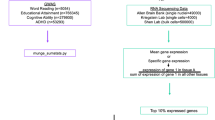
Abstract
Reading is an evolutionarily recent development that recruits and tunes brain circuitry connecting primary- and language-processing regions. We investigated whether metrics of the brain’s physical structure correlate with reading performance and whether genetic variants affect this relationship. To this aim, we used the Adolescent Brain Cognitive Development dataset (n = 9,013) of 9–10-year-olds and focused on 150 measures of cortical surface area (CSA) and thickness. Our results reveal that reading performance is associated with nine measures of brain structure including relevant regions of the reading network. Furthermore, we show that this relationship is partially mediated by genetic factors for two of these measures: the CSA of the entire left hemisphere and, specifically, of the left superior temporal gyrus CSA. These effects emphasize the complex and subtle interplay between genes, brain and reading, which is a partly heritable polygenic skill that relies on a distributed network.




Similar content being viewed by others
Data availability
ABCD data are publicly available through the NIMH Data Archive (https://data-archive.nimh.nih.gov/abcd). The current study analysed the full baseline sample (n ≅ 11,878) from the ABCD data release 3.0 RDS (https://doi.org/10.15154/1520591) and the Genotyping Data from the ABCD Curated Annual Release 3.0 (NDA Study 901; https://doi.org/10.15154/1519007). All variables included in the current study are listed and described in Supplementary Table 1. GWAS summary statistics used in this study are available from the NHGRI-EBI GWAS Catalogue https://www.ebi.ac.uk/gwas/downloads/summary-statistics and the Oxford Brain Imaging Genetics Server, BIG40 (https://open.win.ox.ac.uk/ukbiobank/big40/). Supplementary Table 7 contains the references and field IDs for all analysed traits.
Code availability
The custom code associated with this study is publicly available at https://github.com/amaiacc/MS-brain-reading-genetics/.
References
Sandak, R., Mencl, W. E., Frost, S. J. & Pugh, K. R. The neurobiological basis of skilled and impaired reading: recent findings and new directions. Sci. Stud. Read. 8, 273–292 (2004).
Dehaene, S. & Cohen, L. Cultural recycling of cortical maps. Neuron 56, 384–398 (2007).
Friederici, A. D. The cortical language circuit: from auditory perception to sentence comprehension. Trends Cogn. Sci. 16, 262–268 (2012).
Lau, E. F., Phillips, C. & Poeppel, D. A cortical network for semantics: (de)constructing the N400. Nat. Rev. Neurosci. 9, 920–N933 (2008).
Martin, A., Schurz, M., Kronbichler, M. & Richlan, F. Reading in the brain of children and adults: a meta-analysis of 40 functional magnetic resonance imaging studies. Hum. Brain Mapp. 36, 1963–1981 (2015).
Pugh, K. R. et al. Neurobiological studies of reading and reading disability. J. Commun. Disord. 34, 479–492 (2001).
Carreiras, M. et al. An anatomical signature for literacy. Nature 461, 983–986 (2009).
Dehaene, S., Cohen, L., Morais, J. & Kolinsky, R. Illiterate to literate: behavioural and cerebral changes induced by reading acquisition. Nat. Rev. Neurosci. 16, 234–244 (2015).
Jednoróg, K. et al. The influence of socioeconomic status on children’s brain structure. PLoS ONE 7, e42486 (2012).
Romeo, R. R., Uchida, L. & Christodoulou, J. A. Socioeconomic status and reading outcomes: neurobiological and behavioral correlates. New Dir. Child Adolesc. Dev. https://doi.org/10.1002/cad.20475 (2022).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 5th edn (APA Publishing, 2013).
World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 10th Revision 5th edn (WHO, 2016).
Goswami, U. Phonology, reading development, and dyslexia: a cross-linguistic perspective. Ann. Dyslexia 52, 139–163 (2002).
Ramus, F. Developmental dyslexia: specific phonological deficit or general sensorimotor dysfunction? Curr. Opin. Neurobiol. 13, 212–218 (2003).
Ramus, F., Altarelli, I., Jednoróg, K., Zhao, J. & Covella, L. S. Neuroanatomy of developmental dyslexia: pitfalls and promise. Neurosci. Biobehav. Rev. 84, 434–452 (2018).
Richlan, F. Developmental dyslexia: dysfunction of a left hemisphere reading network. Front. Hum. Neurosci. 6, 120 (2012).
Andreola, C. et al. The heritability of reading and reading-related neurocognitive components: a multi-level meta-analysis. Neurosci. Biobehav. Rev. 121, 175–200 (2021).
Verhoef, E., Shapland, C. Y., Fisher, S. E., Dale, P. S. & Pourcain, B. S., The developmental origins of genetic factors influencing language and literacy: associations with early-childhood vocabulary. J. Child Psychol. Psychiatry 62, 728–738 (2020).
Eising, E. et al. Genome-wide association analyses of individual differences in quantitatively assessed reading- and language-related skills in up to 34,000 people. Proc. Natl Acad. Sci. USA 119, e2202764119 (2022).
Fisher, S. E. & DeFries, J. C. Developmental dyslexia: genetic dissection of a complex cognitive trait. Nat. Rev. Neurosci. 3, 767–780 (2002).
Doust, C. et al. Discovery of 42 genome-wide significant loci associated with dyslexia. Nat. Genet. 54, 1621–1629 (2022); https://doi.org/10.1038/s41588-022-01192-y
Gialluisi, A. et al. Genome-wide association study reveals new insights into the heritability and genetic correlates of developmental dyslexia. Mol. Psychiatry 26, 3004–3017 (2020).
Grasby, K. L. et al. The genetic architecture of the human cerebral cortex. Science 367, eaay6690 (2020).
Elliott, L. T. et al. Genome-wide association studies of brain imaging phenotypes in UK Biobank. Nature 562, 210–216 (2018).
Smith, S. M. et al. An expanded set of genome-wide association studies of brain imaging phenotypes in UK Biobank. Nat. Neurosci. 24, 737–745 (2021).
Landi, N. & Perdue, M. V. Neuroimaging genetics studies of specific reading disability and developmental language disorder: a review. Lang. Linguist. Compass 13, e12349 (2019).
Roeske, D. et al. First genome-wide association scan on neurophysiological endophenotypes points to trans-regulation effects on SLC2A3 in dyslexic children. Mol. Psychiatry 16, 97–107 (2009).
Scarborough, H. S. & Brady, S. A. Toward a common terminology for talking about speech and reading: a glossary of the phon words and some related terms. J. Lit. Res. 34, 299–336 (2002).
Taylor, J. S. H., Rastle, K. & Davis, M. H. Can cognitive models explain brain activation during word and pseudoword reading? A meta-analysis of 36 neuroimaging studies. Psychol. Bull. 139, 766–791 (2013).
Jobard, G., Crivello, F. & Tzourio-Mazoyer, N. Evaluation of the dual route theory of reading: a metanalysis of 35 neuroimaging studies. NeuroImage 20, 693–712 (2003).
Dick, A. S. et al. Meaningful associations in the Adolescent Brain Cognitive Development study. NeuroImage 239, 118262 (2021).
Vigneau, M. et al. Meta-analyzing left hemisphere language areas: phonology, semantics, and sentence processing. NeuroImage 30, 1414–1432 (2006).
Rueckl, J. G. et al. Universal brain signature of proficient reading: evidence from four contrasting languages. Proc. Natl Acad. Sci. USA 112, 15510–15515 (2015).
Destrieux, C., Fischl, B., Dale, A. M. & Halgren, E. A sulcal depth-based anatomical parcellation of the cerebral cortex. NeuroImage 47, S151 (2009).
Hyatt, C. S. et al. The quandary of covarying: a brief review and empirical examination of covariate use in structural neuroimaging studies on psychological variables. NeuroImage 205, 116225 (2020).
Yang, J. et al. Common SNPs explain a large proportion of the heritability for human height. Nat. Genet. 42, 565–569 (2010).
Bulik-Sullivan, B. et al. An atlas of genetic correlations across human diseases and traits. Nat. Genet. 47, 1236–1241 (2015).
Lee, J. J. et al. Gene discovery and polygenic prediction from a genome-wide association study of educational attainment in 1.1 million individuals. Nat. Genet. 50, 1112–1121 (2018).
Howe, L. J. et al. Within-sibship genome-wide association analyses decrease bias in estimates of direct genetic effects. Nat. Genet. 54, 581–592 (2022).
Maier, R. M., Visscher, P. M., Robinson, M. R. & Wray, N. R. Embracing polygenicity: a review of methods and tools for psychiatric genetics research. Psychol. Med. 48, 1055–1067 (2018).
Vuoksimaa, E. et al. Is bigger always better? The importance of cortical configuration with respect to cognitive ability. NeuroImage 129, 356–366 (2016).
Dynak, A. et al. Separating the influences of late talking and dyslexia on brain structure. J. Abnorm. Psychol. 130, 286–296 (2021).
Palmer, C. E. et al. Distinct regionalization patterns of cortical morphology are associated with cognitive performance across different domains. Cereb. Cortex 31, 3856–3871 (2021).
Hickok, G. & Poeppel, D. The cortical organization of speech processing. Nat. Rev. Neurosci. 8, 393–402 (2007).
Atteveldt, N., Formisano, E., Goebel, R. & Blomert, L. Integration of letters and speech sounds in the human brain. Neuron 43, 271–282 (2004).
Boets, B. et al. Intact but less accessible phonetic representations in adults with dyslexia. Science 342, 1251–1254 (2013).
Essen, D. C. V. & Dierker, D. L. Surface-based and probabilistic atlases of primate cerebral cortex. Neuron 56, 209–225 (2007).
Płoński, P. et al. Multi-parameter machine learning approach to the neuroanatomical basis of developmental dyslexia. Hum. Brain Mapp. 38, 900–908 (2016).
Perdue, M. V., Mednick, J., Pugh, K. R. & Landi, N. Gray matter structure is associated with reading skill in typically developing young readers. Cereb. Cortex 30, 5449–5459 (2020).
Richlan, F., Kronbichler, M. & Wimmer, H. Structural abnormalities in the dyslexic brain: a meta-analysis of voxel-based morphometry studies. Hum. Brain Mapp. 34, 3055–3065 (2012).
Altarelli, I. et al. Planum temporale asymmetry in developmental dyslexia: revisiting an old question. Hum. Brain Mapp. 35, 5717–5735 (2014).
Ben-Shachar, M., Dougherty, R. F., Deutsch, G. K. & Wandell, B. A. Differential sensitivity to words and shapes in ventral occipito-temporal cortex. Cereb. Cortex 17, 1604–1611 (2007).
Cohen, L. et al. The visual word form area. Brain 123, 291–307 (2000).
Beelen, C., Vanderauwera, J., Wouters, J., Vandermosten, M. & Ghesquière, P. Atypical gray matter in children with dyslexia before the onset of reading instruction. Cortex 121, 399–413 (2019).
Carreiras, M., Quiñones, I., Hernández-Cabrera, J. A. & Duñabeitia, J. A. Orthographic coding: brain activation for letters, symbols, and digits. Cereb. Cortex 25, 4748–4760 (2015).
Feng, X. et al. A universal reading network and its modulation by writing system and reading ability in French and Chinese children. eLife 9, e54591 (2020).
Lyall, A. E. et al. Dynamic development of regional cortical thickness and surface area in early childhood. Cereb. Cortex 25, 2204–2212 (2014).
Panizzon, M. S. et al. Distinct genetic influences on cortical surface area and cortical thickness. Cereb. Cortex 19, 2728–2735 (2009).
Rakic, P. Specification of cerebral cortical areas. Science 241, 170–176 (1988).
Meer, D. et al. Quantifying the polygenic architecture of the human cerebral cortex: extensive genetic overlap between cortical thickness and surface area. Cereb. Cortex 30, 5597–5603 (2020).
Eyler, L. T. et al. A comparison of heritability maps of cortical surface area and thickness and the influence of adjustment for whole brain measures: a magnetic resonance imaging twin study. Twin Res. Hum. Genet. 15, 304–314 (2012).
Hoogman, M. et al. Brain imaging of the cortex in ADHD: a coordinated analysis of large-scale clinical and population-based samples. Am. J. Psychiatry 176, 531–542 (2019).
Owens, M. M. et al. Multimethod investigation of the neurobiological basis of ADHD symptomatology in children aged 9-10: baseline data from the ABCD study. Transl. Psychiatry 11, 64 (2021).
Alfaro-Almagro, F. et al. Confound modelling in UK Biobank brain imaging. NeuroImage 224, 117002 (2021).
Batouli, S. A. H., Trollor, J. N., Wen, W. & Sachdev, P. S. The heritability of volumes of brain structures and its relationship to age: a review of twin and family studies. Ageing Res. Rev. 13, 1–9 (2014).
Jansen, A. G., Mous, S. E., White, T., Posthuma, D. & Polderman, T. J. C. What twin studies tell us about the heritability of brain development, morphology, and function: a review. Neuropsychol. Rev. 25, 27–46 (2015).
Teeuw, J. et al. Genetic influences on the development of cerebral cortical thickness during childhood and adolescence in a Dutch longitudinal twin sample: the brainscale study. Cereb. Cortex 29, 978–993 (2018).
Visscher, P. M. et al. Statistical power to detect genetic (co)variance of complex traits using SNP data in unrelated samples. PLoS Genet. 10, e1004269 (2014).
Frei, O. et al. Bivariate causal mixture model quantifies polygenic overlap between complex traits beyond genetic correlation. Nat. Commun. 10, 2417 (2019).
Mitchell, B. L. et al. Educational attainment polygenic scores are associated with cortical total surface area and regions important for language and memory. NeuroImage 15, 116691 (2020).
Lett, T. A. et al. Cortical surfaces mediate the relationship between polygenic scores for intelligence and general intelligence. Cereb. Cortex 30, 2708–2719 (2019).
Vuoksimaa, E. et al. The genetic association between neocortical volume and general cognitive ability is driven by global surface area rather than thickness. Cereb. Cortex 25, 2127–2137 (2014).
Marek, S. et al. Reproducible brain-wide association studies require thousands of individuals. Nature 603, 654–660 (2022).
Smith, S. et al. Structural variability in the human brain reflects fine-grained functional architecture at the population level. J. Neurosci. 39, 6136–6149 (2019).
Casey, B. J. et al. The Adolescent brain Cognitive Development (ABCD) study: imaging acquisition across 21 sites. Dev. Cogn. Neurosci. 32, 43–54 (2018).
Littlejohns, T. J. et al. The UK Biobank imaging enhancement of 100,000 participants: rationale, data collection, management and future directions. Nat. Commun. 11, 2624 (2020).
Jednoróg, K. et al. How reliable are gray matter disruptions in specific reading disability across multiple countries and languages? insights from a large-scale voxel-based morphometry study. Hum. Brain Mapp. 36, 1741–1754 (2015).
Loughnan, R. J. et al. Gene-experience correlation during cognitive development: evidence from the Adolescent Brain Cognitive Development (ABCD) Study. https://doi.org/10.1101/637512 (2019).
Walhovd, K. B. et al. Neurodevelopmental origins of lifespan changes in brain and cognition. Proc. Natl Acad. Sci. USA 113, 9357–9362 (2016).
Jernigan, T. L. & Brown, S. A. Introduction. Dev. Cogn. Neurosci. 32, 1–3 (2018).
Auchter, A. M. et al. A description of the ABCD organizational structure and communication framework. Dev. Cogn. Neurosci. 32, 8–15 (2018).
Garavan, H. et al. Recruiting the ABCD sample: design considerations and procedures. Dev. Cogn. Neurosci. 32, 16–22 (2018).
Compton, W. M., Dowling, G. J. & Garavan, H. Ensuring the best use of data. JAMA Pediatr. 173, 809 (2019).
Luciana, M. et al. Adolescent neurocognitive development and impacts of substance use: overview of the Adolescent Brain Cognitive Development (ABCD) baseline neurocognition battery. Dev. Cogn. Neurosci. 32, 67–79 (2018).
Hagler, D. J. et al. Image processing and analysis methods for the Adolescent Brain Cognitive Development study. NeuroImage 202, 116091 (2019).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 67, 1–48 (2015).
Revelle, W. R. Psych: Procedures for psychological, psychometric, and personality research. Software, R package version 2.2.9 (2022); https://CRAN.R-project.org/package=psych
Mowinckel, A. M. & Vidal-Piñeiro, D. Visualization of brain statistics with R Packages ggseg and ggseg3d. Adv. Methods Pract. Psychol. Sci. 3, 466–483 (2020).
Mowinckel, A. M., & Vidal-Piñeiro, D. ggsegDesterieux: Desterieux datasets for the ggseg-plotting tool. R package version 1.0.1.002 (2021); https://github.com/LCBC-UiO/ggsegDesterieux
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Coleman, J. R. I. et al. Quality control, imputation and analysis of genome-wide genotyping data from the Illumina HumanCoreExome microarray. Brief. Funct. Genomics 15, 298–304 (2015).
Patterson, N., Price, A. L. & Reich, D. Population structure and eigenanalysis. PLoS Genet. 2, e190 (2006).
Auton, A. et al. A global reference for human genetic variation. Nature 526, 68–74 (2015).
Choi, S. W. & OReilly, P. F. PRSice-2: polygenic risk score software for biobank-scale data. GigaScience 8, giz082 (2019).
Tingley, D., Yamamoto, T., Hirose, K., Keele, L. & Imai, K., mediation: R package for causal mediation analysis. J. Stat. Softw. 59, 1–38 (2014).
Imai, K., Keele, L. & Tingley, D. A general approach to causal mediation analysis. Psych. Meth. 15, 309–334 (2010).
Acknowledgements
We thank J. Barry and C. Handley for helping with manuscript proofreading. This research is supported by the Basque Government through the BERC 2022–2025 programme and by the Spanish State Research Agency through BCBL Severo Ochoa excellence accreditation no. CEX2020-001010-S. A.C.-C. received funding from the Spanish Ministry of Science and Innovation and the Agencia Estatal de Investigación through Ayudas Juan de la Cierva-Incorporación (ref. no. IJC2018-036023-I), the Programa Fellows Gipuzkoa de atracción y retención de talento from the Diputación Foral de Gipuzkoa. P.M.P-A. is supported by grants from the Spanish Ministry of Science and Innovation (grant no. PID2021-123574NB-I00), from the Basque Government (grant no. PIBA-2021-1-0003) and from the Red guipuzcoana de Ciencia, Tecnología e Innovación of the Diputación Foral de Gipuzkoa (grant no. FA/OF 422/2022). M.C. is supported by the Spanish Ministry of Science and Innovation grant (no. PID2021-122918OB-I00) and ‘La Caixa’ Foundation (grant no. ID 100010434), under the agreement no. HR18-00178-DYSTHAL. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. Data used in the preparation of this article were obtained from the ABCD Study (https://abcdstudy.org/) and are held in the National Institute of Mental Health (NIMH) Data Archive. This is a multisite, longitudinal study designed to recruit more than 10,000 children aged 9–10 and follow them over 10 years into early adulthood. The ABCD Study is supported by the NIH and additional federal partners under award numbers U01DA041022, U01DA041028, U01DA041048, U01DA041089, U01DA041106, U01DA041117, U01DA041120, U01DA041134, U01DA041148, U01DA041156, U01DA041174, U24DA041123 and U24DA041147. A full list of supporters is available at https://abcdstudy.org/federal-partners/. A listing of participating sites and a complete listing of the study investigators can be found at https://abcdstudy.org/principal-investigators/. ABCD consortium investigators designed and implemented the study and/or provided data but did not necessarily participate in the analysis or writing of this report. This manuscript reflects the views of the authors and may not reflect the opinions or views of the NIH or ABCD consortium investigators. The ABCD data repository grows and changes over time. The ABCD data used in this report came from https://doi.org/10.15154/1520591 and https://doi.org/10.15154/1519007 (genotyping data).
Author information
Authors and Affiliations
Contributions
All authors conceived and designed the study. A.C.-C. analysed the data, made figures and wrote the first draft of the paper. All authors discussed the results and contributed towards the writing of the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Human Behaviour thanks Dan Belsky and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figs. 1–15.
Supplementary Table
Supplementary tables 1–10.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carrión-Castillo, A., Paz-Alonso, P.M. & Carreiras, M. Brain structure, phenotypic and genetic correlates of reading performance. Nat Hum Behav 7, 1120–1134 (2023). https://doi.org/10.1038/s41562-023-01583-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-023-01583-z
- Springer Nature Limited
This article is cited by
-
Neuroscience: genes, brains, and reading variability
Communications Psychology (2023)