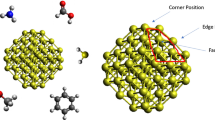
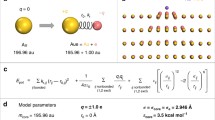
Abstract
Chemists generally believe that covalent and ionic bonds form much stronger links between atoms than the van der Waals force does. However, this is not always so. We present cases in which van der Waals dispersive forces introduce new competitive bonding possibilities rather than just modulating traditional bonding scenarios. Although the new possibilities could arise from any soft–soft chemical interaction, we focus on bonding between gold atoms and alkyl or arylsulfur ligands, RS. Consideration of all the interactions at play in sulfur-protected gold surfaces and gold nanoparticles is necessary to understand their structural, chemical and spectroscopic properties. In turn, such knowledge opens pathways to new chemical entities and innovative nanotechnological devices. Such experimentation is complemented by modern theory, and presented here is a broad overview of computational methods appropriate to fields ranging from gas-phase chemistry to device physics and biochemistry.





Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Dobson, J. F. Beyond pairwise additivity in London dispersion interactions. Int. J. Quantum Chem. 114, 1157–1161 (2014).
Tsoi, S. et al. Van der Waals screening by single-layer graphene and molybdenum disulfide. ACS Nano 8, 12410–12417 (2014).
Schmidbaur, H. & Schier, A. A briefing on aurophilicity. Chem. Soc. Rev. 37, 1931–1951 (2008).
Pyykko, P. Theoretical chemistry of gold. Angew. Chem. Int. Ed. 43, 4412–4456 (2004).
Liptrot, D. J. & Power, P. P. London dispersion forces in sterically crowded inorganic and organometallic molecules. Nat. Rev. Chem. 1, 0004 (2017).
Guo, J. D., Liptrot, D. J., Nagase, S. & Power, P. P. The multiple bonding in heavier group 14 element alkene analogues is stabilized mainly by dispersion force effects. Chem. Sci. 6, 6235–6244 (2015).
Schreiner, P. R. et al. Overcoming lability of extremely long alkane carbon–carbon bonds through dispersion forces. Nature 477, 308–311 (2011).
Fokin, A. A. et al. Stable alkanes containing very long carbon–carbon bonds. J. Am. Chem. Soc. 134, 13641–13650 (2012).
Grimme, S. & Schreiner, P. R. Steric crowding can stabilize a labile molecule: solving the hexaphenylethane riddle. Angew. Chem. Int. Ed. 50, 12639–12642 (2011).
Lyngvi, E., Sanhueza, I. A. & Schoenebeck, F. Dispersion makes the difference: bisligated transition states found for the oxidative addition of Pd(Pt Bu3)2 to Ar–OSO2R and dispersion-controlled chemoselectivity in reactions with Pd[P(iPr)(tBu2)]2 . Organometallics 34, 805–812 (2015).
Wagner, J. P. & Schreiner, P. R. London dispersion decisively contributes to the thermodynamic stability of bulky NHC-coordinated main group compounds. J. Chem. Theory Comput. 12, 231–237 (2016).
Hänninen, M. M., Pal, K., Day, B. M., Pugh, T. & Layfield, R. A. A three-coordinate iron–silylene complex stabilized by ligand–ligand dispersion forces. Dalton Trans. 45, 11301–11305 (2016).
Reimers, J. R., Watts, R. O. & Klein, M. L. Intermolecular potential functions and the properties of water. Chem. Phys. 64, 95–114 (1982).
Kumar, M., Chaudhari, R. V., Subramaniam, B. & Jackson, T. A. Ligand effects on the regioselectivity of rhodium-catalyzed hydroformylation: density functional calculations illuminate the role of long-range noncovalent interactions. Organometallics 33, 4183–4191 (2014).
Wolters, L. P., Koekkoek, R. & Bickelhaupt, F. M. Role of steric attraction and bite-angle flexibility in metal-mediated C–H bond activation. ACS Catal. 5, 5766–5775 (2015).
Schweighauser, L., Strauss, M. A., Bellotto, S. & Wegner, H. A. Attraction or repulsion? London dispersion forces control azobenzene switches. Angew. Chem. Int. Ed. 54, 13436–13439 (2015).
Wagner, C. L. et al. Dispersion-force-assisted disproportionation: a stable two-coordinate copper(II) complex. Angew. Chem. Int. Ed. 55, 10444–10447 (2016).
Lomas, J. R., Baddeley, C. J., Tikhov, M. S. & Lambert, R. M. Ethyne cyclization to benzene over Cu(110). Langmuir 11, 3048–3053 (1995).
Bilic, A., Reimers, J. R., Hush, N. S., Hoft, R. C. & Ford, M. J. Adsorption of benzene on copper, silver, and gold surfaces. J. Chem. Theory Comput. 2, 1093–1105 (2006).
Reimers, J. R., Li, M., Wan, D., Gould, T. & Ford, M. J. in Noncovalent Interactions in Quantum Chemistry and Physics: Theory and Applications (eds Otero de la Roza, A. & DiLabio, G. ) in press (Elsevier, 2017).
Das, P. K., Samanta, S., McQuarters, A. B., Lehnert, N. & Dey, A. Valence tautomerism in synthetic models of cytochrome P450. Proc. Natl Acad. Sci. USA 113, 6611–6616 (2016).
Hersleth, H.-P., Ryde, U., Rydberg, P., Gö rbitz, C. H. & Andersson, K. K. Structures of the high-valent metalion haem–oxygen intermediates in peroxidases, oxygenases and catalases. J. Inorg. Biochem. 100, 460–476 (2006).
Reimers, J. R., McKemmish, L., McKenzie, R. H. & Hush, N. S. Bond angle variations in XH3 [X = N, P, As, Sb, Bi]: the critical role of Rydberg orbitals exposed using a diabatic state model. Phys. Chem. Chem. Phys. 17, 24618–24640 (2015).
Reimers, J. R., McKemmish, L., McKenzie, R. H. & Hush, N. S. A unified diabatic description for electron transfer reactions, isomerization reactions, proton transfer reactions, and aromaticity. Phys. Chem. Chem. Phys. 17, 24598–25617 (2015).
McKemmish, L. K., McKenzie, R. H., Hush, N. S. & Reimers, J. R. Quantum entanglement between electronic and vibrational degrees of freedom in molecules. J. Chem. Phys. 135, 244110 (2011).
McKemmish, L., McKenzie, R. H., Hush, N. S. & Reimers, J. R. Electron–vibration entanglement in the Born–Oppenheimer description of chemical reactions and spectroscopy. Phys. Chem. Chem. Phys. 17, 24666–24682 (2015).
Reimers, J. R., McKemmish, L., McKenzie, R. H. & Hush, N. S. Non-adiabatic effects in thermochemistry, spectroscopy and kinetics: the general importance of all three Born–Oppenheimer breakdown corrections. Phys. Chem. Chem. Phys. 17, 24640–24665 (2015).
Reimers, J. R., Ford, M. J., Halder, A., Ulstrup, J. & Hush, N. S. Gold surfaces and nanoparticles are protected by Au(O)-thiyl species and are destroyed when Au(I)-thiolates form. Proc. Natl Acad. Sci. USA 113, E1424–E1433 (2016).
Brust, M., Walker, M., Bethell, D., Schiffrin, D. J. & Whyman, R. Synthesis of thiol-derivatized gold nanoparticles in a two-phase liquid-liquid system. J. Chem. Soc., Chem. Commun. 801–802 (1994).
Heister, K., Zharnikov, M., Grunze, M. & Johansson, L. S. O. Adsorption of alkanethiols and biphenylthiols on Au and Ag substrates: a high-resolution X-ray photoelectron spectroscopy study. J. Phys. Chem. B 105, 4058–4061 (2001).
Heister, K., Zharnikov, M., Grunze, M., Johansson, L. S. O. & Ulman, A. Characterization of X-ray induced damage in alkanethiolate monolayers by high-resolution photoelectron spectroscopy. Langmuir 17, 8–11 (2001).
Tanaka, A., Takeda, Y., Imamura, M. & Sato, S. Dynamic final-state effect on the Au 4f core-level photoemission of dodecanethiolate-passivated Au nanoparticles on graphite substrates. Phys. Rev. B 68, 195415 (2003).
Corbierre, M. K. & Lennox, R. B. Preparation of thiol-capped gold nanoparticles by chemical reduction of soluble Au(I)-thiolates. Chem. Mater. 17, 5691–5696 (2005).
Shaporenko, A., Zharnikov, M., Feulner, P. & Menzel, D. Quantitative analysis of temperature effects in radiation damage of thiolate-based self-assembled monolayers. J. Phys. Condens. Matter 18, S1677–S1689 (2006).
Park, E. D. & Lee, J. S. Effects of pretreatment conditions on CO oxidation over supported Au catalysts. J. Catal. 186, 1–11 (1999).
Venezia, A. M. et al. Relationship between structure and CO oxidation activity of ceria-supported gold catalysts. J. Phys. Chem. B 109, 2821–2827 (2005).
Casaletto, M. P., Longo, A., Martorana, A., Prestianni, A. & Venezia, A. M. XPS study of supported gold catalysts: the role of Au0 and Au+δ species as active sites. Surf. Interface Anal. 38, 215–218 (2006).
Mikhlin, Y. L., Nasluzov, V. A., Romanchenko, A. S., Shor, A. M. & Pal'yanova, G. A. XPS and DFT studies of the electronic structures of AgAuS and Ag3AuS2 . J. Alloys Compd. 617, 314–321 (2014).
Liao, L. et al. Structure of chiral Au44(2,4-DMBT)26 nanocluster with an 18-electron shell closure. J. Am. Chem. Soc. 138, 10425–10428 (2016).
McNeillie, A., Brown, D. H., Smith, W. E., Gibson, M. & Watson, L. X-Ray photoelectron spectra of some gold compounds. J. Chem. Soc., Dalton Trans. 767–770 (1980).
Behera, M. & Ram, S. Inquiring the mechanism of formation, encapsulation, and stabilization of gold nanoparticles by poly(vinyl pyrrolidone) molecules in 1-butanol. Appl. Nanosci. 4, 247–254 (2014).
Senthilnathan, J., Rao, K. S., Lin, W.-H., Ting, J.-M. & Yoshimura, M. Formation of reusable Au-acetonitrile polymers and N-doped graphene catalyst under UV light via submerged liquid plasma process. J. Mater. Chem. A 3, 3035–3043 (2015).
Zhang, P. & Sham, T. K. X-Ray studies of the structure and electronic behavior of alkanethiolate-capped gold nanoparticles: the interplay of size and surface effects. Phys. Rev. Lett. 90, 245502 (2003).
de la Llave, E., Clarenc, R., Schiffrin, D. J. & Williams, F. J. Organization of alkane amines on a gold surface: structure, surface dipole, and electron transfer. J. Phys. Chem. C 118, 468–475 (2014).
Chaudhuri, A. et al. The structure of the Au(111)/methylthiolate interface. New insights from near-edge X-ray absorption spectroscopy and X-ray standing waves. J. Chem. Phys. 130, 124708 (2009).
Mikhlin, Y. et al. XAS and XPS examination of the Au–S nanostructures produced via the reduction of aqueous gold(III) by sulfide ions. J. Electron Spectrosc. Relat. Phenom. 177, 24–29 (2010).
Corthey, G. et al. Synthesis and characterization of gold@gold(I)–thiomalate core@shell nanoparticles. ACS Nano 4, 3413–3421 (2010).
Simms, G. A., Padmos, J. D. & Zhang, P. Structural and electronic properties of protein/thiolate-protected gold nanocluster with “staple” motif: a XAS, L-DOS, and XPS study.J. Chem. Phys. 131, 214703 (2009).
Pearson, R. G. Hard and soft acids and bases HSAB. 1. Fundamental principles. J. Chem. Educ. 45, 581–587 (1968).
Pearson, R. G. Hard and soft acids and bases — the evolution of a chemical concept. Coord. Chem. Rev. 100, 403–425 (1990).
Ayers, P. W., Parr, R. G. & Pearson, R. G. Elucidating the hard/soft acid/base principle: a perspective based on half-reactions. J. Chem. Phys. 124, 194107 (2006).
Selinger, B. Chemistry in the Marketplace (Allen & Unwin, 1999).
Barton, E. J. et al. ExoMol molecular line lists V: the ro-vibrational spectra of NaCl and KCl. Mon. Not. R. Astron. Soc. 442, 1821–1829 (2014).
Reckien, W., Eggers, M. & Bredow, T. Theoretical study of the adsorption of benzene on coinage metals. Beilstein J. Org. Chem. 10, 1775–1784 (2014).
Liu, W. et al. Quantitative prediction of molecular adsorption: structure and binding of benzene on coinage metals. Phys. Rev. Lett. 115, 036104 (2015).
Hohman, J. N. et al. Exchange reactions between alkanethiolates and alkaneselenols on Au{111}. J. Am. Chem. Soc. 136, 8110–8121 (2014).
Schmøkel, M. S. et al. Testing the concept of hypervalency: charge density analysis of K2SO4 . Inorg. Chem. 51, 8607–8616 (2012).
Tang, Q. & Jiang, D.-E. Insights into the PhC≡C/Au interface. J. Phys. Chem. C 119, 10804–10810 (2014).
Zaba, T. et al. Formation of highly ordered self-assembled monolayers of alkynes on Au(111) substrate. J. Am. Chem. Soc. 136, 11918–11921 (2014).
Li, Y., Silverton, L. C., Haasch, R. & Tong, Y. Y. Alkanetelluroxide-protected gold nanoparticles. Langmuir 24, 7048–7053 (2008).
Kurashige, W. et al. Au25 clusters containing unoxidized tellurolates in the ligand shell. J. Phys. Chem. Lett. 5, 2072–2076 (2014).
Reimers, J. R., Wang, Y., Cankurtaran, B. O. & Ford, M. J. Chemical analysis of the superatom model for sulfur-stabilized gold nanoparticles. J. Am. Chem. Soc. 132, 8378–8384 (2010).
Zhang, J. & Ulstrup, J. Oxygen-free in situ scanning tunnelling microscopy. J. Electroanal. Chem. 599, 213–220 (2007).
McAdon, M. H. & Goddard, W. A. Charge density waves, spin density waves, and Peierls distortions in one-dimensional metals. 1. Hartree–Fock studies of Cu, Ag, Au, Li, and Na. J. Chem. Phys. 88, 277–302 (1988).
McAdon, M. H. & Goddard, W. A. Charge density waves, spin density waves, and Peierls distortions in one-dimensional metals. 2. Generalized valence bond studies of copper, silver, gold, lithium and sodium. J. Phys. Chem. 92, 1352–1365 (1988).
Fang, J. et al. Recent advances in the synthesis and catalytic applications of ligand-protected, atomically precise metal nanoclusters. Coord. Chem. Rev. 322, 1–29 (2016).
De, M., Ghosh, P. S. & Rotello, V. M. Applications of nanoparticles in biology. Adv. Mater. 20, 4225–4241 (2008).
Sapsford, K. E. et al. Functionalizing nanoparticles with biological molecules: developing chemistries that facilitate nanotechnology. Chem. Rev. 113, 1904–2074 (2013).
Saha, K., Agasti, S. S., Kim, C., Li, X. & Rotello, V. M. Gold nanoparticles in chemical and biological sensing. Chem. Rev. 112, 2739–2779 (2012).
Zhang, L. & Wang, E. Metal nanoclusters: new fluorescent probes for sensors and bioimaging. Nano Today 9, 132–157 (2014).
Muthu, M. S., Agrawal, P. & Singh, S. Theranostic nanomedicine of gold nanoclusters: an emerging platform for cancer diagnosis and therapy. Nanomedicine 11, 327–330 (2016).
Whitesides, G. M. & Laibinis, P. E. Wet chemical approaches to the characterization of organic surfaces: self-assembled monolayers, wetting, and the physical-organic chemistry of the solid-liquid interface. Langmuir 6, 87–96 (1990).
Schmidbaur, H. Ludwig Mond Lecture. High-carat gold compounds. Chem. Soc. Rev. 24, 391–400 (1995).
Love, J. C., Estroff, L. A., Kriebel, J. K., Nuzzo, R. G. & Whitesides, G. M. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem. Rev. 105, 1103–1169 (2005).
Hutchings, G. J., Brust, M. & Schmidbaur, H. Gold — an introductory perspective. Chem. Soc. Rev. 37, 1759–1765 (2008).
Skrabalak, S. E. et al. Gold nanocages: synthesis, properties, and applications. Acc. Chem. Res. 41, 1587–1595 (2008).
Sardar, R., Funston, A. M., Mulvaney, P. & Murray, R. W. Gold nanoparticles: past, present, and future. Langmuir 25, 13840–13851 (2009).
Zhao, P., Li, N. & Astruc, D. State of the art in gold nanoparticle synthesis. Coord. Chem. Rev. 257, 638–665 (2013).
Kurashige, W., Niihori, Y., Sharma, S. & Negishi, Y. Precise synthesis, functionalization and application of thiolate-protected gold clusters. Coord. Chem. Rev. 320–321, 238–250 (2016).
Goswami, N., Yao, Q., Chen, T. & Xie, J. Mechanistic exploration and controlled synthesis of precise thiolate–gold nanoclusters. Coord. Chem. Rev. 329, 1–15 (2016).
Pensa, E. et al. The chemistry of the sulfur–gold interface: in search of a unified model. Acc. Chem. Res. 45, 1183–1192 (2012).
Maksymovych, P., Sorescu, D. C. & Yates, J. T. Jr. Gold-adatom-mediated bonding in self-assembled short-chain alkanethiolate species on the Au(111) surface. Phys. Rev. Lett. 97, 146103 (2006).
Jiang, D.-E., Tiago, M. L., Luo, W. D. & Dai, S. The “staple” motif: a key to stability of thiolate-protected gold nanoclusters. J. Am. Chem. Soc. 130, 2777–2779 (2008).
Wang, Y. et al. Chain-branching control of the atomic structure of alkanethiol-based gold–sulfur interfaces. J. Am. Chem. Soc. 133, 14856–14859 (2011).
Yan, J. et al. Controlling the stereochemistry and regularity of butanethiol self-assembled monolayers on Au(111). J. Am. Chem. Soc. 136, 17087–17094 (2014).
Ouyang, R. et al. Intermixed adatom and surface bound adsorbates in regular self-assembled monolayers of racemic 2-butanethiol on Au(111). ChemPhysChem 16, 928–932 (2015).
Grumelli, D., Maza, F. L., Kern, K., Salvarezza, R. C. & Carro, P. Surface structure and chemistry of alkanethiols on Au(100)-(1×1) substrates. J. Phys. Chem. C 120, 291–296 (2016).
Chadha, R. K., Kumar, R. & Tuck, D. G. The direct electrochemical synthesis of thiolato complexes of copper, silver, and gold; the molecular structure of [Cu(SC6H4CH3-o)(1,10-phenanthroline)]2·CH3CN. Can. J. Chem. 65, 1336–1342 (1987).
Wang, Y., Hush, N. S. & Reimers, J. R. Understanding the chemisorption of 2-methyl-2-propanethiol on Au(111). J. Phys. Chem. C 111, 10878–10885 (2007).
Bandyopadhyay, S., Chattopadhyay, S. & Dey, A. The protonation state of thiols in self-assembled monolayers on roughened Ag/Au surfaces and nanoparticles. Phys. Chem. Chem. Phys. 17, 24866–24873 (2015).
Brust, M., Fink, J., Bethell, D., Schiffrin, D. J. & Kiely, C. Synthesis and reactions of functionalized gold nanoparticles. J. Chem. Soc. Chem. Commun. 1655–1656 (1995).
Romero, E. A., Peltier, J. L., Jazzar, R. & Bertrand, G. Catalyst-free dehydrocoupling of amines, alcohols, and thiols with pinacol borane and 9-borabicyclononane (9-BBN). Chem. Commun. 52, 10563–10565 (2016).
Civit, M. G. et al. Ynones merge activation/conjugate addition of chalcogenoborates ArE-Bpin (E = Se, S). Adv. Synth. Catal. 357, 3098–3103 (2015).
Solé, C. & Fernández, E. Alkoxide activation of aminoboranes towards selective amination. Angew. Chem. Int. Ed. 52, 11351–11355 (2013).
Davis, R. E. & Gottbrath, J. A. Boron hydrides. V. Methanolysis sodium borohydride. J. Am. Chem. Soc. 84, 895–898 (1962).
Negishi, Y. & Tsukuda, T. One-pot preparation of subnanometer-sized gold clusters via reduction and stabilization by meso-2,3-dimercaptosuccinic acid. J. Am. Chem. Soc. 125, 4046–4047 (2003).
Schaaff, T. G., Knight, G., Shafigullin, M. N., Borkman, R. F. & Whetten, R. L. Isolation and selected properties of a 10.4 kDa gold:glutathione cluster compound. J. Phys. Chem. B 102, 10643–10646 (1998).
Goulet, P. J. G. & Lennox, R. B. New insights into Brust–Schiffrin metal nanoparticle synthesis. J. Am. Chem. Soc. 132, 9582–9584 (2010).
Li, Y., Zaluzhna, O. & Tong, Y. J. Critical role of water and the structure of inverse micelles in the Brust–Schiffrin synthesis of metal nanoparticles. Langmuir 27, 7366–7370 (2011).
Perala, S. R. K. & Kumar, S. On the mechanism of metal nanoparticle synthesis in the Brust–Schiffrin method. Langmuir 29, 9863–9873 (2013).
Yu, C. et al. Investigation on the mechanism of the synthesis of gold(I) thiolate complexes by NMR. J. Phys. Chem. C 118, 10434–10440 (2014).
Uehara, A. et al. Electrochemical insight into the Brust–Schiffrin synthesis of Au nanoparticles. J. Am. Chem. Soc. 137, 15135–15144 (2015).
Marbella, L. E. et al. Description and role of bimetallic prenucleation species in the formation of small nanoparticle alloys. J. Am. Chem. Soc. 137, 15852–15858 (2015).
Zhu, L. et al. New insight into intermediate precursors of Brust–Schiffrin gold nanoparticles synthesis. J. Phys. Chem. C 117, 11399–11404 (2013).
Graham, T. R., Renslow, R., Govind, N. & Saunders, S. R. Precursor ion–ion aggregation in the Brust–Schiffrin synthesis of alkanethiol nanoparticles. J. Phys. Chem. C 120, 19837–19847 (2016).
Zaluzhna, O., Li, Y., Zangmeister, C., Allison, T. C. & Tong, Y. J. Mechanistic insights on one-phase versus two-phase Brust–Schiffrin method synthesis of Au nanoparticles with dioctyl-diselenides. Chem. Commun. 48, 362–364 (2012).
Perdew, J. P. & Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. B 45, 13244–13249 (1992).
Otero de la Roza, A. & DiLabio, G. (eds) Noncovalent Interactions in Quantum Chemistry and Physics: Theory and Applications in press (Elsevier, 2017).
Mäkinen, V., Koskinen, P. & Häkkinen, H. Modeling thiolate-protected gold clusters with density-functional tight-binding. Eur. Phys. J. D 67, 38 (2013).
Perdew, J. P., Burke, W. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Bilic, A., Reimers, J. R., Hush, N. S. & Hafner, J. Adsorption of ammonia on the gold (111) surface. J. Chem. Phys. 116, 8981–8987 (2002).
Wang, Y., Hush, N. S. & Reimers, J. R. Formation of gold-methanethiyl self-assembled monolayers. J. Am. Chem. Soc. 129, 14532–14533 (2007).
Cafe, P. F. et al. Chemisorbed and physisorbed structures for 1,10-phenanthroline and dipyrido[3,2-a:2ʹ,3ʹ-c]phenazine on Au(111). J. Phys. Chem. C 111, 17285–17296 (2007).
Towler, M. D. in Computational Methods for Large Systems: Electronic Structure Approaches for Biotechnology and Nanotechnology (ed. Reimers, J. R. ) 119–166 (Wiley, 2011).
Al-Hamdani, Y. S., Ma, M., Alfè, D., von Lilienfeld, O. A. & Michaelides, A. Communication: water on hexagonal boron nitride from diffusion Monte Carlo. J. Chem. Phys. 142, 181101 (2015).
Booth, G. H., Grüneis, A., Kresse, G. & Alavi, A. Towards an exact description of electronic wavefunctions in real solids. Nature 493, 365–370 (2013).
Booth, G. H., Thom, A. J. W. & Alavi, A. Fermion Monte Carlo without fixed nodes: a game of life, death, and annihilation in Slater determinant space. J. Chem. Phys. 131, 054106 (2009).
Dubecký, M., Mitas, L. & Jurecˇka, P. Noncovalent interactions by quantum Monte Carlo. Chem. Rev. 116, 5188–5215 (2016).
Grüneis, A. A coupled cluster and Møller–Plesset perturbation theory study of the pressure induced phase transition in the LiH crystal. J. Chem. Phys. 143, 102817 (2015).
Voloshina, E. & Paulus, B. First multireference correlation treatment of bulk metals. J. Chem. Theory Comput. 10, 1698–1706 (2014).
Olsen, T. & Thygesen, K. S. Beyond the random phase approximation: improved description of short-range correlation by a renormalized adiabatic local density approximation. Phys. Rev. B 88, 115131 (2013).
Gould, T. Communication: beyond the random phase approximation on the cheap: improved correlation energies with the efficient “radial exchange hole” kernel. J. Chem. Phys. 137, 111101 (2012).
Goerigk, L. & Grimme, S. Double-hybrid density functionals. WIREs Comput. Mol. Sci. 4, 576–600 (2014).
Grimme, S., Hansen, A., Brandenburg, J. G. & Bannwarth, C. Dispersion-corrected mean-field electronic structure methods. Chem. Rev. 116, 5105–5154 (2016).
Grimme, S. Semiempirical hybrid density functional with perturbative second-order correlation. J. Chem. Phys. 124, 034108 (2006).
Goerigk, L. & Grimme, S. Efficient and accurate double-hybrid-meta-GGA density functionals — evaluation with the extended GMTKN30 database for general main group thermochemistry, kinetics, and noncovalent interactions. J. Chem. Theory Comput. 7, 291–309 (2011).
Chai, J.-D. & Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 10, 6615–6620 (2008).
Yanai, T., Tew, D. P. & Handy, N. C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 393, 51–57 (2004).
Cai, Z.-L., Crossley, M. J., Reimers, J. R., Kobayashi, R. & Amos, R. D. Density-functional theory for charge-transfer: the nature of the N-bands of porphyrins and chlorophylls revealed through CAM-B3LYP, CASPT2, and SAC-CI calculations. J. Phys. Chem. B 110, 15624–15632 (2006).
Zhao, Y. & Truhlar, D. G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Grimme, S., Ehrlich, S. & Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 32, 1456–1465 (2011).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Tkatchenko, A., Distasio, R. A., Car, R. & Scheffler, M. Accurate and efficient method for many-body van der Waals interactions. Phys. Rev. Lett. 108, 236402 (2012).
Ambrosetti, A., Reilly, A. M., Distasio Jr, R. A. & Tkatchenko, A. Long-range correlation energy calculated from coupled atomic response functions. J. Chem. Phys. 140, 18a508 (2014).
Christian, M. S., Otero-de-la-Roza, A. & Johnson, E. R. Surface adsorption from the exchange–hole dipole moment dispersion model. J. Chem. Theory Comput. 12, 3305–3315 (2016).
Vydrov, O. A. & Van Voorhis, T. Nonlocal van der Waals density functional: the simpler the better. J. Chem. Phys. 133, 244103 (2010).
Dion, M., Rydberg, H., Schrö der, E., Langreth, D. C. & Lundqvist, B. I. Van der Waals density functional for general geometries. Phys. Rev. Lett. 92, 246401–246401 (2004).
Lee, K., Murray, É. D., Kong, L., Lundqvist, B. I. & Langreth, D. C. Higher-accuracy van der Waals density functional. Phys. Rev. B 82, 081101 (2010).
Erhard, J., Bleiziffer, P. & Görling, A. Power series approximation for the correlation kernel leading to Kohn–Sham methods combining accuracy, computational efficiency, and general applicability. Phys. Rev. Lett. 117, 143002 (2016).
Eshuis, H., Bates, J. E. & Furche, F. Electron correlation methods based on the random phase approximation. Theor. Chem. Acc. 131, 1084 (2012).
Gillan, M. J., Alfè, D., Bygrave, P. J., Taylor, C. R. & Manby, F. R. Energy benchmarks for water clusters and ice structures from an embedded many-body expansion. J. Chem. Phys. 139, 114101 (2013).
Paulus, B. The method of increments — a wavefunction-based ab initio correlation method for solids. Phys. Rep. 428, 1–52 (2006).
Muller, C. & Paulus, B. Wavefunction-based electron correlation methods for solids. Phys. Chem. Chem. Phys. 14, 7605–7614 (2012).
Stoll, H., Paulus, B. & Fulde, P. An incremental coupled-cluster approach to metallic lithium. Chem. Phys. Lett. 469, 90–93 (2009).
de Lara-Castells, M. P., Mitrushchenkov, A. O. & Stoll, H. Combining density functional and incremental post-Hartree–Fock approaches for van der Waals dominated adsorbate–surface interactions: Ag2/graphene. J. Chem. Phys. 143, 102804 (2015).
de Lara-Castells, M. P. et al. A combined periodic density functional and incremental wave-function-based approach for the dispersion-accounting time-resolved dynamics of 4He nanodroplets on surfaces: 4He/graphene. J. Chem. Phys. 141, 151102 (2014).
Rolik, Z., Szegedy, L., Ladjánszki, I., Ladóczki, B. & Kállay, M. An efficient linear-scaling CCSD(T) method based on local natural orbitals. J. Chem. Phys. 139, 094105 (2013).
Kruse, H. & Grimme, S. A geometrical correction for the inter- and intra-molecular basis set superposition error in Hartree–Fock and density functional theory calculations for large systems. J. Chem. Phys. 136, 154101 (2012).
Goerigk, L. & Reimers, J. R. Efficient methods for the quantum chemical treatment of protein structures: the effects of London-dispersion and basis-set incompleteness on peptide and water-cluster geometries. J. Chem. Theory Comput. 9, 3240–3251 (2013).
Fedorov, D. G., Nagata, T. & Kitaura, K. Exploring chemistry with the fragment molecular orbital method. Phys. Chem. Chem. Phys. 14, 7562–7577 (2012).
Canfield, P., Dahlbom, M. G., Reimers, J. R. & Hush, N. S. Density-functional geometry optimization of the 150000-atom photosystem-I trimer. J. Chem. Phys. 124, 024301 (2006).
Tomasi, J., Mennucci, B. & Cammi, R. Quantum mechanical continuum solvation models. Chem. Rev. 105, 2999–3093 (2005).
Floris, F. M., Tomasi, J. & Pascual Ahuir, J. L. Dispersion and repulsion contributions to the solvation energy: refinements to a simple computational model in the continuum approximation. J. Computat. Chem. 12, 784–791 (1991).
Reimers, J. R. et al. From chaos to order: chain-length dependence of the free energy of formation of tetraalkylporphyrin self-assembled monolayer polymorphs J. Phys. Chem. C 120, 1739–1748 (2016).
Reimers, J. R. et al. A priori calculations of the free energy of formation from solution of polymorphic self-assembled monolayers. Proc. Natl Acad. Sci. USA 112, E6101–E6110 (2015).
Goerigk, L. & Grimme, S. A thorough benchmark of density functional methods for general main group thermochemistry, kinetics, and noncovalent interactions. Phys. Chem. Chem. Phys. 13, 6670–6688 (2011).
Fajín, J. L. C., Teixeira, F., Gomes, J. R. B. & Cordeiro, M. N. D. S. Effect of van der Waals interactions in the DFT description of self-assembled monolayers of thiols on gold. Theor. Chem. Acc. 134, 67 (2015).
Geim, A. K. & Grigorieva, I. V. Van der Waals heterostructures. Nature 499, 419–425 (2013).
Voloshina, E. & Dedkov, Y. S. Graphene on metallic surfaces: problems and perspectives. Phys. Chem. Chem. Phys. 14, 13502–13514 (2012).
Gao, W. & Tkatchenko, A. Sliding mechanisms in multilayered hexagonal boron nitride and graphene: The effects of directionality, thickness, and sliding constraints. Phys. Rev. Lett. 114, 096101 (2015).
Jadzinsky, P. D., Calero, G., Ackerson, C. J., Bushnell, D. A. & Kornberg, R. D. Structure of a thiol monolayer-protected gold nanoparticle at 1. 1 Å resolution. Science 318, 430–433 (2007).
Acknowledgements
Financial support from the Australian Research Council Discovery Projects grant DP160101301, and computational support from National Computational Infrastructure (d63 and no2) and INTERSECT (r88 and sb4) are gratefully acknowledged. The authors also acknowledge helpful discussions with J. Zhang and Q. Chi, Technical University of Denmark.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Reimers, J., Ford, M., Marcuccio, S. et al. Competition of van der Waals and chemical forces on gold–sulfur surfaces and nanoparticles. Nat Rev Chem 1, 0017 (2017). https://doi.org/10.1038/s41570-017-0017
Published:
DOI: https://doi.org/10.1038/s41570-017-0017
- Springer Nature Limited
This article is cited by
-
Determination of urinary spermine using controlled dissolution of polysulfide modified gold electrode
Microchimica Acta (2023)
-
Interaction Between Two Ground-State Atoms
International Journal of Theoretical Physics (2021)
-
Enabling long-lived organic room temperature phosphorescence in polymers by subunit interlocking
Nature Communications (2019)
-
Non-chemisorbed gold–sulfur binding prevails in self-assembled monolayers
Nature Chemistry (2019)
-
Toxic effects and biodistribution of ultrasmall gold nanoparticles
Archives of Toxicology (2017)