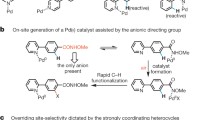
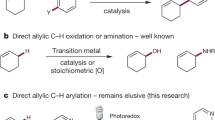
Abstract
The introduction of a trifluoromethyl (CF3) group can dramatically improve a compound’s biological properties. Despite the well-established importance of trifluoromethylated compounds, general methods for the trifluoromethylation of alkyl C–H bonds remain elusive. Here we report the development of a dual-catalytic C(sp3)–H trifluoromethylation through the merger of light-driven, decatungstate-catalysed hydrogen atom transfer and copper catalysis. This metallaphotoredox methodology enables the direct conversion of both strong aliphatic and benzylic C–H bonds into the corresponding C(sp3)–CF3 products in a single step using a bench-stable, commercially available trifluoromethylation reagent. The reaction requires only a single equivalent of substrate and proceeds with excellent selectivity for positions distal to unprotected amines. To demonstrate the utility of this new methodology for late-stage functionalization, we have directly derivatized a broad range of approved drugs and natural products to generate valuable trifluoromethylated analogues. Preliminary mechanistic experiments reveal that a ‘Cu–CF3’ species is formed during this process and the critical C(sp3)–CF3 bond-forming step involves the copper catalyst.






Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information.
References
Meanwell, N. A. Fluorine and fluorinated motifs in the design and application of bioisosteres for drug design. J. Med. Chem. 61, 5822–5880 (2018).
Furet, P. et al. Discovery of NVP-BYL719 a potent and selective phosphatidylinositol-3 kinase α inhibitor selected for clinical evaluation. Bioorg. Med. Chem. Lett. 23, 3741–3748 (2013).
Alonso, C., Martínez de Marigorta, E., Rubiales, G. & Palacios, F. Carbon trifluoromethylation reactions of hydrocarbon derivatives and heteroarenes. Chem. Rev. 115, 1847–1935 (2015).
Charpentier, J., Früh, N. & Togni, A. Electrophilic trifluoromethylation by use of hypervalent iodine reagents. Chem. Rev. 115, 650–682 (2015).
Ji, Y. et al. Innate C–H trifluoromethylation of heterocycles. Proc. Natl Acad. Sci. USA 108, 14411–14415 (2011).
Nagib, D. A. & MacMillan, D. W. C. Trifluoromethylation of arenes and heteroarenes by means of photoredox catalysis. Nature 480, 224–228 (2011).
Wang, X. et al. Copper-catalyzed C(sp 3)–C(sp 3) bond formation using a hypervalent iodine reagent: an efficient allylic trifluoromethylation. J. Am. Chem. Soc. 133, 16410–16413 (2011).
Parsons, A. T. & Buchwald, S. L. Copper-catalyzed trifluoromethylation of unactivated olefins. Angew. Chem. Int. Ed. 50, 9120–9123 (2011).
Xiao, H. et al. Copper-catalyzed late-stage benzylic C(sp 3)–H trifluoromethylation. Chem 5, 940–949 (2019).
Paeth, M. et al. Copper-mediated trifluoromethylation of benzylic Csp 3–H bonds. Chem. Eur. J. 24, 11559–11563 (2018).
Guo, S., AbuSalim, D. I. & Cook, S. P. Aqueous benzylic C–H trifluoromethylation for late-stage functionalization. J. Am. Chem. Soc. 140, 12378–12382 (2018).
Liu, Z. et al. Copper‐catalyzed remote C(sp 3)−H trifluoromethylation of carboxamides and sulfonamides. Angew. Chem. Int. Ed. 58, 2510–2513 (2019).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Shaw, M. H., Shurtleff, V. W., Terrett, J. A., Cuthbertson, J. D. & MacMillan, D. W. C. Native functionality in triple catalytic cross-coupling: sp 3 C-H bonds as latent nucleophiles. Science 352, 1304–1308 (2016).
Heitz, D. R., Tellis, J. C. & Molander, G. A. Photochemical nickel-catalyzed C–H arylation: synthetic scope and mechanistic investigations. J. Am. Chem. Soc. 138, 12715–12718 (2016).
Shields, B. J. & Doyle, A. G. Direct C(sp 3)–H cross coupling enabled by catalytic generation of chlorine radicals. J. Am. Chem. Soc. 138, 12719–12722 (2016).
Shen, Y., Gu, Y. & Martin, R. sp 3 C–H arylation and alkylation enabled by the synergy of triplet excited ketones and nickel catalysts. J. Am. Chem. Soc. 140, 12200–12209 (2018).
McLean, E. B. & Lee, A.-L. Dual copper- and photoredox-catalysed reactions. Tetrahedron 74, 4881–4902 (2018).
Ferraudi, G. Photochemical generation of metastable methylcopper complexes. Oxidation–reduction of methyl radicals by copper complexes. Inorg. Chem. 17, 2506–2508 (1978).
Freiberg, M. & Meyerstein, D. Reactions of aliphatic free radicals with copper cations in aqueous solution. Part 2. Reactions with cupric ions: a pulse radiolysis study. J. Chem. Soc. Faraday Trans. 1, 1825–1837 (1980).
Navon, N., Golub, G., Cohen, H. & Meyerstein, D. Kinetics and reaction mechanisms of copper(i) complexes with aliphatic free radicals in aqueous solutions. A pulse-radiolysis study. Organometallics 14, 5670–5676 (1995).
Casitas, A. & Ribas, X. The role of organometallic copper(iii) complexes in homogeneous catalysis. Chem. Sci. 4, 2301–2318 (2013).
Liang, Y., Zhang, X. & MacMillan, D. W. C. Decarboxylative sp 3 C-N coupling via dual copper and photoredox catalysis. Nature 559, 83–88 (2018).
Kautzky, J. A., Wang, T., Evans, R. W. & MacMillan, D. W. C. Decarboxylative trifluoromethylation of aliphatic carboxylic acids. J. Am. Chem. Soc. 140, 6522–6526 (2018).
Kornfilt, D. J. P. & MacMillan, D. W. C. Copper-catalyzed trifluoromethylation of alkyl bromides. J. Am. Chem. Soc. 141, 6853–6858 (2019).
Duncan, D. C. & Fox, M. A. Early events in decatungstate photocatalyzed oxidations: a nanosecond laser transient absorbance reinvestigation. J. Phys. Chem. A 102, 4559–4567 (1998).
De Waele, V., Poizat, O., Fagnoni, M., Bagno, A. & Ravelli, D. Unraveling the key features of the reactive state of decatungstate anion in hydrogen atom transfer (HAT) photocatalysis. ACS Catal. 6, 7174–7182 (2016).
Tzirakis, M. D., Lykakis, I. N. & Orfanopoulos, M. Decatungstate as an efficient photocatalyst in organic chemistry. Chem. Soc. Rev. 38, 2609–2621 (2009).
Schultz, D. M. et al. Oxyfunctionalization of the remote C–H bonds of aliphatic amines by decatungstate photocatalysis. Angew. Chem. Int. Ed. 56, 15274–15278 (2017).
Laudadio, G. et al. Selective C(sp 3)−H aerobic oxidation enabled by decatungstate photocatalysis in flow. Angew. Chem. Int. Ed. 57, 4078–4082 (2018).
West, J. G., Huang, D. & Sorensen, E. J. Acceptorless dehydrogenation of small molecules through cooperative base metal catalysis. Nat. Commun. 6, 10093 (2015).
Halperin, S. D., Fan, H., Chang, S., Martin, R. E. & Britton, R. A convenient photocatalytic fluorination of unactivated C–H bonds. Angew. Chem. Int. Ed. 53, 4690–4693 (2014).
Zwick, C. R. III & Renata, H. Evolution of biocatalytic and chemocatalytic C–H functionalization strategy in the synthesis of manzacidin C. J. Org. Chem. 83, 7407–7415 (2018).
Ravelli, D., Protti, S. & Fagnoni, M. Decatungstate anion for photocatalyzed ‘window ledge’ reactions. Acc. Chem. Res. 49, 2232–2242 (2016).
Ravelli, D., Fagnoni, M., Fukuyama, T., Nishikawa, T. & Ryu, I. Site-selective C–H functionalization by decatungstate anion photocatalysis: synergistic control by polar and steric effects expands the reaction scope. ACS Catal. 8, 701–713 (2018).
Perry, I. B. et al. Direct arylation of strong aliphatic C–H bonds. Nature 560, 70–75 (2018).
Blakemore, D. C. et al. Organic synthesis provides opportunities to transform drug discovery. Nat. Chem. 10, 383–394 (2018).
Song, K.-S., Liu, L. & Guo, Q.-X. Effects of α-ammonium, α-phosphonium and α-sulfonium groups on C–H bond dissociation energies. J. Org. Chem. 68, 4604–4607 (2003).
Lee, M. & Sanford, M. S. Palladium-catalyzed, terminal-selective C(sp 3)–H oxidation of aliphatic amines. J. Am. Chem. Soc. 137, 12796–12799 (2015).
Howell, J. M., Feng, K., Clark, J. R., Trzepkowski, L. J. & White, M. C. Remote oxidation of aliphatic C–H bonds in nitrogen-containing molecules. J. Am. Chem. Soc. 137, 14590–14593 (2015).
Lee, M. & Sanford, M. S. Remote C(sp 3)–H oxygenation of protonated aliphatic amines with potassium persulfate. Org. Lett. 19, 572–575 (2017).
Tan, X. et al. Silver-catalyzed decarboxylative trifluoromethylation of aliphatic carboxylic acids. J. Am. Chem. Soc. 139, 12430–12433 (2017).
Shen, H. et al. Trifluoromethylation of alkyl radicals in aqueous solution. J. Am. Chem. Soc. 139, 9843–9846 (2017).
Paeth, M. et al. Csp 3–Csp 3 bond-forming reductive elimination from well-defined copper(iii) complexes. J. Am. Chem. Soc. 141, 3153–3159 (2019).
Texier, I., Delaire, J. A. & Giannotti, C. Reactivity of the charge transfer excited state of sodium decatungstate at the nanosecond time scale. Phys. Chem. Chem. Phys. 2, 1205–1212 (2000).
Khan, M. A. & Schwing-Weill, M. J. Stability and electronic spectra of the copper(ii) chloro complexes in aqueous solutions. Inorg. Chem. 15, 2202–2205 (1976).
Kochi, J. K., Bemis, A. & Jenkins, C. L. Mechanism of electron transfer oxidation of alkyl radicals by copper(ii) complexes. J. Am. Chem. Soc. 90, 4616–4625 (1968).
Jenkins, C. L. & Kochi, J. K. Solvolytic routes via alkylcopper intermediates in the electron-transfer oxidation of alkyl radicals. J. Am. Chem. Soc. 94, 843–855 (1972).
Surry, D. S. & Buchwald, S. L. Diamine ligands in copper-catalyzed reactions. Chem. Sci. 1, 13–31 (2010).
Grynkiewicz, G. & Gadzikowska, M. Tropane alkaloids as medicinally useful natural products and their synthetic derivatives as new drugs. Pharmacol. Rep. 60, 439–463 (2008).
Yarmolchuk, V. S. et al. Synthesis and characterization of β‐trifluoromethyl‐substituted pyrrolidines. Eur. J. Org. Chem. 2013, 3086–3093 (2013).
Jacobs, R. T. et al. Synthesis, structure–activity relationships, and pharmacological evaluation of a series of fluorinated 3-benzyl-5-indolecarboxamides: identification of 4-[[5-[((2R)-2-methyl-4,4,4-trifluorobutyl)carbamoyl]-1-methylindol-3-yl]methyl]-3-methoxy-N-[(2-methylphenyl)sulfonyl]benzamide, a potent orally active antagonist of leukotrienes D4 and E4. J. Med. Chem. 37, 1282–1297 (1994).
Blakemore, D. C. et al. Synthesis and in vivo evaluation of 3-substituted gababutins. Bioorg. Med. Chem. Lett. 20, 362–365 (2010).
Zhang, Z. & Tang, W. Drug metabolism in drug discovery and development. Acta Pharm. Sin. B 8, 721–732 (2018).
McLean, T. H. et al. 1-Aminomethylbenzocycloalkanes: conformationally restricted hallucinogenic phenethylamine analogues as functionally selective 5-HT2A receptor agonists. J. Med. Chem. 49, 5794–5803 (2006).
Fukuyama, T. et al. Photocatalyzed site-selective C(sp 3)–H functionalization of alkylpyridines at non-benzylic positions. Org. Lett. 19, 6436–6439 (2017).
Kobayashi, S., Nagayama, S. & Busujima, T. Lewis acid catalysts stable in water. Correlation between catalytic activity in water and hydrolysis constants and exchange rate constants for substitution of inner-sphere water ligands. J. Am. Chem. Soc. 120, 8287–8288 (1998).
Ling, L., Liu, K., Li, X. & Li, Y. General reaction mode of hypervalent iodine trifluoromethylation reagent: a density functional theory study. ACS Catal. 5, 2458–2468 (2015).
Nebra, N. & Grushin, V. V. Distinct mechanism of oxidative trifluoromethylation with a well-defined Cu(ii) fluoride promoter: hidden catalysis. J. Am. Chem. Soc. 136, 16998–17001 (2014).
Yang, G. & Zhang, W. Renaissance of pyridine-oxazolines as chiral ligands for asymmetric synthesis. Chem. Soc. Rev. 47, 1783–1810 (2018).
Acknowledgements
We acknowledge financial support from the National Institute of General Medical Sciences (NIGMS), the NIH (award no. R01 GM078201-05) to D.W.C.M., V.B. and P.J.S., and gifts from MSD, Abbvie, Pfizer and Janssen. P.J.S. and V.B. acknowledge Princeton University, E. Taylor and the Taylor family for an Edward C. Taylor Fellowship. We thank L. Wilson (Lotus Separations) for assistance with compound purification, D. Abrams for helpful discussions, I. Pelczer for assistance with NMR spectroscopy, O. Garry for synthesizing a batch of sodium decatungstate, I. Perry for providing a graphical rendering of the decatungstate anion and R. Martinie, H. Zhong and J. Eng for assistance with EPR spectroscopy. The content is solely the responsibility of the authors and does not necessarily represent the official views of NIGMS.
Author information
Authors and Affiliations
Contributions
P.J.S. and V.B. performed and analysed the experiments. P.J.S., V.B., D.M.S., D.A.D. and D.W.C.M. designed the experiments. Y.-h.L. and E.C.S. performed the computational analysis. P.J.S., V.B. and D.W.C.M. prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Experimental procedures, optimization data, mechanistic studies, computational data and product characterization data.
Rights and permissions
About this article
Cite this article
Sarver, P.J., Bacauanu, V., Schultz, D.M. et al. The merger of decatungstate and copper catalysis to enable aliphatic C(sp3)–H trifluoromethylation. Nat. Chem. 12, 459–467 (2020). https://doi.org/10.1038/s41557-020-0436-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-020-0436-1
- Springer Nature Limited
This article is cited by
-
Nickel-catalysed enantioselective alkene dicarbofunctionalization enabled by photochemical aliphatic C–H bond activation
Nature Catalysis (2024)
-
Red-light-mediated copper-catalyzed photoredox catalysis promotes regioselectivity switch in the difunctionalization of alkenes
Nature Communications (2024)
-
Late-stage synthesis of heterobifunctional molecules for PROTAC applications via ruthenium-catalysed C‒H amidation
Nature Communications (2023)
-
Photochemical diversification of strong C(sp3)–H bonds enabled by allyl bromide and sodium fluoride
Nature Synthesis (2023)
-
Radical C(sp3)–H functionalization and cross-coupling reactions
Nature Reviews Chemistry (2022)