Abstract
Artificial ocean fertilization (AOF) aims to safely stimulate phytoplankton growth in the ocean and enhance carbon sequestration. AOF carbon sequestration efficiency appears lower than natural ocean fertilization processes due mainly to the low bioavailability of added nutrients, along with low export rates of AOF-produced biomass to the deep ocean. Here we explore the potential application of engineered nanoparticles (ENPs) to overcome these issues. Data from 123 studies show that some ENPs may enhance phytoplankton growth at concentrations below those likely to be toxic in marine ecosystems. ENPs may also increase bloom lifetime, boost phytoplankton aggregation and carbon export, and address secondary limiting factors in AOF. Life-cycle assessment and cost analyses suggest that net CO2 capture is possible for iron, SiO2 and Al2O3 ENPs with costs of 2–5 times that of conventional AOF, whereas boosting AOF efficiency by ENPs should substantially enhance net CO2 capture and reduce these costs. Therefore, ENP-based AOF can be an important component of the mitigation strategy to limit global warming.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Main
Recent international climate remediation scenarios rely on massive carbon dioxide removal (CDR) from the atmosphere, in addition to sharp CO2 emission reduction, to keep global warming at less than 2 °C (refs. 1,2). One CDR approach, artificial ocean fertilization (AOF), involves the intentional addition of a limiting nutrient (typically iron) to stimulate phytoplankton growth and CO2 uptake in the oceans (Fig. 1a)2,3,4,5,6,7,8,9. A fraction of the stimulated phytoplankton biomass subsequently sinks, exporting carbon to the deep ocean and potentially the ocean floor, for hundreds to thousands of years.
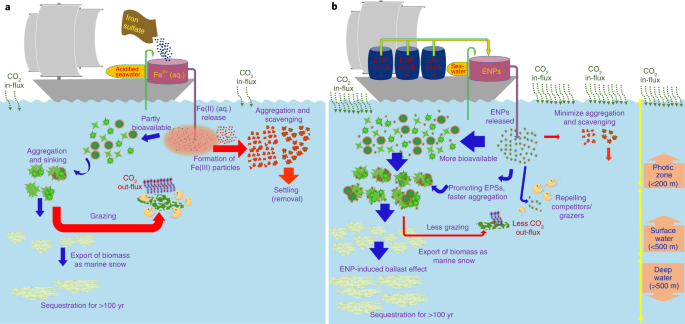
Schematics of ocean fertilization based on the conventional approach using iron sulfate (a) and the new approach with ENPs as proposed in this study (b). In a, after dissolving iron sulfate in acidified seawater onboard the ship, the solution is released into the ocean where the soluble iron (Fe(II) (aq.)) may partly be utilized by phytoplankton and cause a bloom, drawing down CO2 from the atmosphere. The particulate matter that remains from phytoplankton blooms may then undergo aggregation and subsequent sedimentation. If the biomass resulting from this bloom reaches the deep sea (for example, >1000 m), it may sequester carbon for centuries. However, this whole process might be hindered at the stage when the Fe(II) (aq.) is released into the seawater by the formation of particulate iron (oxyhydr)oxides, which can then aggregate and settle out of the photic zone rather than being bioavailable to phytoplankton. The process might also be hindered by low production rates and/or low settling rate of biomass compared with grazing and remineralization rates. In b, the stage of the rapid removal of added nutrients may be minimized by engineering the surface of ENPs, for example, using polymer or bimetallic coating. This may cause added nanoparticle nutrients to become more bioavailable to phytoplankton over a longer period and potentially result in a greater bloom than in the conventional scenario. ENPs may also induce EPS release from phytoplankton cells which can increase phytoplankton aggregation and sedimentation, promote the ballast effect, and thus result in a more effective export of their biomass into the deep ocean.
To date, 13 field-scale experiments employing artificial addition of Fe2+ into the ocean over 25–300 km2 have been conducted, in addition to several field-scale studies monitoring natural occurrences of ocean fertilization by iron4,6,10. These studies have shown that although considerable phytoplankton blooms can be stimulated in most artificial additions, their efficiencies in CO2 drawdown are much less than those observed for naturally occurring fertilization, for example, via terrestrial dust deposition through the atmosphere6. Slow export rates of biomass, especially if not enhanced compared to remineralization rates (the rates at which organic carbon is converted into CO2 by bacteria), undo the potential benefits of AOF. Remineralization can also lead to oxygen depletion in subsurface waters and the production of methane and nitrous oxide with higher global warming potential than CO2 (ref. 11). Nevertheless, with increasingly wide public concerns about the impacts of climate change and the need for negative emission technologies to meet the targets of the 2015 Paris Agreement, recently there has been a renewed interest in AOF1,2,3,4,5.
Due to their relatively high number density, and very high specific surface area and diffusivity, nanoparticles (1–100 nm) are probably bioavailable to phytoplankton in the ocean12,13,14,15. The important role of naturally occurring nanoparticles is already established in natural ocean fertilization12,13,14,16,17,18,19. For instance, natural iron (oxyhydr)oxide nanoparticles have been proposed as a source of bioavailable iron in fluxes from glaciers14,15, continental sediments13, volcanic ashes12 and hydrothermal vent emissions19. Natural nanoparticles may also supply other nutrients such as phosphorus16. These findings suggest that the use of engineered nanoparticles (ENPs) in AOF may lead to desirable efficiencies because of their nanocharacteristics, which resemble those of their natural counterparts. In addition, it is possible to design ENPs to mitigate the drawbacks of the current AOF approach, thereby maximizing their efficiency. If considerable CO2 drawdown is achieved by using ENPs, this may allow applications of the approach as a CDR technology at smaller scales or specific locations, and thus allay some of the concerns regarding risks of geoengineering the entire marine ecosystem and downstream ‘nutrient stealing’.
ENP applications in environmental systems at relatively large scales have been widely studied in the past two decades, such as nanoscale zero-valent iron (NZVI) slurries used for soil/groundwater remediation20,21,22 and the application of various ENPs for enhancing sustainable agriculture23 and aquaculture24,25. In soil/groundwater remediation, despite initial challenges regarding ENP delivery to contaminant source zones and concerns about ENP risks to drinking water resources, the technology has already been commercialized widely around the world20.
Here we summarize possible opportunities for ENPs to address the drawbacks and risks of AOF. We further estimate the environmental impacts and economic costs of ENP use for AOF by conducting life-cycle assessment (LCA; Supplementary Information, section 1) and life-cycle costing (LCC; Supplementary Information, section 2) and evaluate the toxic and beneficial aspects of ENPs to marine ecosystems based on data compiled from 123 studies selected among 265 peer-reviewed studies.
Ocean fertilization challenges and nanotechnology solutions
Bioavailability and phytoplankton growth enhancement
Metallic nanoparticles may be more bioavailable to phytoplankton than soluble forms due to higher local concentrations generated around phytoplankton cells26. This is because of the association of ENPs with planktonic cell surfaces, which may lead to a higher surface concentration of ENPs and their dissolution products and thus more bio-uptake26. Iron added in soluble form in previous AOF experiments immediately formed colloidal iron (oxyhydr)oxides and eventually micrometre-sized particles or aggregates that probably sank out of the photic zone, reducing iron bioavailability (Fig. 1a)4,14,17,18,27,28,29. ENPs, due to their surface modification, will address this problem (Fig. 1b), using polymers (for example, carboxymethyl cellulose) and/or metals (for example, aluminium hydroxide) to control their aggregation and dissolution rates, especially within the critical size range of 10–100 nm, thus tuning their functionality and longevity in environmental media20,21,30.
Numerous studies have demonstrated the stimulating effects of ENPs on algal growth (Fig. 2a,b and Supplementary Table 1). Notable algae population enhancement may be achieved using NZVI, SiO2 and CeO2 ENPs with a mean stimulation effect (cell abundance or growth) increased by 35–756% compared with controls31,32,33,34. In particular, the role of NZVI in enhancing marine microalgae growth was comparable to or higher than commonly used EDTA-Fe at equimolar concentrations of 11.7 µM (ref. 31). This was attributed to similar or higher bioavailability of ENPs, which can bind to extracellular polymeric substances (EPSs) around the algae cell, leading to ENP uptake via endocytosis31. Importantly, algal growth rates were similar for a wide range of NZVI concentrations (1.17–117 µM)31, revealing another advantage of using ENPs: algal growth is less dependent on ENP concentration in contrast to the substantial impact of the concentration when soluble iron is used35,36.
a, Box plot of lower and upper limits of growth stimulation compared to the control observed for different ENPs. QD, quantum dot. b, Minimum and maximum values of the same data (thin bars) compared to the mean values (thick bars). c, Concentration ranges of different ENPs causing toxic impacts (acute or chronic toxicity, including median effective (EC50), lethal (LC50) or inhibitory (IC50) concentrations) on different species related to marine ecosystems such as phytoplankton, zooplankton, fish and crustaceans (magenta and red) compared to concentration ranges resulting in promotion of algae/phytoplankton growth (light and dark green). d, Nominal ranges of mean concentrations investigated for toxicity in different studies (thin orange bars), and the ranges of mean toxic concentrations (EC50, LC50 or IC50, thick orange bars) observed for different ENPs compared to the nominal ranges of mean concentrations investigated in algae growth stimulation studies (thin green bars) with mean concentration ranges resulting in growth stimulation (thick green bars). The numbers beneath the name of each ENP indicate the number of studies involved in the data ranges. Growth stimulation data are related to marine algal species (21% of data), freshwater species (33% of data) and species common between the two environments (46% of data). The detailed dataset and the list of references are available in Supplementary Tables 1 and 6. On each box of the box plots, the middle mark shows the median, and the bottom and top sides indicate the 25th and 75th percentiles, respectively. The ‘+’ markers indicate individual outliers, and the whiskers show the extent of non-outlier data.
On the question as to whether the concentration of added ENPs should be comparable with natural colloid concentrations for ENPs to be bioavailable, we note that in general it is the interactions and thus affinity between species in solution (whether ionic or nanoparticle species) and surfaces (here phytoplankton EPSs) which control the species bioavailability, not the concentrations26,31. Our estimate of the potential ENP concentration range for AOF (as described in the Supplementary Information, section 5) is ~1010–1014 particles per litre, which is similar to or greater than reported background colloid and nanoparticle concentrations in seawater of ~107–1012 particles per litre37,38. This ENP number range is also ~800–1010 times the potential number concentration of phytoplankton being produced (4 × 102–4 × 108 particles per litre; Supplementary Information, section 5), suggesting that added ENPs are not overshadowed by ambient colloids or by potential phytoplankton numbers created during AOF.
Co-limitation by other nutrients
Incomplete bloom development in several AOF experiments was partly attributed to the depletion of secondary nutrients, particularly co-limitation by silicic acid for diatom growth4,6,39. Such depletion of secondary limiting nutrients may be addressed effectively within the context of nanotechnology by using a mixture of ENPs or a versatile core–shell nanocomposite as illustrated in Fig. 3. The enhancement in microalgae cell growth in the presence of silica ENPs has already been demonstrated (Fig. 2a,b). Composite nanoparticles containing all nutrients required by phytoplankton can be designed and manufactured in an efficiently engineered architecture for delivery to phytoplankton (Fig. 3c,d) to minimize the total amount of nutrient mass required for a large-scale AOF scheme, thus lowering some of its risks and side effects. Such utilization of nanocomposites currently prevails in other contexts of nanotechnology such as environmental remediation, agricultural/aquaculture enhancement and microalgae biorefinery21,24,25,40. Phytoplankton nutrient co-limitation by trace metal micronutrients such as zinc, cobalt, manganese and aluminium has been reported41,42, while stimulations of algal growth in the presence of ZnO and Al2O3 ENPs have also been observed, suggesting that ENPs are promising in addressing co-limitation by other nutrients (Fig. 2a,b).
Two possible scenarios in which ENPs can be used in AOF include applying a mixture of ENPs with different coatings (a,b) and the use of a versatile nanocomposite (c,d). In general, in response to ENPs, phytoplankton cells produce EPSs which may promote steric repulsion against attachment34, while polymer bridging attraction and potentially hydrophobic attractions63 may together lead to a loose adherence of ENPs onto phytoplankton surface EPSs69. The latter is favourable for AOF because it can lead to ENP detachment from the cell surface after partial utilization, which may provide availability for the next generations of phytoplankton. Detachment of ENP–EPS complex from phytoplankton cells may also result from further excretion of EPSs by phytoplankton in the presence of ENPs47,64. In the mixed ENP scenario (a,b), nanoparticles such as iron ENPs may supply nutrients for phytoplankton via dissolution while promoting EPS generation, which may aggregate phytoplankton cells and increase sinking velocity. Some ENPs such as SiO2, which can be designed to interact with cells, may attach to the cell surface EPS and further enhance their sinking rate through the ballast effect (b). In the second scenario (c,d), multilayer core–shell ENPs are designed with thicknesses of different layers in accordance with the amount and the time of nutrients required by phytoplankton species at the time when a nutrient becomes limiting (d). All particles may be coated with polymers and/or with metallic layers such as aluminium hydroxide (Al(OH)3)30 to minimize ENP–ENP attachment via creating steric repulsion or screening attractive magnetic forces. These may also control their interactions with cells as well as their dissolution.
Light limitation
Light limitation has been proposed as an important factor in reducing the efficacy of AOF4. Within the agritechnology realm, applying ENPs such as TiO2 and ZnO to foliage has shown an increased solar light absorption and generally enhanced photosynthetic efficiency23. The use of CeO2 ENPs, which are biocompatible with a broad range of microorganisms, has shown promise in filtering harmful ultraviolet light and allowing the absorption of useful visible light, thereby protecting microalgae43,44. Although this might not directly address limitations by the overall spectrum of the light in ocean settings such as the Southern Ocean, ENPs like CeO2 nanoparticles may still improve light-use efficiency by phytoplankton. In general, because ENPs are smaller in size than the wavelength of visible light, they tend to have lower shading effects than larger particulate nutrients such as Fe(III) (oxyhydr)oxides which are naturally formed in conventional AOF. Moreover, light insufficiency hinders photoreduction of colloidal Fe(III) to soluble Fe(II) and thus decreases iron bioavailability for phytoplankton uptake29. This may, in turn, accelerate iron removal in particulate form from the water column. On the other hand, potentially well-dispersed ENPs are generally directly bioavailable to phytoplankton without needing further dissolution under light limiting conditions26,31,33.
Export efficiency to deep sea
Application of ENPs to AOF may also enhance the export efficiency of accumulated phytoplankton biomass. Individual phytoplankton cells, especially non-diatom species and live cells, can have a density lower than that of seawater, reducing their sinking potential45. Phytoplankton sinking rates can be enhanced when materials with a higher density are adsorbed onto cells or incorporated into aggregates to induce a ballast effect. Silica ENPs (at a concentration of 75 mg l−1) were found effective in facilitating the sedimentation and removal of cyanobacteria from the water column46. Uptake of silica increased cyanobacteria density and sinking velocity through the formation of cyanobacteria–SiO2 complexes. Even a 1% increase in the density of phytoplankton can double the sinking speed45. This means the mass ratio of added SiO2 ENPs to phytoplankton biomass generated during the bloom could be just 1:100 to double the biomass export rate via the ballast effect.
Another important factor affecting phytoplankton export is their aggregation tendency. Marine phytoplankton EPSs are critical for the formation of marine snow, which is deemed key to phytoplankton export to the deep sea47. The use of SiO2 and CeO2 ENPs has also been proposed to facilitate oil-spill removal by promoting the production of EPSs by marine phytoplankton, leading to enhanced aggregation and sinking47. Accordingly, SiO2 ENPs might not only address co-limitation by multiple nutrients but also may facilitate the export of the phytoplankton as a ballast agent and phytoplankton EPS promoter. More efficient export of ballasted organic matter may reduce subsurface deoxygenation and methane and nitrous oxide production caused by water column remineralization.
Longevity of biomass production
Crustacean grazers can shorten phytoplankton bloom lifetime and reduce AOF efficiency39,48,49. There are opportunities for ENPs to protect a developing bloom by repelling grazers and competitors, and this may be controlled by tuning the doses of various ENPs considered for AOF. Mesocosm experiments have shown that ENPs such as CuNPs and AuNPs, when added in combination with nutrients (nitrogen and phosphorus), can enhance the bloom continuation by more than 50 days50, due to the adverse effect of ENPs on planktonic species competitors. Further, aluminium oxide ENPs skewed the selective feeding pattern of Daphnia towards algal feed that was not exposed to ENPs51.
Multilayer core–shell ENPs can be designed with a sequence of layers according to the sequence of the micronutrient limitations and subsequent grazing pressure during different stages of phytoplankton bloom development and sinking (Fig. 3c,d). This protects the inner layers of multilayer core–shell ENP against dissolution and therefore dilution in seawater, sustaining the biomass production in surface waters for longer at a lower overall mass of nutrient added. The influence of dilution on the concentration of ENPs is less than that of their soluble form because the diffusivity of ENPs within the size range of 10–100 nm (15.5 down to 1.55 cm2 yr−1 at 25 °C) is generally one to two orders of magnitude less than that of soluble Fe2+ (222.8 cm2 yr−1 at 25 °C) under quiescent conditions52.
Although factors such as biomass sinking, phytoplankton bloom longevity and aggregate size might be countervailing and are currently the subject of ongoing research7,8,53, ENPs’ abilities to enhance phytoplankton growth and promote EPS production (which enhances cell aggregation and biomass export) are all advantageous for carbon dioxide removal. Further research is also required to understand these concomitant factors under natural conditions and in the presence of ENPs.
Overall, although future research may test and develop more functionalized hybrid ENPs specifically made for AOF schemes and investigate their fate and ecological risks in the ocean, among the currently used ENPs there are various candidates, including NZVI, iron oxide, SiO2, ZnO, CeO2, Al2O3 and TiO2 nanoparticles, that may be relevant for future AOF applications (Table 1).
LCA and LCC
We conducted an LCA and an LCC analysis for several relevant ENPs (NZVI, SiO2, Al2O3, ZnO and CeO2), their polymer coatings and AOF operation (transportation of materials, distribution over the ocean and monitoring of the subsequent impacts). We considered a range of ENP synthesis methods including chemical, mechanical and green (plant extract-based54) approaches (Figs. 4 and 5, Supplementary Fig. 1, and Supplementary Tables 2–5). CO2-equivalent (CO2e) emissions per kg ENPs for all processes involved are 38.3 kg on average (ranging from 11.2 to 156 kg) which is 13 times (or 4–53 times) larger than those of iron sulfate conventionally used in AOF (Fig. 4a,b). The total cost for these processes is on average US$72 per kg ENPs (US$21–153; Fig. 5a). These figures will be smaller if we only consider the best synthesis methods for NZVI, SiO2 and Al2O3 for which several synthesis methods are possible (for example, green methods with 12.9 kg CO2e emissions and a cost of US$63.6 per kg ENPs for use in AOF, on average). On average, these lead to 4–10 times larger CO2 emissions compared with conventionally used iron sulfate. Henceforth, we discuss these ENP types only, as these may also be the main ENPs for AOF.
CO2e emissions determined from LCA for the production of NZVI, SiO2, Al2O3, ZnO and CeO2 ENPs by several synthesis methods (a), their polymer coatings and their use in AOF (b). Iron sulfate previously used in AOF has also been included in the analyses for comparison. AOF processes include transportation from the manufacturing site to the distribution region, distribution over the ocean via either ship or aircraft, and monitoring the AOF performance using similar ships to those used for the distribution. Error bars are uncertainties based on 99% confidence intervals of estimates obtained from Monte Carlo analysis70. ENP synthesis methods include sodium borohydride reduction, dithionite reduction, green syntheses using green tea, coffee and Virginia creeper (Parthenocissus tricuspidata) leave extracts, ball milling and electrical wire explosion for NZVI; green syntheses and ball milling for SiO2; green synthesis and ball milling for Al2O3; and ball milling for ZnO and CeO2. CMC, carboxymethyl cellulose.
Costs after (a) and before (b) including the labour costs, capital costs and environmental costs for the production of NZVI, SiO2, Al2O3, ZnO and CeO2 ENPs by various synthesis methods, using ENP polymer coatings and AOF operation processes (delivery and distribution, using ship only). The analysis has also been conducted for iron sulfate for comparison. The purple bars show the ranges of costs estimated, and the blue lines near the middle of the bars indicate the averaged results.
While estimating carbon export is difficult and uncertain, especially over long-term periods53,55, based on the previous field-scale AOF studies, each kilogram of added iron can lead to ~80–800 kg of CO2 removal during the course of a fertilization event56 (values converted from molar ratio given in the reference to mass ratio), although 50–67% of this may be compensated by CO2 release from excess microbial respiration, decreasing the estimate to 40–400 kg of CO2 (refs. 56,57). The lower limit of this range is still ~3-fold higher than the amount of CO2e emissions per kg NZVI, SiO2 and Al2O3 ENPs synthesized using green methods (12.9 kg), and still higher when synthesized with the ball-milling method (30.0 kg). Based on this efficiency range, our analyses show that AOF with iron sulfate costs ~US$0.030–0.300 per kg CO2 removed, which is within previously reported ranges of US$0.008–0.450 (refs. 9,58) and leads to 2.97 kg CO2e emissions per kg iron used as determined here. Although the use of ENPs (NZVI, SiO2 and Al2O3) may increase costs by ~2–5 times (US$0.16–1.6 with green synthesis methods or US$0.07–0.65 with the ball-milling method for NZVI and SiO2) compared with iron sulfate, we have shown here that ENPs may enhance AOF efficiencies much above those of iron sulfate2. Further, our LCC analysis also considers labour, capital and environmental costs in addition to monitoring costs which were rarely included previously58. Our approach is relatively conservative because the labour, capital and environmental costs are mostly embedded in the prices of materials and processes considered in life-cycle inventories while we have also specified these costs separately (Fig. 5a,b). If we drop these additional costs from our analysis, the use of the aforementioned ENPs increases costs over conventional AOF only by around 1.4–2.2 times (US$0.036–0.37 with all green synthesis methods or US$0.024–0.24 with the ball-milling method for NZVI and SiO2; Fig. 5b).
Other potential environmental impacts resulting from our LCA analysis are summarized in Supplementary Table 4, showing that in most cases CeO2 synthesis (using the ball-milling method) causes the largest environmental impacts among different ENP types/synthesis methods. Although green methods in all impact categories, except urban land occupation, are the most environmentally friendly synthesis methods, in a few categories (agricultural land occupation, marine eutrophication and urban land occupation) they can also cause large impacts (Supplementary Table 4). The potential economic costs resulting from four important environmental impact categories, that is, climate change, freshwater eutrophication, ozone depletion and terrestrial acidification, are presented in Supplementary Table 3. These costs range from negligible to US$21 (US$1.14 on average) per kg ENP used and are generally highest for climate change (CO2e emissions) among different categories, except for green synthesis of NZVI and AOF processes (ship only) which show the highest environmental impact in the category of terrestrial acidification (US$2.4 and US$0.37, respectively; Supplementary Table 3). Further discussions on the feasibility of the ENP use in AOF are presented in Supplementary Information, section 3–5.
Toxicity of ENPs in oceans
While natural nanoparticles exist in most ocean settings14,18, the potential adverse environmental risks of adding ENPs to the ocean require rigorous assessment. We collated toxicity data for ENPs from 98 studies (Supplementary Table 6 and Fig. 2c,d). The ranges of ENP concentrations found to be toxic to benthic and planktonic marine species are compared with concentrations found to stimulate algae growth. These data show that ENP concentrations inducing growth stimulation are mostly below the toxic ranges, especially for NZVI and quantum dots with almost no overlap in the enhanced growth versus toxic ranges, and for SiO2, Fe2O3 and CeO2 with slight overlaps (Fig. 2c,d). The concentration range that gives rise to growth stimulation averaged for NZVI, iron oxides, SiO2 and CeO2 is 2.07–44.3 mg l−1 whereas the toxic concentration range averaged for these ENPs is 132–1,090 mg l−1. If the upper threshold of the stimulation concentration range is selected for ENP application, this concentration—which is already well below the lower toxic limit—will drop substantially in the ocean by the time ENPs reach deeper habitats due to dilution, and uptake by phytoplankton bloom and other processes. This may partly alleviate concerns about the potential risks of ENPs to marine ecosystems, although potential accumulations from repeated AOF should also be considered. Further, a concentration of 75 mg l−1 SiO2 ENPs which may induce a ballast effect46 is near the lower limit of the SiO2 ENP toxic concentration range, 73–1,300 mg l−1, suggesting that achieving some levels of ballast effect using ENPs is possible from a toxicology viewpoint.
While the presence of ENPs might induce reactive oxygen species (ROS) production, the promoting effect of several ENPs on algae growth (Fig. 2a,b) suggests that ROS do not induce toxicity within the ENP concentration ranges given above. This is in line with the recent paradigm shift from regarding ROS as creating toxicity to them being essential for biological and physiochemical functions of organisms and impacting nutrient cycling in marine environments59,60. Additionally, some ENPs such as CeO2 may protect against ROS by scavenging them32,33.
Fate and transport of ENPs in oceans
Although the potentially low concentrations of ENPs required for AOF are favourable for minimizing ENP aggregation, ENP surface modifications are still required to reduce aggregation and settling and thereby lengthen residence time in surface waters14,20,52,61. Typically this is achieved using polymers that are inexpensive, efficient, biocompatible, and can be adsorbed onto ENP surfaces via simple physisorption processes and/or using metals that are also aimed to improve ENP performance in a core–shell structure20,21,30,52. Polymers generally provide steric repulsion at the surface of ENPs while a metallic shell may reduce the Hamaker constant or enhance electrostatic repulsion to overcome forces such as van der Waals and magnetic attractions20,21,30,52. Ligands with low molecular weight (for example, diol ether), which are well solvated in a concentrated brine, have shown a considerable steric stabilization effect in seawater conditions for 10 nm SiO2 ENPs61. Some polymers (for example, sulfonated copolymers), which do not bind with divalent cations, provide a steric stabilization effect for iron oxide ENPs against bridging flocculation62. Such polymers might also reduce the heteroaggregation of ENPs with ubiquitous colloids in seawater, although further research is needed to investigate these effects. Heteroaggregation between ENPs and phytoplankton cells due to polymer bridging63 could be one possible pathway for ENP removal from the ocean surface. However, continuous excretion of soluble EPS fraction from phytoplankton64, especially due to the presence of ENPs47, might cause detachment of ENPs from the cell wall (Fig. 3), and thus reduce ENP removal through heteroaggregation with phytoplankton. Such complex mechanisms are currently poorly understood and warrant further research.
Overall, current studies on the fate and transport of ENPs suggest that nanoparticles are generally unlikely to remain in their original form for long in aquatic environments because various mechanisms of aggregation, dissolution and transformation result in their removal from the water column or alter their characteristics such that they become similar to their natural colloidal counterparts ubiquitous in the environment17,18.
Regulatory and public acceptance challenges
There are already concerns about potential adverse effects of AOF, such as enhancement of methane and nitrous oxide production, impacts on the ocean ecosystem or far-field effects on productivity4,6,65. Although operational AOF activities are currently banned under relevant regulatory bodies, for example, the London Convention/London Protocol, the road to conducting small-scale legitimate scientific explorations that meet an environmental assessment framework is still open4,6,66,67. There is growing acceptance that more research is needed to evaluate AOF side effects so that the demands of policy-makers and the public for a clearer understanding of such effects can be met4,6,65. Further, terrestrial applications of ENPs have already gained favour in several cases such as drinking water decontamination and purification68, groundwater remediation20,21,22 and sustainable agriculture/aquaculture23,24,25. Nevertheless, ENP use in AOF is a new realm that also necessitates substantial consideration of potential impacts.
Conclusions and a roadmap for future research
Although ENPs show promise in addressing many of the current AOF limitations such as bioavailability, nutrient/light co-limitation, phytoplankton bloom longevity and carbon export efficiency, our present information and estimations are based on diverse contexts rather than focused studies on the use of ENPs in realistic AOF conditions. Some potential challenges to overcome include public and regulatory concerns about the potential toxicity of ENPs to marine ecosystems under realistic conditions, unknown long-term impacts of ENP additions on the biogeochemistry of the oceans and the tendency of ENPs to aggregate over time within the marine environment. None of these challenges are necessarily insurmountable.
To begin to address such limitations, research is required in the following key areas:
-
Selecting the optimum characteristics of an ideal hybrid ENP or a mixture of several individual ENPs to harvest their combined benefits for the most effective AOF (Fig. 3).
-
Designing and manufacturing novel multicomponent/hybrid ENPs to meet the requirements of AOF including delivery of the optimum limiting factors for phytoplankton growth, physicochemical stability in seawater, limited interactions with natural colloids and selective interaction with phytoplankton. Despite the countervailing effects of some of these requirements, nanotechnology may provide a unique opportunity to address them.
-
Assessing the ecological impact of using ENPs at all scales, ranging from simple bench-scale laboratory tests to mesocosm and field-scale experiments.
-
Further development of ocean biogeochemical models is required to consider a more robust description of AOF-related mechanisms such as ballasting and the burial of biomass in marine sediments53. Such models should also consider the fate and transport of ENPs in the ocean and allow for the optimization of ENP performance in AOF. These models may then be used for testing various AOF scenarios on local and global scales, investigating long-term and far-field impacts, and providing tools for decision making.
-
Understanding the mechanisms of phytoplankton growth enhancement by ENPs and the fate of ENPs and generated biomass in marine environments that are relevant to realistic AOF conditions.
To achieve substantial CO2 removal from the atmosphere via ENP-enabled AOF, long-term repeated additions of ENPs will be needed. Estimating expenses and risks for such an implementation requires determining effective and safe concentration ranges in realistic conditions. This should include considering the fate of added ENPs and exported biomass in deep waters and sediments for understanding the long-term implications of AOF.
Further advancement of the technology may be continued by designing smart ENPs that can target phytoplankton efficiently and enhance phytoplankton aggregation and sinking speed while potentially repelling grazers to protect the biomass export. If part of the ENP mixture or hybrid ENP provides such beneficial activity, this will, in turn, prevent or retard environmental problems such as methane and nitrous oxide production and oxygen consumption.
Overall, ENPs, due to their great number density, specific surface area, diffusion, bioavailability and potential for designing their effective targetability and other functionalities, may provide multiple benefits for application in AOF. These benefits address the challenges of using conventional approaches for AOF (use of dissolved nutrients), which include enhancing bioavailability, nutrient co-delivery, photosynthetic efficiency, export efficiency and phytoplankton bloom longevity. Although our analyses show that CO2e emissions and implementation costs for AOF using selected ENPs (NZVI, SiO2 and Al2O3) are higher by 4–10 and 2–5 times, respectively, than using dissolved iron, these results are based on the worst-case scenario where the use of ENPs leads to similar CO2 removal as achieved by the conventional approach. The potential for ENPs to address the aforementioned challenges with conventional AOF will likely improve the efficiency of AOF and may alleviate the concerns about its implementation. In conclusion, ENP-based AOF may be a remarkable carbon dioxide removal approach to fight climate change.
Data availability
All data used, including toxicity, growth enhancement, LCA and LCC analysis are presented in the Supplementary Information.
References
IPCC Climate Change 2022: Mitigation of Climate Change. Working Group III Contribution to the IPCC Sixth Assessment Report (2022).
A Research Strategy for Ocean-based Carbon Dioxide Removal and Sequestration (National Academies of Sciences, Engineering and Medicine, 2021).
Greenhouse Gas Removal (Royal Society, 2018); https://royalsociety.org/topics-policy/projects/greenhouse-gas-removal/
Williamson, P. et al. Ocean fertilization for geoengineering: a review of effectiveness, environmental impacts and emerging governance. Process Saf. Environ. Prot. 90, 475–488 (2012).
Güssow, K., Proelss, A., Oschlies, A., Rehdanz, K. & Rickels, W. Ocean iron fertilization: why further research is needed. Mar. Policy 34, 911–918 (2010).
Yoon, J.-E. et al. Reviews and syntheses: ocean iron fertilization experiments—past, present, and future looking to a future Korean Iron Fertilization Experiment in the Southern Ocean (KIFES) project. Biogeosciences 15, 5847–5889 (2018).
Smetacek, V. et al. Deep carbon export from a Southern Ocean iron-fertilized diatom bloom. Nature 487, 313–319 (2012).
Boyd, P. W. et al. A mesoscale phytoplankton bloom in the polar Southern Ocean stimulated by iron fertilization. Nature 407, 695–702 (2000).
Boyd, P. W. Implications of large-scale iron fertilization of the oceans: introduction and synthesis. Mar. Ecol. Prog. Ser. 364, 213–218 (2008).
Pollard, R. T. et al. Southern Ocean deep-water carbon export enhanced by natural iron fertilization. Nature 457, 577–580 (2009).
Xiao, K.-Q., Moore, O. W., Babakhani, P., Curti, L. & Peacock, C. L. Mineralogical control on methylotrophic methanogenesis and implications for cryptic methane cycling in marine surface sediment. Nat. Commun. 13, 1–9 (2022).
Langmann, B., Zakšek, K., Hort, M. & Duggen, S. Volcanic ash as fertiliser for the surface ocean. Atmos. Chem. Phys. 10, 3891–3899 (2010).
Homoky, W. B. et al. Iron colloids dominate sedimentary supply to the ocean interior. Proc. Natl Acad. Sci. USA 118, e2016078118 (2021).
Raiswell, R. & Canfield, D. E. The iron biogeochemical cycle past and present. Geochem. Perspect. 1, 1–2 (2012).
Raiswell, R. Iceberg-hosted nanoparticulate Fe in the Southern Ocean: mineralogy, origin, dissolution kinetics and source of bioavailable Fe. Deep Sea Res. 2 Top. Stud. Oceanogr. 58, 1364–1375 (2011).
Saeed, H. et al. Regulation of phosphorus bioavailability by iron nanoparticles in a monomictic lake. Sci. Rep. 8, 1–14 (2018).
Hochella, M. F. et al. Nanominerals, mineral nanoparticles, and Earth systems. Science 319, 1631–1635 (2008).
Hochella, M. F. et al. Natural, incidental, and engineered nanomaterials and their impacts on the Earth system. Science 363, eaau8299 (2019).
Fitzsimmons, J. N. et al. Iron persistence in a distal hydrothermal plume supported by dissolved–particulate exchange. Nat. Geosci. 10, 195–201 (2017).
Phenrat, T. & Lowry, G. V. Nanoscale Zerovalent Iron Particles for Environmental Restoration (Springer, 2019).
O’Carroll, D., Sleep, B., Krol, M., Boparai, H. & Kocur, C. Nanoscale zero valent iron and bimetallic particles for contaminated site remediation. Adv. Water Res. 51, 104–122 (2013).
Park, C. M., Wang, D. & Su, C. in Handbook of Nanomaterials for Industrial Applications (ed Hussain, C. M.) 849–882 (Elsevier, 2018).
Lowry, G. V., Avellan, A. & Gilbertson, L. M. Opportunities and challenges for nanotechnology in the agri-tech revolution. Nat. Nanotechnol. 14, 517 (2019).
Sabo‐Attwood, T., Apul, O. G., Bisesi, J. H. Jr, Kane, A. S. & Saleh, N. B. Nano‐scale applications in aquaculture: opportunities for improved production and disease control. J. Fish. Dis. 44, 359–370 (2021).
Moges, F. D., Patel, P., Parashar, S. K. S. & Das, B. Mechanistic insights into diverse nano-based strategies for aquaculture enhancement: a holistic review. Aquaculture 519, 734770 (2020).
Ponton, D. E. et al. Three-layered silver nanoparticles to trace dissolution and association to a green alga. Nanotoxicology 13, 1149–1160 (2019).
Wells, M. L. & Mayer, L. M. The phttoconversion of colloidal iron oxyhydroxides in seawater. Deep Sea Res. A 38, 1379–1395 (1991).
Bowie, A. R. et al. The fate of added iron during a mesoscale fertilisation experiment in the Southern Ocean. Deep Sea Res. 2 Top. Stud. Oceanogr. 48, 2703–2743 (2001).
Coale, K. H. et al. IronEx-I, an in situ iron-enrichment experiment: experimental design, implementation and results. Deep Sea Res. 2 Top. Stud. Oceanogr. 45, 919–945 (1998).
Hu, Y.-B. & Li, X.-Y. Influence of a thin aluminum hydroxide coating layer on the suspension stability and reductive reactivity of nanoscale zero-valent iron. Appl. Catal. B 226, 554–564 (2018).
Kadar, E., Rooks, P., Lakey, C. & White, D. A. The effect of engineered iron nanoparticles on growth and metabolic status of marine microalgae cultures. Sci. Total Environ. 439, 8–17 (2012).
Deng, X.-Y. et al. Biological effects of TiO2 and CeO2 nanoparticles on the growth, photosynthetic activity, and cellular components of a marine diatom Phaeodactylum tricornutum. Sci. Total Environ. 575, 87–96 (2017).
Sendra, M., Yeste, P. M., Moreno-Garrido, I., Gatica, J. M. & Blasco, J. CeO2 NPs, toxic or protective to phytoplankton? Charge of nanoparticles and cell wall as factors which cause changes in cell complexity. Sci. Total Environ. 590, 304–315 (2017).
Chen, F. et al. Algae response to engineered nanoparticles: current understanding, mechanisms and implications. Environ. Sci.: Nano 6, 1026–1042 (2019).
Huang, X., Wei, L., Huang, Z. & Yan, J. Effect of high ferric ion concentrations on total lipids and lipid characteristics of Tetraselmis subcordiformis, Nannochloropsis oculata and Pavlova viridis. J. Appl. Phycol. 26, 105–114 (2014).
Rajabi Islami, H. & Assareh, R. Effect of different iron concentrations on growth, lipid accumulation, and fatty acid profile for biodiesel production from Tetradesmus obliquus. J. Appl. Phycol. 31, 3421–3432 (2019).
Graca, B., Zgrundo, A., Zakrzewska, D., Rzodkiewicz, M. & Karczewski, J. Origin and fate of nanoparticles in marine water—preliminary results. Chemosphere 206, 359–368 (2018).
Wells, M. L. & Goldberg, E. D. Occurrence of small colloids in sea water. Nature 353, 342–344 (1991).
Boyd, P. W. et al. The decline and fate of an iron-induced subarctic phytoplankton bloom. Nature 428, 549 (2004).
Nguyen, M. K., Moon, J.-Y., Bui, V. K. H., Oh, Y.-K. & Lee, Y.-C. Recent advanced applications of nanomaterials in microalgae biorefinery. Algal Res. 41, 101522 (2019).
Brand, L. E., Sunda, W. G. & Guillard, R. R. L. Limitation of marine phytoplankton reproductive rates by zinc, manganese, and iron. Limnol. Oceanogr. 28, 1182–1198 (1983).
Zhou, L., Tan, Y., Huang, L., Fortin, C. & Campbell, P. G. C. Aluminum effects on marine phytoplankton: implications for a revised iron hypothesis (iron–aluminum hypothesis). Biogeochemistry 139, 123–137 (2018).
Xia, T. et al. Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano 2, 2121–2134 (2008).
Sicard, C. et al. CeO2 nanoparticles for the protection of photosynthetic organisms immobilized in silica gels. Chem. Mater. 23, 1374–1378 (2011).
Sommer, U., Charalampous, E., Genitsaris, S. & Moustaka-Gouni, M. Benefits, costs and taxonomic distribution of marine phytoplankton body size. J. Plankton Res. 39, 494–508 (2017).
Xiong, W. et al. Prevention of cyanobacterial blooms using nanosilica: a biomineralization-inspired strategy. Environ. Sci. Technol. 51, 12717–12726 (2017).
Chiu, M.-H. et al. Effect of engineered nanoparticles on exopolymeric substances release from marine phytoplankton. Nanoscale Res. Lett. 12, 620 (2017).
Martin, P. et al. Iron fertilization enhanced net community production but not downward particle flux during the Southern Ocean iron fertilization experiment LOHAFEX. Glob. Biogeochem. Cycles 27, 871–881 (2013).
Laglera, L. M. et al. Iron partitioning during LOHAFEX: copepod grazing as a major driver for iron recycling in the Southern Ocean. Mar. Chem. 196, 148–161 (2017).
Simonin, M. et al. Engineered nanoparticles interact with nutrients to intensify eutrophication in a wetland ecosystem experiment. Ecol. Appl. 28, 1435–1449 (2018).
Pakrashi, S., Dalai, S., Chandrasekaran, N. & Mukherjee, A. Trophic transfer potential of aluminium oxide nanoparticles using representative primary producer (Chlorella ellipsoides) and a primary consumer (Ceriodaphnia dubia). Aquat. Toxicol. 152, 74–81 (2014).
Baalousha, M. Effect of nanomaterial and media physicochemical properties on nanomaterial aggregation kinetics. NanoImpact 6, 55–68 (2017).
Henson, S. A. et al. Uncertain response of ocean biological carbon export in a changing world. Nat. Geosci. 15, 248–254 (2022).
Kozma, G., Rónavári, A., Kónya, Z. & Kukovecz, A. Environmentally benign synthesis methods of zero-valent iron nanoparticles. ACS Sustain. Chem. Eng. 4, 291–297 (2016).
Boyd, P. W., Claustre, H., Levy, M., Siegel, D. A. & Weber, T. Multi-faceted particle pumps drive carbon sequestration in the ocean. Nature 568, 327–335 (2019).
de Baar, H. J. W., Gerringa, L. J. A., Laan, P. & Timmermans, K. R. Efficiency of carbon removal per added iron in ocean iron fertilization. Mar. Ecol. Prog. Ser. 364, 269–282 (2008).
Aumont, O. & Bopp, L. Globalizing results from ocean in situ iron fertilization studies. Global Biogeochem. Cycles 20, 1–15 (2006).
Harrison, D. P. A method for estimating the cost to sequester carbon dioxide by delivering iron to the ocean. Int. J. Glob. Warm. 5, 231–254 (2013).
Hansel, C. M. & Diaz, J. M. Production of extracellular reactive oxygen species by marine biota. Annu. Rev. Mar. Sci. 13, 177–200 (2021).
Rezayian, M., Niknam, V. & Ebrahimzadeh, H. Oxidative damage and antioxidative system in algae. Toxicol. Rep. 6, 1309–1313 (2019).
Worthen, A. J., Tran, V., Cornell, K. A., Truskett, T. M. & Johnston, K. P. Steric stabilization of nanoparticles with grafted low molecular weight ligands in highly concentrated brines including divalent ions. Soft Matter 12, 2025–2039 (2016).
Bagaria, H. G. et al. Stabilization of iron oxide nanoparticles in high sodium and calcium brine at high temperatures with adsorbed sulfonated copolymers. Langmuir 29, 3195–3206 (2013).
Wang, H., Adeleye, A. S., Huang, Y., Li, F. & Keller, A. A. Heteroaggregation of nanoparticles with biocolloids and geocolloids. Adv. Colloid Interface Sci. 226, 24–36 (2015).
Cheng, Q. et al. Enhanced excretion of extracellular polymeric substances associated with nonylphenol tolerance in Dictyosphaerium sp. J. Hazard. Mater. 395, 122644 (2020).
Oschlies, A., Koeve, W., Rickels, W. & Rehdanz, K. Side effects and accounting aspects of hypothetical large-scale Southern Ocean iron fertilization. Biogeosciences (BG) 7, 4017–4035 (2010).
GESAMP. High Level Review of a Wide Range of Proposed Marine Geoengineering Techniques, Vol 98 (INTL. MAR. ORG., 2019).
Freestone, D. & Rayfuse, R. Ocean iron fertilization and international law. Mar. Ecol. Prog. Ser. 364, 227–233 (2008).
Alvarez, P. J. J., Chan, C. K., Elimelech, M., Halas, N. J. & Villagrán, D. Emerging opportunities for nanotechnology to enhance water security. Nat. Nanotechnol. 13, 634–641 (2018).
Ma, S., Zhou, K., Yang, K. & Lin, D. Heteroagglomeration of oxide nanoparticles with algal cells: effects of particle type, ionic strength and pH. Environ. Sci. Technol. 49, 932–939 (2015).
Goedkoop, M., De Schryver, A., Oele, M., Durksz, S. & de Roest, D. Introduction to LCA with SimaPro 7 (PRé Consultants, 2008).
Acknowledgements
P.B. and C.L.P. acknowledge support from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (Grant agreement No. 725613 MinOrg). B.S.T. received support from the Grantham Foundation for the Protection of the Environment. We thank Christian März, and William B. Homoky for reviewing an early draft of the paper and the anonymous reviewers for their constructive comments which improved the manuscript.
Author information
Authors and Affiliations
Contributions
P.B. conceived the idea. P.B. and T.P. planned the research and compiled the data from toxicity and growth enhancement literature. P.B. and K.S. conducted the LCA and LCC. P.B. wrote the first draft of the manuscript. P.B., M.B. and M.F.H. prepared the figures. M.B., B.S.T. and M.F.H. provided critical feedback. M.F.H. supervised the research. P.B., M.B., C.L.P., B.S.T. and M.F.H. wrote, reviewed and edited the manuscript. All authors read, discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Philip Williamson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary sections 1–5, Figs. 1–3 and Tables 1–6.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Babakhani, P., Phenrat, T., Baalousha, M. et al. Potential use of engineered nanoparticles in ocean fertilization for large-scale atmospheric carbon dioxide removal. Nat. Nanotechnol. 17, 1342–1351 (2022). https://doi.org/10.1038/s41565-022-01226-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-022-01226-w
- Springer Nature Limited
This article is cited by
-
Electrochemical ocean iron fertilization and alkalinity enhancement approach toward CO2 sequestration
npj Ocean Sustainability (2024)