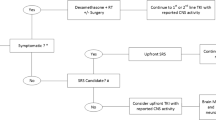
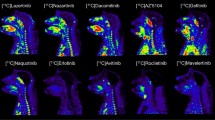
Abstract
The improved survival outcomes of patients with non-small-cell lung cancer (NSCLC), largely owing to the improved control of systemic disease provided by immune-checkpoint inhibitors and novel targeted therapies, have highlighted the challenges posed by central nervous system (CNS) metastases as a devastating yet common complication, with up to 50% of patients developing such lesions during the course of the disease. Early-generation tyrosine-kinase inhibitors (TKIs) often provide robust systemic disease control in patients with oncogene-driven NSCLCs, although these agents are usually unable to accumulate to therapeutically relevant concentrations in the CNS owing to an inability to cross the blood–brain barrier. However, the past few years have seen a paradigm shift with the emergence of several novel or later-generation TKIs with improved CNS penetrance. Such agents have promising levels of activity against brain metastases, as demonstrated by data from preclinical and clinical studies. In this Review, we describe current preclinical and clinical evidence of the intracranial activity of TKIs targeting various oncogenic drivers in patients with NSCLC, with a focus on newer agents with enhanced CNS penetration, leptomeningeal disease and the need for intrathecal treatment options. We also discuss evolving assessment criteria and regulatory considerations for future clinical investigations.
Key points
-
Central nervous system (CNS) metastasis and leptomeningeal disease are clinical challenges in the treatment of patients with advanced-stage non-small-cell lung cancers (NSCLCs) that are often associated with inferior outcomes.
-
Targeted therapies have improved the outcomes of several molecularly defined subgroups of patients with oncogene-driven NSCLCs, although certain small-molecule inhibitors confer only limited levels of CNS benefit, often owing to an inability to cross the blood–brain barrier.
-
Preclinical evaluations of Kpuu (the unbound drug partition coefficient) are commonly used to assess the CNS penetrance of novel agents during drug development.
-
Several newer tyrosine-kinase inhibitors have improved CNS efficacy, including osimertinib in EGFR-mutant NSCLCs and alectinib and lorlatinib in ALK-rearranged NSCLCs, with many others in clinical development.
-
Response Assessment in Neuro-Oncology criteria for response assessment in patients with brain or leptomeningeal metastases are different from RECIST, and future clinical trials should incorporate those criteria into the study design.


Similar content being viewed by others
References
Barnholtz-Sloan, J. S. et al. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J. Clin. Oncol. 22, 2865–2872 (2004).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 69, 7–34 (2019).
D’Antonio, C. et al. Bone and brain metastasis in lung cancer: recent advances in therapeutic strategies. Ther. Adv. Med. Oncol. 6, 101–114 (2014).
Sorensen, J. B., Hansen, H. H., Hansen, M. & Dombernowsky, P. Brain metastases in adenocarcinoma of the lung: frequency, risk groups, and prognosis. J. Clin. Oncol., https://doi.org/10.1200/JCO.1988.6.9.1474 (2016).
Rangachari, D. et al. Brain metastases in patients with EGFR-mutated or ALK-rearranged non-small-cell lung cancers. Lung Cancer 88, 108–111 (2015).
Coelho, J. C. et al. Non-small-cell lung cancer with CNS metastasis: disparities from a real-world analysis (GBOT-LACOG 0417). JCO Glob. Oncol. https://doi.org/10.1200/GO.21.00333 (2022).
Sperduto, P. W. et al. Estimating survival in patients with lung cancer and brain metastases: an update of the Graded Prognostic Assessment for Lung Cancer Using Molecular Markers (Lung-molGPA). JAMA Oncol. 3, 827–831 (2017).
Sperduto, P. W. et al. Graded Prognostic Assessment (GPA) for patients with lung cancer and brain metastases: initial report of the small cell lung cancer GPA and update of the non-small cell lung cancer GPA including the effect of programmed death ligand 1 and other prognostic factors. Int. J. Radiat. Oncol. Biol. Phys. 114, 60–74 (2022).
Zimmermann, S., Dziadziuszko, R. & Peters, S. Indications and limitations of chemotherapy and targeted agents in non-small cell lung cancer brain metastases. Cancer Treat. Rev. 40, 716–722 (2014).
Besse, B. et al. Bevacizumab in patients with nonsquamous non–small cell lung cancer and asymptomatic, untreated brain metastases (BRAIN): a nonrandomized, phase II study. Clin. Cancer Res. 21, 1896–1903 (2015).
Tawbi, H. A. et al. Combined nivolumab and ipilimumab in melanoma metastatic to the brain. N. Engl. J. Med. 379, 722–730 (2018).
Li, J. et al. Concurrent nivolumab and ipilimumab with brain stereotactic radiosurgery for brain metastases from non-small cell lung cancer: a phase I trial. J. Clin. Oncol. 38, 2531 (2020).
Goldberg, S. B. et al. Pembrolizumab for management of patients with NSCLC and brain metastases: long-term results and biomarker analysis from a non-randomised, open-label, phase 2 trial. Lancet Oncol. 21, 655–663 (2020).
Hou, X. et al. Efficacy, safety, and health-related quality of life with camrelizumab plus pemetrexed and carboplatin as first-line treatment for advanced nonsquamous NSCLC with brain metastases (CAP-BRAIN): a multicenter, open-label, single-arm, phase 2 study. J. Thorac. Oncol. https://doi.org/10.1016/j.jtho.2023.01.083 (2023).
Non-small cell lung cancer. National Comprehensive Cancer Network (NCCN) (2023).
Heon, S. et al. The impact of initial gefitinib or erlotinib versus chemotherapy on central nervous system progression in advanced non-small cell lung cancer with EGFR mutations. Clin. Cancer Res. 18, 4406–4414 (2012).
Johung, K. L. et al. Extended survival and prognostic factors for patients with ALK-rearranged non-small-cell lung cancer and brain metastasis. J. Clin. Oncol. 34, 123–129 (2016).
Sperduto, P. W. et al. The effect of gene alterations and tyrosine kinase inhibition on survival and cause of death in patients with adenocarcinoma of the lung and brain metastases. Int. J. Radiat. Oncol. Biol. Phys. 96, 406–413 (2016).
Bohn, J.-P., Pall, G., Stockhammer, G. & Steurer, M. Targeted therapies for the treatment of brain metastases in solid tumors. Target. Oncol. 11, 263–275 (2016).
Welsh, J. W. et al. Phase II trial of erlotinib plus concurrent whole-brain radiation therapy for patients with brain metastases from non-small-cell lung cancer. J. Clin. Oncol. 31, 895–902 (2013).
Reungwetwattana, T. et al. CNS response to osimertinib versus standard epidermal growth factor receptor tyrosine kinase inhibitors in patients with untreated EGFR-mutated advanced non-small-cell lung cancer. J. Clin. Oncol. 36, 3290–3297 (2018).
Shaw, A. T. et al. Lorlatinib in non-small-cell lung cancer with ALK or ROS1 rearrangement: an international, multicentre, open-label, single-arm first-in-man phase 1 trial. Lancet Oncol. 18, 1590–1599 (2017).
Sweeney, M. D., Zhao, Z., Montagne, A., Nelson, A. R. & Zlokovic, B. V. Blood–brain barrier: from physiology to disease and back. Physiol. Rev. 99, 21–78 (2019).
Mo, F., Pellerino, A., Soffietti, R. & Rudà, R. Blood–brain barrier in brain tumors: biology and clinical relevance. Int. J. Mol. Sci. 22, 12654 (2021).
Steeg, P. S. The blood–tumour barrier in cancer biology and therapy. Nat. Rev. Clin. Oncol. 18, 696–714 (2021).
Lockman, P. R. et al. Heterogeneous blood–tumor barrier permeability determines drug efficacy in experimental brain metastases of breast cancer. Clin. Cancer Res. 16, 5664–5678 (2010).
Gril, B. et al. HER2 antibody–drug conjugate controls growth of breast cancer brain metastases in hematogenous xenograft models, with heterogeneous blood–tumor barrier penetration unlinked to a passive marker. Neuro Oncol. 22, 1625–1636 (2020).
Ahn, H. K. et al. ALK inhibitor crizotinib combined with intrathecal methotrexate treatment for non-small cell lung cancer with leptomeningeal carcinomatosis. Lung Cancer 76, 253–254 (2012).
Angeli, E. & Bousquet, G. Brain metastasis treatment: the place of tyrosine kinase inhibitors and how to facilitate their diffusion across the blood–brain barrier. Pharmaceutics 13, 1446 (2021).
Sánchez‐Dengra, B., Gonzalez‐Alvarez, I., Bermejo, M. & Gonzalez‐Alvarez, M. Physiologically based pharmacokinetic (Pbpk) modeling for predicting brain levels of drug in rat. Pharmaceutics 13, 1402 (2021).
Singh Badhan, R. K., Chenel, M. & Penny, J. I. Development of a physiologically-based pharmacokinetic model of the rat central nervous system. Pharmaceutics 6, 97 (2014).
De Lange, E. C. M. Utility of CSF in translational neuroscience. J. Pharmacokinet. Pharmacodyn. 40, 315 (2013).
Varadharajan, S. et al. Exploring in silico prediction of the unbound brain-to-plasma drug concentration ratio: model validation, renewal, and interpretation. J. Pharm. Sci. 104, 1197–1206 (2015).
Wilcox, J. A. & Boire, A. A. Leveraging molecular and immune-based therapies in leptomeningeal metastases. CNS Drugs 37, 45–67 (2023).
Li, B. T. et al. Trastuzumab deruxtecan in HER2-mutant non-small-cell lung cancer. N. Engl. J. Med. 386, 241–251 (2022).
Bartsch, R. et al. Trastuzumab deruxtecan in HER2-positive breast cancer with brain metastases: a single-arm, phase 2 trial. Nat. Med. 28, 1840–1847 (2022).
Kabraji, S. et al. Preclinical and clinical efficacy of trastuzumab deruxtecan in breast cancer brain metastases. Clin. Cancer Res. 29, 174–182 (2023).
Doebele, R. C. et al. Oncogene status predicts patterns of metastatic spread in treatment-naive nonsmall cell lung cancer. Cancer 118, 4502–4511 (2012).
Owen, S. & Souhami, L. The management of brain metastases in non-small cell lung cancer. Front. Oncol. 4, 248 (2014).
de Vries, N. A. et al. Restricted brain penetration of the tyrosine kinase inhibitor erlotinib due to the drug transporters P-gp and BCRP. Invest. N. Drugs 30, 443–449 (2012).
Luo, S., Chen, L., Chen, X. & Xie, X. Evaluation on efficacy and safety of tyrosine kinase inhibitors plus radiotherapy in NSCLC patients with brain metastases. Oncotarget 6, 16725–16734 (2015).
Grommes, C. et al. ‘Pulsatile’ high-dose weekly erlotinib for CNS metastases from EGFR mutant non-small cell lung cancer. Neuro Oncol. 13, 1364–1369 (2011).
Arbour, K. C. et al. Twice weekly pulse and daily continuous-dose erlotinib as initial treatment for patients with epidermal growth factor receptor-mutant lung cancers and brain metastases. Cancer 124, 105–109 (2018).
Yu, H. A. et al. Phase 1 study of twice weekly pulse dose and daily low-dose erlotinib as initial treatment for patients with EGFR-mutant lung cancers. Ann. Oncol. 28, 278–284 (2017).
Ballard, P. et al. Preclinical comparison of osimertinib with other EGFR-TKIs in EGFR-mutant NSCLC brain metastases models, and early evidence of clinical brain metastases activity. Clin. Cancer Res. 22, 5130–5140 (2016).
Colclough, N. et al. Preclinical comparison of the blood–brain barrier permeability of osimertinib with other EGFR TKIs. Clin. Cancer Res. 27, 189–201 (2021).
Wu, Y.-L. et al. CNS efficacy of osimertinib in patients with T790M-positive advanced non-small-cell lung cancer: data from a randomized phase III trial (AURA3). J. Clin. Oncol. 36, 2702–2709 (2018).
Zeng, Q. et al. Discovery and evaluation of clinical candidate AZD3759, a potent, oral active, central nervous system-penetrant, epidermal growth factor receptor tyrosine kinase inhibitor. J. Med. Chem. https://doi.org/10.1021/acs.jmedchem.5b01073 (2015).
Yang, Z. et al. AZD3759, a BBB-penetrating EGFR inhibitor for the treatment of EGFR mutant NSCLC with CNS metastases. Sci. Transl. Med. 8 (2016).
Li, X. et al. Enhanced efficacy of AZD3759 and radiation on brain metastasis from EGFR mutant non-small cell lung cancer. Int. J. Cancer 143, 212–224 (2018).
Ahn, M.-J. et al. Phase I study of AZD3759, a CNS penetrable EGFR inhibitor, for the treatment of non-small-cell lung cancer (NSCLC) with brain metastasis (BM) and leptomeningeal metastasis (LM). J. Clin. Oncol. 34, 9003 (2016).
Wu, Y.-L. et al. Randomized phase 3 study of first-line AZD3759 (zorifertinib) versus gefitinib or erlotinib in EGFR-mutant (EGFR m+) non-small-cell lung cancer (NSCLC) with central nervous system (CNS) metastasis. J. Clin. Oncol. 41, 9001–9001 (2023).
Conti, C. et al. BLU-701 is a Highly Potent, Brain-Penetrant and WT-Sparing Next-generation EGFR TKI for the Treatment of Sensitizing (ex19del, L858R) and C797S Resistance Mutations (Blueprint Medicines Corporation, 2021).
Zapata, A., Chefer, V. I. & Shippenberg, T. S. Microdialysis in rodents. Curr. Protoc. Neurosci. 7, Unit 7.2 (2009).
BDTX-1535 goes after osimertinib resistance. Cancer Discov. 11, 2952–2953 (2021).
Lucas, M. C. et al. 27MO BDTX-1535, a CNS penetrant, irreversible inhibitor of intrinsic and acquired resistance EGFR mutations, demonstrates preclinical efficacy in NSCLC and GBM PDX models. Ann. Oncol. 33, S14 (2022).
Remon, J., Hendriks, L. E. L., Cardona, A. F. & Besse, B. EGFR exon 20 insertions in advanced non-small cell lung cancer: a new history begins. Cancer Treat. Rev. 90, 102105 (2020).
Leal, J. L. et al. EGFR exon 20 insertion mutations: clinicopathological characteristics and treatment outcomes in advanced non-small cell lung cancer. Clin. Lung Cancer 22, e859–e869 (2021).
Syed, Y. Y. Amivantamab: first approval. Drugs 81, 1349–1353 (2021).
Zhou, C. et al. Treatment outcomes and safety of mobocertinib in platinum-pretreated patients with EGFR exon 20 insertion-positive metastatic non-small cell lung cancer. JAMA Oncol. 7, e214761 (2021).
Yun, J. et al. Antitumor activity of amivantamab (JNJ-61186372), an EGFR–MET bispecific antibody, in diverse models of egfr exon 20 insertion-driven NSCLC. Cancer Discov. 10, 1194–1209 (2020).
Park, K. et al. Amivantamab in EGFR exon 20 insertion-mutated non-small-cell lung cancer progressing on platinum chemotherapy: initial results from the CHRYSALIS phase I study. J. Clin. Oncol. 39, 3391–3402 (2021).
Riely, G. J. et al. Activity and safety of mobocertinib (TAK-788) in previously treated non-small cell lung cancer with EGFR exon 20 insertion mutations from a phase I/II trial. Cancer Discov. 11, 1688–1699 (2021).
Pearson, P. G. et al. Abstract 3261: LNG-451, a potent inhibitor of EGFR exon 20 insertion mutations with high CNS exposure. Cancer Res. 82, 3261 (2022).
Wang, M. et al. 987P — Sunvozertinib for NSCLC patients with EGFR exon 20 insertion mutations: preliminary analysis of WU-KONG6, the first pivotal study. in ESMO Congress S448–S554 (Annals of Oncology, 2022).
Junttila, M. R. et al. Abstract 1466: ORIC-114, a brain penetrant, orally bioavailable, irreversible inhibitor selectively targets EGFR and HER2 exon20 insertion mutants and regresses intracranial NSCLC xenograft tumors. Cancer Res. 81, 1466–1466 (2021).
Cornelissen, R. et al. Poziotinib in treatment-naive NSCLC harboring HER2 exon 20 mutations: ZENITH20-4, a multicenter, multicohort, open-label, phase 2 trial (cohort 4). J. Thorac. Oncol. https://doi.org/10.1016/j.jtho.2023.03.016 (2023).
Koivunen, J. P. et al. EML4–ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clin. Cancer Res. 14, 4275–4283 (2008).
Costa, D. B. et al. Clinical experience with crizotinib in patients with advanced ALK-rearranged non-small-cell lung cancer and brain metastases. J. Clin. Oncol. 33, 1881–1888 (2015).
Costa, D. B. et al. CSF concentration of the anaplastic lymphoma kinase inhibitor crizotinib. J. Clin. Oncol. 29, e443–e445 (2011).
Camidge, D. R. et al. Brigatinib versus crizotinib in ALK-positive non-small-cell lung cancer. N. Engl. J. Med. 379, 2027–2039 (2018).
Peters, S. et al. Alectinib versus crizotinib in untreated ALK-positive non-small-cell lung cancer. N. Engl. J. Med. 377, 829–838 (2017).
Shaw, A. T. et al. First-line lorlatinib or crizotinib in advanced ALK-positive lung cancer. N. Engl. J. Med. 383, 2018–2029 (2020).
Chow, L. Q. M. et al. ASCEND-7: efficacy and safety of ceritinib treatment in patients with ALK-positive non-small cell lung cancer metastatic to the brain and/or leptomeninges. Clin. Cancer Res. 1838, 2021 (2022).
Soria, J.-C. et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet 389, 917–929 (2017).
Kodama, T., Tsukaguchi, T., Yoshida, M., Kondoh, O. & Sakamoto, H. Selective ALK inhibitor alectinib with potent antitumor activity in models of crizotinib resistance. Cancer Lett. 351, 215–221 (2014).
Kodama, T. et al. Antitumor activity of the selective ALK inhibitor alectinib in models of intracranial metastases. Cancer Chemother. Pharmacol. 74, 1023–1028 (2014).
Gadgeel, S. et al. Alectinib versus crizotinib in treatment-naive anaplastic lymphoma kinase-positive (ALK+) non-small-cell lung cancer: CNS efficacy results from the ALEX study. Ann. Oncol. 29, 2214–2222 (2018).
Mok, T. et al. Updated overall survival and final progression-free survival data for patients with treatment-naive advanced ALK-positive non-small-cell lung cancer in the ALEX study. Ann. Oncol. 31, 1056–1064 (2020).
Camidge, D. R. et al. Brigatinib versus crizotinib in advanced ALK inhibitor-naive ALK-positive non-small cell lung cancer: second interim analysis of the phase III ALTA-1L trial. J. Clin. Oncol. 38, 3592–3603 (2020).
Horn, L. et al. Ensartinib vs crizotinib for patients with anaplastic lymphoma kinase-positive non-small cell lung cancer. JAMA Oncol. 7, 1617 (2021).
Landi, L. & Cappuzzo, F. Achievements and future developments of ALK-TKIs in the management of CNS metastases from ALK-positive NSCLC. Transl. Lung Cancer Res. 5, 579–587 (2016).
Zou, H. Y. et al. PF-06463922 is a potent and selective next-generation ROS1/ALK inhibitor capable of blocking crizotinib-resistant ROS1 mutations. Proc. Natl Acad. Sci. USA 112, 3493–3498 (2015).
Sun, S., Pithavala, Y. K., Martini, J.-F. & Chen, J. Evaluation of lorlatinib cerebrospinal fluid concentrations in relation to target concentrations for ALK inhibition. J. Clin. Pharmacol. https://doi.org/10.1002/jcph.2056 (2022).
Solomon, B. J. et al. Efficacy and safety of first-line lorlatinib versus crizotinib in patients with advanced, ALK-positive non-small-cell lung cancer: updated analysis of data from the phase 3, randomised, open-label CROWN study. Lancet Respir. Med. 11, 354–366 (2023).
Dagogo-Jack, I. et al. Phase II study of lorlatinib in patients with anaplastic lymphoma kinase-positive lung cancer and CNS-specific relapse. JCO Precis. Oncol. https://doi.org/10.1200/PO.21.00522 (2022).
Murray, B. W. et al. TPX-0131, a potent CNS-penetrant, next-generation inhibitor of wild-type ALK and ALK-resistant mutations. Mol. Cancer Ther. 20, 1499–1507 (2021).
Pelish, H. E. et al. Abstract 1468: NUV-655 (NVL-655) is a selective, brain-penetrant ALK inhibitor with antitumor activity against the lorlatinib-resistant G1202R/L1196M compound mutation. Cancer Res. 81, 1468 (2021).
Ernani, V. & Stinchcombe, T. E. Management of brain metastases in non-small-cell lung cancer. J. Oncol. Pract. 15, 563–570 (2019).
Ou, S.-H. I. & Zhu, V. W. CNS metastasis in ROS1 + NSCLC: an urgent call to action, to understand, and to overcome. Lung Cancer 130, 201–207 (2019).
Wu, Y.-L. et al. Phase II study of crizotinib in East Asian patients with ROS1-positive advanced non-small-cell lung cancer. J. Clin. Oncol. 36, 1405–1411 (2018).
Azelby, C. M., Sakamoto, M. R. & Bowles, D. W. ROS1 targeted therapies: current status. Curr. Oncol. Rep. 23, 94 (2021).
Fischer, H. et al. Entrectinib, a TRK/ROS1 inhibitor with anti-CNS tumor activity: differentiation from other inhibitors in its class due to weak interaction with P-glycoprotein. Neuro Oncol. 22, 819–829 (2020).
Frampton, J. E. Entrectinib: a review in NTRK + solid tumours and ROS1 + NSCLC. Drugs 81, 697–708 (2021).
Menichincheri, M. et al. Discovery of entrectinib: a new 3-aminoindazole as a potent anaplastic lymphoma kinase (ALK), c-ros oncogene 1 kinase (ROS1), and pan-tropomyosin receptor kinases (pan-TRKs) inhibitor. J. Med. Chem. 59, 3392–3408 (2016).
Frampton, J. E. Entrectinib: a review in NTRK + solid tumours and ROS1 + NSCLC. Drugs 81, 697 (2021).
De Braud, F. G. M. et al. Entrectinib in locally advanced/metastatic ROS1 and NTRK fusion-positive non-small cell lung cancer (NSCLC): updated integrated analysis of STARTRK-2, STARTRK-1 and ALKA-372-001. Ann. Oncol. 30, v609 (2019).
Doebele, R. C. et al. TRIDENT-1: a global, multicenter, open-label phase II study investigating the activity of repotrectinib in advanced solid tumors harboring ROS1 or NTRK1-3 rearrangements. J. Clin. Oncol. https://doi.org/10.1200/JCO.2020.38.15_suppl.TPS9637 (2020).
Ou, S. H. I. et al. Efficacy of taletrectinib (AB-106/DS-6051b) in ROS1 + NSCLC: an updated pooled analysis of U.S. and Japan phase 1 studies. JTO Clin. Res. Rep. 2 (2021).
Schneider, J. L. et al. A phase 2 study of lorlatinib in patients with ROS1-rearranged lung cancer with brain-only progression on crizotinib. JTO Clin. Res. Rep. 3, 100347 (2022).
Cascetta, P. et al. RET inhibitors in non-small-cell lung cancer. Cancers (Basel) 13, 4415 (2021).
Gillespie, C. S. et al. Genomic alterations and the incidence of brain metastases in advanced and metastatic non-small cell lung cancer: a systematic review and meta-analysis. J. Thorac. Oncol. https://doi.org/10.1016/j.jtho.2023.06.017 (2023).
Drilon, A. et al. Frequency of brain metastases and multikinase inhibitor outcomes in patients with RET-rearranged lung cancers. J. Thorac. Oncol. 13, 1595–1601 (2018).
Drilon, A. et al. Selpercatinib in patients with RET fusion-positive non-small-cell lung cancer: updated safety and efficacy from the Registrational LIBRETTO-001 phase I/II trial. J. Clin. Oncol. 41, 385–394 (2023).
Murciano-Goroff, Y. R. et al. Central nervous system disease in patients with RET fusion-positive NSCLC treated with selpercatinib. J. Thorac. Oncol. 18, 620–627 (2023).
Gainor, J. F. et al. Pralsetinib for RET fusion-positive non-small-cell lung cancer (ARROW): a multi-cohort, open-label, phase 1/2 study. Lancet Oncol. 22, 959–969 (2021).
Zhou, Q. et al. Efficacy and safety of pralsetinib in patients with advanced RET fusion‐positive non-small cell lung cancer. Cancer https://doi.org/10.1002/cncr.34897 (2023).
Doebele, R. C. et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1–2 trials. Lancet Oncol. 21, 271–282 (2020).
Liu, D. et al. Characterization of on-target adverse events caused by TRK inhibitor therapy. Ann. Oncol. 31, 1207–1215 (2020).
Drilon, A. et al. Efficacy and safety of larotrectinib in patients with tropomyosin receptor kinase fusion-positive lung cancers. JCO Precis. Oncol. 6, e2100418 (2022).
Liu, F. et al. NTRK fusion in non-small cell lung cancer: diagnosis, therapy, and TRK inhibitor resistance. Front Oncol 12, 864666 (2022).
Nagasaka, M. et al. TRUST-II: a global phase II study for taletrectinib in ROS1 fusion-positive lung cancer and other solid tumors. J. Clin. Oncol. 40, TPS8601 (2022).
Besse, B. et al. Abstract P02-01: repotrectinib in patients with NTRK fusion-positive advanced solid tumors: update from the registrational phase 2 TRIDENT-1 trial. Mol. Cancer Ther. 20, P02-01 (2021).
Negrao, M. V. et al. Molecular landscape of BRAF-mutant NSCLC reveals an association between clonality and driver mutations and identifies targetable non-V600 driver mutations. J. Thorac. Oncol. 15, 1611 (2020).
Leonetti, A. et al. BRAF in non-small cell lung cancer (NSCLC): pickaxing another brick in the wall. Cancer Treat. Rev. 66, 82–94 (2018).
Planchard, D. et al. Dabrafenib plus trametinib in patients with previously untreated BRAFV600E-mutant metastatic non-small-cell lung cancer: an open-label, phase 2 trial. Lancet Oncol. 18, 1307–1316 (2017).
Davies, M. A. et al. Dabrafenib plus trametinib in patients with BRAFV600-mutant melanoma brain metastases (COMBI-MB): a multicentre, multicohort, open-label, phase 2 trial. Lancet Oncol. 18, 863–873 (2017).
Mathieu, L. N. et al. FDA approval summary: capmatinib and tepotinib for the treatment of metastatic NSCLC harboring MET exon 14 skipping mutations or alterations.Clin. Cancer Res. 28, 249–254 (2022).
Paik, P. K. et al. Tepotinib in non-small-cell lung cancer with MET exon 14 skipping mutations. N. Engl. J. Med. 383, 931 (2020).
Offin, M. et al. CNS metastases in patients with MET exon 14-altered lung cancers and outcomes with crizotinib. JCO Precis. Oncol. 4, 871–876 (2020).
Drusbosky, L. M., Dawar, R., Rodriguez, E. & Ikpeazu, C. V. Therapeutic strategies in METex14 skipping mutated non-small cell lung cancer. J. Hematol. Oncol. 14, 129 (2021).
Garon, E. B. et al. Abstract CT082: capmatinib in METex14-mutated (mut) advanced non-small cell lung cancer (NSCLC): results from the phase II GEOMETRY mono-1 study, including efficacy in patients (pts) with brain metastases (BM). Cancer Res. 80, CT082 (2020).
Viteri, S. et al. 1286 P activity of tepotinib in brain metastases (BM): preclinical models and clinical data from patients (pts) with MET exon 14 (METex14) skipping NSCLC. Ann. Oncol. 31, S831 (2020).
Le, X. et al. Tepotinib efficacy and safety in patients with MET exon 14 skipping NSCLC: outcomes in patient subgroups from the VISION study with relevance for clinical practice. Clin. Cancer Res. 28, 1117–1126 (2022).
Hartmaier, R. J. et al. Osimertinib + savolitinib to overcome acquired MET-mediated resistance in epidermal growth factor receptor-mutated, MET-amplified non-small cell lung cancer: TATTON. Cancer Discov. 13, 98–113 (2023).
Lee, D. H. et al. ABN401 in patients with NSCLC with MET exon 14 (MET ex14) skipping: result from the pilot expansion study. J. Clin. Oncol. 41, e21148 (2023).
Judd, J. et al. Characterization of KRAS mutation subtypes in non-small cell lung cancer. Mol. Cancer Ther. 20, 2577–2584 (2021).
Reita, D. et al. Direct targeting KRAS mutation in non-small cell lung cancer: focus on resistance. Cancers 14, 1321 (2022).
Sabari, J. K. et al. Activity of adagrasib (MRTX849) in brain metastases: preclinical models and clinical data from patients with KRASG12C-mutant non-small cell lung cancer. Clin. Cancer Res. https://doi.org/10.1158/1078-0432.CCR-22-0383 (2022).
Canon, J. et al. The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature 575, 217–223 (2019).
Skoulidis, F. et al. Sotorasib for lung cancers with KRAS p.G12C mutation. N. Engl. J. Med. 384, 2371–2381 (2021).
Ramalingam, S. S. et al. Efficacy of sotorasib in KRAS p.G12C-mutated NSCLC with stable brain metastases: a post-hoc analysis of CodeBreaK 100. J. Thorac. Oncol. 16, S1123 (2012).
Sabari, J. K. et al. Activity of adagrasib (MRTX849) in brain metastases: preclinical models and clinical data from patients with KRASG12C-mutant non-small cell lung cancer. Clin. Cancer Res. 28, 3318–3328 (2022).
Negrao, M. V. et al. Intracranial efficacy of adagrasib in patients from the KRYSTAL-1 trial with KRASG12C-mutated non-small-cell lung cancer who have untreated CNS metastases. J. Clin. Oncol. https://doi.org/10.1200/JCO.23.00046 (2023).
Sabari, J. K. et al. Activity of adagrasib (MRTX849) in patients with KRASG12C-mutated NSCLC and active, untreated CNS metastases in the KRYSTAL-1 trial. J. Clin. Oncol. 40, LBA9009 (2022).
Yang, J. C. H. et al. Osimertinib in patients with epidermal growth factor receptor mutation-positive non-small-cell lung cancer and leptomeningeal metastases: the BLOOM study. J. Clin. Oncol. 38, 538–547 (2020).
Ahn, M.-J. et al. Osimertinib for patients (pts) with leptomeningeal metastases (LM) associated with EGFRm advanced NSCLC: the AURA LM study. Ann. Oncol. 30, ii48 (2019).
Byrnes, D. M. et al. Incidence of neurological complications secondary to intrathecal chemotherapy used as either prophylaxis or treatment of leptomeningeal carcinomatosis. Blood 128, 5973 (2016).
Fan, C. et al. Efficacy and safety of intrathecal pemetrexed combined with dexamethasone for treating tyrosine kinase inhibitor-failed leptomeningeal metastases from EGFR-mutant NSCLC — a prospective, open-label, single-arm phase 1/2 clinical trial (unique identifier: ChiCTR1800016615). J. Thorac. Oncol. 16, 1359–1368 (2021).
Zagouri, F. et al. Intrathecal administration of trastuzumab for the treatment of meningeal carcinomatosis in HER2-positive metastatic breast cancer: a systematic review and pooled analysis. Breast Cancer Res. Treat. 139, 13–22 (2013).
Figura, N. B. et al. Intrathecal trastuzumab in the management of HER2 + breast leptomeningeal disease: a single institution experience. Breast Cancer Res. Treat. 169, 391–396 (2018).
Figura, N. B. et al. Clinical outcomes of breast leptomeningeal disease treated with intrathecal trastuzumab, intrathecal chemotherapy, or whole brain radiation therapy. Breast Cancer Res. Treat. 175, 781–788 (2019).
Groves, M. D. et al. A multicenter phase II trial of intrathecal topotecan in patients with meningeal malignancies. Neuro Oncol. 10, 208–215 (2008).
Sinicrope, K. D. et al. LPTO-09. Intrathecal topotecan for leptomeningeal metastasis in solid tumors: the MD Anderson experience. Neurooncol. Adv. 1, i8 (2019).
Jaeckle, K. A. et al. Intra‐CSF topotecan in treatment of breast cancer patients with leptomeningeal metastases. Cancer Med. 9, 7935–7942 (2020).
Abbott, N. J. Evidence for bulk flow of brain interstitial fluid: significance for physiology and pathology. Neurochem. Int. 45, 545–552 (2004).
Lin, J. H. CSF as a surrogate for assessing CNS exposure: an industrial perspective. Curr. Drug. Metab. 9, 46–59 (2008).
Soderquist, R. G. & Mahoney, M. J. Central nervous system delivery of large molecules: challenges and new frontiers for intrathecally administered therapeutics. Exp. Opin. Drug Deliv. 7, 285–293 (2010).
Chamberlain, M. C. Treatment of leptomeningeal metastasis with intraventricular administration of depot cytarabine (DTC 101). Arch. Neurol. 50, 261 (1993).
Glantz, M. J. et al. Randomized trial of a slow-release versus a standard formulation of cytarabine for the intrathecal treatment of lymphomatous meningitis. J. Clin. Oncol. 17, 3110–3116 (1999).
Ohyashiki, K., Ohyashiki, J. H., Iwabuchi, A., Ito, H. & Toyama, K. Central nervous system involvement in acute nonlymphocytic leukemia with inv(16)(p13q22). Leukemia 2, 398–399 (1988).
Lin, N. U. et al. Response assessment criteria for brain metastases: proposal from the RANO group. Lancet Oncol. 16, e270–e278 (2015).
Lin, N. U. et al. Challenges relating to solid tumour brain metastases in clinical trials, part 1: patient population, response, and progression. A report from the RANO group. Lancet Oncol. 14, e396–e406 (2013).
Chukwueke, U. N. & Wen, P. Y. Use of the Response Assessment in Neuro-Oncology (RANO) criteria in clinical trials and clinical practice. CNS Oncol. 8, CNS28 (2019).
Chamberlain, M. et al. Leptomeningeal metastases: a RANO proposal for response criteria. Neuro Oncol. 19, 484–492 (2017).
Wen, P. Y. et al. Response assessment in neuro-oncology clinical trials. J. Clin. Oncol. 35, 2439–2449 (2017).
Food and Drug Administration. Evaluating Cancer Drugs in Patients with Central Nervous System Metastases (FDA, 2021).
Zhou, K., Cai, X., Wang, X., Lan, X. & Zhang, X. Efficacy and safety of WBRT + EGFR-TKI versus WBRT only in the treatment of NSCLC patients with brain metastasis: an updated meta-analysis. Thorac. Cancer 13, 563–570 (2022).
Brown, P. D. et al. Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC·3): a multicentre, randomised, controlled, phase 3 trial. Lancet Oncol. 18, 1049–1060 (2017).
Mehrabian, H., Detsky, J., Soliman, H., Sahgal, A. & Stanisz, G. J. Advanced magnetic resonance imaging techniques in management of brain metastases. Front. Oncol. 9, 440 (2019).
Zakaria, R. et al. The role of the immune response in brain metastases: novel imaging biomarkers for immunotherapy. Front. Oncol. 11, 711405 (2021).
Daqqaq, T. S. & Alhasan, A. S. Positron emission tomography and perfusion weighted imaging in the detection of brain tumors recurrence. Neurosciences 27, 131–142 (2022).
Kwee, R. M. & Kwee, T. C. Dynamic susceptibility MR perfusion in diagnosing recurrent brain metastases after radiotherapy: a systematic review and meta-analysis. J. Magn. Reson. Imaging 51, 524–534 (2020).
Jia, C. et al. Brain metastases of non-small cell lung cancer: magnetic resonance spectroscopy for clinical outcome assessment in patients with stereotactic radiotherapy. Onco Targets Ther. 13, 13087–13096 (2020).
Guo, D. et al. A novel score combining magnetic resonance spectroscopy parameters and systemic immune-inflammation index improves prognosis prediction in non-small cell lung cancer patients with brain metastases after stereotactic radiotherapy. Front. Oncol. 12, 762230 (2022).
Najjar, A. M., Johnson, J. M. & Schellingerhout, D. The emerging role of amino acid PET in neuro-oncology. Bioengineering (Basel) 5, 104 (2018).
Otman, H. et al. Delayed [18F]-FDG PET imaging increases diagnostic performance and reproducibility to differentiate recurrence of brain metastases from radionecrosis. Clin. Nucl. Med. 47, 800–806 (2022).
Pauleit, D. et al. PET with O-(2-18F-fluoroethyl)-l-tyrosine in peripheral tumors: first clinical results. J. Nucl. Med. 46, 411–416 (2005).
Celli, M. et al. Diagnostic and prognostic potential of 18F-FET PET in the differential diagnosis of glioma recurrence and treatment-induced changes after chemoradiation therapy. Front. Oncol. 11, 721821 (2021).
Albert, N. L. et al. Response Assessment in Neuro-Oncology working group and European Association for Neuro-Oncology recommendations for the clinical use of PET imaging in gliomas. Neuro Oncol. 18, 1199–1208 (2016).
Chou, J. et al. Immunotherapeutic targeting and PET imaging of DLL3 in small-cell neuroendocrine prostate cancer. Cancer Res. 83, 301–315 (2023).
Lv, G. et al. PET imaging of tumor PD-L1 expression with a highly specific nonblocking single-domain antibody. J. Nucl. Med. 61, 117–122 (2020).
Boire, A. et al. Liquid biopsy in central nervous system metastases: a RANO review and proposals for clinical applications. Neuro Oncol. 21, 571–584 (2019).
Wu, X. et al. Cerebrospinal fluid cell-free DNA-based detection of high level of genomic instability is associated with poor prognosis in NSCLC patients with leptomeningeal metastases. Front. Oncol. 12, 664420 (2022).
Fan, Y. et al. Cell-cycle and DNA-damage response pathway is involved in leptomeningeal metastasis of non-small cell lung cancer. Clin. Cancer Res. 24, 209–216 (2018).
Zheng, M.-M. et al. Genotyping of cerebrospinal fluid associated with osimertinib response and resistance for leptomeningeal metastases in EGFR-mutated NSCLC. J. Thorac. Oncol. 16, 250–258 (2021).
Zheng, M.-M. et al. Clinical utility of cerebrospinal fluid cell-free DNA as liquid biopsy for leptomeningeal metastases in ALK-rearranged NSCLC. J. Thorac. Oncol. 14, 924–932 (2019).
Li, Y. S. et al. Unique genetic profiles from cerebrospinal fluid cell-free DNA in leptomeningeal metastases of EGFR-mutant non-small-cell lung cancer: a new medium of liquid biopsy. Ann. Oncol. 29, 945–952 (2018).
Li, M. et al. Dynamic monitoring of cerebrospinal fluid circulating tumor DNA to identify unique genetic profiles of brain metastatic tumors and better predict intracranial tumor responses in non-small cell lung cancer patients with brain metastases: a prospective cohort study (GASTO 1028). BMC Med. 20, 398 (2022).
Lu, B. Y. et al. Spatially resolved analysis of the T cell immune contexture in lung cancer-associated brain metastases. J. Immunother. Cancer 9, e002684 (2021).
de Groot, J. et al. Window-of-opportunity clinical trial of pembrolizumab in patients with recurrent glioblastoma reveals predominance of immune-suppressive macrophages. Neuro Oncol. 22, 539–549 (2020).
Jing Li, M. P. Stereotactic Radiosurgery Versus Whole-brain Radiation Therapy for Patients with 4-15 Brain Metastases: A Phase III Randomized Controlled Trial (American Society for Radiation Oncology Annual Meeting, 2020).
Magnuson, W. J. et al. Management of brain metastases in tyrosine kinase inhibitor-naive epidermal growth factor receptor-mutant non-small-cell lung cancer: a retrospective multi-institutional analysis. J. Clin. Oncol. 35, 1070–1077 (2017).
Soffietti, R., Ahluwalia, M., Lin, N. & Rudà, R. Management of brain metastases according to molecular subtypes. Nat. Rev. Neurol. 16, 557–574 (2020).
Vogelbaum, M. A. et al. Treatment for brain metastases: ASCO-SNO-ASTRO guideline. J. Clin. Oncol. 40, 492–516 (2022).
White, M. N. et al. Combining osimertinib with chemotherapy in EGFR-mutant NSCLC at progression. Clin. Lung Cancer 22, 201–209 (2021).
Chen, G. et al. Central nervous system efficacy of furmonertinib versus gefitinib in patients with non-small cell lung cancer with epidermal growth factor receptor mutations: results from FURLONG study. J. Clin. Oncol. 40, 9101 (2022).
Deng, Y. et al. The concentration of erlotinib in the cerebrospinal fluid of patients with brain metastasis from non-small-cell lung cancer. Mol. Clin. Oncol. 2, 116–120 (2014).
Yang, J.-J. et al. Icotinib versus whole-brain irradiation in patients with EGFR-mutant non-small-cell lung cancer and multiple brain metastases (BRAIN): a multicentre, phase 3, open-label, parallel, randomised controlled trial. Lancet Respir. Med. 5, 707–716 (2017).
Jung, H. A. et al. Totality outcome of afatinib sequential treatment in patients with EGFR mutation-positive non-small cell lung cancer in South Korea (TOAST): Korean Cancer Study Group (KCSG) LU-19-22. Transl. Lung Cancer Res. 11, 1369–1379 (2022).
Cho, B. C. et al. A phase 1/2 study of lazertinib 240 mg in patients with advanced EGFR T790M-positive NSCLC after previous EGFR tyrosine kinase inhibitors. J. Thorac. Oncol. 17, 558–567 (2022).
Solomon, B. J. et al. Lorlatinib in patients with ALK-positive non-small-cell lung cancer: results from a global phase 2 study. Lancet Oncol. 19, 1654–1667 (2018).
Li, W. et al. TRUST — Updated Efficacy and Safety of Taletrectinib in Patients (pts) with ROS1 + Non-Small Cell Lung Cancer (NSCLC) (European Lung Cancer Congress, 2023).
Ou, S. et al. OA02.03. Clinical activity of lorlatinib in patients with ROS1 + advanced non-small cell lung cancer: phase 2 study cohort EXP-6. J. Thorac. Oncol. 13, S322–S323 (2018).
Frost, N. et al. Lorlatinib in pretreated ALK- or ROS1-positive lung cancer and impact of TP53 co-mutations: results from the German early access program. Ther. Adv. Med. Oncol. 13, 175883592098055 (2021).
Odintsov, I. et al. Comparison of TAS0953/HM06 and selpercatinib in RET fusion-driven preclinical disease models of intracranial metastases. J. Clin. Oncol. 40, 2024 (2022).
Griesinger, F. et al. Safety and efficacy of pralsetinib in RET fusion-positive non-small-cell lung cancer including as first-line therapy: update from the ARROW trial. Ann. Oncol. 33, 1168–1178 (2022).
Acknowledgements
X.L. is supported by the Damon Runyon Foundation and V Foundation for Cancer Research. J.V.H. is supported by NIH R50CA265307, 5R01CA247975 and R01CA234183. K.C. is supported by NIH T32 CA009666. J.Z. is supported by the Cancer Prevention and Research Institute of Texas Multi-Investigator Research Award grant (RP160668), the NIH (NCI) R01CA234629-01 and 5U01CA256780.
Author information
Authors and Affiliations
Contributions
K.P. and X.L. wrote the manuscript. All authors researched data for the manuscript, made a substantial contribution to discussions of content and edited and/or reviewed the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.Z. has acted as a consultant and/or adviser of AstraZeneca, Bristol Myers Squibb, GenePlus, Innovent, Johnson & Johnson and Novartis, and has received research grants from Johnson & Johnson, Merck and Novartis. J.V.H. has acted as an adviser of AstraZeneca, Boehringer Ingelheim, BrightPath Biotherapeutics, Bristol Myers Squibb, Catalyst Biotech, EMD Serono, Foundation Medicine, Genentech/Roche, GlaxoSmithKline, Guardant Health, Hengrui Pharmaceutical, Janssen, Kairos Ventures, Leads Biolabs, Lilly, Mirati Therapeutics, Nexus Health Systems, Novartis, Pneuma Respiratory, Roche, Sanofi/Aventis, Spectrum Pharmaceuticals and Takeda, has received research funding from AstraZeneca, GlaxoSmithKline and Spectrum, is included in a licensing agreement between Spectrum and MD Anderson Cancer Center regarding intellectual property relating to treatments targeting of EGFR and HER2 exon 20 mutations, and declares stock and other ownership interests in Bio-Tree Consulting and Cardinal Spine. X.L. has acted as a consultant and/or adviser of Abbvie, AstraZeneca, Blueprint Medicines, Boehringer Ingelheim, Eli Lilly, EMD Serono (Merck KGaA), Hengrui Therapeutics, Janssen, Novartis, Sensei Biotherapeutics and Spectrum Pharmaceutics, and has received research funding from Boehringer Ingelheim, Eli Lilly, EMD Serono and Regeneron. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks R. Soffietti and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, K., Concannon, K., Li, J. et al. Emerging therapeutics and evolving assessment criteria for intracranial metastases in patients with oncogene-driven non-small-cell lung cancer. Nat Rev Clin Oncol 20, 716–732 (2023). https://doi.org/10.1038/s41571-023-00808-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-023-00808-4
- Springer Nature Limited