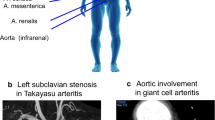
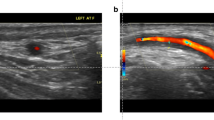
Abstract
Large-vessel vasculitis (LVV) manifests as inflammation of the aorta and its major branches and is the most common primary vasculitis in adults. LVV comprises two distinct conditions, giant cell arteritis and Takayasu arteritis, although the phenotypic spectrum of primary LVV is complex. Non-specific symptoms often predominate and so patients with LVV present to a range of health-care providers and settings. Rapid diagnosis, specialist referral and early treatment are key to good patient outcomes. Unfortunately, disease relapse remains common and chronic vascular complications are a source of considerable morbidity. Although accurate monitoring of disease activity is challenging, progress in vascular imaging techniques and the measurement of laboratory biomarkers may facilitate better matching of treatment intensity with disease activity. Further, advances in our understanding of disease pathophysiology have paved the way for novel biologic treatments that target important mediators of disease in both giant cell arteritis and Takayasu arteritis. This work has highlighted the substantial heterogeneity present within LVV and the importance of an individualized therapeutic approach. Future work will focus on understanding the mechanisms of persisting vascular inflammation, which will inform the development of increasingly sophisticated imaging technologies. Together, these will enable better disease prognostication, limit treatment-associated adverse effects, and facilitate targeted development and use of novel therapies.








Similar content being viewed by others
References
Jennette, J. C. et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 65, 1–11 (2013).
Horton, B. T., Magath, T. B. & Brown, G. E. Arteritis of the temporal vessels: a previously undescribed form. Arch. Intern. Med. 53, 400–409 (1934).
Kogstad, O. A. Polymyalgia rheumatica and its relation to arteritis temporalis. Acta Med. Scand. 178, 591–598 (1965).
Gilmour, J. R. Giant cell chronic arteritis. J. Pathol. Bacteriol. 53, 263–277 (1941).
Hamrin, B., Jonsson, N. & Hellsten, S. Polymyalgia arteritica. Ann. Rheum. Dis. 27, 397–405 (1968).
Blockmans, D. et al. Repetitive 18F-fluorodeoxyglucose positron emission tomography in giant cell arteritis: a prospective study of 35 patients. Arthritis Rheum. 55, 131–137 (2006).
Schmidt, W. A., Natusch, A., Moller, D. E., Vorpahl, K. & Gromnica-Ihle, E. Involvement of peripheral arteries in giant cell arteritis: a color Doppler sonography study. Clin. Exp. Rheumatol. 20, 309–318 (2002).
Hellmich, B. et al. 2018 Update of the EULAR recommendations for the management of large vessel vasculitis. Ann. Rheum. Dis. 79, 19–30 (2020). These guidelines provide a framework for the diagnosis and management of LVV based on the best available evidence.
Takayasu, M. A case with peculiar changes of the retinal central vessels [Japanese]. Acta Soc. Ophthal Jpn. 12, 554–555 (1908).
Nasu, T. Pathology of pulseless disease. A systematic study and critical review of twenty-one autopsy cases reported in Japan. Angiology 14, 225–242 (1963).
Gonzalez-Gay, M. A. et al. Giant cell arteritis in northwestern Spain: a 25-year epidemiologic study. Medicine 86, 61–68 (2007).
Salvarani, C., Crowson, C. S., O’Fallon, W. M., Hunder, G. G. & Gabriel, S. E. Reappraisal of the epidemiology of giant cell arteritis in Olmsted County, Minnesota, over a fifty-year period. Arthritis Rheum. 51, 264–268 (2004). This study is an important example of epidemiological research in GCA.
Kermani, T. A. et al. Increase in age at onset of giant cell arteritis: a population-based study. Ann. Rheum. Dis. 69, 780–781 (2010).
Gran, J. T. & Myklebust, G. The incidence of polymyalgia rheumatica and temporal arteritis in the county of Aust Agder, south Norway: a prospective study 1987-94. J. Rheumatol. 24, 1739–1743 (1997).
Gonzalez-Gay, M. A. et al. Epidemiology of giant cell arteritis and polymyalgia rheumatica. Arthritis Rheum. 61, 1454–1461 (2009).
Muratore, F. et al. Large-vessel giant cell arteritis: a cohort study. Rheumatology 54, 463–470 (2015).
Schmidt, W. A., Seifert, A., Gromnica-Ihle, E., Krause, A. & Natusch, A. Ultrasound of proximal upper extremity arteries to increase the diagnostic yield in large-vessel giant cell arteritis. Rheumatology 47, 96–101 (2008).
Boesen, P. & Sorensen, S. F. Giant cell arteritis, temporal arteritis, and polymyalgia rheumatica in a Danish county. A prospective investigation, 1982–1985. Arthritis Rheum. 30, 294–299 (1987).
Salvarani, C. et al. Epidemiologic and immunogenetic aspects of polymyalgia rheumatica and giant cell arteritis in northern Italy. Arthritis Rheum. 34, 351–356 (1991).
Kobayashi, S. et al. Clinical and epidemiologic analysis of giant cell (temporal) arteritis from a nationwide survey in 1998 in Japan: the first government-supported nationwide survey. Arthritis Rheum. 49, 594–598 (2003).
Tam, S. & Wong, T. C. Temporal arteritis in Hong Kong. Int. J. Rheum. Dis. 11, 163–169 (2008).
Sharma, A., Mohammad, A. J. & Turesson, C. Incidence and prevalence of giant cell arteritis and polymyalgia rheumatica: a systematic literature review. Semin. Arthritis Rheum. 50, 1040–1048 (2020).
Koide, K. Takayasu arteritis in Japan. Heart Vessel. Suppl. 7, 48–54 (1992).
Watts, R., Al-Taiar, A., Mooney, J., Scott, D. & Macgregor, A. The epidemiology of Takayasu arteritis in the UK. Rheumatology 48, 1008–1011 (2009).
Dreyer, L., Faurschou, M. & Baslund, B. A population-based study of Takayasu s arteritis in eastern Denmark. Clin. Exp. Rheumatol. 29, S40–S42 (2011).
Mohammad, A. J. & Mandl, T. Takayasu arteritis in southern Sweden. J. Rheumatol. 42, 853–858 (2015).
Gudbrandsson, B., Molberg, O., Garen, T. & Palm, O. Prevalence, incidence, and disease characteristics of Takayasu Arteritis by ethnic background: data from a large, population-based cohort resident in southern Norway. Arthritis Care Res. 69, 278–285 (2017).
Arnaud, L. et al. Takayasu arteritis in France: a single-center retrospective study of 82 cases comparing white, North African, and black patients. Medicine 89, 1–17 (2010).
Rutter, M., Bowley, J., Lanyon, P. C., Grainge, M. J. & Pearce, F. A. A systematic review and meta-analysis of the incidence rate of Takayasu arteritis. Rheumatology 60, 4982–4990 (2021).
Goel, R. et al. Long-term outcome of 251 patients with Takayasu arteritis on combination immunosuppressant therapy: single centre experience from a large tertiary care teaching hospital in southern India. Semin. Arthritis Rheum. 47, 718–726 (2018).
Danda, D. et al. Clinical course of 602 patients with Takayasu’s arteritis: comparison between childhood-onset versus adult onset disease. Rheumatology 60, 2246–2255 (2020).
Zhang, Z. et al. An observational study of sex differences in Takayasu arteritis in china: implications for worldwide regional differences. Ann. Vasc. Surg. 66, 309–317 (2020).
Watanabe, Y., Miyata, T. & Tanemoto, K. Current clinical features of new patients with Takayasu arteritis observed from cross-country research in Japan: age and sex specificity. Circulation 132, 1701–1709 (2015).
Aeschlimann, F. A. et al. Presentation and disease course of childhood-onset versus adult-onset Takayasu arteritis. Arthritis Rheumatol. 71, 315–323 (2019).
Quinn, K. A. et al. Patterns of clinical presentation in Takayasu’s arteritis. Semin. Arthritis Rheum. 50, 576–581 (2020).
Tomelleri, A. et al. Gender differences in clinical presentation and vascular pattern in patients with Takayasu arteritis. Scand. J. Rheumatol. 48, 482–490 (2019).
Carmona, F. D., Gonzalez-Gay, M. A. & Martin, J. Genetic component of giant cell arteritis. Rheumatology 53, 6–18 (2014).
Mattey, D. L. et al. Association of giant cell arteritis and polymyalgia rheumatica with different tumor necrosis factor microsatellite polymorphisms. Arthritis Rheum. 43, 1749–1755 (2000).
Salvarani, C. et al. Intercellular adhesion molecule 1 gene polymorphisms in polymyalgia rheumatica/giant cell arteritis: association with disease risk and severity. J. Rheumatol. 27, 1215–1221 (2000).
Rueda, B. et al. A functional variant of vascular endothelial growth factor is associated with severe ischemic complications in giant cell arteritis. J. Rheumatol. 32, 1737–1741 (2005).
Palomino-Morales, R. et al. Association between toll-like receptor 4 gene polymorphism and biopsy-proven giant cell arteritis. J. Rheumatol. 36, 1501–1506 (2009).
Serrano, A. et al. Identification of the PTPN22 functional variant R620W as susceptibility genetic factor for giant cell arteritis. Ann. Rheum. Dis. 72, 1882–1886 (2013).
Carmona, F. D. et al. A genome-wide association study identifies risk alleles in plasminogen and P4HA2 associated with giant cell arteritis. Am. J. Hum. Genet. 100, 64–74 (2017). Genome-wide association studies such as this one are providing novel insights into the pathogenesis of GCA.
Weyand, C. & Goronzy, J. Immune mechanisms in medium and large-vessel vasculitis. Nat. Rev. Rheumatol. 9, 731–740 (2013).
Renauer, P. & Sawalha, A. H. The genetics of Takayasu arteritis. Presse Med. 46, e179–e187 (2017).
Terao, C. et al. Genetic determinants and an epistasis of LILRA3 and HLA-B*52 in Takayasu arteritis. Proc. Natl Acad. Sci. USA 115, 13045–13050 (2018).
Seko, Y. et al. Perforin-secreting killer cell infiltration and expression of a 65-kD heat-shock protein in aortic tissue of patients with Takayasu’s arteritis. J. Clin. Invest. 93, 750–758 (1994).
Renauer, P. A. et al. Identification of susceptibility loci in IL6, RPS9/LILRB3, and an intergenic locus on chromosome 21q22 in Takayasu arteritis in a genome-wide association study. Arthritis Rheumatol. 67, 1361–1368 (2015).
Saruhan-Direskeneli, G. et al. Identification of multiple genetic susceptibility loci in Takayasu arteritis. Am. J. Hum. Genet. 93, 298–305 (2013).
Terao, C. et al. Two susceptibility loci to Takayasu arteritis reveal a synergistic role of the IL12B and HLA-B regions in a Japanese population. Am. J. Hum. Genet. 93, 289–297 (2013).
Ortiz-Fernandez, L. et al. Identification of susceptibility loci for Takayasu arteritis through a large multi-ancestral genome-wide association study. Am. J. Hum. Genet. 108, 84–99 (2021). Genome-wide association studies such as this one are providing novel insights into the pathogenesis of TAK.
Carmona, F. D. et al. Analysis of the common genetic component of large-vessel vasculitides through a meta-Immunochip strategy. Sci. Rep. 7, 43953 (2017).
Smeeth, L., Cook, C. & Hall, A. J. Incidence of diagnosed polymyalgia rheumatica and temporal arteritis in the United Kingdom, 1990–2001. Ann. Rheum. Dis. 65, 1093–1098 (2006).
Nordborg, E. & Nordborg, C. Giant cell arteritis: epidemiological clues to its pathogenesis and an update on its treatment. Rheumatology 42, 413–421 (2003).
Ostrowski, R. A., Metgud, S., Tehrani, R. & Jay, W. M. Varicella zoster virus in giant cell arteritis: a review of current medical literature. Neuroophthalmology 43, 159–170 (2019).
Kumar Chauhan, S., Kumar Tripathy, N., Sinha, N., Singh, M. & Nityanand, S. Cellular and humoral immune responses to mycobacterial heat shock protein-65 and its human homologue in Takayasu’s arteritis. Clin. Exp. Immunol. 138, 547–553 (2004).
Pedreira, A. L. S. & Santiago, M. B. Association between Takayasu arteritis and latent or active Mycobacterium tuberculosis infection: a systematic review. Clin. Rheumatol. 39, 1019–1026 (2020).
Hill, C. L. et al. Risk of mortality in patients with giant cell arteritis: a systematic review and meta-analysis. Semin. Arthritis Rheum. 46, 513–519 (2017).
Richards, B. L., March, L. & Gabriel, S. E. Epidemiology of large-vessel vasculidities. Best. Pract. Res. Clin. Rheumatol. 24, 871–883 (2010).
Li, K. J., Semenov, D., Turk, M. & Pope, J. A meta-analysis of the epidemiology of giant cell arteritis across time and space. Arthritis Res. Ther. 23, 82 (2021).
Gran, J. T., Myklebust, G., Wilsgaard, T. & Jacobsen, B. K. Survival in polymyalgia rheumatica and temporal arteritis: a study of 398 cases and matched population controls. Rheumatology 40, 1238–1242 (2001).
Yang, L. et al. Clinical manifestations and longterm outcome for patients with Takayasu arteritis in China. J. Rheumatol. 41, 2439–2446 (2014).
Soto, M. E., Espinola, N., Flores-Suarez, L. F. & Reyes, P. A. Takayasu arteritis: clinical features in 110 Mexican Mestizo patients and cardiovascular impact on survival and prognosis. Clin. Exp. Rheumatol. 26, S9–S15 (2008).
Schmidt, J. et al. Diagnostic features, treatment, and outcomes of Takayasu arteritis in a US cohort of 126 patients. Mayo Clin. Proc. 88, 822–830 (2013).
Park, S. J. et al. Incidence, prevalence, mortality and causes of death in Takayasu arteritis in Korea - a nationwide, population-based study. Int. J. Cardiol. 235, 100–104 (2017).
Mirouse, A. et al. Overall survival and mortality risk factors in Takayasu’s arteritis: a multicenter study of 318 patients. J. Autoimmun. 96, 35–39 (2019).
Goel, R. et al. Cardiovascular and renal morbidity in Takayasu arteritis: a population-based retrospective cohort study from the united kingdom. Arthritis Rheumatol. 73, 504–511 (2021).
Jin, K. et al. NOTCH-induced rerouting of endosomal trafficking disables regulatory T cells in vasculitis. J. Clin. Invest. 131, e136042 (2021).
Miyabe, C. et al. An expanded population of pathogenic regulatory T cells in giant cell arteritis is abrogated by IL-6 blockade therapy. Ann. Rheum. Dis. 76, 898–905 (2017).
Samson, M. et al. Improvement of Treg immune response after treatment with tocilizumab in giant cell arteritis. Clin. Transl. Immunol. 10, e1332 (2021).
Weyand, C. M., Berry, G. J. & Goronzy, J. J. The immunoinhibitory PD-1/PD-L1 pathway in inflammatory blood vessel disease. J. Leukoc. Biol. 103, 565–575 (2018).
Zhang, H. et al. Immunoinhibitory checkpoint deficiency in medium and large vessel vasculitis. Proc. Natl Acad. Sci. USA 114, E970–E979 (2017).
Daxini, A., Cronin, K. & Sreih, A. G. Vasculitis associated with immune checkpoint inhibitors-a systematic review. Clin. Rheumatol. 37, 2579–2584 (2018).
Wen, Z. et al. The microvascular niche instructs T cells in large vessel vasculitis via the VEGF-Jagged1-Notch pathway. Sci. Transl. Med. 9, eaal3322 (2017).
Watanabe, R. et al. MMP (matrix metalloprotease)-9-producing monocytes enable T cells to invade the vessel wall and cause vasculitis. Circ. Res. 123, 700–715 (2018).
Segarra, M. et al. Gelatinase expression and proteolytic activity in giant-cell arteritis. Ann. Rheum. Dis. 66, 1429–1435 (2007).
Segarra, M. et al. Dual function of focal adhesion kinase in regulating integrin-induced MMP-2 and MMP-9 release by human T lymphoid cells. FASEB J. 19, 1875–1877 (2005).
Piggott, K. et al. Blocking the NOTCH pathway inhibits vascular inflammation in large-vessel vasculitis. Circulation 123, 309–318 (2011).
Wang, L. et al. ROS-producing immature neutrophils in giant cell arteritis are linked to vascular pathologies. JCI Insight 5, e139163 (2020).
Cid, M. C. et al. Cell adhesion molecules in the development of inflammatory infiltrates in giant cell arteritis: inflammation-induced angiogenesis as the preferential site of leukocyte-endothelial cell interactions. Arthritis Rheum. 43, 184–194 (2000).
Tombetti, E. & Mason, J. C. Takayasu arteritis: advanced understanding is leading to new horizons. Rheumatology 58, 206–219 (2019).
Coit, P., De Lott, L. B., Nan, B., Elner, V. M. & Sawalha, A. H. DNA methylation analysis of the temporal artery microenvironment in giant cell arteritis. Ann. Rheum. Dis. 75, 1196–1202 (2016).
Mohan, S. V., Liao, Y. J., Kim, J. W., Goronzy, J. J. & Weyand, C. M. Giant cell arteritis: immune and vascular aging as disease risk factors. Arthritis Res. Ther. 13, 231 (2011).
Watanabe, R., Berry, G. J., Liang, D. H., Goronzy, J. J. & Weyand, C. M. Pathogenesis of giant cell arteritis and Takayasu arteritis-similarities and differences. Curr. Rheumatol. Rep. 22, 68 (2020).
Ma-Krupa, W. et al. Activation of arterial wall dendritic cells and breakdown of self-tolerance in giant cell arteritis. J. Exp. Med. 199, 173–183 (2004).
Maleszewski, J. J. et al. Clinical and pathological evolution of giant cell arteritis: a prospective study of follow-up temporal artery biopsies in 40 treated patients. Mod. Pathol. 30, 788–796 (2017).
Weyand, C. M. & Goronzy, J. J. Medium- and large-vessel vasculitis. N. Engl. J. Med. 349, 160–169 (2003).
Pryshchep, O., Ma-Krupa, W., Younge, B. R., Goronzy, J. J. & Weyand, C. M. Vessel-specific Toll-like receptor profiles in human medium and large arteries. Circulation 118, 1276–1284 (2008). This study, and others from the same group, used humanized mouse models of GCA to uncover important aspects of disease pathogenesis.
Kaiser, M., Younge, B., Björnsson, J., Goronzy, J. J. & Weyand, C. M. Formation of new vasa vasorum in vasculitis. Production of angiogenic cytokines by multinucleated giant cells. Am. J. Pathol. 155, 765–774 (1999).
Terrier, B. et al. Interleukin-21 modulates Th1 and Th17 responses in giant cell arteritis. Arthritis Rheum. 64, 2001–2011 (2012).
Watanabe, R. et al. GM-CSF is a pro-inflammatory cytokine in experimental vasculitis of medium and large arteries. Arthritis Rheumatol. https://doi.org/10.1038/s41577-020-0357-7 (2019).
Corbera-Bellalta, M. et al. Blocking GM-CSF receptor α with mavrilimumab reduces infiltrating cells, pro-inflammatory markers, and neoangiogenesis in ex-vivo cultured arteries from patients with giant cell arteritis. Ann. Rheum. Dis. 57, 175–184 (2021).
Deng, J., Younge, B. R., Olshen, R. A., Goronzy, J. J. & Weyand, C. M. Th17 and Th1 T-cell responses in giant cell arteritis. Circulation 121, 906–915 (2010).
Espígol-Frigolé, G. et al. Increased IL-17A expression in temporal artery lesions is a predictor of sustained response to glucocorticoid treatment in patients with giant-cell arteritis. Ann. Rheum. Dis. 72, 1481–1487 (2013).
Saadoun, D. et al. Th1 and Th17 cytokines drive inflammation in Takayasu arteritis. Arthritis Rheumatol. 67, 1353–1360 (2015).
Ciccia, F. et al. Difference in the expression of IL-9 and IL-17 correlates with different histological pattern of vascular wall injury in giant cell arteritis. Rheumatology 54, 1596–1604 (2015).
Zerbini, A. et al. Increased expression of interleukin-22 in patients with giant cell arteritis. Rheumatology 57, 64–72 (2018).
Corbera-Bellalta, M. et al. Blocking interferon γ reduces expression of chemokines CXCL9, CXCL10 and CXCL11 and decreases macrophage infiltration in ex vivo cultured arteries from patients with giant cell arteritis. Ann. Rheum. Dis. 75, 1177–1186 (2016).
Régnier, P. et al. Targeting JAK/STAT pathway in Takayasu’s arteritis. Ann. Rheum. Dis. 79, 951–959 (2020).
Zhang, H. et al. Inhibition of JAK-STAT signaling suppresses pathogenic immune responses in medium and large vessel vasculitis. Circulation 137, 1934–1948 (2018).
Maciejewski-Duval, A. et al. mTOR pathway activation in large vessel vasculitis. J. Autoimmun. 94, 99–109 (2018).
Hadjadj, J. et al. mTOR pathway is activated in endothelial cells from patients with Takayasu arteritis and is modulated by serum immunoglobulin G. Rheumatology 57, 1011–1020 (2018).
Kurata, A. et al. Difference in immunohistochemical characteristics between Takayasu arteritis and giant cell arteritis: It may be better to distinguish them in the same age. Mod. Rheumatol. 29, 992–1001 (2019).
Samson, M. et al. Involvement and prognosis value of CD8+ T cells in giant cell arteritis. J. Autoimmun. 72, 73–83 (2016).
Kaiser, M., Weyand, C. M., Björnsson, J. & Goronzy, J. J. Platelet-derived growth factor, intimal hyperplasia, and ischemic complications in giant cell arteritis. Arthritis Rheum. 41, 623–633 (1998).
Lozano, E., Segarra, M., García-Martínez, A., Hernández-Rodríguez, J. & Cid, M. C. Imatinib mesylate inhibits in vitro and ex vivo biological responses related to vascular occlusion in giant cell arteritis. Ann. Rheum. Dis. 67, 1581–1588 (2008).
Planas-Rigol, E. et al. Endothelin-1 promotes vascular smooth muscle cell migration across the artery wall: a mechanism contributing to vascular remodelling and intimal hyperplasia in giant-cell arteritis. Ann. Rheum. Dis. 76, 1624–1634 (2017).
Le Joncour, A. et al. Mast cells drive pathologic vascular lesions in Takayasu arteritis. J. Allergy Clin. Immunol. https://doi.org/10.1016/j.jaci.2021.05.003 (2021).
Hu, D. et al. Artery tertiary lymphoid organs control aorta immunity and protect against atherosclerosis via vascular smooth muscle cell lymphotoxin β receptors. Immunity 42, 1100–1115 (2015).
Graver, J. C. et al. Massive B-cell infiltration and organization into artery tertiary lymphoid organs in the aorta of large vessel giant cell arteritis. Front. Immunol. 10, 83 (2019).
Inder, S. J. et al. Immunophenotypic analysis of the aortic wall in Takayasu’s arteritis: involvement of lymphocytes, dendritic cells and granulocytes in immuno-inflammatory reactions. Cardiovasc. Surg. 8, 141–148 (2000).
van der Geest, K. S. et al. Disturbed B cell homeostasis in newly diagnosed giant cell arteritis and polymyalgia rheumatica. Arthritis Rheumatol. 66, 1927–1938 (2014).
Mutoh, T. et al. Identification of two major autoantigens negatively regulating endothelial activation in Takayasu arteritis. Nat. Commun. 11, 1253 (2020). This study implicates distinct anti-endothelial cell antibodies in the pathogenesis of TAK.
Desbois, A. C. et al. Specific follicular helper T cell signature in Takayasu arteritis. Arthritis Rheumatol. 73, 1233–1243 (2021).
Mackie, S. L. et al. British Society for Rheumatology guideline on diagnosis and treatment of giant cell arteritis. Rheumatology 59, e1–e23 (2020).
Diamantopoulos, A. P., Haugeberg, G., Lindland, A. & Myklebust, G. The fast-track ultrasound clinic for early diagnosis of giant cell arteritis significantly reduces permanent visual impairment: towards a more effective strategy to improve clinical outcome in giant cell arteritis? Rheumatology 55, 66–70 (2016).
Parikh, M. et al. Prevalence of a normal C-reactive protein with an elevated erythrocyte sedimentation rate in biopsy-proven giant cell arteritis. Ophthalmology 113, 1842–1845 (2006).
Furuta, S., Cousins, C., Chaudhry, A. & Jayne, D. Clinical features and radiological findings in large vessel vasculitis: are Takayasu arteritis and giant cell arteritis 2 different diseases or a single entity? J. Rheumatol. 42, 300–308 (2015).
Luqmani, R. et al. The Role of Ultrasound Compared to Biopsy of Temporal Arteries in the Diagnosis and Treatment of Giant Cell Arteritis (TABUL): a diagnostic accuracy and cost-effectiveness study. Health Technol. Assess. 20, 1–238 (2016). This landmark study evaluated the diagnostic accuracy of ultrasonography and TAB in GCA.
Dejaco, C. et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann. Rheum. Dis. 77, 636–643 (2018).
Turesson, C., Borjesson, O., Larsson, K., Mohammad, A. J. & Knight, A. Swedish Society of Rheumatology 2018 guidelines for investigation, treatment, and follow-up of giant cell arteritis. Scand. J. Rheumatol. 48, 259–265 (2019).
Maz, M. et al. 2021 American College of Rheumatology/Vasculitis Foundation guideline for the management of giant cell arteritis and Takayasu arteritis. Arthritis Rheumatol. 73, 1349–1365 (2021).
Duftner, C. et al. Imaging in diagnosis, outcome prediction and monitoring of large vessel vasculitis: a systematic literature review and meta-analysis informing the EULAR recommendations. RMD Open 4, e000612 (2018).
Barra, L., Kanji, T., Malette, J. & Pagnoux, C. Imaging modalities for the diagnosis and disease activity assessment of Takayasu’s arteritis: a systematic review and meta-analysis. Autoimmun. Rev. 17, 175–187 (2018).
Yamada, I. et al. Takayasu arteritis: diagnosis with breath-hold contrast-enhanced three-dimensional MR angiography. J. Magn. Reson. Imaging 11, 481–487 (2000).
Lariviere, D. et al. Positron emission tomography and computed tomography angiography for the diagnosis of giant cell arteritis: A real-life prospective study. Medicine 95, e4146 (2016).
Stellingwerff, M. D. et al. Different scoring methods of FDG PET/CT in giant cell arteritis: need for standardization. Medicine 94, e1542 (2015).
Grayson, P. C. et al. (18)F-fluorodeoxyglucose-positron emission tomography as an imaging biomarker in a prospective, longitudinal cohort of patients with large vessel vasculitis. Arthritis Rheumatol. 70, 439–449 (2018). This study attempted to define the role of PET imaging in LVV disease monitoring and produced PETVAS.
Soussan, M. et al. Management of large-vessel vasculitis with FDG-PET: a systematic literature review and meta-analysis. Medicine 94, e622 (2015).
Sammel, A. M. et al. Diagnostic accuracy of positron emission tomography/computed tomography of the head, neck, and chest for giant cell arteritis: a prospective, double-blind, cross-sectional study. Arthritis Rheumatol. 71, 1319–1328 (2019).
Nielsen, B. D. et al. Simple dichotomous assessment of cranial artery inflammation by conventional 18F-FDG PET/CT shows high accuracy for the diagnosis of giant cell arteritis: a case-control study. Eur. J. Nucl. Med. Mol. Imaging 46, 184–193 (2019).
Dellavedova, L. et al. The prognostic value of baseline 18F-FDG PET/CT in steroid-naïve large-vessel vasculitis: introduction of volume-based parameters. Eur. J. Nucl. Med. Mol. Imaging 43, 340–348 (2016).
Nielsen, B. D. et al. Three days of high-dose glucocorticoid treatment attenuates large-vessel 18F-FDG uptake in large-vessel giant cell arteritis but with a limited impact on diagnostic accuracy. Eur. J. Nucl. Med. Mol. Imaging 45, 1119–1128 (2018).
Einspieler, I. et al. Imaging large vessel vasculitis with fully integrated PET/MRI: a pilot study. Eur. J. Nucl. Med. Mol. Imaging 42, 1012–1024 (2015).
Martin, O. et al. PET/MRI Versus PET/CT for whole-body staging: results from a single-center observational study on 1,003 sequential examinations. J. Nucl. Med. 61, 1131–1136 (2020).
Wei, W. et al. ImmunoPET: concept, design, and applications. Chem. Rev. 120, 3787–3851 (2020).
Pugliese, F. et al. Imaging of vascular inflammation with [11C]-PK11195 and positron emission tomography/computed tomography angiography. J. Am. Coll. Cardiol. 56, 653–661 (2010).
Kermani, T. A. et al. Disease relapses among patients with giant cell arteritis: a prospective, longitudinal cohort study. J. Rheumatol. 42, 1213–1217 (2015).
Alba, M. A. et al. Relapses in patients with giant cell arteritis: prevalence, characteristics, and associated clinical findings in a longitudinally followed cohort of 106 patients. Medicine 93, 194–201 (2014).
Labarca, C. et al. Predictors of relapse and treatment outcomes in biopsy-proven giant cell arteritis: a retrospective cohort study. Rheumatology 55, 347–356 (2016).
Comarmond, C. et al. Long-term outcomes and prognostic factors of complications in Takayasu arteritis: a multicenter study of 318 patients. Circulation 136, 1114–1122 (2017).
Weyand, C. M., Fulbright, J. W., Hunder, G. G., Evans, J. M. & Goronzy, J. J. Treatment of giant cell arteritis: interleukin-6 as a biologic marker of disease activity. Arthritis Rheum. 43, 1041–1048 (2000).
Rimland, C. A. et al. Outcome measures in large-vessel vasculitis: relationship between patient, physician, imaging, and laboratory-based assessments. Arthritis Care Res. 72, 1296–1304 (2019).
Matsuyama, A. et al. Matrix metalloproteinases as novel disease markers in Takayasu arteritis. Circulation 108, 1469–1473 (2003).
Dagna, L. et al. Pentraxin-3 as a marker of disease activity in Takayasu arteritis. Ann. Intern. Med. 155, 425–433 (2011).
Baldini, M. et al. Selective up-regulation of the soluble pattern-recognition receptor pentraxin 3 and of vascular endothelial growth factor in giant cell arteritis: relevance for recent optic nerve ischemia. Arthritis Rheum. 64, 854–865 (2012).
Tombetti, E., Hysa, E., Mason, J. C., Cimmino, M. A. & Camellino, D. Blood biomarkers for monitoring and prognosis of large vessel vasculitides. Curr. Rheumatol. Rep. 23, 17 (2021).
Soriano, A. et al. Visual loss and other cranial ischaemic complications in giant cell arteritis. Nat. Rev. Rheumatol. 13, 476–484 (2017).
Macchioni, P. et al. Survival predictors in biopsy-proven giant cell arteritis: a northern Italian population-based study. Rheumatology 58, 609–616 (2019).
Delaval, L. et al. Large-vessel vasculitis diagnosed between 50 and 60years: case-control study based on 183 cases and 183 controls aged over 60years. Autoimmun. Rev. 18, 714–720 (2019).
Robson, J. C. et al. The relative risk of aortic aneurysm in patients with giant cell arteritis compared with the general population of the UK. Ann. Rheum. Dis. 74, 129–135 (2015).
Proven, A., Gabriel, S. E., Orces, C., O’Fallon, W. M. & Hunder, G. G. Glucocorticoid therapy in giant cell arteritis: duration and adverse outcomes. Arthritis Rheum. 49, 703–708 (2003).
Maksimowicz-McKinnon, K., Clark, T. M. & Hoffman, G. S. Limitations of therapy and a guarded prognosis in an American cohort of Takayasu arteritis patients. Arthritis Rheum. 56, 1000–1009 (2007).
Chanouzas, D. et al. Intravenous pulse methylprednisolone for induction of remission in severe ANCA associated Vasculitis: a multi-center retrospective cohort study. BMC Nephrol. 20, 58 (2019).
Christ, L. et al. Tocilizumab monotherapy after ultra-short glucocorticoid administration in giant cell arteritis: a proof-of-concept trial. Lancet Rheumatol. https://doi.org/10.1016/S2665-9913(21)00152-1 (2021).
Wilson, J. C. et al. Serious adverse effects associated with glucocorticoid therapy in patients with giant cell arteritis (GCA): a nested case-control analysis. Semin. Arthritis Rheum. 46, 819–827 (2017).
Águeda, A. F. et al. Management of Takayasu arteritis: a systematic literature review informing the 2018 update of the EULAR recommendation for the management of large vessel vasculitis. RMD Open 5, e001020 (2019).
Mainbourg, S. et al. Prevalence of giant cell arteritis relapse in patients treated with glucocorticoids: a meta-analysis. Arthritis Care Res. 72, 838–849 (2020).
Mukhtyar, C. et al. Development of an evidence-based regimen of prednisolone to treat giant cell arteritis - the Norwich regimen. Rheumatol. Adv. Pract. 3, rkz001 (2019).
Hoffman, G. S. et al. Infliximab for maintenance of glucocorticosteroid-induced remission of giant cell arteritis: a randomized trial. Ann. Intern. Med. 146, 621–630 (2007).
Stone, J. H. et al. Trial of tocilizumab in giant-cell arteritis. N. Engl. J. Med. 377, 317–328 (2017). The GiACTA study evaluated the role of tocilizumab in GCA.
Nakaoka, Y. et al. Efficacy and safety of tocilizumab in patients with refractory Takayasu arteritis: results from a randomised, double-blind, placebo-controlled, phase 3 trial in Japan (the TAKT study). Ann. Rheum. Dis. 77, 348–354 (2018).
Langford, C. A. et al. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of Takayasu arteritis. Arthritis Rheumatol. 69, 846–853 (2017). Although the results did not demonstrate efficacy, this was the first randomized controlled trial conducted in TAK, demonstrating feasibility.
Strehl, C. et al. Defining conditions where long-term glucocorticoid treatment has an acceptably low level of harm to facilitate implementation of existing recommendations: viewpoints from an EULAR task force. Ann. Rheum. Dis. 75, 952–957 (2016).
Barra, L. et al. Variations in the clinical practice of physicians managing Takayasu arteritis: a nationwide survey. Open Access. Rheumatol. 9, 91–99 (2017).
Jover, J. A. et al. Combined treatment of giant-cell arteritis with methotrexate and prednisone. a randomized, double-blind, placebo-controlled trial. Ann. Intern. Med. 134, 106–114 (2001).
Hoffman, G. S. et al. A multicenter, randomized, double-blind, placebo-controlled trial of adjuvant methotrexate treatment for giant cell arteritis. Arthritis Rheum. 46, 1309–1318 (2002).
Spiera, R. F. et al. A prospective, double-blind, randomized, placebo controlled trial of methotrexate in the treatment of giant cell arteritis (GCA). Clin. Exp. Rheumatol. 19, 495–501 (2001).
Mahr, A. D. et al. Adjunctive methotrexate for treatment of giant cell arteritis: an individual patient data meta-analysis. Arthritis Rheum. 56, 2789–2797 (2007).
Gérard, A. L. et al. Efficacy and safety of steroid-sparing treatments in giant cell arteritis according to the glucocorticoids tapering regimen: a systematic review and meta-analysis. Eur. J. Intern. Med. 88, 96–103 (2021).
Koster, M. J. et al. Efficacy of methotrexate in real-world management of giant cell arteritis: a case-control study. J. Rheumatol. 46, 501–508 (2019).
Diamantopoulos, A. P., Hetland, H. & Myklebust, G. Leflunomide as a corticosteroid-sparing agent in giant cell arteritis and polymyalgia rheumatica: a case series. Biomed. Res. Int. 2013, 120638 (2013).
Karabayas, M. et al. Evaluation of adjunctive mycophenolate for large vessel giant cell arteritis. Rheumatol. Adv. Pract. 4, rkaa069 (2020).
Ly, K. H. et al. Steroid-sparing effect and toxicity of dapsone treatment in giant cell arteritis: a single-center, retrospective study of 70 patients. Medicine 95, e4974 (2016).
Monti, S. et al. Systematic literature review informing the 2018 update of the EULAR recommendation for the management of large vessel vasculitis: focus on giant cell arteritis. RMD Open 5, e001003 (2019).
de Boysson, H. et al. Is there a place for cyclophosphamide in the treatment of giant-cell arteritis? A case series and systematic review. Semin. Arthritis Rheum. 43, 105–112 (2013).
Misra, D. P., Wakhlu, A., Agarwal, V. & Danda, D. Recent advances in the management of Takayasu arteritis. Int. J. Rheum. Dis. 22, 60–68 (2019).
Villiger, P. M. et al. Tocilizumab for induction and maintenance of remission in giant cell arteritis: a phase 2, randomised, double-blind, placebo-controlled trial. Lancet 387, 1921–1927 (2016).
Strand, V. et al. Health-related quality of life in patients with giant cell arteritis treated with tocilizumab in a phase 3 randomised controlled trial. Arthritis Res. Ther. 21, 64 (2019).
Calderón-Goercke, M. et al. Tocilizumab in giant cell arteritis. Observational, open-label multicenter study of 134 patients in clinical practice. Semin. Arthritis Rheum. 49, 126–135 (2019).
Unizony, S. et al. Clinical outcomes of patients with giant cell arteritis treated with tocilizumab in real-world clinical practice: decreased incidence of new visual manifestations. Arthritis Res. Ther. 23, 8 (2021).
Calderón-Goercke, M. et al. Tocilizumab in giant cell arteritis: differences between the GiACTA trial and a multicentre series of patients from the clinical practice. Clin. Exp. Rheumatol. 38, 112–119 (2020).
Clément, J. et al. Real-world risk of relapse of giant cell arteritis treated with tocilizumab: a retrospective analysis of 43 patients. J. Rheumatol. 48, 1435–1441 (2021).
Stone, J. H. et al. Long-term effect of tocilizumab in patients with giant cell arteritis: open-label extension phase of the Giant Cell Arteritis Actemra (GiACTA) trial. Lancet Rheumatol. 3, E328–E336 (2021).
Stone, J. H. et al. Glucocorticoid dosages and acute-phase reactant levels at giant cell arteritis flare in a randomized trial of tocilizumab. Arthritis Rheumatol. 71, 1329–1338 (2019).
Unizony, S. et al. Tocilizumab for the treatment of large-vessel vasculitis (giant cell arteritis, Takayasu arteritis) and polymyalgia rheumatica. Arthritis Care Res. 64, 1720–1729 (2012).
Xenitidis, T., Horger, M., Zeh, G., Kanz, L. & Henes, J. C. Sustained inflammation of the aortic wall despite tocilizumab treatment in two cases of Takayasu arteritis. Rheumatology 52, 1729–1731 (2013).
Reichenbach, S. et al. Magnetic resonance angiography in giant cell arteritis: results of a randomized controlled trial of tocilizumab in giant cell arteritis. Rheumatology 57, 982–986 (2018).
Schönau, V. et al. Resolution of vascular inflammation in patients with new-onset giant cell arteritis: data from the RIGA study. Rheumatology 60, 3851–3861 (2021).
Sebastian, A. et al. Efficacy and safety of tocilizumab in giant cell arteritis: a single centre NHS experience using imaging (ultrasound and PET-CT) as a diagnostic and monitoring tool. RMD Open 6, e001417 (2020).
Cid, M. C. et al. Mavrilimumab (anti GM-CSF receptor α monoclonal antibody) reduces risk of flare and increases sustained remission in a phase 2 trial of patients with giant cell arteritis. Ann. Rheum. Dis. 80, 31 (2021).
Langford, C. A. et al. A randomized, double-blind trial of abatacept (CTLA-4Ig) for the treatment of giant cell arteritis. Arthritis Rheumatol. 69, 837–845 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04474847 (2021).
Hernández-Rodríguez, J. et al. Tissue production of pro-inflammatory cytokines (IL-1beta, TNFalpha and IL-6) correlates with the intensity of the systemic inflammatory response and with corticosteroid requirements in giant-cell arteritis. Rheumatology 43, 294–301 (2004).
García-Martínez, A. et al. Clinical relevance of persistently elevated circulating cytokines (tumor necrosis factor alpha and interleukin-6) in the long-term followup of patients with giant cell arteritis. Arthritis Care Res. 62, 835–841 (2010).
Martínez-Taboada, V. M. et al. A double-blind placebo controlled trial of etanercept in patients with giant cell arteritis and corticosteroid side effects. Ann. Rheum. Dis. 67, 625–630 (2008).
Seror, R. et al. Adalimumab for steroid sparing in patients with giant-cell arteritis: results of a multicentre randomised controlled trial. Ann. Rheum. Dis. 73, 2074–2081 (2014).
Brack, A. et al. Giant cell vasculitis is a T cell-dependent disease. Mol. Med. 3, 530–543 (1997).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03725202 (2021).
Nakaoka, Y. et al. Long-term efficacy and safety of tocilizumab in refractory Takayasu arteritis: final results of the randomized controlled phase 3 TAKT study. Rheumatol 59, 2427–2434 (2020).
Mekinian, A. et al. Efficacy of biological-targeted treatments in Takayasu arteritis: multicenter, retrospective study of 49 patients. Circulation 132, 1693–1700 (2015).
Misra, D. P., Rathore, U., Patro, P., Agarwal, V. & Sharma, A. Disease-modifying anti-rheumatic drugs for the management of Takayasu arteritis-a systematic review and meta-analysis. Clin. Rheumatol. 40, 4391–4416 (2021).
Youngstein, T. et al. Serial analysis of clinical and imaging indices reveals prolonged efficacy of TNF-α and IL-6 receptor targeted therapies in refractory Takayasu arteritis. Clin. Exp. Rheumatol. 32, S11–S18 (2014).
Gudbrandsson, B., Molberg, Ø. & Palm, Ø. TNF inhibitors appear to inhibit disease progression and improve outcome in Takayasu arteritis; an observational, population-based time trend study. Arthritis Res. Ther. 19, 99 (2017).
Mekinian, A. et al. Efficacy and safety of TNF-α antagonists and tocilizumab in Takayasu arteritis: multicenter retrospective study of 209 patients. Rheumatology https://doi.org/10.1093/rheumatology/keab635 (2021).
Terao, C. et al. Ustekinumab as a therapeutic option for Takayasu arteritis: from genetic findings to clinical application. Scand. J. Rheumatol. 45, 80–82 (2016).
Yachoui, R., Kreidy, M., Siorek, M. & Sehgal, R. Successful treatment with ustekinumab for corticosteroid- and immunosuppressant-resistant Takayasu’s arteritis. Scand. J. Rheumatol. 47, 246–247 (2018).
Pazzola, G. et al. Rituximab therapy for Takayasu arteritis: a seven patients experience and a review of the literature. Rheumatology 57, 1151–1155 (2018).
Kuwabara, S., Tanimura, S., Matsumoto, S., Nakamura, H. & Horita, T. Successful remission with tofacitinib in a patient with refractory Takayasu arteritis complicated by ulcerative colitis. Ann. Rheum. Dis. 79, 1125–1126 (2020).
Saadoun, D. et al. Retrospective analysis of surgery versus endovascular intervention in Takayasu arteritis: a multicenter experience. Circulation 125, 813–819 (2012).
Park, H. S. et al. Long term results of endovascular treatment in renal arterial stenosis from Takayasu arteritis: angioplasty versus stent placement. Eur. J. Radiol. 82, 1913–1918 (2013).
Jeong, H. S., Jung, J. H., Song, G. G., Choi, S. J. & Hong, S. J. Endovascular balloon angioplasty versus stenting in patients with Takayasu arteritis: a meta-analysis. Medicine 96, e7558 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04366596 (2021).
Perera, A. H. et al. Optimizing the outcome of vascular intervention for Takayasu arteritis. Br. J. Surg. 101, 43–50 (2014).
Assie, C., Janvresse, A., Plissonnier, D., Levesque, H. & Marie, I. Long-term follow-up of upper and lower extremity vasculitis related to giant cell arteritis: a series of 36 patients. Medicine 90, 40–51 (2011).
Le Hello, C. et al. Symptomatic lower-limb giant-cell arteritis: Characteristics, management and long-term outcome. J. Med. Vasc. 42, 148–156 (2017).
Alba, M. A. et al. Central nervous system vasculitis: still more questions than answers. Curr. Neuropharmacol. 9, 437–448 (2011).
Guerrero, A. M. et al. Intracranial internal carotid artery angioplasthy and stenting in giant cell arteritis. J. Neuroimaging 25, 307–309 (2015).
Tomasson, G. et al. Risk for cardiovascular disease early and late after a diagnosis of giant-cell arteritis: a cohort study. Ann. Intern. Med. 160, 73–80 (2014).
Ray, J. G., Mamdani, M. M. & Geerts, W. H. Giant cell arteritis and cardiovascular disease in older adults. Heart 91, 324–328 (2005).
Amiri, N., De Vera, M., Choi, H. K., Sayre, E. C. & Avina-Zubieta, J. A. Increased risk of cardiovascular disease in giant cell arteritis: a general population-based study. Rheumatol 55, 33–40 (2016).
Pujades-Rodriguez, M. et al. Associations between polymyalgia rheumatica and giant cell arteritis and 12 cardiovascular diseases. Heart 102, 383–389 (2016).
Pujades-Rodriguez, M., Morgan, A. W., Cubbon, R. M. & Wu, J. Dose-dependent oral glucocorticoid cardiovascular risks in people with immune-mediated inflammatory diseases: a population-based cohort study. PLoS Med. 17, e1003432 (2020).
Ben-Shlomo, Y. et al. Aortic pulse wave velocity improves cardiovascular event prediction: an individual participant meta-analysis of prospective observational data from 17,635 subjects. J. Am. Coll. Cardiol. 63, 636–646 (2014).
Ng, W. F. et al. Takayasu’s arteritis: a cause of prolonged arterial stiffness. Rheumatology 45, 741–745 (2006).
Seyahi, E. et al. Atherosclerosis in Takayasu arteritis. Ann. Rheum. Dis. 65, 1202–1207 (2006).
Narváez, J. et al. Impact of antiplatelet therapy in the development of severe ischemic complications and in the outcome of patients with giant cell arteritis. Clin. Exp. Rheumatol. 26, S57–S62 (2008).
de Souza, A. W. et al. Antiplatelet therapy for the prevention of arterial ischemic events in Takayasu arteritis. Circ. J. 74, 1236–1241 (2010).
Abularrage, C. J. et al. Quality of life of patients with Takayasu’s arteritis. J. Vasc. Surg. 47, 131–136 (2008). discussion 136-7.
Akar, S. et al. Quality of life in patients with Takayasu’s arteritis is impaired and comparable with rheumatoid arthritis and ankylosing spondylitis patients. Clin. Rheumatol. 27, 859–865 (2008).
Rimland, C. A. et al. Outcome measures in large vessel vasculitis: relationship between patient-, physician-, imaging-, and laboratory-based assessments. Arthritis Care Res. 72, 1296–1304 (2020).
Hellmann, D. B. et al. Domains of health-related quality of life important to patients with giant cell arteritis. Arthritis Rheuma. 49, 819–825 (2003).
Sreih, A. G. et al. Health-related outcomes of importance to patients with Takayasu’s arteritis. Clin. Exp. Rheumatol. 36, 51–57 (2018).
Aydin, S. Z., Direskeneli, H. & Merkel, P. A. Assessment of disease activity in large-vessel vasculitis: results of an international Delphi exercise. J. Rheumatol. 44, 1928–1932 (2017).
Aitken, M. & Basu, N. Improving quality of life in vasculitis patients. Rheumatology 59, iii132–iii135 (2020).
Barra, L. et al. Impact of vasculitis on employment and income. Clin. Exp. Rheumatol. 36, 58–64 (2018).
Koster, M. J., Warrington, K. J. & Matteson, E. L. Morbidity and mortality of large-vessel vasculitides. Curr. Rheumatol. Rep. 22, 86 (2020).
Wen, Z. et al. NADPH oxidase deficiency underlies dysfunction of aged CD8+ Tregs. J. Clin. Invest. 126, 1953–1967 (2016).
Nadkarni, S. et al. Investigational analysis reveals a potential role for neutrophils in giant-cell arteritis disease progression. Circ. Res. 114, 242–248 (2014).
Kong, X. & Sawalha, A. H. Takayasu arteritis risk locus in IL6 represses the anti-inflammatory gene GPNMB through chromatin looping and recruiting MEF2-HDAC complex. Ann. Rheum. Dis. 78, 1388–1397 (2019).
Keser, G., Aksu, K. & Direskeneli, H. Discrepancies between vascular and systemic inflammation in large vessel vasculitis: an important problem revisited. Rheumatology 57, 784–790 (2018).
Guleria, A. et al. NMR-based serum metabolomics discriminates Takayasu arteritis from healthy individuals: a proof-of-principle study. J. Proteome Res. 14, 3372–3381 (2015).
Cui, X. et al. Novel biomarkers for the precisive diagnosis and activity classification of Takayasu arteritis. Circ. Genom. Precis. Med. 12, e002080 (2019).
Bolha, L. et al. Identification of microRNAs and their target gene networks implicated in arterial wall remodelling in giant cell arteritis. Rheumatology 59, 3540–3552 (2020).
Grayson, P. C. et al. 18F-fluorodeoxyglucose-positron emission tomography as an imaging biomarker in a prospective, longitudinal cohort of patients with large vessel vasculitis. Arthritis Rheumatol. 70, 439–449 (2018).
Youngstein, T. et al. FDG uptake by prosthetic arterial grafts in large vessel vasculitis is not specific for active disease. JACC 10, 1042–1052 (2017).
Ćorović, A., Wall, C., Mason, J. C., Rudd, J. H. F. & Tarkin, J. M. Novel positron emission tomography tracers for imaging vascular inflammation. Curr. Cardiol. Rep. 22, 119 (2020).
Lamare, F. et al. Detection and quantification of large-vessel inflammation with 11C-(R)-PK11195 PET/CT. J. Nucl. Med. 52, 33–39 (2011).
Pugliese, F. et al. Imaging of vascular inflammation with [11C]-PK11195 and PET/CT angiography. J. Am. Coll. Cardiol. 56, 33–39 (2010).
Tarkin, J. M. et al. Novel approach to imaging active Takayasu arteritis using somatostatin receptor positron emission tomography/magnetic resonance imaging. Circ. Cardiovasc. Imaging 13, e010389 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04071691 (2021).
Tombetti, E. et al. Novel angiographic scores for evaluation of large vessel vasculitis. Sci. Rep. 8, 15979 (2018).
Nakagomi, D. et al. Development of a score for assessment of radiologic damage in large-vessel vasculitis (Combined Arteritis Damage Score, CARDS). Clin. Exp. Rheumatol. 35, 139–145 (2017).
Goel, R. et al. Derivation of an angiographically based classification system in Takayasu’s arteritis: an observational study from India and North America. Rheumatology 59, 1118–1127 (2020).
Gribbons, K. B. et al. Patterns of arterial disease in Takayasu’s arteritis and giant cell arteritis. Arthritis Care Res. 72, 1615–1624 (2020).
Tarzi, R. M., Mason, J. C. & Pusey, C. D. Issues in trial design for ANCA-associated and large-vessel vasculitis. Nat. Rev. 10, 502–510 (2014).
Sreih, A. G. et al. Development of a core set of outcome measures for large-vessel vasculitis: report from OMERACT 2016. J. Rheumatol. 44, 1933–1937 (2017). The OMERACT group are attempting to define outcome measures for use in LVV clinical studies.
Luqmani, R. A. et al. Birmingham Vasculitis Activity Score (BVAS) in systemic necrotizing vasculitis. QJM 87, 671–678 (1994).
Kerr, G. S. et al. Takayasu arteritis. Ann. Intern. Med. 120, 919–929 (1994).
Aydin, S. Z. et al. Assessment of disease activity and progression in Takayasu’s arteritis with Disease Extent Index-Takayasu. Rheumatology 49, 1889–1893 (2010).
Misra, R. et al. Development and initial validation of the Indian Takayasu Clinical Activity Score (ITAS2010). Rheumatology 52, 1795–1801 (2013).
Gribbons, K. B. et al. Diagnostic assessment strategies and disease subsets in giant cell arteritis: data from an international observational cohort. Arthritis Rheumatol. 72, 667–676 (2020).
Grayson, P. C. et al. Distribution of arterial lesions in Takayasu’s arteritis and giant cell arteritis. Ann. Rheum. Dis. 71, 1329–1334 (2012).
Mader, T. H., Werner, R. P., Chamberlain, D. G. & Doornbos, D. Giant cell arteritis in Alaska Natives. Can. J. Ophthalmol. 44, 53–56 (2009).
Smith, C. A., Fidler, W. J. & Pinals, R. S. The epidemiology of giant cell arteritis. Report of a ten-year study in Shelby County, Tennessee. Arthritis Rheum. 26, 1214–1219 (1983).
Hall, S. et al. Takayasu arteritis. A study of 32 North American patients. Medicine 64, 89–99 (1985).
Ing, E. B. et al. The incidence of giant cell arteritis in Ontario, Canada. Can. J. Ophthalmol. 54, 119–124 (2019).
Martinez, P. & et al. Incidence and prevalence of polymyalgia rheumatic and giant cell arteritis: a 15-year study in a health care management organization [abstract]. Arthritis Rheumatol. 68 (Suppl. 10), 1190 (2016).
Brekke, L. K. et al. Incidence of giant cell arteritis in Western Norway 1972-2012: a retrospective cohort study. Arthritis Res. Ther. 19, 278 (2017).
Tomasson, G., Bjornsson, J., Zhang, Y., Gudnason, V. & Merkel, P. A. Cardiovascular risk factors and incident giant cell arteritis: a population-based cohort study. Scand. J. Rheumatol. 48, 213–217 (2019).
Dreyer, L., Faurschou, M. & Baslund, B. A population-based study of Takayasu´s arteritis in eastern Denmark. Clin. Exp. Rheumatol. 29, S40–S42 (2011).
Catanoso, M. et al. Incidence, prevalence, and survival of biopsy-proven giant cell arteritis in northern Italy during a 26-year period. Arthritis Care Res. 69, 430–438 (2017).
Pucelj, N. P. et al. The incidence of giant cell arteritis in Slovenia. Clin. Rheumatol. 38, 285–290 (2019).
Romero-Gómez, C. et al. Epidemiological study of primary systemic vasculitides among adults in southern Spain and review of the main epidemiological studies. Clin. Exp. Rheumatol. 33 (Suppl. 89), S-11-8 (2015).
Saritas, F., Donmez, S., Direskeneli, H. & Pamuk, O. N. The epidemiology of Takayasu arteritis: a hospital-based study from northwestern part of Turkey. Rheumatol. Int. 36, 911–916 (2016).
Friedman, G., Friedman, B. & Benbassat, J. Epidemiology of temporal arteritis in Israel. Isr. J. Med. Sci. 18, 241–244 (1982).
Bas-Lando, M. et al. The incidence of giant cell arteritis in Jerusalem over a 25-year period: annual and seasonal fluctuations. Clin. Exp. Rheumatol. 25, S15–S17 (2007).
Nesher, G., Ben-Chetrit, E., Mazal, B. & Breuer, G. S. The incidence of primary systemic vasculitis in Jerusalem: a 20-year hospital-based retrospective study. J. Rheumatol. 43, 1072–1077 (2016).
el-Reshaid, K., Varro, J., al-Duwairi, Q. & Anim, J. T. Takayasu’s arteritis in Kuwait. J. Trop. Med. Hyg. 98, 299–305 (1995).
Dunstan, E. et al. Epidemiology of biopsy-proven giant cell arteritis in South Australia. Intern. Med. J. 44, 32–39 (2014).
Makin, K., Isbel, M. & Nossent, J. Frequency, presentation, and outcome of Takayasu arteritis in Western Australia. Mod. Rheumatol. 27, 1019–1023 (2017).
Abdul-Rahman, A. M., Molteno, A. C. & Bevin, T. H. The epidemiology of giant cell arteritis in Otago, New Zealand: a 9-year analysis. N. Z. Med. J. 124, 44–52 (2011).
Li, L., Neogi, T. & Jick, S. Mortality in patients with giant cell arteritis: a cohort study in UK primary care. Arthritis Care Res. 70, 1251–1256 (2018).
Ben-Shabat, N. et al. Mortality among patients with giant cell arteritis: a large-scale population-based cohort study. J. Rheumatol. 47, 1385–1391 (2020).
Kermani, T. A. et al. Large-vessel involvement in giant cell arteritis: a population-based cohort study of the incidence-trends and prognosis. Ann. Rheum. Dis. 72, 1989–1994 (2013).
Michailidou, D. et al. Clinical symptoms and associated vascular imaging findings in Takayasu’s arteritis compared to giant cell arteritis. Ann. Rheum. Dis. 79, 262–267 (2020).
Uy, C. P. et al. The impact of integrated noninvasive imaging in the management of Takayasu arteritis. JACC Cardiovasc. Imaging 14, 495–500 (2021).
Spira, D., Xenitidis, T., Henes, J. & Horger, M. MRI parametric monitoring of biological therapies in primary large vessel vasculitides: a pilot study. Br. J. Radiol. 89, 20150892 (2016).
Quinn, K. A. et al. Comparison of magnetic resonance angiography and (18)F-fluorodeoxyglucose positron emission tomography in large-vessel vasculitis. Ann. Rheum. Dis. 77, 1165–1171 (2018).
Prieto-González, S. et al. Effect of glucocorticoid treatment on computed tomography angiography detected large-vessel inflammation in giant-cell arteritis. A prospective, longitudinal study. Medicine 94, e486 (2015).
Dweck, M. R. et al. Hybrid magnetic resonance imaging and positron emission tomography with fluorodeoxyglucose to diagnose active cardiac sarcoidosis. JACC Cardiovasc. Imaging 11, 94–107 (2018).
Abgral, R. et al. Clinical utility of combined FDG-PET/MR to assess myocardial disease. JACC Cardiovasc. Imaging 10, 594–597 (2017).
Laurent, C. et al. PET/MRI in large-vessel vasculitis: clinical value for diagnosis and assessment of disease activity. Sci. Rep. 9, 12388 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02902731 (2020).
Weyand, C. M., Hicok, K. C., Hunder, G. G. & Goronzy, J. J. Tissue cytokine patterns in patients with polymyalgia rheumatica and giant cell arteritis. Ann. Intern. Med. 121, 484–491 (1994).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04930094 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04633447 (2021).
Espígol-Frigolé, G. et al. Expression and function of IL12/23 related cytokine subunits (p35, p40, and p19) in giant-cell arteritis lesions: contribution of p40 to Th1- and Th17-mediated inflammatory pathways. Front Immunol. 9, 809 (2018).
Espígol-Frigolé, G. et al. Identification of IL-23p19 as an endothelial proinflammatory peptide that promotes gp130-STAT3 signaling. Sci. Signal. 9, ra28 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03711448 (2021).
Conway, R. et al. Ustekinumab for refractory giant cell arteritis: a prospective 52-week trial. Semin. Arthritis Rheum. 48, 523–528 (2018).
Matza, M. A., Fernandes, A. D., Stone, J. H. & Unizony, S. H. Ustekinumab for the treatment of giant cell arteritis. Arthritis Care Res. 73, 893–897 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03841734 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03892785 (2021).
Schmidt, W. A. et al. A multicentre, randomised, double-blind, placebo-controlled, parallel-group study to evaluate the efficacy and safety of sirukumab in the treatment of giant cell arteritis. Rheumatol. Ther. 7, 793–810 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04299971 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04161898 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04882072 (2021).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04564001 (2020).
Acknowledgements
D.P. and M.K. are funded by Clinical Academic Fellowships from the Chief Scientist Office, Scotland (CAF/19/01 and CAF/21/05, respectively). M.C.C. is funded by the Ministerio de Ciencia e Innovación/AEI//10.13039/501100011033 (PID2020-114909RB-I00) and the International Vasculitis Foundation. N.D. is supported by a Senior Clinical Research Fellowship from the Chief Scientist Office (SCAF/19/02). J.C.M., S.P.M. and T.Y. acknowledge infrastructure support from the Imperial National Institute for Health Research (NIHR) Biomedical Research Centre. P.C.G. is supported by the Intramural Research Program at the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Author information
Authors and Affiliations
Contributions
Introduction (N.D., D.P., J.C.M. and T.Y.); Epidemiology (N.D., D.P., R.G., S.P.M. and T.Y.); Mechanisms/pathophysiology (N.D., D.P., M.K., C.S.G., C.M.W. and T.Y.); Diagnosis/screening/prevention (N.D., D.P., P.C.G. and T.Y.); Management (N.D., D.P., N.B., M.C.C. and T.Y.); Quality of life (N.D., D.P., R.G., S.P.M., C.O. and T.Y.); Outlook (N.D., D.P., J.C.M. and T.Y.); Overview of the Primer (N.D., D.P., J.C.M. and T.Y.).
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Disease Primers thanks Christian Dejaco, who co-reviewed with Milena Bond; David Saadoun; Maxime Samson; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pugh, D., Karabayas, M., Basu, N. et al. Large-vessel vasculitis. Nat Rev Dis Primers 7, 93 (2021). https://doi.org/10.1038/s41572-021-00327-5
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-021-00327-5
- Springer Nature Limited
This article is cited by
-
Single-cell analysis in rheumatic and allergic diseases: insights for clinical practice
Nature Reviews Immunology (2024)
-
18F-FDG-PET/MR imaging to monitor disease activity in large vessel vasculitis
Nature Communications (2024)
-
Elevated serum 25-hydroxyvitamin D: a potential indicator of remission in Takayasu arteritis patients with normal ESR and CRP levels
Clinical Rheumatology (2024)
-
Vascular Interventions in Takayasu Arteritis
Current Treatment Options in Rheumatology (2024)
-
64Cu-DOTATATE, the growing signal for PET imaging of vascular inflammation?
Journal of Nuclear Cardiology (2023)