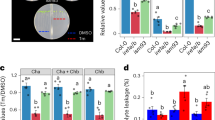
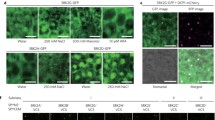
Abstract
Inositol-requiring enzyme 1 (IRE1) is the most conserved transducer of the unfolded protein response that produces either adaptive or death signals depending on the amplitude and duration of its activation. Here, we report that SQUAMOSA PROMOTER-BINDING PROTEIN-LIKE 6 (SPL6)-deficient plants displayed hyperactivation of the endoplasmic reticulum (ER) stress sensor IRE1, leading to cell death in rice panicles, indicating that SPL6 is an essential survival factor for the suppression of persistent or intense ER stress conditions. Importantly, knockdown of the hyperactivated mRNA level of IRE1 rescues panicle apical abortion in the spl6-1 transgenic plants harbouring the IRE1-RNAi constructs, establishing the genetic linkage between the hyperactivation of IRE1 and cell death in spl6-1. Our findings reveal a novel cell survival machinery in which SPL6 represses the transcriptional activation of the ER stress sensor IRE1 in control of ER stress signalling outputs that hinge on a balance between adaptive and death signals for determining cell fates during ER stress.






Similar content being viewed by others
References
Ron, D. & Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 8, 519–529 (2007).
Hetz, C. The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat. Rev. Mol. Cell Biol. 13, 89–102 (2012).
Howell, S. H. Endoplasmic reticulum stress responses in plants. Annu. Rev. Plant Biol. 64, 477–499 (2013).
Walter, P. & Ron, D. The unfolded protein response: from stress pathway to homeostatic regulation. Science 334, 1081–1086 (2011).
Deng, Y. et al. Heat induces the splicing by IRE1 of a mRNA encoding a transcription factor involved in the unfolded protein response in Arabidopsis. Proc. Natl Acad. Sci. USA 108, 7247–7252 (2011).
Nagashima, Y. et al. Arabidopsis IRE1 catalyses unconventional splicing of bZIP60 mRNA to produce the active transcription factor. Sci. Rep. 1, 29 (2011).
Hayashi, S., Wakasa, Y., Takahashi, H., Kawakatsu, T. & Takaiwa, F. Signal transduction by IRE1-mediated splicing of bZIP50 and other stress sensors in the endoplasmic reticulum stress response of rice. Plant J. 69, 946–956 (2012).
Lu, S.-J. et al. Conservation of IRE1-regulated bZIP74 mRNA unconventional splicing in rice (Oryza sativa L.) involved in ER stress responses. Mol. Plant 5, 504–514 (2012).
Rao, R. V., Ellerby, H. M. & Bredesen, D. E. Coupling endoplasmic reticulum stress to the cell death program. Cell Death Differ. 11, 372–380 (2004).
Maurel, M., Chevet, E., Tavernier, J. & Gerlo, S. Getting RIDD of RNA: IRE1 in cell fate regulation. Trends Biochem. Sci. 39, 245–254 (2014).
Chen, Y. & Brandizzi, F. IRE1: ER stress sensor and cell fate executor. Trends Cell Biol. 23, 547–555 (2013).
Klein, J., Saedler, H. & Huijser, P. A new family of DNA binding proteins includes putative transcriptional regulators of the Antirrhinum majus floral meristem identity gene SQUAMOSA. Mol. Gen. Genet. 250, 7–16 (1996).
Wu, G. & Poethig, R. S. Temporal regulation of shoot development in Arabidopsis thaliana by miR156 and its target SPL3. Development 133, 3539–3547 (2006).
Usami, T., Horiguchi, G., Yano, S. & Tsukaya, H. The more and smaller cells mutants of Arabidopsis thaliana identify novel roles for SQUAMOSA PROMOTER BINDING PROTEIN-LIKE genes in the control of heteroblasty. Development 136, 955–964 (2009).
Wang, J.-W., Czech, B. & Weigel, D. miR156-regulated SPL transcription factors define an endogenous flowering pathway in Arabidopsis thaliana. Cell 138, 738–749 (2009).
Yamaguchi, A. et al. The microRNA-regulated SBP-box transcription factor SPL3 is a direct upstream activator of LEAFY, FRUITFULL, and APETALA1. Dev. Cell 17, 268–278 (2009).
Cardon, G. H., Hohmann, S., Nettesheim, K., Saedler, H. & Huijser, P. Functional analysis of the Arabidopsis thaliana SBP-box gene SPL3: a novel gene involved in the floral transition. Plant J. 12, 367–377 (1997).
Wang, H. et al. The origin of the naked grains of maize. Nature 436, 714–719 (2005).
Manning, K. et al. A naturally occurring epigenetic mutation in a gene encoding an SBP-box transcription factor inhibits tomato fruit ripening. Nat. Genet. 38, 948–952 (2006).
Unte, U. S. et al. SPL8, an SBP-Box gene that affects pollen sac development in Arabidopsis. Plant Cell 15, 1009–1019 (2003).
Jiao, Y. et al. Regulation of OsSPL14 by OsmiR156 defines ideal plant architecture in rice. Nat. Genet. 42, 541–544 (2010).
Miura, K. et al. OsSPL14 promotes panicle branching and higher grain productivity in rice. Nat. Genet. 42, 545–549 (2010).
Zhu, Z. F. et al. Genetic control of inflorescence architecture during rice domestication. Nat. Commun. 4, 2200 (2013).
Chuck, G. S., Brown, P. J., Meeley, R. & Hake, S. Maize SBP-box transcription factors unbranched2 and unbranched3 affect yield traits by regulating the rate of lateral primordia initiation. Proc. Natl Acad. Sci. USA 111, 18775–18780 (2014).
Preston, J. C. & Hileman, L. C. Functional evolution in the plant SQUAMOSA-PROMOTER BINDING PROTEIN-LIKE (SPL) gene family. Front. Plant Sci. 4, 80 (2013).
Xie, K., Wu, C. & Xiong, L. Genomic organization, differential expression, and interaction of SQUAMOSA promoter-binding-like transcription factors and microRNA156 in rice. Plant Physiol. 142, 280–293 (2006).
Rhoades, M. W. et al. Prediction of plant microRNA targets. Cell 110, 513–520 (2002).
Yang, J. et al. A killer-protector system regulates both hybrid sterility and segregation distortion in rice. Science 337, 1336–1340 (2012).
Birkenbihl, R. P., Jach, G., Saedler, H. & Huijser, P. Functional dissection of the plant-specific SBP-domain: overlap of the DNA-binding and nuclear localization domains. J. Mol. Biol. 352, 585–596 (2005).
Schroder, M. & Kaufman, R. J. The mammalian unfolded protein response. Annu. Rev. Biochem. 74, 739–789 (2005).
Woehlbier, U. & Hetz, C. Modulating stress responses by the UPRosome: a matter of life and death. Trends Biochem. Sci. 36, 329–337 (2011).
Danial, N. N. & Korsmeyer, S. J. Cell death: critical control points. Cell 116, 205–219 (2004).
Zuppini, A., Navazio, L. & Mariani, P. Endoplasmic reticulum stress-induced programmed cell death in soybean cells. J. Cell Sci. 117, 2591–2598 (2004).
Iwata, Y. & Koizumi, N. Unfolded protein response followed by induction of cell death in cultured tobacco cells treated with tunicamycin. Planta 220, 804–807 (2005).
Watanabe, N. & Lam, E. BAX inhibitor-1 modulates endoplasmic reticulum stress-mediated programmed cell death in Arabidopsis. J. Biol. Chem. 283, 3200–3210 (2008).
Qiang, X., Zechmann, B., Reitz, M. U., Kogel, K.-H. & Schaefer, P. The mutualistic fungus Piriformospora indica colonizes Arabidopsis roots by inducing an endoplasmic reticulum stress-triggered caspase-dependent cell death. Plant Cell 24, 794–809 (2012).
Yang, Z.-T. et al. The membrane-associated transcription factor NAC089 controls ER-stress-induced programmed cell death in plants. PLoS Genet. 10, e1004243 (2014).
Deng, Y., Srivastava, R. & Howell, S. H. Endoplasmic reticulum (ER) stress response and its physiological roles in plants. Int. J. Mol. Sci. 14, 8188–8212 (2013).
Iwata, Y. & Koizumi, N. Plant transducers of the endoplasmic reticulum unfolded protein response. Trends Plant Sci. 17, 720–727 (2012).
Akter, M. B. et al. Fine mapping and candidate gene analysis of a new mutant gene for panicle apical abortion in rice. Euphytica 197, 387–398 (2014).
Yamagishi, J., Miyamoto, N., Hirotsu, S., Laza, R. C. & Nemoto, K. QTLs for branching, floret formation, and pre-flowering floret abortion of rice panicle in a temperate japonica × tropical japonica cross. Theor. Appl. Genet. 109, 1555–1561 (2004).
Tan, C. J., Sun, Y. J., Xu, H. S. & Yu, S. B. Identification of quantitative trait locus and epistatic interaction for degenerated spikelets on the top of panicle in rice. Plant Breed. 130, 177–184 (2011).
Cheng, Z.-J. et al. Fine mapping of qPAA8, a gene controlling panicle apical development in rice. J. Integr. Plant Biol. 53, 710–718 (2011).
Chen, Y. & Brandizzi, F. AtIRE1A/AtIRE1B and AGB1 independently control two essential unfolded protein response pathways in Arabidopsis. Plant J. 69, 266–277 (2012).
Wakasa, Y., Hayashi, S., Ozawa, K. & Takaiwa, F. Multiple roles of the ER stress sensor IRE1 demonstrated by gene targeting in rice. Sci. Rep. 2, 944 (2012).
Toki, S. et al. Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J. 47, 969–976 (2006).
Yu, H.-D. et al. Downregulation of chloroplast RPS1 negatively modulates nuclear heat-responsive expression of HsfA2 and its target genes in Arabidopsis. PLoS Genet. 8, e1002669 (2012).
Wu, G. et al. The sequential action of miR156 and miR172 regulates developmental timing in Arabidopsis. Cell 138, 750–759 (2009).
Phan, H. A., Iacuone, S., Li, S. F. & Parish, R. W. The MYB80 transcription factor is required for pollen development and the regulation of tapetal programmed cell death in Arabidopsis thaliana. Plant Cell 23, 2209–2224 (2011).
Guan, Q., Yue, X., Zeng, H. & Zhu, J. The protein phosphatase RCF2 and its interacting partner NAC019 are critical for heat stress-responsive gene regulation and thermotolerance in Arabidopsis. Plant Cell 26, 438–453 (2014).
Acknowledgements
This study was supported by the Chinese Academy of Sciences (Strategic Priority Research Program XDPB0404), the Ministry of Science and Technology of China (National Key R&D Program of China, 2016YFD0100405), the National Natural Science Foundation of China (31770314 and 31570260) and the Chinese Academy of Sciences (XDA08010203). We thank the Rice T-DNA Insertion Sequence Database for providing the rice mutant lines; H.-T. Liu, X.-F. Yang and S.-Q. Zhao for suggestions and technical assistance; X.-Y. Gao, X.-S. Gao, J.-Q. Li and Z.-P. Zhang for assistance with electron microscopy and confocal microscopy.
Author information
Authors and Affiliations
Contributions
F.-Q.G. and Q.-L.W. supervised the project and designed the experiments. Q.-L.W., A.-Z.S., S.-T.C. and L.-S.C. performed the experiments. Q.-L.W., A.-Z.S. and S.-T.C. analysed the data and interpreted the results. F.-Q.G. and Q.-L.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–22 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Wang, QL., Sun, AZ., Chen, ST. et al. SPL6 represses signalling outputs of ER stress in control of panicle cell death in rice. Nature Plants 4, 280–288 (2018). https://doi.org/10.1038/s41477-018-0131-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-018-0131-z
- Springer Nature Limited