Abstract
While most alien species fail to establish, some invade native communities and become widespread. Our understanding of invasion success is derived mainly from pairwise interactions between aliens and natives, while interactions among more than two species remain largely unexplored. Here, we experimentally tested whether and how a third plant species, either native or alien, affected the competitive outcomes between alien and native plants through its soil legacy. We first conditioned soil with one of ten species (six natives and four aliens) or without plants. We then grew on these 11 soils five aliens and five natives without competition, or with intra- or interspecific competition. We found that aliens were not more competitive than natives when grown on soil conditioned by other natives or on non-conditioned soil. However, aliens were more competitive than natives on soil conditioned by other aliens (that is, invasional meltdown). Soil conditioning did not change competitive outcomes by affecting the strength of competition between later plants. Instead, soil conditioned by aliens pushed competitive outcomes towards later aliens by affecting the growth of aliens less negatively than that of natives. Microbiome analysis verified this finding, as we showed that the soil-legacy effects of a species on later species were less negative when their fungal endophyte communities were less similar, and that fungal endophyte communities were less similar between two aliens than between aliens and natives. Our study reveals invasional meltdown in multispecies communities and identifies soil microorganisms as a driver of the invasion success of alien plants.





Similar content being viewed by others
Data availability
The data from this study are freely available at https://doi.org/10.6084/m9.figshare.12682382. The raw sequencing data are deposited at the Sequence Read Archive at NCBI under accession number PRJNA647659.
Code availability
The R script for analysing and plotting are freely available at https://github.com/ecozhang/Invasional-meltdown and permanently deposited at https://doi.org/10.6084/m9.figshare.12682382.
References
van Kleunen, M., Bossdorf, O. & Dawson, W. The ecology and evolution of alien plants. Annu. Rev. Ecol. Evol. Syst. 49, 25–47 (2018).
Seebens, H. et al. No saturation in the accumulation of alien species worldwide. Nat. Commun. 8, 14435 (2017).
Vilà, M. et al. Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 14, 702–708 (2011).
Elton, C. S. The Ecology of Invasion by Animals and Plants (Univ. of Chicago Press, 1958).
Kuebbing, S. E. & Nunez, M. A. Invasive non-native plants have a greater effect on neighbouring natives than other non-natives. Nat. Plants 2, 16134 (2016).
Golivets, M. & Wallin, K. F. Neighbour tolerance, not suppression, provides competitive advantage to non-native plants. Ecol. Lett. 21, 745–759 (2018).
Zhang, Z. & van Kleunen, M. Common alien plants are more competitive than rare natives but not than common natives. Ecol. Lett. 22, 1378–1386 (2019).
White, E. M., Wilson, J. C. & Clarke, A. R. Biotic indirect effects: a neglected concept in invasion biology. Divers. Distrib. 12, 443–455 (2006).
Sotomayor, D. A. & Lortie, C. J. Indirect interactions in terrestrial plant communities: emerging patterns and research gaps. Ecosphere 6, art103 (2015).
Aschehoug, E. T. & Callaway, R. M. Diversity increases indirect interactions, attenuates the intensity of competition, and promotes coexistence. Am. Nat. 186, 452–459 (2015).
Feng, Y. & van Kleunen, M. Phylogenetic and functional mechanisms of direct and indirect interactions among alien and native plants. J. Ecol. 104, 1136–1148 (2016).
Stotz, G. C. et al. Not a melting pot: plant species aggregate in their non‐native range. Glob. Ecol. Biogeogr. 29, 482–490 (2019).
Wardle, D. A. & Peltzer, D. A. Impacts of invasive biota in forest ecosystems in an aboveground–belowground context. Biol. Invasions 19, 3301–3316 (2017).
Kulmatiski, A., Beard, K. H. & Stark, J. M. Soil history as a primary control on plant invasion in abandoned agricultural fields. J. Appl. Ecol. 43, 868–876 (2006).
Simberloff, D. & Von Holle, B. Positive interactions of nonindigenous species: invasional meltdown? Biol. Invasions 1, 21–32 (1999).
Simberloff, D. Invasional meltdown 6 years later: important phenomenon, unfortunate metaphor, or both? Ecol. Lett. 9, 912–919 (2006).
Braga, R. R., Gómez-Aparicio, L., Heger, T., Vitule, J. R. S. & Jeschke, J. M. Structuring evidence for invasional meltdown: broad support but with biases and gaps. Biol. Invasions 20, 923–936 (2018).
Maynard, D. S., Miller, Z. R. & Allesina, S. Predicting coexistence in experimental ecological communities. Nat. Ecol. Evol. 4, 91–100 (2020).
May, R. M. Will a large complex system be stable? Nature 238, 413–414 (1972).
Godoy, O., Stouffer, D. B., Kraft, N. J. B. & Levine, J. M. Intransitivity is infrequent and fails to promote annual plant coexistence without pairwise niche differences. Ecology 98, 1193–1200 (2017).
Vandermeer, J. H. The competitive structure of communities: an experimental approach with protozoa. Ecology 50, 362–371 (1969).
Friedman, J., Higgins, L. M. & Gore, J. Community structure follows simple assembly rules in microbial microcosms. Nat. Ecol. Evol. 1, 0109 (2017).
Case, T. J. & Bender, E. A. Testing for higher order interactions. Am. Nat. 118, 920–929 (1981).
Levine, J. M., Bascompte, J., Adler, P. B. & Allesina, S. Beyond pairwise mechanisms of species coexistence in complex communities. Nature 546, 56–64 (2017).
Prince, E. K., Myers, T. L., Naar, J. & Kubanek, J. Competing phytoplankton undermines allelopathy of a bloom-forming dinoflagellate. Proc. R. Soc. B 275, 2733–2741 (2008).
Tilman, D. Resource Competition and Community Structure (Princeton Univ. Press, 1982).
Dawson, W., Fischer, M. & van Kleunen, M. Common and rare plant species respond differently to fertilisation and competition, whether they are alien or native. Ecol. Lett. 15, 873–880 (2012).
Godoy, O., Valladares, F. & Castro-Díez, P. Multispecies comparison reveals that invasive and native plants differ in their traits but not in their plasticity. Funct. Ecol. 25, 1248–1259 (2011).
Liu, Y. J. & van Kleunen, M. Nitrogen acquisition of Central European herbaceous plants that differ in their global naturalization success. Funct. Ecol. 33, 566–575 (2019).
Holt, R. D. Predation, apparent competition, and the structure of prey communities. Theor. Popul. Biol. 12, 197–229 (1977).
Bever, J. D., Westover, K. M. & Antonovics, J. Incorporating the soil community into plant population dynamics: the utility of the feedback approach. J. Ecol. 85, 561–573 (1997).
Kulmatiski, A., Beard, K. H., Stevens, J. R. & Cobbold, S. M. Plant-soil feedbacks: a meta-analytical review. Ecol. Lett. 11, 980–992 (2008).
Lekberg, Y. et al. Relative importance of competition and plant-soil feedback, their synergy, context dependency and implications for coexistence. Ecol. Lett. 21, 1268–1281 (2018).
Latz, E. et al. Plant diversity improves protection against soil-borne pathogens by fostering antagonistic bacterial communities. J. Ecol. 100, 597–604 (2012).
Kardol, P., Cornips, N. J., van Kempen, M. M. L., Bakx-Schotman, J. M. T. & van der Putten, W. H. Microbe-mediated plant–soil feedback causes historical contingency effects in plant community assembly. Ecol. Monogr. 77, 147–162 (2007).
Dawson, W., Schrama, M. & Austin, A. Identifying the role of soil microbes in plant invasions. J. Ecol. 104, 1211–1218 (2016).
Callaway, R. M., Thelen, G. C., Rodriguez, A. & Holben, W. E. Soil biota and exotic plant invasion. Nature 427, 731–733 (2004).
Ke, P. J. & Wan, J. Effects of soil microbes on plant competition: a perspective from modern coexistence theory. Ecol. Monogr. 90, e01391 (2020).
Kuebbing, S. E., Classen, A. T., Call, J. J., Henning, J. A. & Simberloff, D. Plant–soil interactions promote co-occurrence of three nonnative woody shrubs. Ecology 96, 2289–2299 (2015).
Callaway, R. M. et al. Novel weapons: invasive plant suppresses fungal mutualists in America but not in its native Europe. Ecology 89, 1043–1055 (2008).
Darwin, C. On the Origin of Species (J. Murray, 1859).
Keane, R. M. & Crawley, M. J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 17, 164–170 (2002).
Mangla, S. & Callaway, R. M. Exotic invasive plant accumulates native soil pathogens which inhibit native plants. J. Ecol. 96, 58–67 (2008).
Saul, W. C. & Jeschke, J. M. Eco-evolutionary experience in novel species interactions. Ecol. Lett. 18, 236–245 (2015).
van Kleunen, M. et al. Global exchange and accumulation of non-native plants. Nature 525, 100–103 (2015).
Pyšek, P. et al. Naturalized alien flora of the world. Preslia 89, 203–274 (2017).
Essl, F. et al. Drivers of the relative richness of naturalized and invasive plant species on Earth. AoB PLANTS 11, plz051 (2019).
Seebens, H. et al. Global rise in emerging alien species results from increased accessibility of new source pools. Proc. Natl Acad. Sci. USA 115, E2264–E2273 (2018).
Adler, P. B. et al. Competition and coexistence in plant communities: intraspecific competition is stronger than interspecific competition. Ecol. Lett. 21, 1319–1329 (2018).
Mangan, S. A. et al. Negative plant–soil feedback predicts tree-species relative abundance in a tropical forest. Nature 466, 752–755 (2010).
Dal Co, A., van Vliet, S., Kiviet, D. J., Schlegel, S. & Ackermann, M. Short-range interactions govern the dynamics and functions of microbial communities. Nat. Ecol. Evol. 4, 366–375 (2020).
Reinhart, K. O., Packer, A., Van der Putten, W. H. & Clay, K. Plant–soil biota interactions and spatial distribution of black cherry in its native and invasive ranges. Ecol. Lett. 6, 1046–1050 (2003).
Liu, H. & Stiling, P. Testing the enemy release hypothesis: a review and meta-analysis. Biol. Invasions 8, 1535–1545 (2006).
Zhang, Z. et al. Contrasting effects of specialist and generalist herbivores on resistance evolution in invasive plants. Ecology 99, 866–875 (2018).
Chun, Y. J., van Kleunen, M. & Dawson, W. The role of enemy release, tolerance and resistance in plant invasions: linking damage to performance. Ecol. Lett. 13, 937–946 (2010).
Dickie, I. A. et al. The emerging science of linked plant–fungal invasions. New Phytol. 215, 1314–1332 (2017).
Shipunov, A., Newcombe, G., Raghavendra, A. K. H. & Anderson, C. L. Hidden diversity of endophytic fungi in an invasive plant. Am. J. Bot. 95, 1096–1108 (2008).
Hardoim, P. R. et al. The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol. Rev. 79, 293–320 (2015).
Busby, P. E., Peay, K. G. & Newcombe, G. Common foliar fungi of Populus trichocarpa modify Melampsora rust disease severity. New Phytol. 209, 1681–1692 (2016).
Großkopf, T. & Soyer, O. S. Synthetic microbial communities. Curr. Opin. Microbiol. 18, 72–77 (2014).
Divíšek, J. et al. Similarity of introduced plant species to native ones facilitates naturalization, but differences enhance invasion success. Nat. Commun. 9, 4631 (2018).
Feng, Y., Fouqueray, T. D., van Kleunen, M. & Cornelissen, H. Linking Darwin’s naturalisation hypothesis and Elton’s diversity–invasibility hypothesis in experimental grassland communities. J. Ecol. 107, 794–805 (2019).
Li, S. P. et al. The effects of phylogenetic relatedness on invasion success and impact: deconstructing Darwin’s naturalisation conundrum. Ecol. Lett. 18, 1285–1292 (2015).
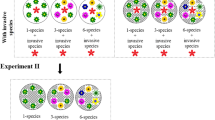
van Kleunen, M., Dawson, W., Bossdorf, O. & Fischer, M. The more the merrier: multi-species experiments in ecology. Basic Appl. Ecol. 15, 1–9 (2014).
FloraWeb (Bundesamt für Naturschutz, 2003); http://www.floraweb.de/
Richardson, D. M. et al. Naturalization and invasion of alien plants: concepts and definitions. Divers. Distrib. 6, 93–107 (2000).
Brinkman, E. P., Van der Putten, W. H., Bakker, E.-J. & Verhoeven, K. J. F. Plant–soil feedback: experimental approaches, statistical analyses and ecological interpretations. J. Ecol. 98, 1063–1073 (2010).
Rinella, M. J. & Reinhart, K. O. Toward more robust plant–soil feedback research. Ecology 99, 550–556 (2018).
Zhang, Z., Liu, Y., Brunel, C. & van Kleunen, M. Evidence for Elton’s diversity–invasibility hypothesis from belowground. Ecology https://doi.org/10.1002/ecy.3187 (accepted).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 41, e1 (2013).
Orgiazzi, A. et al. Unravelling soil fungal communities from different Mediterranean land-use backgrounds. PLoS ONE 7, e34847 (2012).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–13 (2011).
Callahan, B. J. et al. DADA2: high-resolution sample inference from Illumina amplicon data. Nat. Methods 13, 581–583 (2016).
Nilsson, R. H. et al. The UNITE database for molecular identification of fungi: handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res. 47, D259–D264 (2018).
Nguyen, N. H. et al. FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol. 20, 241–248 (2016).
R: A language and environment for statistical computing v.3.6.1 (R Foundation for Statistical Computing, 2019); http://www.R-project.org/
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R Core Team. nlme: Linear and nonlinear mixed effect s models. R package version 3.1-140 (2019).
Gibson, D., Connolly, J., Hartnett, D. & Weidenhamer, J. Designs for greenhouse studies of interactions between plants. J. Ecol. 87, 1–16 (1999).
Aschehoug, E. T., Brooker, R., Atwater, D. Z., Maron, J. L. & Callaway, R. M. The mechanisms and consequences of interspecific competition among plants. Annu. Rev. Ecol. Syst. 47, 263–281 (2016).
Hart, S. P., Burgin, J. R. & Marshall, D. J. Revisiting competition in a classic model system using formal links between theory and data. Ecology 93, 2015–2022 (2012).
Zuur, A., Ieno, E., Walker, N., Saveliev, A. & Smith, G. Mixed Effects Models and Extensions in Ecology with R (Springer, 2009).
Schielzeth, H. Simple means to improve the interpretability of regression coefficients. Methods Ecol. Evol. 1, 103–113 (2010).
Bennett, J. A. & Klironomos, J. Mechanisms of plant–soil feedback: interactions among biotic and abiotic drivers. New Phytol. 222, 91–96 (2019).
Oksanen, J. et al. vegan: Community Ecology Package. R package version 2.5-6 (2019).
Wei, T. & Simko, V. corrplot: Visualization of a correlation matrix. R package version 0.84 (2017).
Acknowledgements
We thank L. Arnold, S. Berg, O. Ficht, M. Fuchs, S. Gommel, E. Mamonova, V. Pasqualetto, C. Rabung, B. Rüter, B. Speißer, H. Vahlenkamp and E. Werner for practical assistance, and J. Levine and R. Rohr for early discussion. Z.Z. was funded by the China Scholarship Council (grant no. 201606100049) and supported by the International Max Planck Research School for Organismal Biology. Y.L. was funded by the Chinese Academy of Sciences (grant nos. Y9H1011001 and Y9B7041001).
Author information
Authors and Affiliations
Contributions
Z.Z. conceived the idea. Z.Z., Y.L. and M.v.K. designed the experiment. Z.Z., Y.L. and C.B. performed the experiment. Z.Z. analysed the data and wrote the manuscript with input from all other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
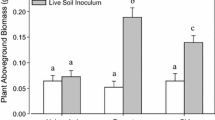
Extended Data Fig. 1 Effects of soil-conditioning treatments on belowground and total biomass of alien (orange) and native (purple) test species that were grown alone.
Mean values (± SEs) were calculated based on biomass of plants grown alone. For the soil-conditioning treatments, ‘non-conditioned’ refers to soil that was not conditioned by any plant, ‘home’ to soil conditioned by the same species as the test species, and ‘alien’ and ‘native’ to soils conditioned by other species than the test species, which were alien or native, respectively.
Extended Data Fig. 2 Effects of soil-conditioning treatments on soil community compositions of bacteria and fungi.
Nonmetric multidimensional scaling (NMDS) was used to visualize differences in the soil microbial communities of the plant species. Data points represent soil samples. Ellipses represent means ± 1 SDs for soil conditioned by aliens (orange) or natives (purple), or not conditioned by plants (grey). The different colors used for the points indicate different species. Soil was either alive (a,c) or sterilized (b,d) before the conditioning treatment.
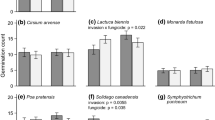
Extended Data Fig. 3 Effects of soil-conditioning treatments on diversity of soil bacteria and fungi and relative abundance of fungal pathogens.
Species richness and Shannon diversity were calculated as diversity metrics. Soil was either alive or sterilized before the conditioning treatment.
Extended Data Fig. 4 Effects of diversity of bacteria and fungi, and relative abundance of fungal pathogens on soil-legacy effects.
Purple dots represent native test species, and orange dots represent alien test species. Negative values of soil-legacy effects indicate that plants grew worse on conditioned soil than on non-conditioned soil. No significant relationship was found. Note that relative abundance of all bacteria and fungi is 1, and thus their correlations to soil-legacy effect was not tested.
Extended Data Fig. 6 Graphical summary of effects of soil-conditioning plants on competitive outcomes between alien and native plants.
a, In pairwise competition, aliens and natives had similar aboveground biomass when grown alone, and suppressed each other equally, as indicated by the same thickness of the red arrows. Consequently, aliens were as competitive as natives (that is natives had similar aboveground biomass when in competition). b, soil conditioned by natives suppressed later aliens and natives equally, as indicated by the same thickness of the blue arrows. Consequently, soil conditioned by natives did not change the competitive outcomes. c, soil conditioned by aliens suppressed later aliens less than natives, as indicated by the thinner blue arrow towards aliens. Consequently, aliens changed the competitive outcomes, favoring later aliens over natives. Effects of soil conditioned by plants on strength of competition (that is red arrows) are not shown, as they did not change competitive outcomes between alien and native plants (for example see Fig. 3c).
Extended Data Fig. 7 Dissimilarities of soil communities within and between species, when sterilized field soil was used as inoculum.
a, e, bacterial communities; b, f, fungal communities, c, g, fungal pathogen communities; d, h, fungal endophyte communities. See Fig. 4 for detailed descriptions.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6, Tables 1–17 and details on statistical analyses.
Rights and permissions
About this article
Cite this article
Zhang, Z., Liu, Y., Brunel, C. et al. Soil-microorganism-mediated invasional meltdown in plants. Nat Ecol Evol 4, 1612–1621 (2020). https://doi.org/10.1038/s41559-020-01311-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-020-01311-0
- Springer Nature Limited
This article is cited by
-
Invasive and native plants show different root responses to feedback-mediated soil heterogeneity
Plant and Soil (2024)
-
Negative conspecific plant-soil feedback on alien plants co-growing with natives is partly mitigated by another alien
Plant and Soil (2024)
-
Core-predominant gut fungus Kazachstania slooffiae promotes intestinal epithelial glycolysis via lysine desuccinylation in pigs
Microbiome (2023)
-
Impact of Parthenium hysterophorus L. invasion on soil fungal communities in the Yellow River Delta
Annals of Microbiology (2023)
-
An exotic plant species indirectly facilitates a secondary exotic plant through increased soil salinity
Biological Invasions (2023)