey, A., Jackson, W.T. Viruses and autophagy: bend, but don’t break. Nat Rev Microbiol 22, 309–321 (2024). https://doi.org/10.1038/s41579-023-00995-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41579-023-00995-y
- Springer Nature Limited
This article is cited by
-
The roles and mechanisms of endoplasmic reticulum stress-mediated autophagy in animal viral infections
Veterinary Research (2024)