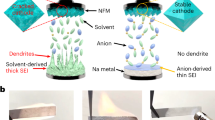
Abstract
Sodium-ion batteries (NIBs) have attracted worldwide attention for next-generation energy storage systems. However, the severe instability of the solid–electrolyte interphase (SEI) formed during repeated cycling hinders the development of NIBs. In particular, the SEI dissolution in NIBs with a high-voltage cathode is more severe than in the case of Li-ion batteries (LIBs) and leads to continuous side reactions, electrolyte depletion and irreversible capacity loss, making NIBs less stable than LIBs. Here we report a rational electrolyte design to suppress the SEI dissolution and enhance NIB performance. Our electrolyte lowers the solvation ability for SEI components and facilitates the formation of insoluble SEI components, which minimizes the SEI dissolution. In addition to the stable SEI on a hard carbon (HC) anode, we also show a stable interphase formation on a NaNi0.68Mn0.22Co0.1O2 (NaNMC) cathode. Our HC||NaNMC full cell with this electrolyte demonstrates >90% capacity retention after 300 cycles when charged to 4.2 V. This study enables high-voltage NIBs with long cycling performance and provides a guiding principle in electrolyte design for sodium-ion batteries.





Similar content being viewed by others
Data availability
All relevant data are included in the paper and its Supplementary Information.
References
Usiskin, R. et al. Fundamentals, status and promise of sodium-based batteries. Nat. Rev. Mater. 6, 1020–1035 (2021).
Hu, Y.-S. & Lu, Y. 2019 Nobel prize for the Li-ion batteries and new opportunities and challenges in Na-ion batteries. ACS Energy Lett. 4, 2689–2690 (2019).
Yabuuchi, N., Kubota, K., Dahbi, M. & Komaba, S. Research development on sodium-ion batteries. Chem. Rev. 114, 11636–11682 (2014).
Pan, H., Hu, Y.-S. & Chen, L. Room-temperature stationary sodium-ion batteries for large-scale electric energy storage. Energy Environ. Sci. 6, 2338–2360 (2013).
Hwang, J.-Y., Myung, S.-T. & Sun, Y.-K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 46, 3529–3614 (2017).
Deng, J., Luo, W. B., Chou, S. L., Liu, H. K. & Dou, S. X. Sodium‐ion batteries: from academic research to practical commercialization. Adv. Energy Mater. 8, 1701428 (2018).
Xu, G. L. et al. Challenges in developing electrodes, electrolytes, and diagnostics tools to understand and advance sodium‐ion batteries. Adv. Energy Mater. 8, 1702403 (2018).
Palomares, V. et al. Na-ion batteries, recent advances and present challenges to become low cost energy storage systems. Energy Environ. Sci. 5, 5884–5901 (2012).
Kundu, D., Talaie, E., Duffort, V. & Nazar, L. F. The emerging chemistry of sodium ion batteries for electrochemical energy storage. Angew. Chem. Int. Ed. 54, 3431–3448 (2015).
Zhou, Q. et al. Thermal stability of high power 26650-type cylindrical Na-ion batteries. Chin. Phys. Lett. 38, 076501 (2021).
Zhao, C. et al. Rational design of layered oxide materials for sodium-ion batteries. Science 370, 708–711 (2020).
Yabuuchi, N. et al. P2-type Nax[Fe1/2Mn1/2]O2 made from earth-abundant elements for rechargeable Na batteries. Nat. Mater. 11, 512–517 (2012).
Wang, J. et al. A high‐energy NASICON‐type cathode material for Na‐ion batteries. Adv. Energy Mater. 10, 1903968 (2020).
Deng, J. et al. High energy density sodium‐ion battery with industrially feasible and air‐stable O3‐type layered oxide cathode. Adv. Energy Mater. 8, 1701610 (2018).
Kamiyama, A. et al. MgO‐template synthesis of extremely high capacity hard carbon for Na‐ion battery. Angew. Chem. Int. Ed. 60, 5114–5120 (2021).
Dou, X. et al. Hard carbons for sodium-ion batteries: structure, analysis, sustainability, and electrochemistry. Mater. Today 23, 87–104 (2019).
Wang, L. et al. Rhombohedral Prussian white as cathode for rechargeable sodium-ion batteries. J. Am. Chem. Soc. 137, 2548–2554 (2015).
Jache, B. & Adelhelm, P. Use of graphite as a highly reversible electrode with superior cycle life for sodium-ion batteries by making use of co-intercalation phenomena. Angew. Chem. 126, 10333–10337 (2014).
House, R. A. et al. Superstructure control of first-cycle voltage hysteresis in oxygen-redox cathodes. Nature 577, 502–508 (2020).
Wen, Y. et al. Expanded graphite as superior anode for sodium-ion batteries. Nat. Commun. 5, 4033 (2014).
Rakov, D. A. et al. Engineering high-energy-density sodium battery anodes for improved cycling with superconcentrated ionic-liquid electrolytes. Nat. Mater. 19, 1096–1101 (2020).
Li, Y. et al. Ultralow-concentration electrolyte for Na-ion batteries. ACS Energy Lett. 5, 1156–1158 (2020).
Li, K. et al. Evolution of the electrochemical interface in sodium ion batteries with ether electrolytes. Nat. Commun. 10, 725 (2019).
Ponrouch, A. et al. Non-aqueous electrolytes for sodium-ion batteries. J. Mater. Chem. A 3, 22–42 (2015).
Wang, J. et al. Fire-extinguishing organic electrolytes for safe batteries. Nat. Energy 3, 22–29 (2018).
Mogensen, R., Colbin, S., Menon, A. S., Björklund, E. & Younesi, R. Sodium bis (oxalato) borate in trimethyl phosphate: a fire-extinguishing, fluorine-free, and low-cost electrolyte for full-cell sodium-ion batteries. ACS Appl. Energy Mater. 3, 4974–4982 (2020).
Mogensen, R., Buckel, A., Colbin, S. & Younesi, R. A wide-temperature-range, low-cost, fluorine-free battery electrolyte based on sodium bis (oxalate) borate. Chem. Mater. 33, 1130–1139 (2021).
Zeng, Z. et al. A safer sodium‐ion battery based on nonflammable organic phosphate electrolyte. Adv. Sci. 3, 1600066 (2016).
Liu, X. et al. High capacity and cycle-stable hard carbon anode for nonflammable sodium-ion batteries. ACS Appl. Mater. Interfaces 10, 38141–38150 (2018).
Mogensen, R., Brandell, D. & Younesi, R. Solubility of the solid electrolyte interphase (SEI) in sodium ion batteries. ACS Energy Lett. 1, 1173–1178 (2016).
Ma, L. A., Naylor, A. J., Nyholm, L. & Younesi, R. Strategies of mitigating dissolution of solid electrolyte interphases in sodium‐ion batteries. Angew. Chem. 133, 4905–4913 (2021).
Song, J., Xiao, B., Lin, Y., Xu, K. & Li, X. Interphases in sodium‐ion batteries. Adv. Energy Mater. 8, 1703082 (2018).
Moshkovich, M., Gofer, Y. & Aurbach, D. Investigation of the electrochemical windows of aprotic alkali metal (Li, Na, K) salt solutions. J. Electrochem. Soc. 148, E155–E167 (2001).
Yan, G. et al. Assessment of the electrochemical stability of carbonate-based electrolytes in Na-ion batteries. J. Electrochem. Soc. 165, A1222–A1230 (2018).
Jin, Y. et al. Highly reversible sodium ion batteries enabled by stable electrolyte-electrode interphases. ACS Energy Lett. 5, 3212–3220 (2020).
Yao, Y. X. et al. Regulating interfacial chemistry in lithium‐ion batteries by a weakly solvating electrolyte. Angew. Chem. Int. Ed. 60, 4090–4097 (2021).
Xu, K., Ding, M. S., Zhang, S., Allen, J. L. & Jow, T. R. Evaluation of fluorinated alkyl phosphates as flame retardants in electrolytes for Li-ion batteries: I. Physical and electrochemical properties. J. Electrochem. Soc. 150, A161–A169 (2003).
Flamme, B. et al. Guidelines to design organic electrolytes for lithium-ion batteries: environmental impact, physicochemical and electrochemical properties. Green Chem. 19, 1828–1849 (2017).
Cresce, A. V. et al. Solvation behavior of carbonate-based electrolytes in sodium ion batteries. Phys. Chem. Chem. Phys. 19, 574–586 (2017).
Song, J. et al. Controlling surface phase transition and chemical reactivity of O3-layered metal oxide cathodes for high-performance Na-ion batteries. ACS Energy Lett. 5, 1718–1725 (2020).
You, Y. & Manthiram, A. Progress in high‐voltage cathode materials for rechargeable sodium‐ion batteries. Adv. Energy Mater. 8, 1701785 (2018).
Wang, Q. et al. Reaching the energy density limit of layered O3‐NaNi0.5Mn0.5O2 electrodes via dual Cu and Ti substitution. Adv. Energy Mater. 9, 1901785 (2019).
Acknowledgements
This work was supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Vehicle Technologies Office, Advanced Battery Materials Research (BMR) programme of the US Department of Energy (DOE) under contract number DE-AC05-76RL01830. The cathode material development was supported by the US DOE Office of Electricity (contract number 70247 A). The TEM and XPS were performed using EMSL (grid.436923.9), a DOE Office of Science User Facility sponsored by the Office of Biological and Environmental Research located at Pacific Northwest National Laboratory (PNNL). TEM sample preparation was performed in the Radiological Microscopy Suite (RMS), located in the Radiochemical Processing Laboratory (RPL) at PNNL. PNNL is operated by Battelle for the DOE under contract DE-AC05-76RL01830.
Author information
Authors and Affiliations
Contributions
J.-G.Z. proposed the research. Y.J. designed experiments, performed the electrochemical measurements, characterized materials and analysed the data. Y.X., B.E.M. and C.W. performed the FIB scanning electron microscope and TEM. P.G. performed the simulation. M.H.E. performed the XPS measurements. B.X. and X.L. synthesized the NaNMC cathode materials. L.Z. performed the viscosity measurement. P.M.L.L., T.D.V. and R.Y. prepared electrodes. X.C. and J.H. helped the gas generation test. J.L. contributed to the discussion and provided suggestions. Y.J., X.L. and J.-G.Z. wrote the manuscript with input from all other co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Energy thanks Philipp Adelhelm, Reza Younesi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–20, Notes 1–12 and Tables 1 and 2.
Rights and permissions
About this article
Cite this article
Jin, Y., Le, P.M.L., Gao, P. et al. Low-solvation electrolytes for high-voltage sodium-ion batteries. Nat Energy 7, 718–725 (2022). https://doi.org/10.1038/s41560-022-01055-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41560-022-01055-0
- Springer Nature Limited
This article is cited by
-
Tailoring planar strain for robust structural stability in high-entropy layered sodium oxide cathode materials
Nature Energy (2024)
-
Safe electrolyte for long-cycling alkali-ion batteries
Nature Sustainability (2024)
-
A weakly coordinating-intervention strategy for modulating Na+ solvation sheathes and constructing robust interphase in sodium-metal batteries
Nature Communications (2024)
-
Microstrain screening towards defect-less layered transition metal oxide cathodes
Nature Nanotechnology (2024)
-
Recent Progress in Sodium-Ion Batteries: Advanced Materials, Reaction Mechanisms and Energy Applications
Electrochemical Energy Reviews (2024)