Abstract
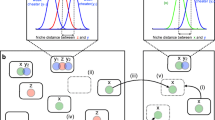
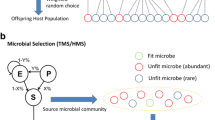
Understanding the ecological and evolutionary processes determining the outcome of biological invasions has been the subject of decades of research with most work focusing on macro-organisms. In the context of microbes, invasions remain poorly understood despite being increasingly recognized as important. To shed light on the factors affecting the success of microbial community invasions, we perform simulations using an individual-based nearly neutral model that combines ecological and evolutionary processes. Our simulations qualitatively recreate many empirical patterns and lead to a description of five general rules of invasion: (1) larger communities evolve better invaders and better defenders; (2) where invader and resident fitness difference is large, invasion success is essentially deterministic; (3) propagule pressure contributes to invasion success, if and only if, invaders and residents are competitively similar; (4) increasing the diversity of invaders has a similar effect to increasing the number of invaders; and (5) more diverse communities more successfully resist invasion.






Similar content being viewed by others
Data availability
All data presented in this paper has been deposited in a public repository and can be accessed at https://github.com/vilacelestin/vilaetal2019.
Code availability
All code presented in this paper has been deposited in a public repository and can be accessed at https://github.com/vilacelestin/vilaetal2019.
References
Ehrenfeld, J. G. Ecosystem consequences of biological invasions. Annu. Rev. Ecol. Evol. Syst. 41, 59–80 (2010).
Lowry, E. et al. Biological invasions: a field synopsis, systematic review, and database of the literature. Ecol. Evol. 3, 182–196 (2012).
Chapman, D., Purse, B. V., Roy, H. E. & Bullock, J. M. Global trade networks determine the distribution of invasive non-native species. Glob. Ecol. Biogeogr. 26, 907–917 (2017).
Gorby, G. L., Robinson, E. N. Jr, Barley, L. R., Clemens, C. M. & McGee, Z. A. Microbial invasion: a covert activity? Can. J. Microbiol. 34, 507–512 (1988).
Cossart, P. & Sansonetti, P. J. Bacterial invasion: the paradigms of enteroinvasive pathogens. Science 304, 242–248 (2004).
Mallon, C. A., van Elsas, J. D. & Salles, J. F. Microbial invasions: the process, patterns, and mechanisms. Trends Microbiol. 23, 719–729 (2015).
Baas Becking, L. G. M. Geobiologie of Inleiding tot de Milieukunde (Van Stockum & Zoon, 1934)
Martiny, J. B. H. et al. Microbial biogeography: putting microorganisms on the map. Nat. Rev. Microbiol. 4, 102–112 (2006).
O’Malley, M. A. The nineteenth century roots of ‘everything is everywhere’. Nat. Rev. Microbiol. 5, 647–651 (2007).
Barberán, A., Casamayor, E. O. & Fierer, N. The microbial contribution to macroecology. Front. Microbiol. 5, 203 (2014).
Horner-Devine, M. C., Lage, M., Hughes, J. B. & Bohannan, B. J. M. A taxa–area relationship for bacteria. Nature 432, 750–753 (2004).
Fierer, N. in Accessing Uncultivated Microorganisms: From the Environment to Organisms and Genomes and Back (ed. Zengler, K.) Ch. 6 (ASM Press, 2008).
Hejda, M., Pyšek, P. & Jarošík, V. Impact of invasive plants on the species richness, diversity and composition of invaded communities. J. Ecol. 97, 393–403 (2009).
Litchman, E. Invisible invaders: non-pathogenic invasive microbes in aquatic and terrestrial ecosystems. Ecol. Lett. 13, 1560–1572 (2010).
Acosta, F., Zamor, R. M., Najar, F. Z., Roe, B. A. & Hambright, K. D. Dynamics of an experimental microbial invasion. Proc. Natl Acad. Sci. USA 112, 11594–11599 (2015).
Rivett, D. W. et al. Elevated success of multispecies bacterial invasions impacts community composition during ecological succession. Ecol. Lett. 21, 516–524 (2018).
Jani, A. J. & Briggs, C. J. The pathogen Batrachochytrium dendrobatidis disturbs the frog skin microbiome during a natural epidemic and experimental infection. Proc. Natl Acad. Sci. USA 111, E5049–E5058 (2014).
Wei, Z. et al. Trophic network architecture of root-associated bacterial communities determines pathogen invasion and plant health. Nat. Commun. 6, 8413 (2015).
Koch, H. & Schmid-Hempel, P. Socially transmitted gut microbiota protect bumble bees against an intestinal parasite. Proc. Natl Acad. Sci. USA 108, 19288–19292 (2011).
Elton, C. S. The Ecology of Invasions by Animals and Plants (Springer, 1958).
Schulze, E.-D. & Mooney, H. A. in Biodiversity and Ecosystem Function (eds Schulze, E.-D. & Mooney, H. A.) 497–510 (Springer, 1994).
Jessup, C. M. et al. Big questions, small worlds: microbial model systems in ecology. Trends Ecol. Evol. 19, 189–197 (2004).
Prosser, J. I. et al. The role of ecological theory in microbial ecology. Nat. Rev. Microbiol. 5, 384–392 (2007).
Lenski, R. E., Rose, M. R., Simpson, S. C. & Tadler, S. C. Long-term experimental evolution in Escherichia coli. I. adaptation and divergence during 2,000 generations. Am. Nat. 138, 1315–1341 (1991).
Wright, E. S. & Vetsigian, K. H. Inhibitory interactions promote frequent bistability among competing bacteria. Nat. Commun. 7, 11274 (2016).
Higgins, L. M., Friedman, J., Shen, H. & Gore, J. Co-occurring soil bacteria exhibit a robust competitive hierarchy and lack of non-transitive interactions. Preprint at https://www.biorxiv.org/content/10.1101/175737v1 (2017).
Foster, K. R. & Bell, T. Competition, not cooperation, dominates interactions among culturable microbial species. Curr. Biol. 22, 1845–1850 (2012).
Coyte, K. Z., Schluter, J. & Foster, K. R. The ecology of the microbiome: networks, competition, and stability. Science 350, 663–666 (2015).
Momeni, B., Xie, L. & Shou, W. Lotka–Volterra pairwise modeling fails to capture diverse pairwise microbial interactions. eLife 6, e25051 (2017).
Goldford, J. E. et al. Emergent simplicity in microbial community assembly. Science 361, 469–474 (2018).
Jousset, A., Schulz, W., Scheu, S. & Eisenhauer, N. Intraspecific genotypic richness and relatedness predict the invasibility of microbial communities. ISME J. 5, 1108–1114 (2011).
Eisenhauer, N., Scheu, S. & Jousset, A. Bacterial diversity stabilizes community productivity. PLoS ONE 7, e34517 (2012).
Mallon, C. A. et al. Resource pulses can alleviate the biodiversity–invasion relationship in soil microbial communities. Ecology 96, 915–926 (2015).
Yang, T. et al. Resource availability modulates biodiversity–invasion relationships by altering competitive interactions. Environ. Microbiol. 19, 2984–2991 (2017).
Jones, M. L., Ramoneda, J., Rivett, D. W. & Bell, T. Biotic resistance shapes the influence of propagule pressure on invasion success in bacterial communities. Ecology 98, 1743–1749 (2017).
Rillig, M. C. et al. Interchange of entire communities: microbial community coalescence. Trends Ecol. Evol. 30, 470–476 (2015).
Lu, N., Sanchez-Gorostiaga, A., Tikhonov, M. & Sanchez, A. Cohesiveness in microbial community coalescence. Preprint at https://www.biorxiv.org/content/10.1101/282723v1 (2018).
Hellweger, F. L., Clegg, R. J., Clark, J. R., Plugge, C. M. & Kreft, J.-U. Advancing microbial sciences by individual-based modelling. Nat. Rev. Microbiol. 14, 461–471 (2016).
Volterra, V. Variations and fluctuations of the number of individuals in animal species living together. ICES J. Mar. Sci. 3, 3–51 (1928).
Mounier, J. et al. Microbial interactions within a cheese microbial community. Appl. Environ. Microbiol. 74, 172–181 (2008).
MacArthur, R. Species packing and competitive equilibrium for many species. Theor. Popul. Biol. 1, 1–11 (1970).
Chesson, P. MacArthur’s consumer-resource model. Theor. Popul. Biol. 37, 26–38 (1990).
Bashan, A. et al. Universality of human microbial dynamics. Nature 534, 259–262 (2016).
Wade, M. J. et al. Perspectives in mathematical modelling for microbial ecology. Ecol. Model. 321, 64–74 (2016).
Zuñiga, C., Zaramela, L. & Zengler, K. Elucidation of complexity and prediction of interactions in microbial communities. Microb. Biotechnol. 10, 1500–1522 (2017).
Kinnunen, M. et al. A conceptual framework for invasion in microbial communities. ISME J. 10, 2773–2775 (2016).
Kinnunen, M., Dechesne, A., Albrechtsen, H.-J. & Smets, B. F. Stochastic processes govern invasion success in microbial communities when the invader is phylogenetically close to resident bacteria. ISME J. 12, 2748–2756 (2018).
Phillips, B. L., Brown, G. P., Webb, J. K. & Shine, R. Invasion and the evolution of speed in toads. Nature 439, 803 (2006).
Sanchez, A. & Gore, J. Feedback between population and evolutionary dynamics determines the fate of social microbial populations. PLoS Biol. 11, e1001547 (2013).
Faillace, C. A. & Morin, P. J. Evolution alters the consequences of invasions in experimental communities. Nat. Ecol. Evol. 1, 13 (2016).
Hubbell, S. The Unified Neutral Theory of Biodiversity and Biogeography (MPB-32) (Princeton Univ. Press, 2001).
Fisher, C. K. & Mehta, P. The transition between the niche and neutral regimes in ecology. Proc. Natl Acad. Sci. USA 111, 13111–13116 (2014).
O’Dwyer, J. P. & Chisholm, R. A mean field model for competition: from neutral ecology to the Red Queen. Ecol. Lett. 17, 961–969 (2014).
Rosindell, J., Harmon, L. J. & Etienne, R. S. Unifying ecology and macroevolution with individual-based theory. Ecol. Lett. 18, 472–482 (2015).
Gilpin, M. Community-level competition: asymmetrical dominance. Proc. Natl Acad. Sci. USA 91, 3252–3254 (1994).
Rillig, M. C., Tsang, A. & Roy, J. Microbial community coalescence for microbiome engineering. Front. Microbiol. 7, 1967 (2016).
Sierocinski, P. et al. A single community dominates structure and function of a mixture of multiple methanogenic communities. Curr. Biol. 27, 3390–3395 (2017).
Fisher, R. A. The Genetical Theory of Natural Selection (Clarendon Press, 1930).
Ohta, T. Slightly deleterious mutant substitutions in evolution. Nature 246, 96–98 (1973).
Desai, M. M., Fisher, D. S. & Murray, A. W. The speed of evolution and maintenance of variation in asexual populations. Curr. Biol. 17, 385–394 (2007).
Doolittle, W. F. & Papke, R. T. Genomics and the bacterial species problem. Genome Biol. 7, 116 (2006).
van Elsas, J. D. et al. Microbial diversity determines the invasion of soil by a bacterial pathogen. Proc. Natl Acad. Sci. USA 109, 1159–1164 (2012).
Rouzine, I. M., Brunet, E. & Wilke, C. O. The traveling-wave approach to asexual evolution: Muller’s ratchet and speed of adaptation. Theor. Popul. Biol. 73, 24–46 (2008).
Wiser, M. J., Ribeck, N. & Lenski, R. E. Long-term dynamics of adaptation in asexual populations. Science 342, 1364–1367 (2013).
Lenski, R. E. et al. Sustained fitness gains and variability in fitness trajectories in the long-term evolution experiment with Escherichia coli. Proc. Biol. Sci. 282, 20152292 (2015).
MacArthur, R. H. & Wilson, E. O. The Theory of Island Biogeography (Princeton Univ. Press, 1967).
Muller, H. J. Some genetic aspects of sex. Am. Nat. 66, 118–138 (1932).
Gerrish, P. J. & Lenski, R. E. in Mutation and Evolution (eds. Woodruff, R. C. & Thompson, J. N.) 127–144 (Springer, 1998).
Park, S.-C. & Krug, J. Clonal interference in large populations. Proc. Natl Acad. Sci. USA 104, 18135–18140 (2007).
Rosindell, J., Hubbell, S. P., He, F., Harmon, L. J. & Etienne, R. S. The case for ecological neutral theory. Trends Ecol. Evol. 27, 203–208 (2012).
Guisan, A. & Zimmermann, N. E. Predictive habitat distribution models in ecology. Ecol. Model. 135, 147–186 (2000).
Uden, D. R., Allen, C. R., Angeler, D. G., Corral, L. & Fricke, K. A. Adaptive invasive species distribution models: a framework for modeling incipient invasions. Biol. Invasions 17, 2831–2850 (2015).
Barbet-Massin, M., Rome, Q., Villemant, C. & Courchamp, F. Can species distribution models really predict the expansion of invasive species? PLoS ONE 13, e0193085 (2018).
Miralles, R., Gerrish, P. J., Moya, A. & Elena, S. F. Clonal interference and the evolution of RNA viruses. Science 285, 1745–1747 (1999).
Tilman, D., Wedin, D. & Knops, J. Productivity and sustainability influenced by biodiversity in grassland ecosystems. Nature 379, 718 (1996).
Tilman, D. Resource competition and community structure. Monogr. Popul. Biol. 17, 1–296 (1982).
Tilman, D. Niche tradeoffs, neutrality, and community structure: a stochastic theory of resource competition, invasion, and community assembly. Proc. Natl Acad. Sci. USA 101, 10854–10861 (2004).
Loreau, M. & Hector, A. Partitioning selection and complementarity in biodiversity experiments. Nature 412, 72–76 (2001).
De Roy, K. et al. Environmental conditions and community evenness determine the outcome of biological invasion. Nat. Commun. 4, 1383 (2013).
Houseman, G. R., Foster, B. L. & Brassil, C. E. Propagule pressure–invasibility relationships: testing the influence of soil fertility and disturbance with Lespedeza cuneata. Oecologia 174, 511–520 (2014).
Ketola, T., Saarinen, K. & Lindström, L. Propagule pressure increase and phylogenetic diversity decrease community’s susceptibility to invasion. BMC Ecol. 17, 15 (2017).
Barney, J. N., Ho, M. W. & Atwater, D. Z. Propagule pressure cannot always overcome biotic resistance: the role of density-dependent establishment in four invasive species. Weed Res. 56, 208–218 (2016).
Dillon, R. J., Vennard, C. T., Buckling, A. & Charnley, A. K. Diversity of locust gut bacteria protects against pathogen invasion. Ecol. Lett. 8, 1291–1298 (2005).
Fargione, J. E. & Tilman, D. Diversity decreases invasion via both sampling and complementarity effects: diversity causes invader underyielding. Ecol. Lett. 8, 604–611 (2005).
Jiang, L. & Morin, P. J. Productivity gradients cause positive diversity–invasibility relationships in microbial communities. Ecol. Lett. 7, 1047–1057 (2004).
Ratzke, C. & Gore, J. Modifying and reacting to the environmental pH can drive bacterial interactions. PLoS Biol. 16, e2004248 (2018).
Simberloff, D. The role of propagule pressure in biological invasions. Annu. Rev. Ecol. Evol. Syst. 40, 81–102 (2009).
Huey, R. B., Gilchrist, G. W., Carlson, M. L., Berrigan, D. & Serra, L. Rapid evolution of a geographic cline in size in an introduced fly. Science 287, 308–309 (2000).
Ochocki, B. M. & Miller, T. E. X. Rapid evolution of dispersal ability makes biological invasions faster and more variable. Nat. Commun. 8, 14315 (2017).
Wilson, E. O. & Peter, F. M. Diversity and Biological Invasions of Oceanic Islands (National Academies Press, 1988).
Carlquist, S. J. Island Biology (Columbia Univ. Press, 1974).
Colautti, R. I., Bailey, S. A., van Overdijk, C. D. A., Amundsen, K. & MacIsaac, H. J. Characterised and projected costs of nonindigenous species in canada. Biol. Invasions 8, 45–59 (2006).
Rosindell, J. & Harmon, L. J. A unified model of species immigration, extinction and abundance on islands. J. Biogeogr. 40, 1107–1118 (2013).
Rosindell, J., Cornell, S. J., Hubbell, S. P. & Etienne, R. S. Protracted speciation revitalizes the neutral theory of biodiversity. Ecol. Lett. 13, 716–727 (2010).
Buckling, A. & Rainey, P. B. The role of parasites in sympatric and allopatric host diversification. Nature 420, 496–499 (2002).
Hansen, S. K., Rainey, P. B., Haagensen, J. A. J. & Molin, S. Evolution of species interactions in a biofilm community. Nature 445, 533–536 (2007).
Kerr, B., Riley, M. A., Feldman, M. W. & Bohannan, B. J. M. Local dispersal promotes biodiversity in a real-life game of rock–paper–scissors. Nature 418, 171–174 (2002).
Mickalide, H. & Kuehn, S. Higher-order interaction inhibits bacterial invasion of a phototroph-predator microbial community. Preprint at https://www.biorxiv.org/content/10.1101/564260v1 (2019).
Li, M. et al. Facilitation promotes invasions in plant‐associated microbial communities. Ecol. Lett. 22, 149–158 (2019).
Niehaus, L. et al. Microbial coexistence through chemical-mediated interactions. Nat. Commun. 10, 2052 (2019).
Acknowledgements
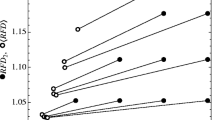
J.V. and M.P. were postgraduate students on the Computational Methods in Ecology and Evolution course at Imperial College, London. J.R. was funded by fellowships from the Natural Environment Research Council (NE/I021179, NE/L011611/1). T.B. was supported by a Royal Society University Research Fellowship. We thank members of the A. Sanchez and R. Chisholm Laboratories for useful discussions about the work. We thank N. Kristensen for comments on the manuscript. We thank A. Jousset and D. Hambright for providing us with access to data for re-plotting in Fig. 6. Our simulations were performed using the high-performance computing facility at Imperial College, London. This study is a contribution to Imperial College’s Grand Challenges in Ecosystems and the Environment initiative.
Author information
Authors and Affiliations
Contributions
All authors contributed to designing the study. J.C.C.V. performed the analyses with input from J.R. and with reference to code written by M.P. J.C.CV. and J.R. wrote the paper. All authors revised the paper. J.C.C.V. and M.P. were postgraduate students in the research group of J.R. M.L.J. was a PhD student in the group of T.B.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, Table 1 and Figs. 1–7.
Rights and permissions
About this article
Cite this article
Vila, J.C.C., Jones, M.L., Patel, M. et al. Uncovering the rules of microbial community invasions. Nat Ecol Evol 3, 1162–1171 (2019). https://doi.org/10.1038/s41559-019-0952-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-019-0952-9
- Springer Nature Limited
This article is cited by
-
Relative dispersion ratios following fecal microbiota transplant elucidate principles governing microbial migration dynamics
Nature Communications (2024)
-
Associations of Batrachochytrium dendrobatidis with skin bacteria and fungi on Asian amphibian hosts
ISME Communications (2023)
-
Solutions in microbiome engineering: prioritizing barriers to organism establishment
The ISME Journal (2022)
-
Aquaculture rearing systems induce no legacy effects in Atlantic cod larvae or their rearing water bacterial communities
Scientific Reports (2022)
-
Microbial invasion of a toxic medium is facilitated by a resident community but inhibited as the community co-evolves
The ISME Journal (2022)