Abstract
Fuelled by innovation, optical microscopy plays a critical role in the life sciences and medicine, from basic discovery to clinical diagnostics. However, optical microscopy is limited by typical penetration depths of a few hundred micrometres for in vivo interrogations in the visible spectrum. Optoacoustic microscopy complements optical microscopy by imaging the absorption of light, but it is similarly limited by penetration depth. In this Review, we summarize progress in the development and applicability of optoacoustic mesoscopy (OPAM); that is, optoacoustic imaging with acoustic resolution and wide-bandwidth ultrasound detection. OPAM extends the capabilities of optical imaging beyond the depths accessible to optical and optoacoustic microscopy, and thus enables new applications. We explain the operational principles of OPAM, its placement as a bridge between optoacoustic microscopy and optoacoustic macroscopy, and its performance in the label-free visualization of tissue pathophysiology, such as inflammation, oxygenation, vascularization and angiogenesis. We also review emerging applications of OPAM in clinical and biological imaging.


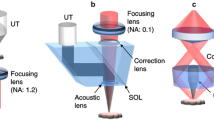
Dominik Soliman; panel c reproduced from ref. 32, Springer Nature Ltd.



Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Helmchen, F. & Denk, W. Deep tissue two-photon microscopy. Nat. Methods 2, 932–940 (2005).
Theer, P., Hasan, M. T. & Denk, W. Two-photon imaging to a depth of 1000 μm in living brains by use of a Ti: Al 2 O 3 regenerative amplifier. Opt. Lett. 28, 1022–1024 (2003).
Smith, A. M., Mancini, M. C. & Nie, S. Second window for in vivo imaging. Nat. Nanotechnol. 4, 710 (2009).
Hong, G. et al. Ultrafast fluorescence imaging in vivo with conjugated polymer fluorophores in the second near-infrared window. Nat. Commun. 5, 4206 (2014).
Vakoc, B. J. et al. Three-dimensional microscopy of the tumor microenvironment in vivo using optical frequency domain imaging. Nat. Med. 15, 1219–1223 (2009).
Drexler, W. et al. Ultrahigh-resolution ophthalmic optical coherence tomography. Nat. Med. 7, 502 (2001).
Fujimoto, J. G. Optical coherence tomography for ultrahigh resolution in vivo imaging. Nat. Biotechnol. 21, 1361 (2003).
Čižmár, T., Mazilu, M. & Dholakia, K. In situ wavefront correction and its application to micromanipulation. Nat. Photon. 4, 388–394 (2010).
Chaigne, T. et al. Controlling light in scattering media non-invasively using the photoacoustic transmission matrix. Nat. Photon. 8, 58–64 (2014).
Vellekoop, I. M., Lagendijk, A. & Mosk, A. Exploiting disorder for perfect focusing. Nat. Photon. 4, 320–322 (2010).
Katz, O., Small, E. & Silberberg, Y. Looking around corners and through thin turbid layers in real time with scattered incoherent light. Nat. Photon. 6, 549–553 (2012).
Ertürk, A. et al. Three-dimensional imaging of solvent-cleared organs using 3DISCO. Nat. Prot. 7, 1983 (2012).
Hama, H. et al. ScaleS: an optical clearing palette for biological imaging. Nat. Neurosci. 18, 1518 (2015).
Dodt, H.-U. et al. Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nat. Methods 4, 331 (2007).
Farkas, D. L. Invention and commercialization in optical bioimaging. Nat. Biotechnol. 21, 1269–1271 (2003).
Weissleder, R. & Pittet, M. J. Imaging in the era of molecular oncology. Nature 452, 580–589 (2008).
Vinegoni, C., Pitsouli, C., Razansky, D., Perrimon, N. & Ntziachristos, V. In vivo imaging of Drosophila melanogaster pupae with mesoscopic fluorescence tomography. Nat. Methods 5, 45 (2008).
Sharpe, J. et al. Optical projection tomography as a tool for 3D microscopy and gene expression studies. Science 296, 541–545 (2002).
Ntziachristos, V., Ripoll, J., Wang, L. V. & Weissleder, R. Looking and listening to light: the evolution of whole-body photonic imaging. Nat. Biotechnol. 23, 313–320 (2005).
Weissleder, R. A clearer vision for in vivo imaging. Nat. Biotechnol. 19, 316–316 (2001).
Maugh, T. H. Photoacoustic spectroscopy: New uses for an old technique. Science 188, 38–39 (1975).
Rosencwaig, A. & Gersho, A. Theory of the photoacoustic effect with solids. J. Appl. Physics 47, 64–69 (1976).
Taruttis, A. & Ntziachristos, V. Advances in real-time multispectral optoacoustic imaging and its applications. Nat. Photon. 9, 219–227 (2015).
Taruttis, A., van Dam, G. M. & Ntziachristos, V. Mesoscopic and macroscopic optoacoustic imaging of cancer. Cancer Res. 75, 1548–1559 (2015).
Wang, L. V. & Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 13, 627 (2016).
Wang, L. V. & Hu, S. Photoacoustic tomography: in vivo imaging from organelles to organs. Science 335, 1458–1462 (2012).
Maslov, K., Zhang, H. F., Hu, S. & Wang, L. V. Optical-resolution photoacoustic microscopy for in vivo imaging of single capillaries. Opt. Lett. 33, 929–931 (2008).
Yao, J., Maslov, K. I., Zhang, Y., Xia, Y. & Wang, L. V. Label-free oxygen-metabolic photoacoustic microscopy in vivo. J. Biomed. Opt. 16, 076003 (2011).
Ntziachristos, V. Going deeper than microscopy: the optical imaging frontier in biology. Nat. Methods 7, 603–614 (2010).
Strohm, E. M., Berndl, E. S. L. & Kolios, M. C. Probing Red Blood Cell Morphology Using High-Frequency Photoacoustics. Biophys. J. 105, 59–67 (2013).
Strohm, E. M., Berndl, E. S. L. & Kolios, M. C. High frequency label-free photoacoustic microscopy of single cells. Photoacoustics 1, 49–53 (2013).
Aguirre, J. S. et al. Precision assessment of label-free psoriasis biomarkers with ultra-broadband optoacoustic mesoscopy. Nat. Biomed. Eng. 1, 0068 (2017).
Alanentalo, T. et al. High-resolution three-dimensional imaging of islet-infiltrate interactions based on optical projection tomography assessments of the intact adult mouse pancreas. J. Biomed. Opt. 13, 054070 (2008).
Piras, D., Xia, W., Steenbergen, W., van Leeuwen, T. G. & Manohar, S. Photoacoustic imaging of the breast using the twente photoacoustic mammoscope: present status and future perspectives. IEEE J. Quantum Elect. 16, 730–739 (2010).
Diot, G. et al. Multispectral optoacoustic tomography (MSOT) of human breast cancer. Clin. Cancer Res. 23, 6912–6922 (2017).
Oraevsky, A. et al. Full-view 3D imaging system for functional and anatomical screening of the breast. in Proc. SPIE 10494 https://doi.org/10.1117/12.2318802 (2018).
Knieling, F. et al. Multispectral Optoacoustic Tomography for Assessment of Crohn’s Disease Activity. New Engl. J. Med. 376, 1292–1294 (2017).
Reber, J. et al. Non-invasive measurement of brown fat metabolism based on optoacoustic imaging of hemoglobin gradients. Cell Metab. 27, 689–701 (2018).
Weber, J., Beard, P. C. & Bohndiek, S. E. Contrast agents for molecular photoacoustic imaging. Nat. Methods 13, 639–650 (2016).
Xu, M. & Wang, L. V. Photoacoustic imaging in biomedicine. Rev. Sci. Instrum. 77, 041101 (2006).
Li, M.-L., Zhang, H. F., Maslov, K., Stoica, G. & Wang, L. V. Improved in vivo photoacoustic microscopy based on a virtual-detector concept. Opt. Lett. 31, 474–476 (2006).
Westervelt, P. J. & Larson, R. S. Laser‐excited broadside array. J. Acoust. Soc. Am. 54, 121–122 (1973).
Diebold, G., Sun, T. & Khan, M. Photoacoustic monopole radiation in one, two, and three dimensions. Phys. Rev. Lett. 67, 3384 (1991).
Ntziachristos, V. & Razansky, D. Molecular imaging by means of multispectral optoacoustic tomography (MSOT). Chemical reviews 110, 2783–2794 (2010).
Xia, J., Yao, J. & Wang, L. V. Photoacoustic tomography: principles and advances. Electromag. Waves 147, 1 (2014).
Deán-Ben, X. L. & Razansky, D. On the link between the speckle free nature of optoacoustics and visibility of structures in limited-view tomography. Photoacoustics 4, 133–140 (2016).
Omar, M., Gateau, J. & Ntziachristos, V. Raster-scan optoacoustic mesoscopy in the 25–125 MHz range. Opt. Lett. 38, 2472–2474 (2013).
Omar, M., Soliman, D., Gateau, J. & Ntziachristos, V. Ultrawideband reflection-mode optoacoustic mesoscopy. Opt. Lett. 39, 3911–3914 (2014).
Laufer, J. et al. In vivo photoacoustic imaging of mouse embryos. J. Biomed. Opt. 17, 061220 (2012).
Jathoul, A. P. et al. Deep in vivo photoacoustic imaging of mammalian tissues using a tyrosinase-based genetic reporter. Nat. Photon. (2015).
Park, J. et al. Delay-multiply-and-sum-based synthetic aperture focusing in photoacoustic microscopy. J. Biomed. Opt. 21, 036010 (2016).
Laufer, J., Zhang, E., Raivich, G. & Beard, P. Three-dimensional noninvasive imaging of the vasculature in the mouse brain using a high resolution photoacoustic scanner. Appl. Opt. 48, 299–306 (2009).
Zhang, E. Z. et al. Multimodal photoacoustic and optical coherence tomography scanner using an all optical detection scheme for 3D morphological skin imaging. Biomed. Opt. Exp. 2, 2202–2215 (2011).
Aguirre, J. et al. Broadband mesoscopic optoacoustic tomography reveals skin layers. Opt. Lett. 39, 6297–6300 (2014).
Gateau, J., Chekkoury, A. & Ntziachristos, V. High-resolution optoacoustic mesoscopy with a 24 MHz multidetector translate-rotate scanner. J. Biomed. Opt. 18, 106005 (2013).
Vionnet, L. et al. 24-MHz scanner for optoacoustic imaging of skin and burn. IEEE Trans. Med. Imag. 33, 535–545 (2014).
Zemp, R., Song, L., Bitton, R., Shung, K. & Wang, L. Realtime photoacoustic microscopy of murine cardiovascular dynamics. Opt. Exp. 16, 18551–18556 (2008).
Wang, L., Maslov, K., Xing, W., Garcia-Uribe, A. & Wang, L. V. Video-rate functional photoacoustic microscopy at depths. J. Biomed. Opt. 17, 106007 (2012).
Zhang, H. F., Maslov, K., Stoica, G. & Wang, L. V. Functional photoacoustic microscopy for high-resolution and noninvasive in vivo imaging. Nat. Biotechnol. 24, 848 (2006).
Fiolka, R., Wicker, K., Heintzmann, R. & Stemmer, A. Simplified approach to diffraction tomography in optical microscopy. Opt. Exp. 17, 12407–12417 (2009).
Wicker, K., Mandula, O., Best, G., Fiolka, R. & Heintzmann, R. Phase optimisation for structured illumination microscopy. Opt. Exp. 21, 2032–2049 (2013).
Betzig, E. et al. Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006).
Manley, S. et al. High-density mapping of single-molecule trajectories with photoactivated localization microscopy. Nat. Methods 5, 155–157 (2008).
Prevedel, R. et al. Simultaneous whole-animal 3D imaging of neuronal activity using light-field microscopy. Nat. Methods 11, 727–730 (2014).
Gustafsson, M. G. Surpassing the lateral resolution limit by a factor of two using structured illumination microscopy. J. Microsc. 198, 82–87 (2000).
Kim, T. et al. White-light diffraction tomography of unlabelled live cells. Nat. Photon. 8, 256–263 (2014).
Keller, P. J. Imaging morphogenesis: technological advances and biological insights. Science 340, 1234168 (2013).
Razansky, D. et al. Multispectral opto-acoustic tomography of deep-seated fluorescent proteins. in vivo. Nat. Photon. 3, 412–417 (2009).
Gateau, J., Caballero, M. Á. A., Dima, A. & Ntziachristos, V. Three‐dimensional optoacoustic tomography using a conventional ultrasound linear detector array: Whole‐body tomographic system for small animals. Med. Phys. 40 (2013).
Szabo, T. L. Diagnostic ultrasound imaging: inside out (Acad. Press, 2004).
Cobbold, R. S. Foundations of biomedical ultrasound (Oxford Univ. Press, 2006).
Zhang, E. Z. & Beard, P. C. A miniature all-optical photoacoustic imaging probe. in Proc. SPIE 7899 https://doi.org/10.1117/12.874883 (2011).
Nuster, R. et al. Hybrid photoacoustic and ultrasound section imaging with optical ultrasound detection. J. Biophoton. 6, 549–559 (2013).
Deán-Ben, X. L., Razansky, D. & Ntziachristos, V. The effects of acoustic attenuation in optoacoustic signals. Phys. Med. Biol. 56, 6129 (2011).
Schwarz, M. S. et al. Optoacoustic dermoscopy of the human skin: tuning excitation energy for optimal detection bandwidth with fast and deep imaging in vivo. IEEE Trans. Med. Imaging 36, 1287–1296 (2017).
Laufer, J. et al. In vivo preclinical photoacoustic imaging of tumor vasculature development and therapy. J. Biomed. Opt. 17, 056016 (2012).
Wissmeyer, G., Soliman, D., Shnaiderman, R., Rosenthal, A. & Ntziachristos, V. All-optical optoacoustic microscope based on wideband pulse interferometry. Opt. Lett. 41, 1953–1956 (2016).
Rosenthal, A. et al. Sensitive interferometric detection of ultrasound for minimally invasive clinical imaging applications. Laser Photon. Rev. 8, 450–457 (2014).
Schwarz, M., Buehler, A. & Ntziachristos, V. Isotropic high resolution optoacoustic imaging with linear detector arrays in bi‐directional scanning. J. Biophoton. 8, 60–70 (2015).
Chekkoury, A. et al. Optical mesoscopy without the scatter: broadband multispectral optoacoustic mesoscopy. Biomed. Opt. Exp. 6, 3134–3148 (2015).
Rebling, J., Warshavski, O., Meynier, C. & Razansky, D. Optoacoustic characterization of broadband directivity patterns of capacitive micromachined ultrasonic transducers. J. Biomed. Opt. 22, 041005 (2017).
Oraevsky, A. A. & Karabutov, A. A. Ultimate sensitivity of time-resolved optoacoustic detection. in Proc. SPIE 3916, 228–239 (2000).
Zhang, E., Laufer, J. & Beard, P. Backward-mode multiwavelength photoacoustic scanner using a planar Fabry-Perot polymer film ultrasound sensor for high-resolution three-dimensional imaging of biological tissues. Appl. Opt. 47, 561–577 (2008).
Beard, P. C., Hurrell, A. M. & Mills, T. N. Characterization of a polymer film optical fiber hydrophone for use in the range 1 to 20 MHz: A comparison with PVDF needle and membrane hydrophones. IEEE T. Ultrason. Ferr. 47, 256–264 (2000).
Maswadi, S. M. et al. All-optical optoacoustic microscopy based on probe beam deflection technique. Photoacoustics 4, 91–101 (2016).
Paltauf, G., Schmidt-Kloiber, H., Köstli, K. & Frenz, M. Optical method for two-dimensional ultrasonic detection. Appl. Phys. Lett. 75, 1048–1050 (1999).
Omar, M. et al. Optical imaging of post-embryonic zebrafish using multi orientation raster scan optoacoustic mesoscopy. Light Sci. Appl. 6, e16186 (2017).
Xu, M. & Wang, L. V. Universal back-projection algorithm for photoacoustic computed tomography. Phys. Rev. E 71, 016706 (2005).
Kostli, K. et al. Optoacoustic imaging using a three-dimensional reconstruction algorithm. IEEE J. Sel. Top. Quant. 7, 918–923 (2001).
Karaman, M., Li, P.-C. & O’Donnell, M. Synthetic aperture imaging for small scale systems. IEEE T. Ultrason. Ferr. 42, 429–442 (1995).
Köstli, K. P. & Beard, P. C. Two-dimensional photoacoustic imaging by use of Fourier-transform image reconstruction and a detector with an anisotropic response. Appl. Opt. 42, 1899–1908 (2003).
Oraevsky, A. A., Andreev, V. G., Karabutov, A. A. & Esenaliev, R. O. Two-dimensional optoacoustic tomography: transducer array and image reconstruction algorithm. in Proc. SPIE 3601 Laser Tissue Interaction X. https://doi.org/10.1117/12.350007 (1999).
Kruger, R. A., Liu, P., Fang, Y. & Appledorn, C. R. Photoacoustic ultrasound (PAUS)—reconstruction tomography. Med. Phys. 22, 1605–1609 (1995).
Rosenthal, A., Razansky, D. & Ntziachristos, V. Fast semi-analytical model-based acoustic inversion for quantitative optoacoustic tomography. IEEE T. Med. Imag. 29, 1275–1285 (2010).
Caballero, M. Á. A., Rosenthal, A., Gateau, J., Razansky, D. & Ntziachristos, V. Model-based optoacoustic imaging using focused detector scanning. Opt. Lett. 37, 4080–4082 (2012).
Dean-Ben, X. L., Buehler, A., Ntziachristos, V. & Razansky, D. Accurate model-based reconstruction algorithm for three-dimensional optoacoustic tomography. IEEE T. Med. Imag. 31, 1922–1928 (2012).
Aguirre, J. et al. A low memory cost model based reconstruction algorithm exploiting translational symmetry for photoacoustic microscopy. Biomed. Opt. Exp. 4, 2813–2827 (2013).
Mohajerani, P., Tzoumas, S., Rosenthal, A. & Ntziachristos, V. Optical and optoacoustic model-based tomography: Theory and current challenges for deep tissue imaging of optical contrast. IEEE Signal Proc. Mag. 32, 88–100 (2015).
Anastasio, M. A., Zhang, J., Modgil, D. & La Rivière, P. J. Application of inverse source concepts to photoacoustic tomography. Inverse Problems 23, S21 (2007).
Huang, C., Wang, K., Nie, L., Wang, L. V. & Anastasio, M. A. Full-wave iterative image reconstruction in photoacoustic tomography with acoustically inhomogeneous media. IEEE T. Med. Imag. 32, 1097–1110 (2013).
Anastasio, M. A. et al. Half-time image reconstruction in thermoacoustic tomography. IEEE T. Med. Imag. 24, 199–210 (2005).
La Rivière, P. J., Zhang, J. & Anastasio, M. A. Image reconstruction in optoacoustic tomography for dispersive acoustic media. Opt. Lett. 31, 781–783 (2006).
Wang, K. & Anastasio, M. A. in Handbook of Mathematical Methods in Imaging 781–815 (Springer, New York, 2011).
Wang, K., Su, R., Oraevsky, A. A. & Anastasio, M. A. Investigation of iterative image reconstruction in three-dimensional optoacoustic tomography. Phys. Med. Biol. 57, 5399 (2012).
Cox, B. T., Arridge, S. R., Köstli, K. P. & Beard, P. C. Two-dimensional quantitative photoacoustic image reconstruction of absorption distributions in scattering media by use of a simple iterative method. Appl. Opt. 45, 1866–1875 (2006).
Tzoumas, S. et al. Eigenspectra optoacoustic tomography achieves quantitative blood oxygenation imaging deep in tissues. Nat. Commun. 7, 12121 (2016).
Cox, B., Laufer, J. G., Arridge, S. R. & Beard, P. C. Quantitative spectroscopic photoacoustic imaging: a review. J. Biomed. Opt. 17, 061202 (2012).
Tzoumas, S., Deliolanis, N., Morscher, S. & Ntziachristos, V. Unmixing molecular agents from absorbing tissue in multispectral optoacoustic tomography. IEEE T. Med. Imag. 33, 48–60 (2014).
Schwarz, M., Buehler, A., Aguirre, J. & Ntziachristos, V. Three‐dimensional multispectral optoacoustic mesoscopy reveals melanin and blood oxygenation in human skin. in vivo. J. Biophoton. 9, 55–60 (2016).
Mohajerani, P., Kellnberger, S. & Ntziachristos, V. Frequency domain optoacoustic tomography using amplitude and phase. Photoacoustics 2, 111–118 (2014).
Kellnberger, S., Deliolanis, N. C., Queirós, D., Sergiadis, G. & Ntziachristos, V. In vivo frequency domain optoacoustic tomography. Opt. Lett. 37, 3423–3425 (2012).
Telenkov, S., Mandelis, A., Lashkari, B. & Forcht, M. Frequency-domain photothermoacoustics: Alternative imaging modality of biological tissues. J. Appl. Phys. 105, 102029 (2009).
VanderLaan, D., Karpiouk, A., Yeager, D. & Emelianov, S. Real-time intravascular ultrasound and photoacoustic imaging. IEEE T. Ultrason. Ferr. 64, 141–149 (2016).
Wu, M. et al. Real-time volumetric lipid imaging in vivo by intravascular photoacoustics at 20 frames per second. Biomed. Optics Exp. 8, 943–953 (2017).
Beziere, N. et al. Optoacoustic imaging and staging of inflammation in a murine model of arthritis. Arthritis Rheumatol. 66, 2071–2078 (2014).
Mallidi, S., Luke, G. P. & Emelianov, S. Photoacoustic imaging in cancer detection, diagnosis, and treatment guidance. Trends Biotechnol. 29, 213–221 (2011).
Liu, Y., Nie, L. & Chen, X. Photoacoustic molecular imaging: from multiscale biomedical applications towards early-stage theranostics. Trends Biotechnol. 34, 420–433 (2016).
Beziere, N. et al. Dynamic imaging of PEGylated indocyanine green (ICG) liposomes within the tumor microenvironment using multi-spectral optoacoustic tomography (MSOT). Biomaterials 37, 415–424 (2015).
Lozano, N., Al-Ahmady, Z. S., Beziere, N. S., Ntziachristos, V. & Kostarelos, K. Monoclonal antibody-targeted PEGylated liposome-ICG encapsulating doxorubicin as a potential theranostic agent. Int. J. Pharm. 482, 2–10 (2015).
Wilson, K., Homan, K. & Emelianov, S. Biomedical photoacoustics beyond thermal expansion using triggered nanodroplet vaporization for contrast-enhanced imaging. Nat. Commun. 3, 618 (2012).
Luke, G. P., Yeager, D. & Emelianov, S. Y. Biomedical applications of photoacoustic imaging with exogenous contrast agents. Ann. Biomed. Eng. 40, 422–437 (2012).
Copland, J. A. et al. Bioconjugated gold nanoparticles as a molecular based contrast agent: implications for imaging of deep tumors using optoacoustic tomography. Mol. Imag. Biol. 6, 341–349 (2004).
Conjusteau, A. et al. Metallic nanoparticles as optoacoustic contrast agents for medical imaging. In Proc. SPIE 6086 Photons Plus Ultrasound: Imaging and Sensing https://doi.org/10.1117/12.658065 (2006).
Gindy, M. E. & Prud’homme, R. K. Multifunctional nanoparticles for imaging, delivery and targeting in cancer therapy. Expert Opin. Drug Del. 6, 865–878 (2009).
Haedicke, K. et al. Sonophore labeled RGD: a targeted contrast agent for optoacoustic imaging. Photoacoustics 6, 1–8 (2017).
Stritzker, J. et al. Vaccinia virus-mediated melanin production allows MR and optoacoustic deep tissue imaging and laser-induced thermotherapy of cancer. Proc. Natl Acad. Sci. USA 110, 3316–3320 (2013).
Paproski, R. J., Forbrich, A. E., Wachowicz, K., Hitt, M. M. & Zemp, R. J. Tyrosinase as a dual reporter gene for both photoacoustic and magnetic resonance imaging. Biomed. Opt. Exp. 2, 771–780 (2011).
Stiel, A. C. et al. High-contrast imaging of reversibly switchable fluorescent proteins via temporally unmixed multispectral optoacoustic tomography. Opt. Lett. 40, 367–370 (2015).
Yao, J. et al. Multiscale photoacoustic tomography using reversibly switchable bacterial phytochrome as a near-infrared photochromic probe. Nat. Methods 13, 67–73 (2016).
Bost, W., Lemor, R. & Fournelle, M. Optoacoustic Imaging of subcutaneous microvasculature with a class one laser. IEEE T. Med. Imag. 33, 1900–1904 (2014).
Kirillin, M., Perekatova, V., Turchin, I. & Subochev, P. Fluence compensation in raster-scan optoacoustic angiography. Photoacoustics 8, 59–67 (2017).
Zhang, E., Laufer, J., Pedley, R. & Beard, P. In vivo high-resolution 3D photoacoustic imaging of superficial vascular anatomy. Phys. Med. Biol. 54, 1035 (2009).
Aguirre, J. et al. The potential of photoacoustic microscopy as a tool to characterize the in vivo degradation of surgical sutures. Biomed. Opt. Exp. 5, 2856–2869 (2014).
Nam, S. Y., Chung, E., Suggs, L. J. & Emelianov, S. Y. Combined ultrasound and photoacoustic imaging to noninvasively assess burn injury and selectively monitor a regenerative tissue-engineered construct. Tissue Eng. Part C Me. 21, 557–566 (2015).
Aizawa, K., Sato, S., Saitoh, D., Ashida, H. & Obara, M. Photoacoustic monitoring of burn healing process in rats. J. Biomed. Opt. 13, 064020 (2008).
Ida, T. et al. Real-time photoacoustic imaging system for burn diagnosis. J. Biomed. Opt. 19, 086013 (2014).
Zhou, Y., Xing, W., Maslov, K. I., Cornelius, L. A. & Wang, L. V. Handheld photoacoustic microscopy to detect melanoma depth in vivo. Opt. Lett. 39, 4731–4734 (2014).
Zhou, Y. et al. Handheld photoacoustic probe to detect both melanoma depth and volume at high speed in vivo. J. Biophoton. 8, 961–967 (2015).
Welzel, J., Lankenau, E., Birngruber, R. & Engelhardt, R. Optical coherence tomography of the human skin. J. Am. Acad. Dermatol. 37, 958–963 (1997).
Welzel, J. Optical coherence tomography in dermatology: a review. Skin Re. Technol. 7, 1–9 (2001).
Zhao, Y. et al. Phase-resolved optical coherence tomography and optical Doppler tomography for imaging blood flow in human skin with fast scanning speed and high velocity sensitivity. Opt. Lett. 25, 114–116 (2000).
Vogt, M. & Ermert, H. Development and evaluation of a high-frequency ultrasound-based system for in vivo strain imaging of the skin. IEEE T. Ultrason. Ferr. 52, 375–385 (2005).
Turnbull, D. H. et al. A 40–100 MHz B-scan ultrasound backscatter microscope for skin imaging. Ultrasound Med. Biol. 21, 79–88 (1995).
Serup, J., Keiding, J., Fullerton, A., Gniadecka, M. & Gniadecki, R. in Handbook of non-invasive methods and the skin. (eds Serup, J., Jemec, G. B. E. & Grove, G. L.) 473–491 (Taylor & Francis, 2006).
Mintz, G. S. et al. American College of Cardiology clinical expert consensus document on standards for acquisition, measurement and reporting of intravascular ultrasound studies (IVUS). J. Am. Coll. Cardiol. 37, 1478–1492 (2001).
Nair, A. et al. Coronary plaque classification with intravascular ultrasound radiofrequency data analysis. Circulation 106, 2200–2206 (2002).
Jang, I.-K. et al. Visualization of coronary atherosclerotic plaques in patients using optical coherence tomography: comparison with intravascular ultrasound. J. Am. Coll. Cardiol. 39, 604–609 (2002).
VanderLaan, D., Karpiouk, A. B., Yeager, D. & Emelianov, S. Real-time intravascular ultrasound and photoacoustic imaging. IEEE T. Ultrason. Ferr. 64, 141–149 (2017).
Jansen, K. et al. Spectroscopic intravascular photoacoustic imaging of lipids in atherosclerosis. J. Biomed. Opt. 19, 026006 (2014).
Wang, P. et al. High-speed intravascular photoacoustic imaging of lipid-laden atherosclerotic plaque enabled by a 2-kHz barium nitrite raman laser. Sci. Rep. 4, 6889 (2014).
Jansen, K., Wu, M., van der Steen, A. F. & van Soest, G. Lipid detection in atherosclerotic human coronaries by spectroscopic intravascular photoacoustic imaging. Opt. Exp. 21, 21472–21484 (2013).
Wang, B. et al. In vivo intravascular ultrasound-guided photoacoustic imaging of lipid in plaques using an animal model of atherosclerosis. Ultrasound Med. Biol. 38, 2098–2103 (2012).
Zhang, J., Yang, S., Ji, X., Zhou, Q. & Xing, D. Characterization of lipid-rich aortic plaques by intravascular photoacoustic tomography. J. Am. Coll. Cardiol. 64, 385–390 (2014).
Hui, J. et al. Real-time intravascular photoacoustic-ultrasound imaging of lipid-laden plaque in human coronary artery at 16 frames per second. Sci. Rep. 7, 1417 (2017).
Ansari, R., Beard, P. C., Zhang, E. Z. & Desjardins, A. E. Photoacoustic endoscopy probe using a coherent fibre-optic bundle and Fabry-Pérot ultrasound sensor. In Proc. SPIE 9708 Photons Plus Ultrasound: Imaging and Sensing https://doi.org/10.1117/12.2209687 (2016).
He, H., Wissmeyer, G., Ovsepian, S. V., Buehler, A. & Ntziachristos, V. Hybrid optical and acoustic resolution optoacoustic endoscopy. Optics Letters 41, 2708–2710 (2016).
Yang, J.-M. et al. Simultaneous functional photoacoustic and ultrasonic endoscopy of internal organs in vivo. Nat. Med. 18, 1297–1302 (2012).
Yang, J. M. et al. Three-dimensional photoacoustic endoscopic imaging of the rabbit esophagus. PloS ONE 10, e0120269 (2015).
Yang, J.-M. et al. Photoacoustic endoscopy. Opt. Lett. 34, 1591–1593 (2009).
He, H., Buehler, A. & Ntziachristos, V. Optoacoustic endoscopy with curved scanning. Opt. Lett. 40, 4667–4670 (2015).
Chen, S.-L., Ling, T., Baac, H. W. & Guo, L. J. Photoacoustic endoscopy using polymer microring resonators. In Proc. SPIE 7899, Photons Plus Ultrasound: Imaging and Sensing https://doi.org/10.1117/12.874205 (2011).
Yuan, Y., Yang, S. & Xing, D. Preclinical photoacoustic imaging endoscope based on acousto-optic coaxial system using ring transducer array. Opt. Lett. 35, 2266–2268 (2010).
Barendse, R. et al. Endoscopic mucosal resection vs transanal endoscopic microsurgery for the treatment of large rectal adenomas. Colorectal Dis. 14, 191–196 (2012).
Soetikno, R. M., Gotoda, T., Nakanishi, Y. & Soehendra, N. Endoscopic mucosal resection. Gastrointest. Endosc. 57, 567–579 (2003).
Yang, Y. et al. Integrated optical coherence tomography, ultrasound and photoacoustic imaging for ovarian tissue characterization. Biomed. Opt. Exp. 2, 2551–2561 (2011).
Omar, M., Schwarz, M., Soliman, D., Symvoulidis, P. & Ntziachristos, V. Pushing the optical imaging limits of cancer with multi-frequency-band raster-scan optoacoustic mesoscopy (RSOM). Neoplasia 17, 208–214 (2015).
Chekkoury, A. et al. High-resolution multispectral optoacoustic tomography of the vascularization and constitutive hypoxemia of cancerous tumors. Neoplasia 18, 459–467 (2016).
Ellenbroek, S. I. & Van Rheenen, J. Imaging hallmarks of cancer in living mice. Nat. Rev. Cancer 14, 406–418 (2014).
Kim, C. et al. In vivo molecular photoacoustic tomography of melanomas targeted by bioconjugated gold nanocages. ACS Nano 4, 4559–4564 (2010).
Deán-Ben, X. L. et al. Functional optoacoustic neuro-tomography for scalable whole-brain monitoring of calcium indicators. Light Sci. Appl. 5, e16201 (2016).
Deán-Ben, X. L. & Razansky, D. Adding fifth dimension to optoacoustic imaging: volumetric time-resolved spectrally enriched tomography. Light Sci. Appl. 3, e137 (2014).
Stein, E. W., Maslov, K. & Wang, L. V. Noninvasive, in vivo imaging of blood-oxygenation dynamics within the mouse brain using photoacoustic microscopy. J. Biomed. Opt. 14, 020502 (2009).
Stein, E. W., Maslov, K. & Wang, L. V. Noninvasive, in vivo imaging of the mouse brain using photoacoustic microscopy. J. Appl. Phys. 105, 102027 (2009).
Johannes Rebling, J., Estrada, H., Zwack, M., Sela, G., Gottschalk, S., and Razansky, D., Hybrid ultrasound and dual-wavelength optoacoustic biomicroscopy for functional neuroimaging. In Proc. SPIE 10064, Photons Plus Ultrasound: Imaging and Sensing https://doi.org/10.1117/12.2250635 (2017).
Estrada, H., Turner, J., Kneipp, M. & Razansky, D. Real-time optoacoustic brain microscopy with hybrid optical and acoustic resolution. Laser Phys. Lett. 11, 045601 (2014).
Liao, L.-D. et al. Imaging brain hemodynamic changes during rat forepaw electrical stimulation using functional photoacoustic microscopy. Neuroimage 52, 562–570 (2010).
Liao, L.-D. et al. Transcranial imaging of functional cerebral hemodynamic changes in single blood vessels using in vivo photoacoustic microscopy. J. Cerebr. Blood F. Met. 32, 938–951 (2012).
Deng, Z., Wang, Z., Yang, X., Luo, Q. & Gong, H. In vivo imaging of hemodynamics and oxygen metabolism in acute focal cerebral ischemic rats with laser speckle imaging and functional photoacoustic microscopy. J. Biomed. Opt. 17, 081415 (2012).
Yao, J. & Wang, L. V. Photoacoustic brain imaging: from microscopic to macroscopic scales. Neurophotonics 1, 011003–011003 (2014).
Wang, D., Wu, Y. & Xia, J. Review on photoacoustic imaging of the brain using nanoprobes. Neurophotonics 3, 010901–010901 (2016).
Hu, S. Listening to the brain with photoacoustics. IEEE J. Sel. T. Quant. 22, 117–126 (2016).
Kim, K. et al. Photoacoustic imaging of early inflammatory response using gold nanorods. Appl. Phys. Lett. 90, 223901 (2007).
Ha, S., Carson, A., Agarwal, A., Kotov, N. A. & Kim, K. Detection and monitoring of the multiple inflammatory responses by photoacoustic molecular imaging using selectively targeted gold nanorods. Biomed. Opt. Exp. 2, 645–657 (2011).
Seeger, M., Karlas, A., Soliman, D., Pelisek, J. & Ntziachristos, V. Multimodal optoacoustic and multiphoton microscopy of human carotid atheroma. Photoacoustics 4, 102–111 (2016).
Estrada, H., Sobol, E., Baum, O. & Razansky, D. Hybrid optoacoustic and ultrasound biomicroscopy monitors’ laser-induced tissue modifications and magnetite nanoparticle impregnation. Laser Phys. Lett. 11, 125601 (2014).
Lin, H. C. A. et al. Selective plane illumination optical and optoacoustic microscopy for postembryonic imaging. Laser Photon. Rev. 9, 29–34 (2015).
Soliman, D., Tserevelakis, G. J., Omar, M. & Ntziachristos, V. Combining microscopy with mesoscopy using optical and optoacoustic label-free modes. Sci. Rep. 5, 12902 (2015).
Tserevelakis, G. J., Soliman, D., Omar, M. & Ntziachristos, V. Hybrid multiphoton and optoacoustic microscope. Opt. Lett. 39, 1819–1822 (2014).
Li, G., Li, L., Zhu, L., Xia, J. & Wang, L. V. Multiview Hilbert transformation for full-view photoacoustic computed tomography using a linear array. J. Biomed. Opt. 20, 066010 (2015).
Harrison, T. et al. Combined photoacoustic and ultrasound biomicroscopy. Opt. Exp. 17, 22041–22046 (2009).
Xi, L., Zhou, L. & Jiang, H. C-scan photoacoustic microscopy for in vivo imaging of Drosophila pupae. Appl. Phys. Lett. 101, 013702 (2012).
Xie, B. et al. Optoacoustic detection of early therapy-induced tumor cell death using a targeted imaging agent. Clin. Cancer Res. 23, 6893–6903 (2017).
Tomaszewski, M. R. et al. Oxygen enhanced optoacoustic tomography (OE-OT) reveals vascular dynamics in murine models of prostate cancer. Theranostics 7, 2900 (2017).
Jose, J. et al. Initial results of imaging melanoma metastasis in resected human lymph nodes using photoacoustic computed tomography. J. Biomed. Opt. 16, 096021 (2011).
Pramanik, M., Ku, G., Li, C. & Wang, L. V. Design and evaluation of a novel breast cancer detection system combining both thermoacoustic (TA) and photoacoustic (PA) tomography. Med. Phys. 35, 2218–2223 (2008).
Kolkman, R. G. M. et al. Photoacoustic mammography laboratory prototype: imaging of breast tissue phantoms. J. Biomed. Opt. 9, 1172 (2004).
Jose, J., Manohar, S., Kolkman, R. G., Steenbergen, W. & van Leeuwen, T. G. Imaging of tumor vasculature using Twente photoacoustic systems. J. Biophoton. 2, 701–717 (2009).
Heijblom, M. et al. Imaging tumor vascularization for detection and diagnosis of breast cancer. Technol. Cancer Res. T. 10, 607–623 (2011).
Heijblom, M. et al. Visualizing breast cancer using the Twente photoacoustic mammoscope: what do we learn from twelve new patient measurements? Opt. Exp. 20, 11582–11597 (2012).
Gurka, M. K. et al. Identification of pancreatic tumors in vivo with ligand-targeted, pH responsive mesoporous silica nanoparticles by multispectral optoacoustic tomography. J. Control. Release 231, 60–67 (2016).
Valluru, K. S. & Willmann, J. K. Clinical photoacoustic imaging of cancer. Ultrasonography 35, 267 (2016).
Stoffels, I. et al. Metastatic status of sentinel lymph nodes in melanoma determined noninvasively with multispectral optoacoustic imaging. Sci. Transl Med. 7, 317ra199 (2015).
Neuschmelting, V., Lockau, H., Ntziachristos, V., Grimm, J. & Kircher, M. F. Lymph node micrometastases and in-transit metastases from melanoma: in vivo detection with multispectral optoacoustic imaging in a mouse model. Radiology 280, 137–150 (2016).
Waldner, M. J. et al. Multispectral optoacoustic tomography in Crohn’s disease: noninvasive imaging of disease activity. Gastroenterology 151, 238–240 (2016).
Knieling, F. et al. Multispectral optoacoustic tomography in Crohn’s disease — a first-in-human diagnostic clinical trial. J. Nuclear Med. 58, 379–379 (2017).
Lev-Tov, H. Dive deep, stay focused! Sci. Transl Med. 9, eaan4292 (2017).
Allen, T. J., Hall, A., Dhillon, A. P., Owen, J. S. & Beard, P. C. Spectroscopic photoacoustic imaging of lipid-rich plaques in the human aorta in the 740 to 1400 nm wavelength range. J Biomed. Opt. 17, 061209 (2012).
Liotta, L. A., Steeg, P. S. & Stetler-Stevenson, W. G. Cancer metastasis and angiogenesis: an imbalance of positive and negative regulation. Cell 64, 327–336 (1991).
Filonov, G. S. et al. Deep‐tissue photoacoustic tomography of a genetically encoded near‐infrared fluorescent probe. Angew. Chem. Int. Edit. 51, 1448–1451 (2012).
Deán-Ben, X. L. & Razansky, D. Functional optoacoustic human angiography with handheld video rate three dimensional scanner. Photoacoustics 1, 68–73 (2013).
Zabihian, B. et al. In vivo dual-modality photoacoustic and optical coherence tomography imaging of human dermatological pathologies. Biomed. Opt. Exp. 6, 3163–3178 (2015).
Castelino, R. F., Hynes, M., Munding, C. E., Telenkov, S. & Foster, F. S. Combined frequency domain photoacoustic and ultrasound imaging for intravascular applications. Biomed. Opt. Exp. 7, 4441–4449 (2016).
Ji, X., Xiong, K., Yang, S. & Xing, D. Intravascular confocal photoacoustic endoscope with dual-element ultrasonic transducer. Opt. Exp. 23, 9130–9136 (2015).
Jansen, K., Van der Steen, A. F., Van Beusekom, H. M., Oosterhuis, J. W. & Van Soest, G. Intravascular photoacoustic imaging of human coronary atherosclerosis. Opt. Lett. 36, 597–599 (2011).
Karpiouk, A. B., Wang, B. & Emelianov, S. Y. Development of a catheter for combined intravascular ultrasound and photoacoustic imaging. Rev. Sci. Instrum. 81, 014901 (2010).
Yang, J.-M. et al. A 2.5-mm diameter probe for photoacoustic and ultrasonic endoscopy. Opt. Exp. 20, 23944–23953 (2012).
Subochev, P. et al. Simultaneous photoacoustic and optically mediated ultrasound microscopy: phantom study. Opt. Lett. 37, 4606–4608 (2012).
Subochev, P., Orlova, A., Shirmanova, M., Postnikova, A. & Turchin, I. Simultaneous photoacoustic and optically mediated ultrasound microscopy: an in vivo study. Biomed. Opt. Exp. 6, 631–638 (2015).
Subochev, P., Fiks, I. & Frenz, M. Simultaneous triple-modality imaging of diffuse reflectance, optoacoustic pressure and ultrasonic scattering using an acoustic-resolution photoacoustic microscope: feasibility study. Laser Phys. Lett. 13, 025605 (2016).
Subochev, P. Cost-effective imaging of optoacoustic pressure, ultrasonic scattering, and optical diffuse reflectance with improved resolution and speed. Opt. Lett. 41, 1006–1009 (2016).
Liu, M. et al. In vivo three dimensional dual wavelength photoacoustic tomography imaging of the far-red fluorescent protein E2-Crimson expressed in adult zebrafish. Biomed. Opt. Exp. 4, 1846–1855 (2013).
Acknowledgements
The authors would like to thank Chapin Rodriguez for proofreading the manuscript and for his valuable suggestions, and Luis Den Bean for his useful comments and suggestions. V.N. acknowledges funding from the Deutsche Forschungsgemeinschaft, Germany (Leibniz Prize 2013; NT 3/10-1), from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 687866 (INNODERM), and from the European Union’s Horizon 2020 research and innovation programme under grant agreement No 732720 (ESOTRAC). The content of this manuscript reflects only the authors’ view, and the European commission is not responsible for any use that may be made of the information provided.
Author information
Authors and Affiliations
Contributions
All authors contributed to writing the paper, revising it and approving the final version.
Corresponding author
Ethics declarations
Competing interests
V.N. is a shareholder in iThera Medical GmbH, a company that commercializes optoacoustic mesoscopy. The company did not provide support for this work.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary discussion.
Rights and permissions
About this article
Cite this article
Omar, M., Aguirre, J. & Ntziachristos, V. Optoacoustic mesoscopy for biomedicine. Nat Biomed Eng 3, 354–370 (2019). https://doi.org/10.1038/s41551-019-0377-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0377-4
- Springer Nature Limited
This article is cited by
-
Correction of high-rate motion for photoacoustic microscopy by orthogonal cross-correlation
Scientific Reports (2024)
-
Bragg grating etalon-based optical fiber for ultrasound and optoacoustic detection
Nature Communications (2024)
-
A flexible optoacoustic blood ‘stethoscope’ for noninvasive multiparametric cardiovascular monitoring
Nature Communications (2023)
-
Dermal features derived from optoacoustic tomograms via machine learning correlate microangiopathy phenotypes with diabetes stage
Nature Biomedical Engineering (2023)
-
Opening a window to skin biomarkers for diabetes stage with optoacoustic mesoscopy
Light: Science & Applications (2023)