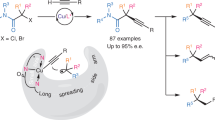
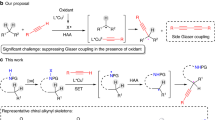
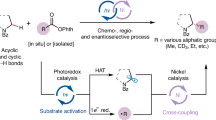
Abstract
Although α-chiral C(sp3)–S bonds are of enormous importance in organic synthesis and related areas, the transition-metal-catalysed enantioselective C(sp3)–S bond construction still represents an underdeveloped domain probably due to the difficult heterolytic metal–sulfur bond cleavage and notorious catalyst-poisoning capability of sulfur nucleophiles. Here we demonstrate the use of chiral tridentate anionic ligands in combination with Cu(I) catalysts to enable a biomimetic enantioconvergent radical C(sp3)–S cross-coupling reaction of both racemic secondary and tertiary alkyl halides with highly transformable sulfur nucleophiles. This protocol not only exhibits a broad substrate scope with high enantioselectivity but also provides universal access to a range of useful α-chiral alkyl organosulfur compounds with different sulfur oxidation states, thus providing a complementary approach to known asymmetric C(sp3)–S bond formation methods. Mechanistic results support a biomimetic radical homolytic substitution pathway for the critical C(sp3)–S bond formation step.




Similar content being viewed by others
Data availability
Data relating to the materials and methods, optimization studies, experimental procedures, mechanistic studies, DFT calculations, HPLC spectra, NMR spectra, and mass spectrometry are available in the Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition numbers CCDC 2212974 (1), 2213037 (52) and 2213038 (83). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Toru, T. & Bolm, C. (eds) Organosulfur Chemistry in Asymmetric Synthesis (Wiley, 2008).
Dénès, F., Pichowicz, M., Povie, G. & Renaud, P. Thiyl radicals in organic synthesis. Chem. Rev. 114, 2587–2693 (2014).
Dunbar, K. L., Scharf, D. H., Litomska, A. & Hertweck, C. Enzymatic carbon–sulfur bond formation in natural product biosynthesis. Chem. Rev. 117, 5521–5577 (2017).
Wang, N., Saidhareddy, P. & Jiang, X. Construction of sulfur-containing moieties in the total synthesis of natural products. Nat. Prod. Rep. 37, 246–275 (2020).
Scott, K. A. & Njardarson, J. T. Analysis of US FDA-approved drugs containing sulfur atoms. Top. Curr. Chem. 376, 5 (2018).
Lamberth, C. Sulfur chemistry in crop protection. J. Sulfur Chem. 25, 39–62 (2004).
Subramanian, H., Moorthy, R. & Sibi, M. P. Thiyl radicals: from simple radical additions to asymmetric catalysis. Angew. Chem. Int. Ed. 53, 13660–13662 (2014).
McGarrigle, E. M. et al. Chalcogenides as organocatalysts. Chem. Rev. 107, 5841–5883 (2007).
Mellah, M., Voituriez, A. & Schulz, E. Chiral sulfur ligands for asymmetric catalysis. Chem. Rev. 107, 5133–5209 (2007).
Margalef, J. et al. Evolution in heterodonor P–N, P–S and P–O chiral ligands for preparing efficient catalysts for asymmetric catalysis. From design to applications. Coord. Chem. Rev. 446, 214120 (2021).
Broderick, J. B., Duffus, B. R., Duschene, K. S. & Shepard, E. M. Radical S-adenosylmethionine enzymes. Chem. Rev. 114, 4229–4317 (2014).
Nakai, T. et al. The radical S-adenosyl-l-methionine enzyme QhpD catalyzes sequential formation of intra-protein sulfur-to-methylene carbon thioether bonds. J. Biol. Chem. 290, 11144–11166 (2015).
Chauhan, P., Mahajan, S. & Enders, D. Organocatalytic carbon–sulfur bond-forming reactions. Chem. Rev. 114, 8807–8864 (2014).
Kikuchi, J. & Terada, M. Enantioconvergent substitution reactions of racemic electrophiles by organocatalysis. Chem. Eur. J. 27, 10215–10225 (2021).
Zhang, X. & Tan, C.-H. Stereospecific and stereoconvergent nucleophilic substitution reactions at tertiary carbon centers. Chem 7, 1451–1486 (2021).
Yu, J.-S. et al. Catalytic enantioselective construction of sulfur-containing tetrasubstituted carbon stereocenters. ACS Catal. 6, 5319–5344 (2016).
Zhu, C., Cai, Y. & Jiang, H. Recent advances for the synthesis of chiral sulfones with the sulfone moiety directly connected to the chiral center. Org. Chem. Front. 8, 5574–5589 (2021).
Cheng, Q. et al. Iridium-catalyzed asymmetric allylic substitution reactions. Chem. Rev. 119, 1855–1969 (2019).
Choi, J., Martín-Gago, P. & Fu, G. C. Stereoconvergent arylations and alkenylations of unactivated alkyl electrophiles: catalytic enantioselective synthesis of secondary sulfonamides and sulfones. J. Am. Chem. Soc. 136, 12161–12165 (2014).
He, S.-J. et al. Nickel-catalyzed enantioconvergent reductive hydroalkylation of olefins with α-heteroatom phosphorus or sulfur alkyl electrophiles. J. Am. Chem. Soc. 142, 214–221 (2020).
Xia, X. & Wang, Z. Cr-catalyzed diastereo- and enantioselective synthesis of β-hydroxy sulfides and selenides. ACS Catal. 12, 11152–11158 (2022).
Sundaravelu, N., Sangeetha, S. & Sekar, G. Metal-catalyzed C–S bond formation using sulfur surrogates. Org. Biomol. Chem. 19, 1459–1482 (2021).
Hegedus, L. L. & McCabe, R. W. Catalyst poisoning. Catal. Rev. Sci. Eng. 23, 377–476 (2007).
Evans, D. A., Miller, S. J., Lectka, T. & von Matt, P. Chiral bis(oxazoline)copper(II) complexes as Lewis acid catalysts for the enantioselective Diels–Alder reaction. J. Am. Chem. Soc. 121, 7559–7573 (1999).
Trost, B. M., Krische, M. J., Radinov, R. & Zanoni, G. On asymmetric induction in allylic alkylation via enantiotopic facial discrimination. J. Am. Chem. Soc. 118, 6297–6298 (1996).
Cai, A. & Kleij, A. W. Regio‐ and enantioselective preparation of chiral allylic sulfones featuring elusive quaternary stereocenters. Angew. Chem. Int. Ed. 58, 14944–14949 (2019).
Khan, A., Zhao, H., Zhang, M., Khan, S. & Zhao, D. Regio- and enantioselective synthesis of sulfone-bearing quaternary carbon stereocenters by Pd-catalyzed allylic substitution. Angew. Chem. Int. Ed. 59, 1340–1345 (2020).
Zhang, Q., Dong, D. & Zi, W. Palladium-catalyzed regio- and enantioselective hydrosulfonylation of 1,3-dienes with sulfinic acids: scope, mechanism, and origin of selectivity. J. Am. Chem. Soc. 142, 15860–15869 (2020).
Li, M.-M., Cheng, L., Xiao, L.-J., Xie, J.-H. & Zhou, Q.-L. Palladium-catalyzed asymmetric hydrosulfonylation of 1,3-dienes with sulfonyl hydrazides. Angew. Chem. Int. Ed. 60, 2948–2951 (2021).
Pritzius, A. B. & Breit, B. Asymmetric rhodium-catalyzed addition of thiols to allenes: synthesis of branched allylic thioethers and sulfones. Angew. Chem. Int. Ed. 54, 3121–3125 (2015).
Yang, X.-H., Davison, R. T. & Dong, V. M. Catalytic hydrothiolation: regio- and enantioselective coupling of thiols and dienes. J. Am. Chem. Soc. 140, 10443–10446 (2018).
Han, X., Wang, M., Liang, Y., Zhao, Y. & Shi, Z. Regio- and enantioselective nucleophilic addition to gem-difluoroallenes. Nat. Synth. 1, 227–234 (2022).
Ueda, M. & Hartwig, J. F. Iridium-catalyzed, regio- and enantioselective allylic substitution with aromatic and aliphatic sulfinates. Org. Lett. 12, 92–94 (2010).
He, Z.-T. & Hartwig, J. F. Enantioselective α-functionalizations of ketones via allylic substitution of silyl enol ethers. Nat. Chem. 11, 177–183 (2019).
Roggen, M. & Carreira, E. M. Enantioselective allylic thioetherification: the effect of phosphoric acid diester on iridium-catalyzed enantioconvergent transformations. Angew. Chem. Int. Ed. 51, 8652–8655 (2012).
Xu, B. et al. Highly enantioselective S–H bond insertion cooperatively catalyzed by dirhodium complexes and chiral spiro phosphoric acids. Chem. Sci. 5, 1442–1448 (2014).
Cavell, K. J., Hill, J. O. & Magee, R. J. Standard enthalpy of formation of bis(diethyldithiocarbamato)copper(II) at 298 K and the copper–sulphur bond energy. J. Chem. Soc. Dalton Trans. 1638–1640 (1980).
Jarrett, J. T. The biosynthesis of thiol- and thioether-containing cofactors and secondary metabolites catalyzed by radical S-adenosylmethionine enzymes. J. Biol. Chem. 290, 3972–3979 (2015).
Taylor, A. M., Farrar, C. E. & Jarrett, J. T. 9-Mercaptodethiobiotin is formed as a competent catalytic intermediate by Escherichia coli biotin synthase. Biochemistry 47, 9309–9317 (2008).
Cicchillo, R. M. et al. Escherichia coli lipoyl synthase binds two distinct [4Fe–4S] clusters per polypeptide. Biochemistry 43, 11770–11781 (2004).
Grove, T. L. et al. Structural insights into thioether bond formation in the biosynthesis of sactipeptides. J. Am. Chem. Soc. 139, 11734–11744 (2017).
MacMahon, T. J., Jackson, T. C. & Freiser, B. S. A gas-phase study of FeSn+ (n = 1–6). J. Am. Chem. Soc. 111, 421–427 (2002).
He, J. et al. Catalytic decarboxylative radical sulfonylation. Chem 6, 1149–1159 (2020).
Choi, J. & Fu, G. C. Transition metal–catalyzed alkyl-alkyl bond formation: another dimension in cross-coupling chemistry. Science 356, eaaf7230 (2017).
Cherney, A. H., Kadunce, N. T. & Reisman, S. E. Enantioselective and enantiospecific transition-metal-catalyzed cross-coupling reactions of organometallic reagents to construct C–C bonds. Chem. Rev. 115, 9587–9652 (2015).
Dong, X.-Y., Li, Z.-L., Gu, Q.-S. & Liu, X.-Y. Ligand development for copper-catalyzed enantioconvergent radical cross-coupling of racemic alkyl halides. J. Am. Chem. Soc. 144, 17319–17329 (2022).
Dong, X.-Y. et al. A general asymmetric copper-catalysed Sonogashira C(sp3)–C(sp) coupling. Nat. Chem. 11, 1158–1166 (2019).
Wang, F.-L. et al. Mechanism-based ligand design for copper-catalysed enantioconvergent C(sp3)–C(sp) cross-coupling of tertiary electrophiles with alkynes. Nat. Chem. 14, 949–957 (2022).
Zhang, Y.-F. et al. Enantioconvergent Cu-catalyzed radical C–N coupling of racemic secondary alkyl halides to access α-chiral primary amines. J. Am. Chem. Soc. 143, 15413–15419 (2021).
Chen, J.-J. et al. Enantioconvergent Cu-catalysed N-alkylation of aliphatic amines. Nature 618, 294–300 (2023).
Wang, L.-L. et al. A general copper-catalysed enantioconvergent radical Michaelis–Becker-type C(sp3)–P cross-coupling. Nat. Synth. 2, 430–438 (2023).
Lin, Q. et al. Efficient C(sp3alkyl)–SCF3 bond formations via copper-mediated trifluoromethylthiolation of alkyl halides. Org. Biomol. Chem. 12, 5500–5508 (2014).
Bulman Page, P. C., Wilkes, R. D. & Reynolds, D. in Comprehensive Organic Functional Group Transformations (eds Katritzky, A. R., Meth-Cohn, O., & Rees, C. W.) 113–275 (Elsevier, 1995).
Sladojevich, F., Trabocchi, A., Guarna, A. & Dixon, D. J. A new family of cinchona-derived amino phosphine precatalysts: application to the highly enantio- and diastereoselective silver-catalyzed isocyanoacetate aldol reaction. J. Am. Chem. Soc. 133, 1710–1713 (2011).
Mampuys, P., McElroy, C. R., Clark, J. H., Orru, R. V. A. & Maes, B. U. W. Thiosulfonates as emerging reactants: synthesis and applications. Adv. Synth. Catal. 362, 3–64 (2020).
Zilbeyaz, K., Oztekin, A. & Kutluana, E. G. Design and synthesis of garlic-related unsymmetrical thiosulfonates as potential Alzheimer’s disease therapeutics: in vitro and in silico study. Bioorg. Med. Chem. 40, 116194 (2021).
Trost, B. M. & Li, C.-J. (eds) Modern Alkyne Chemistry: Catalytic and Atom-Economic Transformations (Wiley, 2015).
Gómez, J. E., Cristòfol, À. & Kleij, A. W. Copper-catalyzed enantioselective construction of tertiary propargylic sulfones. Angew. Chem. Int. Ed. 58, 3903–3907 (2019).
Gao, X., Xiao, Y.-L., Zhang, S., Wu, J. & Zhang, X. Copper-catalyzed enantioselective trifluoromethylthiolation of secondary propargyl sulfonates. CCS Chem. 3, 1463–1471 (2021).
Liu, L. et al. Copper-catalyzed intermolecular enantioselective radical oxidative C(sp3)–H/C(sp)–H cross-coupling with rationally designed oxazoline-derived N,N,P(O)-ligands. Angew. Chem. Int. Ed. 60, 26710–26717 (2021).
Su, B., Lee, T. & Hartwig, J. F. Iridium-catalyzed, β-selective C(sp3)–H silylation of aliphatic amines to form silapyrrolidines and 1,2-amino alcohols. J. Am. Chem. Soc. 140, 18032–18038 (2018).
Cheng, X., Lu, H. & Lu, Z. Enantioselective benzylic C–H arylation via photoredox and nickel dual catalysis. Nat. Commun. 10, 3549 (2019).
Kainz, Q. M. et al. Asymmetric copper-catalyzed C–N cross-couplings induced by visible light. Science 351, 681–684 (2016).
Wang, Z., Yin, H. & Fu, G. C. Catalytic enantioconvergent coupling of secondary and tertiary electrophiles with olefins. Nature 563, 379–383 (2018).
Wang, Z., Yang, Z.-P. & Fu, G. C. Quaternary stereocentres via catalytic enantioconvergent nucleophilic substitution reactions of tertiary alkyl halides. Nat. Chem. 13, 236–242 (2021).
Deng, Q.-H., Melen, R. L. & Gade, L. H. Anionic chiral tridentate N-donor pincer ligands in asymmetric catalysis. Acc. Chem. Res. 47, 3162–3173 (2014).
Dong, J., Krasnova, L., Finn, M. G. & Sharpless, K. B. Sulfur(VI) fluoride exchange (SuFEx): another good reaction for click chemistry. Angew. Chem. Int. Ed. 53, 9430–9448 (2014).
Acknowledgements
The authors highly appreciate the help of C. Yu and S. Chen from SUSTech in preparing the image of protein CteB in Fig. 1a and the assistance of SUSTech Core Research Facilities. Financial support from the National Natural Science Foundation of China (22025103, 92256301 and 21831002), the National Key R&D Program of China (2021YFF0701604 and 2021YFF0701704), Guangdong Innovative Program (2019BT02Y335), Shenzhen Science and Technology Program (KQTD20210811090112004 and JCYJ20220818100604009), and Shenzhen Special Funds (JCYJ20200109141001789) is gratefully acknowledged. We appreciate the assistance of SUSTech Core Research Facilities. Calculations were performed on the high‐performance computing system at the Department of Chemistry, Zhejiang University.
Author information
Authors and Affiliations
Contributions
Y.T., X.-T.L., J.C. and A.G. designed the experiments and analysed the data. Y.T., X.-T.L., J.C., A.G., N.-Y.Y., Z.L., K.-X.G., W.Z. and H.-T.W. performed the experiments. X.H. designed the DFT calculations. J.-R.L. performed the DFT calculations. Y.T., Z.-L.L., Q.-S.G., and X.-Y.L. wrote the manuscript. X.-Y.L. conceived and supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Chaozhong Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–17 and Tables 1–13, experimental procedures, synthetic procedures, characterization data, DFT calculations, and mechanistic discussion.
Supplementary Data 1
Crystallographic data for compound 1; CCDC reference 2212974.
Supplementary Data 2
Crystallographic data for compound 52; CCDC reference 2213037.
Supplementary Data 3
Crystallographic data for compound 83; CCDC reference 2213038.
Supplementary Data 4
Tables of energies and coordinates in xyz format.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tian, Y., Li, XT., Liu, JR. et al. A general copper-catalysed enantioconvergent C(sp3)–S cross-coupling via biomimetic radical homolytic substitution. Nat. Chem. 16, 466–475 (2024). https://doi.org/10.1038/s41557-023-01385-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-023-01385-w
- Springer Nature Limited