Abstract
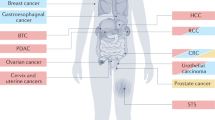
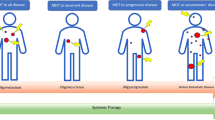
The elimination of metastases remains one of the major challenges in the curative treatment of patients with cancer. Therefore, most patients with metastatic disease typically receive systemic agents, which prolong survival and alleviate symptoms but are rarely curative. The oligometastatic paradigm challenges the prevailing view of metastasis as a disseminated process and proposes the existence of a spectrum of biological virulence within metastatic lesions. In this Perspectives article, we present evidence supporting our hypothesis that integrated clinical and molecular classification of metastases along the spectrum of disease influences the staging and treatment of patients with cancer.


Similar content being viewed by others
References
Hellman, S. & Weichselbaum, R. R. Oligometastases. J. Clin. Oncol. 13, 8–10 (1995).
Weichselbaum, R. R. & Hellman, S. Oligometastases revisited. Nat. Rev. Clin. Oncol. 8, 378–382 (2011).
Ost, P. et al. Surveillance or metastasis-directed therapy for oligometastatic prostate cancer recurrence: a prospective, randomized, multicenter phase II trial. J. Clin. Oncol. 36, 446–453 (2018).
Gomez, D. R. et al. Local consolidative therapy versus maintenance therapy or observation for patients with oligometastatic non-small-cell lung cancer without progression after first-line systemic therapy: a multicentre, randomised, controlled, phase 2 study. Lancet Oncol. 17, 1672–1682 (2016).
Iyengar, P. et al. Consolidative radiotherapy for limited metastatic non-small-cell lung cancer: a phase 2 randomized clinical trial. JAMA Oncol. 4, e173501 (2018).
Gomez, D. R. et al. Local consolidative therapy vs. maintenance therapy or observation for patients with oligometastatic non–small-cell lung cancer: long-term results of a multi-institutional, phase II, randomized study. J. Clin. Oncol. https://doi.org/10.1200/JCO.19.00201 (2019).
Ruers, T. et al. Radiofrequency ablation combined with systemic treatment versus systemic treatment alone in patients with non-resectable colorectal liver metastases: a randomized EORTC Intergroup phase II study (EORTC 40004). Ann. Oncol. 23, 2619–2626 (2012).
Ruers, T. et al. Local treatment of unresectable colorectal liver metastases: results of a randomized phase II trial. J. Natl Cancer Inst. 109, djx015 (2017).
Palma, D. A. et al. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): a randomised, phase 2, open-label trial. Lancet https://doi.org/10.1016/S0140-6736(18)32487-5 (2019).
Palma, D. A. et al. Stereotactic ablative radiotherapy for comprehensive treatment of oligometastatic tumors (SABR-COMET): study protocol for a randomized phase II trial. BMC Cancer 12, 305 (2012).
Singh, D. et al. Is there a favorable subset of patients with prostate cancer who develop oligometastases? Int. J. Radiat. Oncol. Biol. Phys. 58, 3–10 (2004).
deSouza, N. M. et al. Strategies and technical challenges for imaging oligometastatic disease: Recommendations from the European Organisation for Research and Treatment of Cancer imaging group. Eur. J. Cancer 91, 153–163 (2018).
Joice, G. A. et al. Oligometastatic prostate cancer: shaping the definition with molecular imaging and an improved understanding of tumor biology. Curr. Opin. Urol. 27, 533–541 (2017).
Wong, H. S. et al. Comparative study between (68) Ga-prostate-specific membrane antigen positron emission tomography and conventional imaging in the initial staging of prostate cancer. J. Med. Imaging Radiat. Oncol. 62, 816–822 (2018).
Ashworth, A. B. et al. An individual patient data metaanalysis of outcomes and prognostic factors after treatment of oligometastatic non-small-cell lung cancer. Clin. Lung Cancer 15, 346–355 (2014).
Fong, Y. et al. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann. Surg. 230, 309–318 (1999).
Pastorino, U. et al. Long-term results of lung metastasectomy: prognostic analyses based on 5206 cases. J. Thorac. Cardiovasc. Surg. 113, 37–49 (1997).
Lussier, Y. A. et al. Oligo- and polymetastatic progression in lung metastasis(es) patients is associated with specific microRNAs. PLOS ONE 7, e50141 (2012).
Hong, J. C. et al. Classification for long-term survival in oligometastatic patients treated with ablative radiotherapy: A multi-institutional pooled analysis. PLOS ONE 13, e0195149 (2018).
Obenauf, A. C. & Massague, J. Surviving at a distance: organ-specific metastasis. Trends Cancer 1, 76–91 (2015).
Massague, J. & Obenauf, A. C. Metastatic colonization by circulating tumour cells. Nature 529, 298–306 (2016).
Lambert, A. W., Pattabiraman, D. R. & Weinberg, R. A. Emerging biological principles of metastasis. Cell 168, 670–691 (2017).
Bartel, D. P. Metazoan microRNAs. Cell 173, 20–51 (2018).
Lussier, Y. A. et al. MicroRNA expression characterizes oligometastasis(es). PLOS ONE 6, e28650 (2011).
Uppal, A. et al. 14q32-encoded microRNAs mediate an oligometastatic phenotype. Oncotarget 6, 3540–3552 (2015).
Holder, J. L. Jr. et al. A child with an inherited 0.31 Mb microdeletion of chromosome 14q32.33: further delineation of a critical region for the 14q32 deletion syndrome. Am. J. Med. Genet. A 158, 1962–1966 (2012).
Briggs, T. A. et al. Temple syndrome as a result of isolated hypomethylation of the 14q32 imprinted DLK1/MEG3 region. Am. J. Med. Genet. A 170, 170–175 (2016).
Kagami, M. et al. Deletions and epimutations affecting the human 14q32.2 imprinted region in individuals with paternal and maternal upd(14)-like phenotypes. Nat. Genet. 40, 237–242 (2008).
Kagami, M. et al. The IG-DMR and the MEG3-DMR at human chromosome 14q32.2: hierarchical interaction and distinct functional properties as imprinting control centers. PLOS Genet. 6, e1000992 (2010).
Stadtfeld, M. et al. Aberrant silencing of imprinted genes on chromosome 12qF1 in mouse induced pluripotent stem cells. Nature 465, 175–181 (2010).
Oshima, G. et al. DNA methylation controls metastasis-suppressive 14q32-encoded miRNAs. Cancer Res. https://doi.org/10.1158/0008-5472.CAN-18-0692 (2018).
Jamal-Hanjani, M. et al. Tracking the evolution of non-small-cell lung cancer. N. Engl. J. Med. 376, 2109–2121 (2017).
Haffner, M. C. et al. Tracking the clonal origin of lethal prostate cancer. J. Clin. Invest. 123, 4918–4922 (2013).
Turajlic, S. et al. Tracking cancer evolution reveals constrained routes to metastases: TRACERx renal. Cell 173, 581–594 (2018).
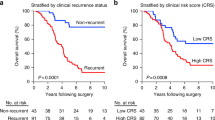
Pitroda, S. P. et al. Integrated molecular subtyping defines a curable oligometastatic state in colorectal liver metastasis. Nat. Commun. 9, 1793 (2018).
Guinney, J. et al. The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350−1356 (2015).
Wang, B. et al. Similarity network fusion for aggregating data types on a genomic scale. Nat. Methods 11, 333–337 (2014).
Van den Eynde, M. et al. The link between the multiverse of immune microenvironments in metastases and the survival of colorectal cancer patients. Cancer Cell 34, 1012–1026 (2018).
Angelova, M. et al. Evolution of metastases in space and time under immune selection. Cell 175, 751–765 (2018).
Pages, F. et al. In situ cytotoxic and memory T cells predict outcome in patients with early-stage colorectal cancer. J. Clin. Oncol. 27, 5944–5951 (2009).
Mlecnik, B. et al. The tumor microenvironment and Immunoscore are critical determinants of dissemination to distant metastasis. Sci. Transl Med. 8, 327ra26 (2016).
Cohen, R. et al. Immunotherapy and metastatic colorectal cancers with microsatellite instability or mismatch repair deficiency. Bull. Cancer 106, 137–142 (2018).
Irahara, N. et al. NRAS mutations are rare in colorectal cancer. Diagn. Mol. Pathol. 19, 157–163 (2010).
Johnson, D. B. et al. Impact of NRAS mutations for patients with advanced melanoma treated with immune therapies. Cancer Immunol. Res. 3, 288–295 (2015).
Wu, Y. M. et al. Inactivation of CDK12 delineates a distinct immunogenic class of advanced prostate cancer. Cell 173, 1770–1782 (2018).
Robinson, D. R. et al. Integrative clinical genomics of metastatic cancer. Nature 548, 297–303 (2017).
Huang, L. et al. Molecular classification of lymph node metastases subtypes predict for survival in head and neck cancer. Clin. Cancer Res. 25, 1795–1808 (2018).
Huang, S. H. & O’Sullivan, B. Overview of the 8th edn TNM classification for head and neck cancer. Curr. Treat. Opt. Oncol. 18, 40 (2017).
Louis, D. N. et al. The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 131, 803–820 (2016).
Parker, C. C. et al. Radiotherapy to the primary tumour for newly diagnosed, metastatic prostate cancer (STAMPEDE): a randomised controlled phase 3 trial. Lancet 392, 2353–2366 (2018).
Sweeney, C. J. et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer. N. Engl. J. Med. 373, 737–746 (2015).
Gundem, G. et al. The evolutionary history of lethal metastatic prostate cancer. Nature 520, 353–357 (2015).
Corcoran, R. B. & Chabner, B. A. Application of cell-free DNA analysis to cancer treatment. N. Engl. J. Med. 379, 1754–1765 (2018).
Radwan, N. et al. A phase II randomized trial of Observation versus stereotactic ablative RadiatIon for OLigometastatic prostate CancEr (ORIOLE). BMC Cancer 17, 453 (2017).
Newton, P. K. et al. Spatiotemporal progression of metastatic breast cancer: a Markov chain model highlighting the role of early metastatic sites. NPJ Breast Cancer 1, 15018 (2015).
Acknowledgements
This work was supported by the Virginia and D. K. Ludwig Fund for Cancer Research, an NIH National Cancer Institute (NCI) R21 grant (CA195075-01A1) and a generous gift from the Foglia Family Foundation.
Reviewer information
Nature Reviews Clinical Oncology thanks P. Ost, S. Siva and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Both authors made a substantial contribution to all aspects of the preparation of this manuscript.
Corresponding author
Ethics declarations
Competing interests
R.R.W. is a consultant and/or adviser for Aettis, AstraZeneca, Genus, ImmunoVir, Merck Serono, Nano Proteagen, Reflexion Pharmaceuticals, RiMO and Shuttle Pharmaceuticals, has been a guest speaker sponsored by Boehringer Ingelheim and has equity and/or intellectual property rights with Boost Therapeutics, Oncosenescence, Reflexion Pharmaceuticals and RiMO. S.P.P. and R.R.W. have a patent entitled “Methods and kits for diagnosis and triage of patients with colorectal liver metastases” pending.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
US NIH ClinicalTrials.gov database: https://www.clinicaltrials.gov
Rights and permissions
About this article
Cite this article
Pitroda, S.P., Weichselbaum, R.R. Integrated molecular and clinical staging defines the spectrum of metastatic cancer. Nat Rev Clin Oncol 16, 581–588 (2019). https://doi.org/10.1038/s41571-019-0220-6
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-019-0220-6
- Springer Nature Limited
This article is cited by
-
Oligometastasis in breast cancer—current status and treatment options from a radiation oncology perspective
Strahlentherapie und Onkologie (2022)
-
Long-term survival after pancreatic metastasis resection from breast cancer: a systematic literature review
Surgical Case Reports (2021)
-
X-change symposium: status and future of modern radiation oncology—from technology to biology
Radiation Oncology (2021)
-
Benefits of local consolidative treatment in oligometastases of solid cancers: a stepwise-hierarchical pooled analysis and systematic review
npj Precision Oncology (2021)
-
Relevanz stereotaktischer ablativer Strahlentherapie bei systemisch metastasierten Patienten
Strahlentherapie und Onkologie (2019)