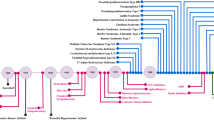
Abstract
The known genetic architecture of blood pressure now comprises >30 genes, with rare variants resulting in monogenic forms of hypertension or hypotension and >1,477 common single-nucleotide polymorphisms (SNPs) being associated with the blood pressure phenotype. Monogenic blood pressure syndromes predominantly involve the renin–angiotensin–aldosterone system and the adrenal glucocorticoid pathway, with a smaller fraction caused by neuroendocrine tumours of the sympathetic and parasympathetic nervous systems. The SNPs identified in genome-wide association studies (GWAS) as being associated with the blood pressure phenotype explain only approximately 27% of the 30–50% estimated heritability of blood pressure, and the effect of each SNP on the blood pressure phenotype is small. A paucity of SNPs from GWAS are mapped to known genes causing monogenic blood pressure syndromes. For example, a GWAS signal mapped to the gene encoding uromodulin has been shown to affect blood pressure by influencing sodium homeostasis, and the effects of another GWAS signal were mediated by endothelin. However, the majority of blood pressure-associated SNPs show pleiotropic associations. Unravelling these associations can potentially help us to understand the underlying biological pathways. In this Review, we appraise the current knowledge of blood pressure genomics, explore the causal pathways for hypertension identified in Mendelian randomization studies and highlight the opportunities for drug repurposing and pharmacogenomics for the treatment of hypertension.
Key points
-
The genetic architecture of blood pressure encompasses approximately 30 genes, with rare variants involved in blood pressure dysregulation and >1,477 common single-nucleotide polymorphisms (SNPs) associated with blood pressure.
-
Monogenic forms of blood pressure disorders involve both germline and somatic variants, with the latter predominantly seen in patients with neoplasms associated with hypertension.
-
Most of the SNPs identified in genome-wide association studies (GWAS) as being associated with blood pressure are pleiotropic and mapped to non-coding regions of the genome, which makes functional mapping challenging.
-
Although translating GWAS signals into causal mechanisms and clinical applications has been slow, Mendelian randomization studies are substantially improving our understanding of known epidemiological correlations between blood pressure and other traits.
-
Pharmacogenomic studies of drug–gene interactions might offer a route to the early clinical translation of GWAS signals, with two clinical trials involving blood pressure GWAS SNPs currently ongoing.



Similar content being viewed by others
References
Canon, W. B. Organization for physiological homeostasis. Physiol. Rev. 9, 399–431 (1929).
Williams, B. et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur. Heart J. 39, 3021–3104 (2018).
Lewington, S. et al. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 360, 1903–1913 (2002).
Whelton, P. K. et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 138, e426–e483 (2018).
Evans, J. G. & Rose, G. Hypertension. Br. Med. Bull. 27, 37–42 (1971).
Forouzanfar, M. H. et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990-2015. JAMA 317, 165–182 (2017).
Frohlich, E. D., Dustan, H. P. & Bumpus, F. M. Irvine H. Page: 1901-1991. The celebration of a leader. Hypertension 18, 443–445 (1991).
Padmanabhan, S., Caulfield, M. & Dominiczak, A. F. Genetic and molecular aspects of hypertension. Circ. Res. 116, 937–959 (2015).
Franklin, S. S. et al. Hemodynamic patterns of age-related changes in blood pressure. The Framingham Heart Study. Circulation 96, 308–315 (1997).
Carvalho, J. J. et al. Blood pressure in four remote populations in the INTERSALT Study. Hypertension 14, 238–246 (1989).
Oliver, W. J., Cohen, E. L. & Neel, J. V. Blood pressure, sodium intake, and sodium related hormones in the Yanomamo Indians, a “no-salt” culture. Circulation 52, 146–151 (1975).
Bursztyn, M. Occupational and environmental influences on hypertension. J. Hum. Hypertens. 34, 202–206 (2020).
Padmanabhan, S. & Joe, B. Towards precision medicine for hypertension: a review of genomic, epigenomic, and microbiomic effects on blood pressure in experimental rat models and humans. Physiol. Rev. 97, 1469–1528 (2017).
Evangelou, E. et al. Genetic analysis of over 1 million people identifies 535 new loci associated with blood pressure traits. Nat. Genet. 50, 1412–1425 (2018).
Giri, A. et al. Trans-ethnic association study of blood pressure determinants in over 750,000 individuals. Nat. Genet. 51, 51–62 (2019).
Havlik, R. J. et al. Blood pressure aggregation in families. Am. J. Epidemiol. 110, 304–312 (1979).
Kupper, N. et al. Heritability of daytime ambulatory blood pressure in an extended twin design. Hypertension 45, 80–85 (2005).
Salfati, E., Morrison, A. C., Boerwinkle, E. & Chakravarti, A. Direct estimates of the genomic contributions to blood pressure heritability within a population-based cohort (ARIC). PLoS One 10, e0133031 (2015).
Luft, F. C. Twins in cardiovascular genetic research. Hypertension 37, 350–356 (2001).
Niiranen, T. J. et al. Risk for hypertension crosses generations in the community: a multi-generational cohort study. Eur. Heart J. 38, 2300–2308 (2017).
Ference, B. A. et al. Clinical effect of naturally random allocation to lower systolic blood pressure beginning before the development of hypertension. Hypertension 63, 1182–1188 (2014).
Timpson, N. J., Greenwood, C. M. T., Soranzo, N., Lawson, D. J. & Richards, J. B. Genetic architecture: the shape of the genetic contribution to human traits and disease. Nat. Rev. Genet. 19, 110–124 (2018).
Zeng, J. et al. Signatures of negative selection in the genetic architecture of human complex traits. Nat. Genet. 50, 746–753 (2018).
Funder, J. W. Primary aldosteronism. Hypertension 74, 458–466 (2019).
Ji, W. et al. Rare independent mutations in renal salt handling genes contribute to blood pressure variation. Nat. Genet. 40, 592–599 (2008).
Baranowski, E. S., Arlt, W. & Idkowiak, J. Monogenic disorders of adrenal steroidogenesis. Horm. Res. Paediatr. 89, 292–310 (2018).
Ceccato, F. & Mantero, F. Monogenic forms of hypertension. Endocrinol. Metab. Clin. North Am. 48, 795–810 (2019).
Seidel, E., Schewe, J. & Scholl, U. I. Genetic causes of primary aldosteronism. Exp. Mol. Med. 51, 131 (2019).
Tadjine, M., Lampron, A., Ouadi, L. & Bourdeau, I. Frequent mutations of beta-catenin gene in sporadic secreting adrenocortical adenomas. Clin. Endocrinol. 68, 264–270 (2008).
Seyberth, H. W., Weber, S. & Komhoff, M. Bartter’s and Gitelman’s syndrome. Curr. Opin. Pediatr. 29, 179–186 (2017).
Laghmani, K. et al. Polyhydramnios, transient antenatal Bartter’s syndrome, and MAGED2 mutations. N. Engl. J. Med. 374, 1853–1863 (2016).
Maass, P. G. et al. PDE3A mutations cause autosomal dominant hypertension with brachydactyly. Nat. Genet. 47, 647–653 (2015).
Pillai, S., Gopalan, V., Smith, R. A. & Lam, A. K. Updates on the genetics and the clinical impacts on phaeochromocytoma and paraganglioma in the new era. Crit. Rev. Oncol. Hematol. 100, 190–208 (2016).
Roman-Gonzalez, A. & Jimenez, C. Malignant pheochromocytoma-paraganglioma: pathogenesis, TNM staging, and current clinical trials. Curr. Opin. Endocrinol. Diabetes Obes. 24, 174–183 (2017).
Dahia, P. L. Pheochromocytoma and paraganglioma pathogenesis: learning from genetic heterogeneity. Nat. Rev. Cancer 14, 108–119 (2014).
Fishbein, L. et al. Comprehensive molecular characterization of pheochromocytoma and paraganglioma. Cancer Cell 31, 181–193 (2017).
Gusev, A. et al. Integrative approaches for large-scale transcriptome-wide association studies. Nat. Genet. 48, 245–252 (2016).
Mancuso, N. et al. Integrating gene expression with summary association statistics to identify genes associated with 30 complex traits. Am. J. Hum. Genet. 100, 473–487 (2017).
Boyle, E. A., Li, Y. I. & Pritchard, J. K. An expanded view of complex traits: from polygenic to omnigenic. Cell 169, 1177–1186 (2017).
Freund, M. K. et al. Phenotype-specific enrichment of mendelian disorder genes near GWAS regions across 62 complex traits. Am. J. Hum. Genet. 103, 535–552 (2018).
Visscher, P. M. et al. 10 Years of GWAS discovery: biology, function, and translation. Am. J. Hum. Genet. 101, 5–22 (2017).
Martin, A. R. et al. Clinical use of current polygenic risk scores may exacerbate health disparities. Nat. Genet. 51, 584–591 (2019).
Claussnitzer, M. et al. A brief history of human disease genetics. Nature 577, 179–189 (2020).
Padmanabhan, S. et al. Genome-wide association study of blood pressure extremes identifies variant near UMOD associated with hypertension. PLoS Genet. 6, e1001177 (2010).
Graham, L. A. et al. Validation of uromodulin as a candidate gene for human essential hypertension. Hypertension 63, 551–558 (2014).
Trudu, M. et al. Common noncoding UMOD gene variants induce salt-sensitive hypertension and kidney damage by increasing uromodulin expression. Nat. Med. 19, 1655–1660 (2013).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03354897?term=NCT03354897 (2017).
Gupta, R. M. et al. A genetic variant associated with five vascular diseases is a distal regulator of endothelin-1 gene expression. Cell 170, 522–533.e15 (2017).
Nikpay, M. et al. A comprehensive 1,000 Genomes-based genome-wide association meta-analysis of coronary artery disease. Nat. Genet. 47, 1121–1130 (2015).
Dhaun, N. & Webb, D. J. Endothelins in cardiovascular biology and therapeutics. Nat. Rev. Cardiol. 16, 491–502 (2019).
Barton, M. & Yanagisawa, M. Endothelin: 30 years from discovery to therapy. Hypertension 74, 1232–1265 (2019).
Iglarz, M. et al. Pharmacology of macitentan, an orally active tissue-targeting dual endothelin receptor antagonist. J. Pharmacol. Exp. Ther. 327, 736–745 (2008).
Verweij, P., Danaietash, P., Flamion, B., Menard, J. & Bellet, M. Randomized dose-response study of the new dual endothelin receptor antagonist aprocitentan in hypertension. Hypertension 75, 956–965 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03541174 (2018).
Ford, T. J. et al. Genetic dysregulation of endothelin-1 is implicated in coronary microvascular dysfunction. Eur. Heart J. 41, 3239–3252 (2020).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT04097314?term=NCT04097314 (2019).
Ren, M. et al. The biological impact of blood pressure-associated genetic variants in the natriuretic peptide receptor C gene on human vascular smooth muscle. Hum. Mol. Genet. 27, 199–210 (2018).
Ng, F. L. et al. Increased NBCn1 expression, Na+/HCO3- co-transport and intracellular pH in human vascular smooth muscle cells with a risk allele for hypertension. Hum. Mol. Genet. 26, 989–1002 (2017).
Zhang, R. et al. A blood pressure-associated variant of the SLC39A8 gene influences cellular cadmium accumulation and toxicity. Hum. Mol. Genet. 25, 4117–4126 (2016).
International Schizophrenia Consortium, et al. Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature 460, 748–752 (2009).
Torkamani, A., Wineinger, N. E. & Topol, E. J. The personal and clinical utility of polygenic risk scores. Nat. Rev. Genet. 19, 581–590 (2018).
International Consortium for Blood Pressure Genome-Wide Association Studies, et al. Genetic variants in novel pathways influence blood pressure and cardiovascular disease risk. Nature 478, 103–109 (2011).
Ettehad, D. et al. Blood pressure lowering for prevention of cardiovascular disease and death: a systematic review and meta-analysis. Lancet 387, 957–967 (2016).
Ference, B. A. et al. Association of genetic variants related to combined exposure to lower low-density lipoproteins and lower systolic blood pressure with lifetime risk of cardiovascular disease. JAMA 322, 1381–1391 (2019).
Khera, A. V. et al. Genetic risk, adherence to a healthy lifestyle, and coronary disease. N. Engl. J. Med. 375, 2349–2358 (2016).
Knowles, J. W. et al. Impact of a genetic risk score for coronary artery disease on reducing cardiovascular risk: a pilot randomized controlled study. Front. Cardiovasc. Med. 4, 53 (2017).
Loos, R. J. Genetics: genome-wide risk profiles - will they change your life(style)? Nat. Rev. Endocrinol. 7, 252–254 (2011).
Pazoki, R. et al. Genetic predisposition to high blood pressure and lifestyle factors: associations with midlife blood pressure levels and cardiovascular events. Circulation 137, 653–661 (2018).
Marquez-Luna, C., Loh, P. R., South Asian Type 2 Diabetes (SAT2D) Consortium; SIGMA Type 2 Diabetes Consortium & Price, A. L. Multiethnic polygenic risk scores improve risk prediction in diverse populations. Genet. Epidemiol. 41, 811–823 (2017).
Gurdasani, D., Barroso, I., Zeggini, E. & Sandhu, M. S. Genomics of disease risk in globally diverse populations. Nat. Rev. Genet. 20, 520–535 (2019).
Chatterjee, N., Shi, J. & Garcia-Closas, M. Developing and evaluating polygenic risk prediction models for stratified disease prevention. Nat. Rev. Genet. 17, 392–406 (2016).
MacArthur, J. et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 45, D896–D901 (2017).
Kamat, M. A. et al. PhenoScanner V2: an expanded tool for searching human genotype-phenotype associations. Bioinformatics 35, 4851–4853 (2019).
Pingault, J. B. et al. Using genetic data to strengthen causal inference in observational research. Nat. Rev. Genet. 19, 566–580 (2018).
Neter, J. E., Stam, B. E., Kok, F. J., Grobbee, D. E. & Geleijnse, J. M. Influence of weight reduction on blood pressure: a meta-analysis of randomized controlled trials. Hypertension 42, 878–884 (2003).
Timpson, N. J. et al. Does greater adiposity increase blood pressure and hypertension risk?: Mendelian randomization using the FTO/MC4R genotype. Hypertension 54, 84–90 (2009).
Bell, J. A. et al. Associations of body mass and fat indexes with cardiometabolic traits. J. Am. Coll. Cardiol. 72, 3142–3154 (2018).
Roerecke, M. et al. The effect of a reduction in alcohol consumption on blood pressure: a systematic review and meta-analysis. Lancet Public Health 2, e108–e120 (2017).
Macgregor, S. et al. Associations of ADH and ALDH2 gene variation with self report alcohol reactions, consumption and dependence: an integrated analysis. Hum. Mol. Genet. 18, 580–593 (2009).
Chen, L., Smith, G. D., Harbord, R. M. & Lewis, S. J. Alcohol intake and blood pressure: a systematic review implementing a Mendelian randomization approach. PLoS Med. 5, e52 (2008).
Holmes, M. V. et al. Association between alcohol and cardiovascular disease: Mendelian randomisation analysis based on individual participant data. BMJ 349, g4164 (2014).
Millwood, I. Y. et al. Conventional and genetic evidence on alcohol and vascular disease aetiology: a prospective study of 500 000 men and women in China. Lancet 393, 1831–1842 (2019).
Horikoshi, M. et al. Genome-wide associations for birth weight and correlations with adult disease. Nature 538, 248–252 (2016).
Barker, D. J., Osmond, C., Golding, J., Kuh, D. & Wadsworth, M. E. Growth in utero, blood pressure in childhood and adult life, and mortality from cardiovascular disease. BMJ 298, 564–567 (1989).
Warrington, N. M. et al. Maternal and fetal genetic effects on birth weight and their relevance to cardio-metabolic risk factors. Nat. Genet. 51, 804–814 (2019).
Langenberg, C., Hardy, R., Kuh, D. & Wadsworth, M. E. Influence of height, leg and trunk length on pulse pressure, systolic and diastolic blood pressure. J. Hypertens. 21, 537–543 (2003).
Bourgeois, B. et al. Associations between height and blood pressure in the United States population. Medicine 96, e9233 (2017).
London, G. M., Guerin, A. P., Pannier, B. M., Marchais, S. J. & Metivier, F. Body height as a determinant of carotid pulse contour in humans. J. Hypertens. Suppl. 10, S93–S95 (1992).
Lai, F. Y. et al. Adult height and risk of 50 diseases: a combined epidemiological and genetic analysis. BMC Med. 16, 187 (2018).
Marouli, E. et al. Mendelian randomisation analyses find pulmonary factors mediate the effect of height on coronary artery disease. Commun. Biol. 2, 119 (2019).
Shrine, N. et al. New genetic signals for lung function highlight pathways and chronic obstructive pulmonary disease associations across multiple ancestries. Nat. Genet. 51, 481–493 (2019).
Palatini, P. Role of elevated heart rate in the development of cardiovascular disease in hypertension. Hypertension 58, 745–750 (2011).
Larsson, S. C., Drca, N., Mason, A. M. & Burgess, S. Resting heart rate and cardiovascular disease. Circ. Genom. Precis. Med. 12, e002459 (2019).
Wheeler, J. G., Mussolino, M. E., Gillum, R. F. & Danesh, J. Associations between differential leucocyte count and incident coronary heart disease: 1764 incident cases from seven prospective studies of 30,374 individuals. Eur. Heart J. 25, 1287–1292 (2004).
Lassale, C. et al. Elements of the complete blood count associated with cardiovascular disease incidence: Findings from the EPIC-NL cohort study. Sci. Rep. 8, 3290 (2018).
Shah, A. D., Denaxas, S., Nicholas, O., Hingorani, A. D. & Hemingway, H. Low eosinophil and low lymphocyte counts and the incidence of 12 cardiovascular diseases: a CALIBER cohort study. Open Heart 3, e000477 (2016).
Jae, S. Y. et al. Higher blood hematocrit predicts hypertension in men. J. Hypertens. 32, 245–250 (2014).
Paul, L. et al. Hematocrit predicts long-term mortality in a nonlinear and sex-specific manner in hypertensive adults. Hypertension 60, 631–638 (2012).
Atsma, F. et al. Hemoglobin level is positively associated with blood pressure in a large cohort of healthy individuals. Hypertension 60, 936–941 (2012).
Schaffer, A. et al. Impact of red blood cells count on the relationship between high density lipoproteins and the prevalence and extent of coronary artery disease: a single centre study [corrected]. J. Thromb. Thrombolysis 40, 61–68 (2015).
Astle, W. J. et al. The allelic landscape of human blood cell trait variation and links to common complex disease. Cell 167, 1415–1429.e19 (2016).
Gladwin, M. T., Crawford, J. H. & Patel, R. P. The biochemistry of nitric oxide, nitrite, and hemoglobin: role in blood flow regulation. Free Radic. Biol. Med. 36, 707–717 (2004).
Forstermann, U., Xia, N. & Li, H. Roles of vascular oxidative stress and nitric oxide in the pathogenesis of atherosclerosis. Circ. Res. 120, 713–735 (2017).
Natanson, C., Kern, S. J., Lurie, P., Banks, S. M. & Wolfe, S. M. Cell-free hemoglobin-based blood substitutes and risk of myocardial infarction and death: a meta-analysis. JAMA 299, 2304–2312 (2008).
Sansanayudh, N. et al. Mean platelet volume and coronary artery disease: a systematic review and meta-analysis. Int. J. Cardiol. 175, 433–440 (2014).
Kaplan, G. A. & Keil, J. E. Socioeconomic factors and cardiovascular disease: a review of the literature. Circulation 88, 1973–1998 (1993).
Davies, N. M., Dickson, M., Davey Smith, G., van den Berg, G. J. & Windmeijer, F. The causal effects of education on health outcomes in the UK Biobank. Nat. Hum. Behav. 2, 117–125 (2018).
Carter, A. R. et al. Understanding the consequences of education inequality on cardiovascular disease: Mendelian randomisation study. BMJ 365, l1855 (2019).
Walker, V. M., Kehoe, P. G., Martin, R. M. & Davies, N. M. Repurposing antihypertensive drugs for the prevention of Alzheimer’s disease: a Mendelian randomization study. Int. J. Epidemiol. https://doi.org/10.1093/ije/dyz155 (2019).
Padmanabhan, S., Aman, A. & Dominiczak, A. F. Recent findings in the genetics of blood pressure: how to apply in practice or is a moonshot required? Curr. Hypertens. Rep. 20, 54 (2018).
Nelson, M. R. et al. The support of human genetic evidence for approved drug indications. Nat. Genet. 47, 856–860 (2015).
Wishart, D. S. et al. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 46, D1074–D1082 (2018).
Davis, A. P. et al. The Comparative Toxicogenomics Database: update 2019. Nucleic Acids Res. 47, D948–D954 (2019).
Pushpakom, S. et al. Drug repurposing: progress, challenges and recommendations. Nat. Rev. Drug Discov. 18, 41–58 (2019).
Jhamb, D., Magid-Slav, M., Hurle, M. R. & Agarwal, P. Pathway analysis of GWAS loci identifies novel drug targets and repurposing opportunities. Drug Discov. Today 24, 1232–1236 (2019).
Tragante, V. et al. Druggability of coronary artery disease risk loci. Circ. Genom. Precis. Med. 11, e001977 (2018).
Stasch, J. P., Pacher, P. & Evgenov, O. V. Soluble guanylate cyclase as an emerging therapeutic target in cardiopulmonary disease. Circulation 123, 2263–2273 (2011).
O’Connor, C. M. et al. Effect of nesiritide in patients with acute decompensated heart failure. N. Engl. J. Med. 365, 32–43 (2011).
Cowie, M. R. & Fisher, M. SGLT2 inhibitors: mechanisms of cardiovascular benefit beyond glycaemic control. Nat. Rev. Cardiol. (2020).
Rizvi, S. M. et al. Invokana (Canagliflozin) as a dual inhibitor of acetylcholinesterase and sodium glucose co-transporter 2: advancement in Alzheimer’s disease-diabetes type 2 linkage via an enzoinformatics study. CNS Neurol. Disord. Drug Targets 13, 447–451 (2014).
DrugBank. Olanzapine. DrugBank https://www.drugbank.ca/drugs/DB00334 (2020).
DrugBank. Topiramate. DrugBank https://www.drugbank.ca/drugs/DB00273 (2020).
Minari, J., Brothers, K. B. & Morrison, M. Tensions in ethics and policy created by National Precision Medicine Programs. Hum. Genomics 12, 22 (2018).
Krzywinski, M. et al. Circos: an information aesthetic for comparative genomics. Genome Res. 19, 1639–1645 (2009).
Acknowledgements
The authors thank Stefanie Lip (University of Glasgow, UK) for designing Fig. 1 for submission. S.P. is funded by the Medical Research Council (MR/M016560/1), the British Heart Foundation (PG/12/85/29925, CS/16/1/31878, RE/18/6/34217), Health Data Research UK and Chief Scientist Office, Scotland. A.F.D. acknowledges funding from UK Research and Innovation Strength in Places Fund (SIPF) 35049.
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, discussed its content, wrote the manuscript, and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Cardiology thanks D. Arnett, M. Irvin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Comparative Toxicogenomics Database: http://ctdbase.org/
DrugBank: https://www.drugbank.com/
GWAS Catalog: https://www.ebi.ac.uk/gwas/
PhenoScanner: http://www.phenoscanner.medschl.cam.ac.uk/
Trans-Omics for Precision Medicine programme: https://www.nhlbi.nih.gov/science/trans-omics-precision-medicine-topmed-program
Glossary
- Genome-wide association studies
-
(GWAS). Hypothesis-free methods that involve rapidly scanning hundreds of thousands of common genetic variations across the DNA of large numbers of individuals to find genetic variants that are associated with a particular disease.
- Single-nucleotide polymorphisms
-
(SNPs). Naturally occurring, single-base substitutions in the human genome with a population frequency >1%. SNPs occur approximately once every 1,000 nucleotides throughout the genome, which means that roughly 4–5 million SNPs occur in a person’s genome.
- Purifying selection
-
Natural selection can be of two types based on its effect on the fate of genetic variations: purifying (negative) selection and positive (Darwinian) selection. Purifying selection is the most prevalent form of selection because it leads to the constant elimination of deleterious variants that are produced in each generation.
- Phenome-wide association studies
-
(PheWAS). Studies in which the association between single-nucleotide polymorphisms (SNPs) or other types of DNA variant is tested across a large number of different phenotypes. The direction of inference in a PheWAS is from a SNP to multiple phenotypes, whereas in genome-wide association studies it is from one phenotype to multiple SNPs.
- Pleiotropy
-
A phenomenon whereby a genetic variant influences multiple traits and can involve a variant having effects on two or more traits via independent pathways (variants in FBN1 cause Marfan syndrome, with abnormalities in the heart, blood vessels, eyes, bones and joints) or because the effect on one trait is causally related to variation in another trait (variants that increase LDL-cholesterol levels are also associated with coronary artery disease).
- Heterozygote advantage
-
When the heterozygous genotype has a higher relative fitness than either the homozygous dominant or homozygous recessive genotype. A classic example is sickle-cell anaemia, in which sickle-cell phenotype carriers have a heterozygote advantage over the reproductive fitness of normal homozygotes in malaria-endemic regions.
- Tag SNPs
-
Linkage disequilibrium results in a high degree of correlation among nearby single-nucleotide polymorphisms (SNPs), whereby most SNP sites convey redundant information and can be omitted for cost-effectiveness during genotyping. Tag SNPs are used to tag a particular haplotype in a region of the genome and genome-wide association studies (GWAS) SNP arrays use a set of representative (tag) SNPs that sufficiently represent the genomic diversity in the study population.
- Linkage disequilibrium
-
The non-random association of alleles at two or more loci in a general population. This property has multiple uses, including detecting sites of past selection in human populations. The fine-scale pattern of linkage disequilibrium shows that the human genome is composed of haplotype blocks within which most or all single-nucleotide polymorphisms (SNPs) are in high linkage disequilibrium, which led to the development of efficient designs of SNP arrays for genome-wide association studies.
- Polygenic risk score
-
A single-value estimate of an individual’s genetic liability to a phenotype, calculated as a sum of the genome-wide genotypes weighted by corresponding genotype effect size estimates derived from genome-wide association studies (GWAS) data. Classic polygenic risk scores include a reduced set of single-nucleotide polymorphisms (SNPs), for instance, only SNPs with a GWAS P value below a specified threshold. Other methods include millions of SNPs, explicitly modelling the correlation structure between SNPs without identifying a minimal subset of SNPs for prediction.
- Mendelian randomization
-
Studies using genetic variation as a natural experiment to investigate the causal relationships between potentially modifiable risk factors and diseases or phenotypes. The idea behind Mendelian randomization is that, because genetic variants are fixed at conception, they are not affected by confounding or reverse causation, which blight causal inferences in conventional observational studies.
Rights and permissions
About this article
Cite this article
Padmanabhan, S., Dominiczak, A.F. Genomics of hypertension: the road to precision medicine. Nat Rev Cardiol 18, 235–250 (2021). https://doi.org/10.1038/s41569-020-00466-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-020-00466-4
- Springer Nature Limited
This article is cited by
-
Hypertension is a genetic condition—a quantum dilemma
Journal of Human Hypertension (2024)
-
Decreased CNNM2 expression in prefrontal cortex affects sensorimotor gating function, cognition, dendritic spine morphogenesis and risk of schizophrenia
Neuropsychopharmacology (2024)
-
Investigation and management of young-onset hypertension: British and Irish hypertension society position statement
Journal of Human Hypertension (2024)
-
Precision Cardio-oncology: Update on Omics-Based Diagnostic Methods
Current Treatment Options in Oncology (2024)
-
An individualized Bayesian method for estimating genomic variants of hypertension
BMC Genomics (2023)