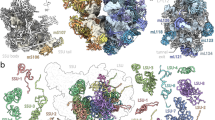
Abstract
The vast majority of eukaryotic cells contain mitochondria, essential powerhouses and metabolic hubs1. These organelles have a bacterial origin and were acquired during an early endosymbiosis event2. Mitochondria possess specialized gene expression systems composed of various molecular machines, including the mitochondrial ribosomes (mitoribosomes). Mitoribosomes are in charge of translating the few essential mRNAs still encoded by mitochondrial genomes3. While chloroplast ribosomes strongly resemble those of bacteria4,5, mitoribosomes have diverged significantly during evolution and present strikingly different structures across eukaryotic species6,7,8,9,10. In contrast to animals and trypanosomatids, plant mitoribosomes have unusually expanded ribosomal RNAs and have conserved the short 5S rRNA, which is usually missing in mitoribosomes11. We have previously characterized the composition of the plant mitoribosome6, revealing a dozen plant-specific proteins in addition to the common conserved mitoribosomal proteins. In spite of the tremendous recent advances in the field, plant mitoribosomes remained elusive to high-resolution structural investigations and the plant-specific ribosomal features of unknown structures. Here, we present a cryo-electron microscopy study of the plant 78S mitoribosome from cauliflower at near-atomic resolution. We show that most of the plant-specific ribosomal proteins are pentatricopeptide repeat proteins (PPRs) that deeply interact with the plant-specific rRNA expansion segments. These additional rRNA segments and proteins reshape the overall structure of the plant mitochondrial ribosome, and we discuss their involvement in the membrane association and mRNA recruitment prior to translation initiation. Finally, our structure unveils an rRNA-constructive phase of mitoribosome evolution across eukaryotes.




Similar content being viewed by others
Data availability
The cryo-EM maps of the mitoribosome have been deposited at the Electron Microscopy Data Bank (EMDB-10654), containing the full mitoribosome map along with the focused LSU from the full mitoribosome, the focused SSU body, the focused SSU head and the SSU head extension from the multi-body refinement. The atomic model of the full mitoribosome have been deposited in the PDB (6XYW).
References
Spinelli, J. B. & Haigis, M. C. The multifaceted contributions of mitochondria to cellular metabolism. Nat. Cell Biol. 20, 745–754 (2018).
Gray, M. W. Mosaic nature of the mitochondrial proteome: implications for the origin and evolution of mitochondria. Proc. Natl Acad. Sci. USA 112, 10133–10138 (2015).
Bieri, P., Greber, B. J. & Ban, N. High-resolution structures of mitochondrial ribosomes and their functional implications. Curr. Opin. Struct. Biol. 49, 44–53 (2018).
Boerema, A. P. et al. Structure of the chloroplast ribosome with chl-RRF and hibernation-promoting factor. Nat. Plants 4, 212–217 (2018).
Bieri, P., Leibundgut, M., Saurer, M., Boehringer, D. & Ban, N. The complete structure of the chloroplast 70S ribosome in complex with translation factor pY. EMBO J. 36, 475–486 (2017).
Waltz, F. et al. Small is big in Arabidopsis mitochondrial ribosome. Nat. Plants 5, 106–117 (2019).
Desai, N., Brown, A., Amunts, A. & Ramakrishnan, V. The structure of the yeast mitochondrial ribosome. Science 355, 528–531 (2017).
Ramrath, D. J. F. et al. Evolutionary shift toward protein-based architecture in trypanosomal mitochondrial ribosomes. Science 362, eaau7735 (2018).
Brown, A. et al. Structures of the human mitochondrial ribosome in native states of assembly. Nat. Struct. Mol. Biol. 24, 866–869 (2017).
Greber, B. J. et al. The complete structure of the 55S mammalian mitochondrial ribosome. Science 348, 303–308 (2015).
Unseld, M., Marienfeld, J. R., Brandt, P. & Brennicke, A. The mitochondrial genome of Arabidopsis thaliana contains 57 genes in 366,924 nucleotides. Nat. Genet. 15, 57–61 (1997).
Nakane, T., Kimanius, D., Lindahl, E. & Scheres, S. H. Characterisation of molecular motions in cryo-EM single-particle data by multi-body refinement in RELION. eLife 7, e42166 (2018).
Amunts, A., Brown, A., Toots, J., Scheres, S. H. W. & Ramakrishnan, V. The structure of the human mitochondrial ribosome. Science 348, 95–98 (2015).
Leontiadou, F., Triantafillidou, D. & Choli-Papadopoulou, T. On the characterization of the putative S20-thx operon of Thermus thermophilus. Biol. Chem. 382, 1001–1006 (2001).
Kummer, E. et al. Unique features of mammalian mitochondrial translation initiation revealed by cryo-EM. Nature 560, 263–267 (2018).
Zoschke, R. & Bock, R. Chloroplast translation: structural and functional organization, operational control, and regulation. Plant Cell 30, 745–770 (2018).
Sloan, D. B. et al. Cytonuclear integration and co-evolution. Nat. Rev. Genet. 19, 635–648 (2018).
Englmeier, R., Pfeffer, S. & Förster, F. Structure of the human mitochondrial ribosome studied in situ by cryoelectron tomography. Structure 25, 1574–1581 (2017).
Pfeffer, S., Woellhaf, M. W., Herrmann, J. M. & Förster, F. Organization of the mitochondrial translation machinery studied in situ by cryoelectron tomography. Nat. Commun. 6, 6019 (2015).
Barkan, A. et al. A combinatorial amino acid code for RNA recognition by pentatricopeptide repeat proteins. PLoS Genet. 8, 4–11 (2012).
Hammani, K. et al. Helical repeats modular proteins are major players for organelle gene expression. Biochimie 100, 141–150 (2014).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Zhang, K. Gctf: real-time CTF determination and correction. J. Struct. Biol. 193, 1–12 (2016).
Zivanov, J. et al. New tools for automated high-resolution cryo-EM structure determination in RELION-3. eLife 7, e42166 (2018).
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nat. Methods 11, 63–65 (2014).
Arenz, S. et al. Structures of the orthosomycin antibiotics avilamycin and evernimicin in complex with the bacterial 70S ribosome. Proc. Natl Acad. Sci. USA 113, 7527–7532 (2016).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Goddard, T. D. et al. UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci. 27, 14–25 (2018).
Ott, M., Amunts, A. & Brown, A. Organization and regulation of mitochondrial protein synthesis. Annu. Rev. Biochem. 85, 77–101 (2016).
Zimmermann, L. et al. A completely reimplemented MPI bioinformatics toolkit with a new hhpred server at its core. J. Mol. Biol. 430, 2237–2243 (2018).
Acknowledgements
We thank A. Bezault for assistance with the Talos Arctica electron microscope; L. Kuhn, J. Chicher and P. Hamman of the Strasbourg Espanade proteomic platform for the proteomic analysis; and M. Sissler for her comments during the article redaction. This work has benefitted from the facilities and expertise of the Biophysical and Structural Chemistry platform (BPCS) at IECB, CNRS UMS3033, Inserm US001, University of Bordeaux. This work was supported by the Centre National de la Recherche Scientifique, the University of Strasbourg, by Agence Nationale de la Recherche (ANR) grants (MITRA, ANR-16-CE11-0024-02, to P.G. and Y.H.) and by the LabEx consortium MitoCross in the frame of the French National Program Investissement d’Avenir (ANR-11-LABX-0057_MITOCROSS), as well as by a European Research Council Starting Grant (TransTryp ID 759120, to Y.H.).
Author information
Authors and Affiliations
Contributions
P.G., F.W. and Y.H. designed and coordinated the experiments. F.W. purified the mitochondria and mitochondrial ribosomes. H.S. acquired the cryo-EM data. H.S. and Y.H. processed the cryo-EM results. H.S., A.B. and F.W. built the atomic models. F.W., H.S. and Y.H. interpreted the structure. P.G., F.W., H.S. and Y.H. wrote and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Data processing workflow.
Graphical summary of the processing workflow described in Methods, with 2D classes presented in a for both datasets and 3D processing, presented in b, with ResMap of the full mitoribosome and SSU only before further processing. c FSC curves of the full mitoribosome and SSU before and after focused classification and multibody refinement.
Extended Data Fig. 2 Multibody refinement and additional SSU head domain movement amplitude.
a Views of the two extreme states of the head and head extension, relative to the body of the SSU, calculated using the multibody refinement implemented in RELION 3.0 (ref. 12), showing the movement in two different planes. b All ten states reveals a movement amplitude of 30°.
Extended Data Fig. 3 Secondary structure diagram of the plant 18S rRNA.
2D representation of the 18S rRNA colored by domain. The rRNA expansions specific to the plant mitoribosome are highlighted in cyan. Extensions that could not be modelled are indicated by dashed lines. A simplified secondary structure diagram of the E. coli 16S rRNA is also shown in the black frame, helices not present in the plant mitoribosome are shown in gray. Secondary structure templates were obtained from the RiboVision suite (http://apollo.chemistry.gatech.edu/RiboVision).
Extended Data Fig. 4 Secondary structure diagram of the plant 26S and 5S rRNA.
2D representation of the 26S rRNA colored by domain. 5S rRNA is shown in dark green. The rRNA expansions specific to the plant mitoribosome are highlighted in cyan. Extensions that could not be modelled are indicated by dashed lines. Simplified secondary structure diagram of the E. coli 23S rRNA is also shown in the black frame, helices not present in the plant mitoribosome are shown in gray. Secondary structure templates were obtained from the RiboVision suite (http://apollo.chemistry.gatech.edu/RiboVision).
Extended Data Fig. 5 The central protuberance of the plant mitoribosome contains a 5S rRNA.
Atomic model of the central protuberance of the plant mitoribosome. Protein components are each indicated in different colors and the 5S rRNA is shown in blue. The plant mitoribosome, contrary to yeast, mammalian and trypanosoma mitoribosome has a 5S rRNA in its CP, like in bacteria. However, the mitochondria specific proteins mL40, mL46, mL59/64 and mL60 are present, augmenting the overall volume of the CP. Moreover, this indicates that the loss of the 5S in yeast, mammals and trypanosoma occurred after the acquisition of these proteins, which seem to constitute core components of the ancestral mitoribosome.
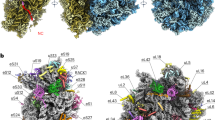
Extended Data Fig. 6 rPPR proteins in their respective densities.
a–h All rPPR present in the model in their respective filtered densities. Assignment of rPPRs was made mainly thanks to their numbers of repeats that constitute reliable ‘finger prints’. rPPR 4, 6 and 9 share the same number of repeats, in which case their assignment was possible thanks to the analysis of their bulky side-chains. a, c and d are designated rPPR* as the local resolution could not allow to distinguished them, even based on the number of repeats as rPPR1, 3a and 3b all are predicted to have 10 repeats. e-f Detailed with of selected side-chain in their densities that allowed unambiguous assignment. g mS83, even though resolved at low resolution, it was identified based on its unique number of repeats (6), which correlates with the predicted number of repeats from the TPRpred software31.
Extended Data Fig. 7 Specificities of plant mitoribosome proteins.
a Compared view of uS4m and bacterial uS4. The N-terminal part is shown in yellow and the C-terminal part in blue. The additional domain of uS4m is shown in red. This additional domain makes a part of the SSU body protuberance. b Similar to uS4m, uS3m also present a large additional domain. Due to low resolution in this area only part of the insertion was modelized and the rest is represented as Unknown5, however there is no doubt that the 250 amino-acids missing would constitute the large density observed on the head of the SSU, shown here in brown. N-terminal parts of the proteins are shown in yellow and C-terminal parts in blue. The additional domain of uS3m is shown in red. c In plant mitochondria, the uL2m protein was already speculated to be composed of two parts (32). The structure confirmed this hypothesis. The N-terminal part of the protein (red) is encoded by a mitochondrial gene and the C-terminal part of the protein (yellow) is encoded by a nuclear gene.
Extended Data Fig. 8 5’UTR of the mitochondrial mRNAs.
a Alignment of sequences surrounding the initiation codons of the 17, out the 33, protein coding genes encoded in the Arabidopsis mitochondrial genome. A characteristic AxAAA consensus is observed and illustrated by the WebLogo (https://weblogo.berkeley.edu/logo.cgi) representation in b.
Extended Data Fig. 9 Proteins of the plant mitochondrial ribosome.
Initial proteome analysis can be found in Waltz et al6. The r-proteins are colored by conservation with the bacterial ribosome (blue) other mitoribosomes (yellow) or specific to the plant mitoribosome (red). uL1m (At2g42710) and bL12m (At3g06040) were not observed in our reconstruction, but those proteins are localized on highly mobile part of the ribosome, respectively L1 and L7/L12 stalks, and found in the mass-spectrometry data to similar level as the other r-proteins, confirming their presence in the mitoribosome. rPPR* designate either rPPR1 (At1g61870), rPPR3a (At1g55890) or rPPR3b (At3g13160). Moreover, mS83, not previously characterized as a PPR protein is also renamed rPPR10. mS31/mS46 protein could not be identified but the density is highly similar to mS31/mS46 yeast and mammalian proteins. m designated proteins encoded in the mitochondrial genome.
Extended Data Fig. 10 Intersubunit bridges list.
The bridges are colored by conservation with the bacterial ribosome (blue) other mitoribosomes (yellow) or specific to the plant mitoribosome (red).In our reconstructions, the CP and SSU head contacts could not be clearly observed, except for mB8, thus B1 bridges were not listed here but are most likely present.
Supplementary information
Rights and permissions
About this article
Cite this article
Waltz, F., Soufari, H., Bochler, A. et al. Cryo-EM structure of the RNA-rich plant mitochondrial ribosome. Nat. Plants 6, 377–383 (2020). https://doi.org/10.1038/s41477-020-0631-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-020-0631-5
- Springer Nature Limited
This article is cited by
-
Structural basis of LRPPRC–SLIRP-dependent translation by the mitoribosome
Nature Structural & Molecular Biology (2024)
-
Mitoribosome structure with cofactors and modifications reveals mechanism of ligand binding and interactions with L1 stalk
Nature Communications (2024)
-
Structure of the actively translating plant 80S ribosome at 2.2 Å resolution
Nature Plants (2023)
-
Structure of a mitochondrial ribosome with fragmented rRNA in complex with membrane-targeting elements
Nature Communications (2022)
-
The P-type pentatricopeptide repeat protein DWEORG1 is a non-previously reported rPPR protein of Arabidopsis mitochondria
Scientific Reports (2022)