Abstract
Stem cells in plants and animals are the source of new tissues and organs. In plants, stem cells are maintained in the central zone (CZ) of multicellular meristems, and large shoot meristems with an increased stem cell population hold promise for enhancing yield. The mobile homeodomain transcription factor WUSCHEL (WUS) is a central regulator of stem cell function in plant shoot meristems. Despite its central importance, the factors that directly modulate WUS protein stability have been a long-standing question. Here, we show that the peptidase DA1 physically interacts with and cleaves the WUS protein, leading to its destabilization. Furthermore, our results reveal that cytokinin signaling represses the level of DA1 protein in the shoot apical meristem, thereby increasing the accumulation of WUS protein. Consistent with these observations, loss of DA1 function results in larger shoot apical meristems with an increased stem cell population and also influences cytokinin-induced enlargement of shoot apical meristem. Collectively, our findings uncover a previously unrecognized mechanism by which the repression of DA1 by cytokinin signaling stabilizes WUS, resulting in the enlarged shoot apical meristems with the increased stem cell number during plant growth and development.
Similar content being viewed by others
Introduction
Plants and animals have a similar stem cell microenvironment, although they have evolved independently of each other1,2. Unlike most animals, plants can generate new shoot and root organs throughout their lifespan by continuous production of pluripotent stem cells in specific structures or tissues called meristems1,3. The Shoot Apical Meristem (SAM) is the source of all post-embryonic cells in aerial parts of a plant4. The Arabidopsis SAM contains three distinct layers of cells (L1-L3), which differentiate into epidermis, ground tissues, and vascular tissues of stem, leaf, and flower, respectively5. The SAM can be divided into different functional domains according to its functions and cytological features. The region containing pluripotent stem cells is called the Central Zone (CZ), in which cells divide slowly and form Peripheral Zones (PZ). The Organizing Center (OC) is a group of cells beneath the CZ region, which specifically initiates and maintains the activity of stem cells6,7,8. Meristem size is determined by the number of stem cells, and these influence the size of organs derived from the meristem9,10,11. The homeodomain transcription factor WUSCHEL (WUS) has a central function in maintaining meristem stem cells7. WUS is specifically expressed in the OC, and the null mutations in WUS result in loss of stem cell properties and premature termination of the shoot apical meristem12,13. Conversely, induction of WUS in the central zone leads to increased apical meristem size14. WUS expression is inhibited by a signaling cascade involving the diffusible peptide CLAVATA3 (CLV3) that binds to the transmembrane receptor kinase CLV1 and receptor-like protein CLV215,16,17,18. Loss of function of this cascade leads to larger meristems due to WUS over-expression19,20,21. WUS protein also moves from the OC to the CZ to activate the expression of CLV322,23. Thus, they form a feedback loop that balances stem cell maintenance and cell differentiation in the shoot apical meristem24. WUS functions as a heterodimer with HAIRY MERISTEM (HAM) transcription factors to regulate the stem cell microenvironment by regulating common downstream genes25. WUS-HAM heterodimers confine CLV3 expression to the outer apical meristem layer26. WUS also interacts with SHOOT MERISTEMLESS (STM) to regulate CLV3 expression and control stem cell number27. In addition, several transcriptional regulators have been described to regulate shoot stem cell function by influencing CLV-WUS signaling in Arabidopsis28,29,30,31,32. Despite the central role of WUS in the initiation and maintenance of stem cell fate, modulation of WUS protein levels remains poorly understood.
The phytohormones cytokinin and auxin play important roles in the regulation of shoot meristems. Cytokinin signaling promotes the enlargement of the SAM and increases shoot stem cell number by activating the expression of the WUS gene33. In contrast, WUS can repress the transcription of several two-component ARABIDOPSIS RESPONSE REGULATORS genes (ARRs)34. Recently, cytokinin signaling has also been reported to stabilize WUS protein, resulting in the accumulation of WUS protein29. Therefore, cytokinin signaling promotes the accumulation of WUS protein through both transcriptional and post-translational regulation of WUS. However, how cytokinin signaling promotes WUS protein accumulation remains unclear due to the lack of an established mechanism for WUS degradation.
The cleavage of proteins is an important way to determine the functions of targeted proteins in plants and animals. We have previously shown that the peptidase DA1 is a central regulator of seed and organ size35,36. Here we show that the peptidase DA1 directly cleaves and destabilizes WUS protein in the SAM. Cytokinin signaling influences this process by repressing DA1 protein levels. Consequently, the da1-1 mutant produces larger shoot apical meristems with an increased stem cell population, and DA1 activity is required for cytokinin-induced enlargement of shoot apical meristem. Our findings reveal an important genetic and molecular mechanism linking cleavage of WUS by DA1 to cytokinin signaling and apical meristem size control.
Results
DA1 regulates shoot stem cell function and SAM size
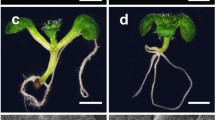
The peptidase DA1 limits seed and organ growth in Arabidopsis35,36. The da1-1 and da1-1Ler mutants formed larger seeds, leaves, inflorescences and flowers than their parental lines Col-0 and Ler, respectively (Supplementary Fig. 1)35,36. We also observed that sepal, petal, and stamen numbers and carpel sizes in da1-1 and da1-1Ler were increased compared with those in their parental lines (Supplementary Fig. 1). Enlarged inflorescences and an increased number of floral organs are associated with larger shoot meristems37. We therefore examined the shoot apical meristem (SAM) size of da1-1Ler and da1-1. As shown in Supplementary Fig. 2, da1-1Ler has a larger SAM and inflorescence meristem (IM) compared with Ler. Similarly, the SAM in da1-1 was obviously larger than that in Col-0 (Fig. 1a, b). The quantification data also showed that the average area of the da1-1 SAM was significantly increased compared with that of Col-0 (Fig. 1b and Supplementary Fig. 3). We previously demonstrated that the genomic fragment containing the DA1 gene (DA1COM) complemented the large seed and organ phenotypes of da1-136. The genomic fragment (DA1COM) also complemented the enlarged SAM phenotype of da1-1 (Fig. 1a, b). A previous study showed that over-expression of DA1 in 35 S:GFP-DA1 plants led to smaller organs38. Similarly, 35 S:GFP-DA1 seedlings also had smaller SAM than the wild type (Fig. 1a, b). Therefore, DA1 negatively regulates shoot meristem size. Expression of DA1 in the SAM was assessed in a pDA1:GUS transgenic line36. As expected, DA1 is strongly expressed in the SAM region (Fig. 1c), consistent with its role in defining SAM size. We also observed that DA1 is strongly expressed in younger leaves (Fig. 1c and Supplementary Fig. 4), consistent with a previous report36. An increased number of cells expressing the stem cell marker gene CLV3 were seen in the da1-1 SAM compared to the wild type SAM (Fig. 1d, e), indicating that da1-1 increases stem cell production in SAM. Thus, these results demonstrated that DA1 is involved in the regulation of shoot stem cell function and meristem size.
a SAMs of Col-0, da1-1, DA1COM and 35S:GFP-DA1 (n = 38). DA1COM represents that the da1-1 mutant was transformed with the DA1 genomic DNA. b The average SAM area of Col-0, da1-1, DA1COM and 35 S:GFP-DA1 (n = 38). The measured region of SAM was shown in Supplementary Fig. 3. Data are mean ± s.e.m. relative to the wild-type value (100%). One-way ANOVA with Tukey’s multiple comparison test was used for statistical analyses(P < 0.05). c Images of SAMs from pDA1:GUS plants(n = 15). d Images of SAMs from pCLV3:YFP (n = 19)(left) and pCLV3:YFP; da1-1Ler (n = 20)(right) plants. YFP (yellow) fluorescence was shown. e The number of fluorescing cells in SAMs of the pCLV3:YFP (n = 19) and pCLV3:YFP; da1-1Ler (n = 20) plants. Data are mean ± s.e.m. relative to the pCLV3:YFP value. P values are from two-sided Student’s t tests. Scale bars, 20 μm (a, c). 10 μm (d). All the plants were grown for 6 days in long-day conditions. The experiments were done with similar results in at least two independent replicates. Source data are provided as a Source Data file.
DA1 acts genetically with WUS to control shoot stem cell function and SAM size
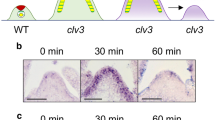
The regulation of SAM size and stem cell population depends on a WUS-CLV3 feedback loop7,12,19,20,21. SAMs of clv mutants are significantly enlarged, and the number of floral organs in clv mutants is increased, similar to SAM phenotypes observed in da1-1 and da1-1Ler mutants. To explore genetic relationships between DA1 and CLV genes in SAM size control, we generated da1-1Ler clv1-1, da1-1Ler clv2-1 and da1-1Ler clv3-2 double mutants. As shown in Supplementary Fig. 5, da1-1Ler strongly enhanced the numbers of clv1-1, clv2-1, and clv3-2 sepals, petals, stamens, and carpels, respectively, suggesting a synergistic genetic interaction between DA1 and CLV genes in floral organ number control. SAM sizes of clv1-1, clv2-1, and clv3-2 were dramatically enhanced by the da1-1Ler (Fig. 2a, b). These analyses indicated that the da1-1Ler mutation synergistically enhanced the large SAM phenotype of clv1-1, clv2-1, and clv3-2 mutants, suggesting that DA1 may act redundantly or in parallel with CLV1, CLV2, and CLV3 to control SAM size and stem cell population39.
a SAMs of Ler (n = 55), da1-1Ler(n = 56), clv1-1(n = 63), da1-1Ler clv1-1(n = 62), clv2-1(n = 47), da1-1Ler clv2-1(n = 58), clv3-2 (n = 63), and da1-1Ler clv3-2(n = 21). b The average SAM area of Ler (n = 55), da1-1Ler(n = 56), clv1-1(n = 63), da1-1Ler clv1-1(n = 62), clv2-1(n = 47), da1-1Ler clv2-1(n = 58), clv3-2 (n = 63), and da1-1Ler clv3-2(n = 21). c SAMs of Ler, da1-1Ler, wus-1 and da1-1Ler wus-1(n = 20). d SAM of Ler, da1-1Ler, wus-7 and da1-1Ler wus-7(n = 46). e The average area of Ler, da1-1Ler, wus-7, and da1-1Ler wus-7(n = 46) SAMs. f The relative expression levels of a set of genes, which have been reported to be upregulated or downregulated by the inducible activation of WUS. SAMs of 6-day-old Col-0 and da1-1 were used to perform quantitative RT-PCR assay. Data was normalized with ACTIN2. Data are mean ± S.D (n = 3 for three biological repeats) relative to the wild-type value. P values are from two-sided Student’s t tests. **P < 0.01 compared with the wild type (Col-0). Data in b and e are presented as mean values ± s.e.m. One-way ANOVA with Tukey’s multiple comparison test was used for statistical analyses (P < 0.05). Scale bars, 50 μm (a, c, d). All the plants were grown for 6 days in long-day conditions. The experiments in a, c, d were done with similar results in at least two independent replicates. Source data are provided as a Source Data file.
These synergistic genetic interactions between DA1 and the CLV pathway suggested that they may share a common downstream component39. The CLV pathway restricts expression of WUS, a central regulator of shoot stem cell proliferation7,12,19,20,21. To assess whether WUS mediates the effect of DA1 on SAM size, we generated a da1-1Ler wus-1 double mutant. The wus-1 mutant is not able to form a normal shoot apical meristem12. Similarly, the da1-1Ler wus-1 double mutant did not form a normal shoot apical meristem (Fig. 2c and Supplementary Fig. 6a). The da1-1Ler wus-1 double mutant exhibited similar plant growth phenotypes to wus-1 (Fig. 2c and Supplementary Fig. 6a, b). We also observed that the SAM region of the da1-1Ler wus-1 double mutant was obviously wider than that of the wus-1 single mutant (Fig. 2c), suggesting that DA1 and WUS may have partially overlapped function in SAM size control. As DA1 has several diverse substrates35,40,41, it is plausible that some of its substrates might be involved in the regulation of SAM size. Considering that wus-1 is a very strong allele and can not form SAM, we further used the weak wus-7 allele to conduct additional genetic analyses. The wus-7 seedlings formed small distorted shoot apical meristems compared to the wild type (Fig. 2d). The da1-1Ler wus-7 double mutant had an overall similar SAM shape to the wus-7 single mutant (Fig. 2d), but the quantification of SAM size showed the wus-7 mutation partially suppressed the large SAM phenotype of da1-1Ler (Fig. 2d, e). As wus-7 is not a null allele, it is possible that any remaining activity of WUS in wus-7 may still be influenced by da1-1. Thus, it seems reasonable that the da1-1Ler wus-7 double mutant had slightly larger SAM than wus-7. These genetic analyses indicated that the large SAM of da1-1 partially depends on the functional WUS. Quantitative RT-PCR results revealed a common set of genes that are regulated by inducible activation of WUS and by da1-1 (Fig. 2f)42, while WUS expression was not affected by da1-1 mutation (Fig. 2f). These results provided further evidence for possible common roles of DA1 and WUS in regulating shoot stem cell function.
DA1 physically interacts with and cleaves WUS
As DA1 acts genetically with WUS, and WUS expression was not changed in the da1-1 mutant, we asked whether DA1 could directly interact with WUS. Yeast two hybrid assays showed that DA1 interacted with WUS in yeast cells (Fig. 3a). The WUS protein comprises a homeodomain, a dimerization domain, the WUS-box and the EARL motif, we then asked which domain of WUS could interact with DA1. Surprisingly, we found that most of deletion versions of WUS fused to BD autoactivated the reporter gene (Supplementary Fig. 7a). In addition, the full-length DA1 or the UIM domains fused to BD also autoactivated the reporter gene (Supplementary Fig. 7b), consistent with a previous report41. We therefore used DA1-LIM + C (LIM domains and the peptidase domain) fused to BD to test the interactions between DA1 and the domains of WUS, respectively. Interestingly, we found that the DA1-LIM + C interacted with the C terminal region of WUS (the dimerization domain, WUS-box and EARL motif), although the DA1-LIM + C did not interact with any single domain of WUS (Supplementary Fig. 7c), respectively. We further found that WUS did interact with AD-DA1-LIM in yeast cells (Supplementary Fig. 7d).
a The indicated construct pairs were co-transformed into yeast strain Y2H Gold (Clontech). Interactions were examined on the control media DDO (SD/-Leu/-Trp) and selective media QDO (SD/-Ade/-His/-Leu/-Trp). b DA1 directly interacts with WUS in vitro. GST-DA1 was pulled down (PD) by MBP-WUS immobilized on amylose resin and analyzed by immunoblotting (IB) with an anti-GST antibody. c WUS interacts with DA1 in vivo. N. benthamiana leaves were transformed by injection of Agrobacterium tumefaciens GV3101 cells harboring 35S:GFP-WUS and 35S:MYC-DA1 plasmids. MYC-DA1 was detected in the immunoprecipitated GFP-WUS complex, indicating that there is a physical association between DA1 and WUS in vivo. d Co-immunoprecipitation analyses showing the interactions between WUS and DA1 in Arabidopsis. Total protein extracts of pWUS:WUS-HA; 35 S:GFP and pWUS:WUS-HA; 35 S:GFP-DA1 plants were incubated with GFP-Trap agarose beads. Precipitates were detected by western blot with anti-GFP and anti-HA antibody. e, f Arabidopsis da1-ko1 dar1-1 mesophyll protoplasts were cotransformed with plasmids expressing WUS-FLAG or FLAG-WUS with HA-DA1 and HA-DA1pep, respectively. The specific cleavage products of WUS-FLAG and FLAG-WUS were indicated by red arrowheads. IP immunoprecipitation; IN input; IB immunoblot. Experiments in b–f were repeated independently at least twice with similar results. Source data are provided as a Source Data file.
To further confirm the interaction between DA1 and WUS, we performed in vitro pull-down assays. GST-DA1 and MBP-WUS fusion proteins were expressed in E.coli, respectively. Pull-down assays showed that GST-DA1 can bind MBP-WUS, but not the negative control MBP (Fig. 3b). The interaction between DA1 and WUS was then examined in planta by transient expression of 35 S:GFP-WUS together with 35S:MYC-DA1 in N. benthamiana leaves. The co-immunoprecipitation (Co-IP) analysis showed that MYC-DA1 was detected in the immunoprecipitation complex of GFP-WUS, but not in that of the negative control GFP (Fig. 3c), indicating that DA1 associates with WUS in vivo. We further investigated whether DA1 associated with WUS in Arabidopsis. We crossed 35 S:GFP-DA1 transgenic line with pWUS:WUS-HA and generated 35 S:GFP-DA1; pWUS:WUS-HA lines. Co-immunoprecipitation (Co-IP) assay revealed that WUS-HA was immunoprecipited with GFP-DA1, but not with the GFP only (Fig. 3d), indicating that DA1 associates with WUS in Arabidopsis. The additional bands below the full-length GFP-WUS or GFP-DA1 in Co-IP assays might be the degraded products as observed in several previous studies (Fig. 3c, d)35,40,43. Finally, bimolecular fluorescence complementation (BiFC) assays in N. benthamiana leaves also showed that DA1 associates with WUS in nuclei (Supplementary Fig. 8). Taken together, these results revealed that DA1 interacts with WUS in vitro and in vivo.
DA1 is a metallopeptidase that cleaves diverse growth regulatory proteins35. As DA1 genetically and physically interacts with WUS, we tested whether DA1 could cleave WUS by transient co-expression in protoplasts35. When WUS-FLAG was transiently co-expressed with HA-DA1, but not HA-DA1pep (no peptidase activity)35, the specific cleavage product of WUS-FLAG was detected (Fig. 3e and Supplementary Fig. 9a). When we put the FLAG tag in the N terminus of WUS (FLAG-WUS), similar results were obtained (Fig. 3f and Supplementary Fig. 9b). Thus, these results indicated that the peptidase DA1 can cleave WUS.
DA1 destabilizes WUS
DA1 peptidase cleaves its substrates and causes their destabilization35. Because our results showed that DA1 physically interacts with and cleaves WUS, this prompted us to investigate the stability of WUS protein in da1-1 plants. In transgenic pWUS:WUS-GFP plants, GFP was observed in the OC of SAM and migrated into the CZ (Fig. 4a), consistent with previous reports22,23. In pWUS:WUS-GFP; da1-1 plants, GFP florescence was observed in the OC and the CZ of the SAM (Fig. 4b). Cells with WUS-GFP were significantly increased in the SAM of pWUS:WUS-GFP; da1-1 compared with those in the SAM of pWUS:WUS-GFP (Fig. 4a, b, e). Similar results were also observed in inflorescence meristems (IMs) of pWUS:WUS-GFP; da1-1 (Fig. 4c, d, f). These results indicated that SAMs and IMs of da1-1 plants contain more WUS proteins than those of the wild type.
a, b SAMs from pWUS:WUS-GFP (a)(n = 14) and pWUS:WUS-GFP; da1-1 (b)(n = 10) transgenic plants. Plants were grown for 9 days in long-day conditions. c, d IMs from pWUS:WUS-GFP (c) (n = 19) and pWUS:WUS-GFP; da1-1 (d) (n = 14) transgenic plants. Plants were grown for 35 days in long-day conditions. e The fluorescing cell number of SAMs from pWUS:WUS-GFP (n = 14) and pWUS:WUS-GFP; da1-1 (n = 10) plants. f The fluorescing cell number of IMs in pWUS:WUS-GFP (n = 19) and pWUS:WUS-GFP; da1-1 (n = 14) plants. g, h SAMs from pWUS:WUS-GFP; pER8:MYC-DA1 plants treated without β -estradiol for 12 h (mock) (n = 8) and with β -estradiol for 12 h (treated) (n = 10). i, j SAMs from pWUS:WUS-GFP treated without β -estradiol for 12 h (mock) (n = 12) and with β -estradiol for 12 h (treated) (n = 12). k The protein levels of MYC-DA1 in pWUS:WUS-GFP; pER8:MYC-DA1 transgenic plants treated with or without (mock) β -estradiol for 12 h. Total protein extracts were subjected to immunoblot assays using anti-MYC, and anti-ACTIN (as loading control) antibodies. l The fluorescing cell number of SAMs from pWUS:WUS-GFP; pER8:MYC-DA1 plants treated without β-estradiol for 12 h (mock) (n = 8) and with β-estradiol for 12 h (n = 10). m The fluorescing cell number of SAMs from pWUS:WUS-GFP plants treated without β -estradiol for 12 h (mock) (n = 12) and with β -estradiol 12 h (treated) (n = 12). n Quantification of WUS-GFP mRNA levels in pWUS:WUS-GFP; pER8:MYC-DA1 plants without β -estradiol for 12 h (mock) and with β -estradiol for 12 h (treated). Data are mean ± s.e.m with three biological replicates. o Quantification of WUS-GFP mRNA levels in pWUS:WUS-GFP plants treated without β -estradiol for 12 h (mock) and with β -estradiol for 12 h (treated). Expression level of WUS-GFP at mock was set at 1. Data are mean ± s.e.m with three biological replicates. Data in e, f, l, and m are presented as mean values ± s.e.m. P values are from two-sided Student’s t tests. GFP (green) fluorescence were shown in a–d, g–j. Scale bars, 10 μm (a–d, g–j). Experiments in a–d, g–k were repeated independently at least twice with similar results. Source data are provided as a Source Data file.
The large SAM of da1-1 may contain more cells with WUS expression. To distinguish between a general increase in WUS-expressing cells in the larger SAM of da1-1 plants and the direct influence of DA1 activity on WUS protein levels within the SAM, we generated transgenic lines of the estradiol-inducible form of DA1 (pER8:MYC-DA1) and then crossed with pWUS:WUS-GFP to obtain pWUS:WUS-GFP; pER8:MYC-DA1 plants. The pWUS:WUS-GFP; pER8:MYC-DA1 seedlings treated with β -estradiol for 12 h led to the strong accumulation of MYC-DA1 protein (Fig. 4k). Conversely, cells with WUS-GFP in the SAMs of pWUS:WUS-GFP; pER8:MYC-DA1 were dramatically decreased at 12 h after induction (Fig. 4g, h, l). By contrast, cells with WUS-GFP in the SAMs of pWUS:WUS-GFP (a negative control) was not changed at 12 h after β -estradiol treatment (Fig. 4i, j, m). The few cells with WUS-GFP in the SAMs of pWUS:WUS-GFP; pER8:MYC-DA1 after β -estradiol treatment were not caused by the reduction of WUS-GFP gene expression, as the levels of WUS mRNA were similar in plants with or without β -estradiol induction (Fig. 4n, o). Considering that the β -estradiol treatment for 12 h did not obviously affect SAM size (Fig. 4g–j), the decreased cells with WUS-GFP in pWUS:WUS-GFP; pER8:MYC-DA1 SAM after β -estradiol induction were not caused by SAM size. Thus, DA1 activity destabilizes WUS proteins in the Arabidopsis SAM.
Specific expression of DA1 driven by the WUS promoter represses the large SAM phenotype of da1-1
WUS is expressed in the OC, and WUS protein moves from the OC to neighboring stem cells. DA1 is expressed in the entire shoot apical meristem. These observations suggested that DA1 may cleave and destabilize WUS in the OC and neighboring cells. Therefore, specific expression of DA1 using the WUS promoter may, at least in part, rescue the large SAM phenotype of da1-1. As predicted, the shoot apical meristems of transgenic da1-1 seedlings expressing DA1 driven by the WUS promoter were smaller than those of da1-1 (Fig. 5a–d, i). Surprisingly, we occasionally observed that transgenic plants exhibited deformed SAMs (Fig. 5e, f), like those observed in the wus-7 mutant. It is possible that the cleavage of WUS by DA1 in the OC strongly reduces the stability of WUS in these transgenic plants, resulting in a wus mutant-like phenotype. Consistent with this, specific expression of DA1pep driven by the WUS promoter did not repress the large SAM phenotype of da1-1 and also did not form distorted SAMs (Fig. 5g–i). These results further established a direct functional relationship between DA1 activity and WUS function.
a Morphologies of da1-1 SAMs. b–d Morphologies of pWUS:DA1; da1-1 SAMs. e, f Morphologies of abnormal SAMs in pWUS:DA1; da1-1. g, h Morphologies of pWUS:DA1pep; da1-1 SAMs(n = 18). i The average SAM area of da1-1, pWUS:DA1; da1-1 and pWUS:DA1pep; da1-1 (n = 18). Data are mean ± s.e.m relative to the da1-1 value (100%). One-way ANOVA with Dunnett’s multiple comparison test was used for statistical analyses (P < 0.05). Scale bars, 20 μm (a–h). All plants were grown for 6 days in long-day conditions. The experiments in a–h were done with similar results in at least two independent replicates. Source data are provided as a Source Data file.
DA1 is involved in cytokinin-induced accumulation of WUS protein and enlargement of the SAM
Cytokinin signaling has been shown to activate the transcription of the WUS gene and increase the stability of WUS protein through post-translational regulation29,33,34. Nonetheless, it remains unclear how cytokinin stabilizes WUS protein due to the lack of an established mechanism for WUS degradation and cleavage. Given that DA1 can cleave and destabilize WUS, we tested whether DA1 is involved in the regulation of cytokinin-promoting stability of WUS protein. We first investigated the effect of cytokinin on DA1 protein level using 35S:GFP-DA1 transgenic plants. We treated 35S:GFP-DA1 plants with or without 6-benzylaminopurine (6-BA) for 8 h because treatment with 6-BA for 8 h did not obviously change the SAM size (Supplementary Fig. 10). Compared with mock-treatment, exogenous application of 6-BA significantly reduced the abundance of GFP-DA1 protein (Fig. 6a, b and Supplementary Fig. 11), while 6-BA did not affect mRNA levels of GFP-DA1 in 35 S:GFP-DA1 (Fig. 6c), indicating that cytokinin signaling can repress DA1 protein levels. We also examined transcription of the DA1 gene in Col-0 when treated with 6-BA for 8 h. Expression level of the DA1 gene in 6-BA treated samples was similar to that in untreated samples (Fig. 6d). These results indicated that cytokinin signaling represses the accumulation of DA1 protein. It is possible that cytokinin represses the accumulation of DA1 protein in the SAM, in turn decreasing WUS cleavage and increasing its accumulation. To test this, we treated pWUS:WUS-GFP seedlings with 6-BA and observed cells with WUS-GFP in the SAMs. When treated with 6-BA for 8 h, cells with WUS-GFP in pWUS:WUS-GFP SAMs were strongly increased, whereas cells with WUS-GFP in the SAMs of pWUS:WUS-GFP; da1-1 were only slightly increased (Fig. 6e–h, m). By contrast, the transcripts of WUS-GFP in pWUS:WUS-GFP and pWUS:WUS-GFP; da1-1 plants were induced by 6-BA in a similar manner (Fig. 6n). Considering that 6-BA treatment for 8 h did not significantly influence the SAM size (Fig. 6e–h and Supplementary Fig. 10), cytokinin-induced accumulation of WUS protein involves DA1 activity.
a, b 6-BA treatment decreases the level of DA1 protein. a Total protein extracts were detected by anti-GFP and anti-ACTIN (loading control) antibodies b Quantification of GFP-DA1 protein levels in a. c, d Quantification of mRNA levels in 35 S:GFP-DA1 or Col-0 treated with or without 6-BA. e SAMs from pWUS:WUS-GFP transgenic plants (n = 14). f SAMs from pWUS:WUS-GFP transgenic plants treated with 6-BA (n = 13). g SAMs from pWUS:WUS-GFP; da1-1 transgenic plants(n = 9). h SAMs from pWUS:WUS-GFP; da1-1 transgenic plants treated with 6-BA (n = 13). i SAMs of 6-day-old Col-0 plants. j SAMs of 6-day-old Col-0 plants treated with 6-BA. k SAMs of 6-day-old da1-1 plants. l SAMs of 6-day-old da1-1 plants treated with 6-BA (n = 30). m The fluorescing cell number of SAMs in pWUS:WUS-GFP (n = 14), pWUS:WUS-GFP + 6-BA (n = 13), pWUS:WUS-GFP; da1-1 (n = 9), and pWUS:WUS-GFP; da1-1+6-BA (n = 13) plants. n Quantification of WUS-GFP mRNA levels. pWUS:WUS-GFP (C), pWUS:WUS-GFP+6-BA (C+6), pWUS:WUS-GFP; da1-1 (d) and pWUS:WUS-GFP; da1-1 + 6-BA (d+6). o The average area of SAMs of Col-0, Col-0 + 6-BA, da1-1 and da1-1 + 6-BA SAMs (n = 30). p A model for cytokinin-DA1-WUS module in controlling the shoot stem cell function and SAM size. DA1 represses WUS to regulate shoot stem cell function and SAM size, while Cytokinin represses DA1 level in the SAM. Cytokinin promotes the WUS accumulation partially through the repression of DA1. The da1-1 mutation caused the WUS accumulation, resulting in the large SAMs with increased stem cell population. Cytokinin also promotes WUS expression. Data in b, c, d and n are mean ± s.e.m with three biological repeats. P values are from two-sided Student’s t tests(b–d) and one-way ANOVA with Tukey’s multiple comparison test (n). Data in m and o are presented as mean values ± s.e.m relative to the mock value (100%). P values are from one-way ANOVA with Tukey’s multiple comparison test (P < 0.05). GFP (green) fluorescence were shown. Scale bars, 20 μm (e–l). The experiments in e–m and o were repeated independently at least twice with similar results. Source data are provided as a Source Data file.
Cytokinin induces the enlargement of the SAM33,44,45. To test whether DA1 is involved in this enlargement, we treated Col-0 and da1-1 seedlings with 6-BA for three days and measured their SAM sizes. The 6-BA treatment increased the SAM size of Col-0 by 43.6%, while the SAM size was increased by 23.5% in da1-1 (Fig. 6i–l, o). This indicated that the reduced DA1 function is partially required for cytokinin-promoting SAM enlargement. Consistent with our results, a previous study showed that cytokinin signaling may also promote the enlargement of SAM partially by influencing mRNA level of WUS28. Taken together, these results revealed that cytokinin promotes SAM enlargement by inducing the expression of the WUS gene and increasing the stability of the WUS protein partially by repressing DA1 accumulation.
Discussion
Plant organs originate from the populations of stem cells in the meristems during post-embryonic development. WUS protein is a central regulator in the maintenance of stem cells in the shoot meristem. The regulation of protein stability is crucial for stem cell function in animals46, but how WUS protein levels are modulated to control plant stem cell function remains unclear. We have previously shown that the peptidase DA1 limits seed and organ growth in Arabidopsis36. In this study, we demonstrated that DA1 physically interacts with and cleaves WUS (Fig. 3), causing its destabilization. Mutations in the peptidase domain of DA1 disrupted the cleavage to WUS, indicating that the peptidase activity of DA1 is required for WUS stability (Fig. 3e, f). The da1-1 seedlings have large shoot apical meristems with an increased stem cell number (Fig. 1). Our biochemical, genetic and gene expression analyses support the conclusion that DA1 and WUS function, at least in part, in a common pathway to control the sizes of shoot apical meristem and stem cell populations (Fig. 2c–e). Although the WUS gene is expressed in the OC, WUS protein can move to neighboring cells to control stem cell function. Our results showed that DA1 is expressed in the whole SAM (Fig. 1c), indicating that DA1 and WUS share overlapped expression domains. Similarly, several other factors that are not specifically expressed in stem cell regions play key roles in stem cell maintenance47,48,49. In addition, specific expression of DA1 driven by the WUS promoter strongly represses the large SAM phenotype of da1-1 (Fig. 5), further supporting their close relationship. Thus, our findings reveal a previously unknown mechanism that the peptidase DA1-mediated cleavage and destabilization of WUS regulates shoot meristem size and shoot stem cell population. Considering that homologs of DA1 and WUS exist in different plant species, it is possible that utilization of DA1 and WUS homologs in different plant species could be a common theme in the regulation of shoot meristem size and plant growth and development. For example, repression of poplar vascular cambial cell division by PagDA1 involves promoting PagWOX4 degradation50. Our results show this likely involves PagWOX4 cleavage by DA1 and subsequent degradation.
Phytohormones are crucial for the maintenance of shoot stem cell homeostasis. Cytokinin signaling induces the transcription of the WUS gene and promotes the stability of WUS protein in the SAM29,33,34. Nonetheless, the underlying mechanism by which cytokinin stabilizes the WUS protein is still unknown due to the lack of an established mechanism for the cleavage and degradation of WUS protein. In this study, we found that cytokinin signaling represses the abundance of DA1 protein in the SAM (Fig. 6a, b and Supplementary Fig. 11). As DA1 can cleave and destabilize WUS, it is possible that cytokinin signaling promotes the accumulation of WUS protein partially by repressing DA1 protein. Supporting this notion, exogenous cytokinin treatment experiments showed that DA1 is involved in the regulation of cytokinin-induced accumulation of WUS protein and enlargement of the SAM (Fig. 6). Therefore, our findings define a regulatory mechanism in which the cleavage of WUS by DA1 connects cytokinin signaling and shoot stem cell number and meristem size regulation in Arabidopsis (Fig. 6p). However, we cannot rule out the possibility that CK could also affect WUS stability through another unknown mechanism as CK treatment can still induce the accumulation of the WUS protein and the enlargement of the SAM in da1-1, although the ratio of induction in da1-1 was less than that in Col-0 (Fig. 6). In addition, it remains unclear how cytokinin signaling regulates DA1 protein levels. Considering that the phosphorylation of DA1 influences its stability51, it will be a worthwhile challenge to investigate whether cytokinin receptors could interact with and phosphorylate DA1 in the future. A previous study showed that light and metabolic signals are able to activate the expression of the WUS gene, which also likely involves cytokinin signaling52. It will be interesting to examine whether the cytokinin-DA1-WUS module could mediate environmental cues (light and nutrients) to regulate stem cell function.
Larger meristems with an increased stem cell population hold promise for increasing yield in different plant species9,10,11. Our findings place DA1 at the crucial position of organ size, meristem size, and stem cell population regulation. Given that mutations in DA1 increase seed size in several crops53,54,55, the regulation of fundamental stem cell proliferation by the DA1-WUS module has the potential to improve crop yield.
Methods
Plant materials and growth conditions
All the plant materials were used in Col-0 or Ler background as indicated. The da1-1, da1-1Ler, da1-ko1 dar1-1, DA1COM, 35S:GFP-DA1, wus-7 and pWUS:WUS-GFP lines were used in previous studies35,36,41,49,56,57,58. The seeds of clv1-1 (NW45), clv2-1 (NW46), clv3-2 (N8066) and wus-1 (N15), were obtained from the Nottingham Arabidopsis Stock Centre (NASC). The da1-1Ler clv1-1, da1-1Ler clv2-1, da1-1Ler clv3-2, da1-1Ler wus-1 and da1-1Ler wus-7 double mutants were generated by crossing da1-1Ler with clv1-1, clv2-1, clv3-2, wus-1 and wus-7, respectively. The pWUS:WUS-GFP; da1-1 was generated by crossing da1-1 with pWUS:WUS-GFP. The pWUS:WUS-GFP; MYC-DA1-PER8 was generated by crossing pWUS:WUS-GFP with MYC-DA1-PER8. All these mutants and double mutants were identified by allele-specific genotyping, transgenic antibiotic resistance screening or GFP signal detection. The primers used above were listed in Supplementary Data 1.
Seeds were sterilized in 100% isopropanol for 1 min and then 5%(v/v) sodium hypochlorite for 10 min. After sufficiently washed with sterile water, seeds were dispersed on 1/2 MS medium containing 1% (w/v) Suc with 0.9% agar and placed at 4°C for 3d. For long-day condition, plants were grown in controlled environmental chamber or in green house with 16 h light /8 h dark. For β -estradiol treatment, plants were grown for 7 days in 1/2MS solid medium and transferred to 1/2MS solid medium with 20 μM β -estradiol for 12 hours.
Confocal microscope observation
For SAM fluorescence signals detection, the shoot apices were detached and prepared from pWUS:WUS-GFP, pWUS:WUS-GFP; da1-1, and pWUS:WUS-GFP; MYC-DA1-PER8 grown under long day condition (16 h light/ 8 h dark) according to a previous study7. Samples were observed using a Zeiss LSM 980 confocal microscopy, and the images used for fluorescencing cells counting were taken by meristem confocal sections at suitable location. All the cell numbers with fluorescence signaling were then quantified by Image J.
Differential interference contrast microscope
For shoot meristem size observation, 6-d-old and 9-d-old plants were harvested and cleared in clearing solution (Chloral hydrate 80 g, water 30 ml, Glycerol 10 ml) for 12 hrs. Samples were observed with DIC microscope (Leica DM2500, Germany) and photographed using cooled CCD digital imaging system (Olympus BH2, Japan).
SAM size measurement
For SAM size measurement, the measurement range takes L1 layer as the boundary and the junction of SAM and primordia on both sides as the starting points. The details can be found in Supplementary Fig. 3. SAMs were measured using Image J.
Scanning electron microscopy
Samples were fixed in FAA solution with vacuum treatment for 30 min. Fixed samples were dehydrated with a gradual ethanol as 70%, 85%, 95%, and 100% (v/ v), then dried by critical-point drying (Hitachi HCP-2), and subsequently coated with a gold layer. The sputter-coated samples were ready for observation and imaging using scanning electron microscope (Hitachi S-3000N).
Quantitative real-time PCR analysis
6-d-old aboveground part of Col-0 and da1-1 were ground in liquid nitrogen, and the RNAprep pure kit was used to extract total RNA following the manufacturer’s instructions (TIANGEN, Cat. No: DP439-H). cDNAs were synthesized using the FastQuant RT Super Mix kit (TIANGEN, Cat. No: KR108). qPCR reactions were assayed on a Lightcycler 480 machine (Roche Applied Science, USA) using 2×SYBR Green supermix kit (Vazyme, China). All individual reactions were done by three biological replicates, and the data were normalized to the ACTIN2 gene. All primers were listed in Supplementary Data 1.
Yeast two-hybrid assay
The Matchmaker Gold Yeast Two-Hybrid System was employed to perform the yeast two-hybrid assay in yeast strain AH109. AD-DA1 and DA1-LIM + C-BD were used in previous studies4. The CDSs of WUS amplified using primers WUS-BD-F/R (Supplementary Data 1) was inserted into pGBKT7 vector (NcoI and PstI digestion) to get BD-WUS constructs using GBclonart Seamless Clone Kit (GB2001-48, Genebank Biosciences). The CDSs of WUS-HD, WUS-DD, WUS-C and WUS-C2 were inserted into pGADT7 vector (ECOR1 and SAC1 digestion) to get AD-WUS-HD, AD-WUS-DD, AD-WUS-C, and AD-WUS-C2 constructs. Primers are listed in the Supplementary Data 1. The Different combinations of plasmids as indicated were co-transformed in AH109 and grown on SD/-Trp/-Leu plates for 3 days. The protein interactions were then selected by spotting the yeast on SD medium with minus Trp/Leu/His/Ade and grown for 3 or more days.
In vivo co-immunoprecipitation
The genomic sequence of WUS was inserted into PMDC32 (in which the 35 S promotor was cleaved) to generate pWUS:WUS-HA. The construct primers are listed in the supplemental Data 1. The respective transgenic Arabidopsis plants of 35 S:GFP-DA1 and pWUS:WUS-HA were obtained by agrobacterium tumefaciens-mediated transformation. pWUS:WUS-HA; 35S:GFP-DA1; and pWUS:WUS-HA; 35S:GFP plants came from crossing pWUS-WUS-HA with 35S:GFP-DA1 and 35S:GFP, respectively.
Aboveground part of pWUS:WUS-HA;35 S:GFP-DA1, and pWUS:WUS-HA;35S:GFP 10-d-old plants were ground in liquid nitrogen. Total proteins were isolated with buffer (50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1% Triton X-100, 5% glycerol, 1 mM EDTA, 1× Roche protease inhibitor cocktail) and incubated with GFP-Trap®_A agarose beads (Chromotek, Cat. No: gta-20) for 1 h with agitation at 4 °C. Beads were washed four times with wash buffer (50 mM Tris [pH 7.5], 150 mM NaCl, 10% glycerol, 0.1% TritonX-100, 1 mM EDTA, and protease inhibitor cocktail). After adding 1×SDS loading buffer, the beads were heated at 98 °C for 5 min, and the corresponding proteins were subjected to sepatate in a 10% or 8% (w/ v) SDS-polyacrylamide gel, and detected with anti-GFP antibody (Abmart, Cat. No: M20004, dilution, 1:5000) and anti-HA antibody (Cwbio, Cat. No: 01239/34220, dilution, 1:5000).
In vitro Pull-down assay
All the constructs were made by infusion cloning kit (GBclonart Seamless Clone Kit, GB2001-48, Genebank Biosciences). The CDS of WUS was infused into pMALC2-MBP digested with SalI and HindIII to generate the MBP-WUS. The CDSs of DA1 was cloned into the pGEX4T-1 vector digested with ECORI and SALI to generate the GST-DA1. Plasmids were transferred into E. coli BL21 (DE3) cells to expression the tag fused proteins. All proteins were induced with 0.4 mM Isopropyl β -D-1-thiogalactopyranoside (IPTG) at different conditions depending on proteins. The bacterial cells were then resuspended with TGH buffer (50 mM HEPES (pH 7.5), 1 mM EGTA, 150 mM NaCl, 1% (v/v) Triton X-100 and 10%(v/v) glycerol). The bacterial suspensions were sonicated on ice for 3 min at 20 amplitudes and centrifugated at 12,000×g for 10 min. The different combinations of MBP-WUS and GST-DA1 proteins with MBP agarose beads (NEB, Cat. No: E8037s) were incubated for 1 h at 4 °C with agitation, respectively. Beads were washed 5 times with washing buffer (50 mM HEPES (pH 7.5), 1 mM EGTA, 150 mM NaCl, 0.5% (v/v) TritonX-100, 1 mM PMSF and 10% (v/v) glycerol). After adding 1×SDS-loading buffer, the beads were heated 5 min at 98 °C and separated by SDS-PAGE gel. The immunoprecipitates were detected by GST antibody (Abmart, Cat. No: M20007M, 1:5000) and MBP antibody (NEB, Cat. No: #E8032, 1:10,000).
Bimolecular fluorescence complementation assay
The CDS of DA1 was amplified by specific primers YN-DA1-F/R, fused with the N-terminal fragment of YFP (nYFP), and subcloned into the linearized pGWB414 vector (digested with XbaI and SalI) using in-fusion enzyme (Genebank Biosciences). The CDS of WUS was amplified by specific primers YC-WUS-F/R, fused with the C-terminal fragment of YFP (cYFP), and subcloned into the linearized pGWB414 vector (digested with XbaI and SalI) using in-fusion enzyme (Genebank Biosciences). Primers used are listed in the supplemental information (Supplementary Data 1). Different combinations of Agrobacterium GV3101 containing the above plasmids were cotransformed into N. benthamiana leaves. After 48 h, YFP fluorescence was observed in leaves using an LSM710 confocal laser scanning microscope (Zeiss)
Cleavage of WUS
The 35 S:HA-DA1 and 35 S:HA-DA1pep constructs were used in previous studies35,59. The cDNA sequences of WUS was inserted into pW1211 and pW1266 to generate 35 S:WUS-FLAG and 35 S:FLAG-WUS by gateway cloning (Invitrogen), respectively. Primers are listed in the Supplementary Data 1.
The rosettes of da1-kol dar1-1 were harvested before bolting to prepare protoplast. The ~0.5 mm fragments of leaves were incubated in enzyme solution (0.3% Macerozyme R-10, 10 mM CaCl2, 0.4 M mannitol, 1.25% Cellulose RS, 20 mM MES at pH 5.7, 5 mM β-Mercatoethanol and 0.1% BSA) for 4 hrs in dark with gentle agitation. After digestion, the W5 solution (154 mM NaCl, 5 mM KCl, 2 mM MES pH 5.7 and 125 mM CaCl2) was added, and followed 10–30 s with strongly shaking. By filtering through 40 μm nylon mesh with three to five washes using W5 solution, the protoplasts were collected by centrifugation at 100 × g for 5 min. The protoplasts were washed twice with W5 buffer and resuspended in MMG solution (0.4 M mannitol, 15 mM MgCl2, and 4 mM MES pH 5.7) around 2× 106 cells mL−1. The 35S:HA-DA1, 35S:HA-DA1pep, 35S:WUS-FLAG, and 35S:FLAG-WUS plasmids were extracted with Plasmid Maxprep Kit (Vigorous, Cat. No: N001). The combinations of constructs (10 μg) were added into 300 μL protoplasts with freshly prepared 330 μL polyethylene glycol (PEG) solution (0.1 M CaCl2, 0.4 M mannitol, 40% w/v PEG4000), and incubated for 20 min on bench. The transfected protoplasts were then resuspended in W5 solution and incubated for 12-16 hrs at 28 °C in the dark. Total proteins were isolated with extraction buffer (50 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.1% Triton, 0.2% NP-40, 5 mM EDTA, and protease inhibitor cocktail). Proteins were detected by western blots with antibody against FLAG (Abmart, Cat. No: M20008, 1:5000) and HA (Cwbio, Cat. No: 01239/34220, dilution, 1:5000).
The effect of 6-BA treatment on meristem size
For 6-benzylaminopurine (6-BA) treatment, 100 μM stock solutions of 6-BA (Sigma-Aldrich) in ddH2O were dissolved in 1/2MS medium containing 1% (w/v) Suc with 0.9% agar prewarmed at 50 °C to a final concentration of 100 nM. For SAM size measurement, seedlings were grown on 1/2MS solid medium in long-day conditions (16 h light and 8 h dark at 22 °C) for 3 days, then transferred to 1/2MS solid medium with 100 nM 6-BA for 3 days or 8.67 days after seed stratification and then transferred to 1/2MS solid medium with 100 nM 6-BA for 8 h.
Statistics analysis
All data are shown as the mean ± s.e.m. unless indicated otherwise. Statistical analysis was performed using GraphPad Prism 7 software (GraphPad Software, Inc. San Diego, CA, USA). All details on statistics have been indicated in figure legends. The exact P-value was included in the figures. No statistical method was used to predetermine sample size. Images were analyzed with ImageJ.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All materials in this study are available from the corresponding author upon request. The authors declare that all data supporting the findings of this study are available within the article and its Supplementary Information files. Arabidopsis reference genome (TAIR10) was used in this study. Source data are provided with this paper.
References
Scheres, B. Stem-cell niches: nursery rhymes across kingdoms. Nat. Rev. Mol. Cell Biol. 8, 345–354 (2007).
Dinneny, J. R. & Benfey, P. N. Plant stem cell niches: standing the test of time. Cell 132, 553–557 (2008).
Weigel, D. & Jurgens, G. Stem cells that make stems. Nature 415, 751–754 (2002).
Jurgens, G. Apical-basal pattern formation in Arabidopsis embryogenesis. EMBO J. 20, 3609–3616 (2001).
Satina, S., Blakeslee, A. F. & Avery, A. G. Demonstration of the three germ layers in the shoot apex of Datura by means of induced polyploidy in periclinal chimeras. Am. J. Bot. 27, 895–905 (1940).
Aichinger, E., Kornet, N., Friedrich, T. & Laux, T. Plant stem cell niches. Annu. Rev. Plant. Biol. 63, 615–636 (2012).
Mayer, K. F. et al. Role of WUSCHEL in regulating stem cell fate in the Arabidopsis shoot meristem. Cell 95, 805–815 (1998).
Laufs, P., Grandjean, O., Jonak, C., Kieu, K. & Traas, J. Cellular parameters of the shoot apical meristem in Arabidopsis. Plant Cell 10, 1375–1390 (1998).
Hu, J. et al. The elite alleles of OsSPL4 regulate grain size and increase grain yield in rice. Rice 14, 90 (2021).
Rodriguez-Leal, D., Lemmon, Z. H., Man, J., Bartlett, M. E. & Lippman, Z. B. Engineering quantitative trait variation for crop improvement by genome editing. Cell 171, 470–480.e478 (2017).
Bommert, P., Nagasawa, N. S. & Jackson, D. Quantitative variation in maize kernel row number is controlled by the FASCIATED EAR2 locus. Nat. Genet. 45, 334–337 (2013).
Laux, T., Mayer, K. F. X., Berger, J. & Jurgens, G. The WUSCHEL gene is required for shoot and floral meristem integrity in Arabidopsis. Development 122, 87–96 (1996).
Gallois, J. L., Woodward, C., Reddy, G. V. & Sablowski, R. Combined SHOOT MERISTEMLESS and WUSCHEL trigger ectopic organogenesis in Arabidopsis. Development 129, 3207–3217 (2002).
Yadav, R. K., Tavakkoli, M. & Reddy, G. V. WUSCHEL mediates stem cell homeostasis by regulating stem cell number and patterns of cell division and differentiation of stem cell progenitors. Development 137, 3581–3589 (2010).
Fletcher, L. C., Brand, U., Running, M. P., Simon, R. & Meyerowitz, E. M. Signaling of cell fate decisions by CLAVATA3 in Arabidopsis shoot meristems. Science 283, 1911–1914 (1999).
Brand, U., Fletcher, J. C., Hobe, M., Meyerowitz, E. M. & Simon, R. Dependence of stem cell fate in Arabidopsis on a feedback loop regulated by CLV3 activity. Science 289, 617–619 (2000).
Rojo, E., Sharma, V. K., Kovaleva, V., Raikhel, N. V. & Fletcher, J. C. CLV3 is localized to the extracellular space, where it activates the Arabidopsis CLAVATA stem cell signaling pathway. Plant Cell 14, 969–977 (2002).
Ogawa, M., Shinohara, H., Sakagami, Y. & Matsubayashi, Y. Arabidopsis CLV3 peptide directly binds CLV1 ectodomain. Science 319, 294 (2008).
Kayes, J. M. & Clark, S. E. CLAVATA2, a regulator of meristem and organ development in Arabidopsis. Development 125, 3843–3851 (1998).
Clark, S. E., Running, M. P. & Meyerowitz, E. M. Clavata3 is a specific regulator of shoot and floral meristem development affecting the same processes as Clavata1. Development 121, 2057–2067 (1995).
Clark, S. E., Running, M. P. & Meyerowitz, E. M. Clavata1, a regulator of meristem and flower development in Arabidopsis. Development 119, 397–418 (1993).
Yadav, R. K. et al. WUSCHEL protein movement mediates stem cell homeostasis in the Arabidopsis shoot apex. Gene Dev. 25, 2025–2030 (2011).
Daum, G., Medzihradszky, A., Suzaki, T. & Lohmann, J. U. A mechanistic framework for noncell autonomous stem cell induction in Arabidopsis. Proc. Natl Acad. Sci. USA 111, 14619–14624 (2014).
Schoof, H. et al. The stem cell population of Arabidopsis shoot meristems is maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100, 635–644 (2000).
Zhou, Y. et al. Control of plant stem cell function by conserved interacting transcriptional regulators. Nature 517, 377–380 (2015).
Zhou, Y. et al. HAIRY MERISTEM with WUSCHEL confines CLAVATA3 expression to the outer apical meristem layers. Science 361, 502–506 (2018).
Su, Y. H. et al. Integration of pluripotency pathways regulates stem cell maintenance in the Arabidopsis shoot meristem. Proc. Natl Acad. Sci. USA 117, 22561–22571 (2020).
Meng, W. J. et al. Type-B ARABIDOPSIS RESPONSE REGULATORs specify the shoot stem cell niche by dual regulation of WUSCHEL. Plant Cell 29, 1357–1372 (2017).
Snipes, S. A. et al. Cytokinin stabilizes WUSCHEL by acting on the protein domains required for nuclear enrichment and transcription. PLoS Genet. 14, e1007351 (2018).
Zhao, Z. et al. Hormonal control of the shoot stem-cell niche. Nature 465, 1089–1092 (2010).
Luo, L. J., Zeng, J., Wu, H. J., Tian, Z. X. & Zhao, Z. A molecular framework for auxin-controlled homeostasis of shoot stem cells in Arabidopsis. Mol Plant 11, 899–913 (2018).
Zubo, Y. O. et al. Cytokinin induces genome-wide binding of the type-B response regulator ARR10 to regulate growth and development in Arabidopsis. Proc. Natl Acad. Sci. USA 114, E5995–E6004 (2017).
Gordon, S. P., Chickarmane, V. S., Ohno, C. & Meyerowitz, E. M. Multiple feedback loops through cytokinin signaling control stem cell number within the Arabidopsis shoot meristem. Proc. Natl Acad. Sci. USA 106, 16529–16534 (2009).
Leibfried, A. et al. WUSCHEL controls meristem function by direct regulation of cytokinin-inducible response regulators. Nature 438, 1172–1175 (2005).
Dong, H. et al. Ubiquitylation activates a peptidase that promotes cleavage and destabilization of its activating E3 ligases and diverse growth regulatory proteins to limit cell proliferation in Arabidopsis. Gene Dev. 31, 197–208 (2017).
Li, Y., Zheng, L., Corke, F., Smith, C. & Bevan, M. W. Control of final seed and organ size by the DA1 gene family in Arabidopsis thaliana. Genes Dev. 22, 1331–1336 (2008).
Kitagawa, M. & Jackson, D. Control of meristem size. Annu. Rev. Plant Biol. 70, 269–291 (2019).
Vanhaeren, H. et al. UBP12 and UBP13 negatively regulate the activity of the ubiquitin-dependent peptidases DA1, DAR1 and DAR2. Elife https://doi.org/10.7554/eLife.52276 (2020).
Perez-Perez, J. M., Candela, H. & Micol, J. L. Understanding synergy in genetic interactions. Trends Genet. 25, 368–376 (2009).
Du, L. et al. The ubiquitin receptor DA1 regulates seed and organ size by modulating the stability of the ubiquitin-specific protease UBP15/SOD2 in Arabidopsis. Plant Cell 26, 665–677 (2014).
Peng, Y. et al. The ubiquitin receptors DA1, DAR1, and DAR2 redundantly regulate endoreduplication by modulating the stability of TCP14/15 in Arabidopsis. Plant Cell 27, 649–662 (2015).
Busch, W. et al. Transcriptional control of a plant stem cell niche. Dev. Cell 18, 849–861 (2010).
Jia, T., Li, F., Liu, S., Dou, J. & Huang, T. DnaJ proteins regulate wus expression in shoot apical meristem of Arabidopsis. Plants (Basel) https://doi.org/10.3390/plants10010136 (2021).
Yang, W. et al. Molecular mechanism of cytokinin-activated cell division in Arabidopsis. Science 371, 1350–1355 (2021).
Chickarmane, V. S., Gordon, S. P., Tarr, P. T., Heisler, M. G. & Meyerowitz, E. M. Cytokinin signaling as a positional cue for patterning the apical-basal axis of the growing Arabidopsis shoot meristem. Proc Natl Acad Sci USA 109, 4002–4007 (2012).
Strikoudis, A., Guillamot, M. & Aifantis, I. Regulation of stem cell function by protein ubiquitylation. EMBO Rep. 15, 365–382 (2014).
Han, P., Li, Q. & Zhu, Y. X. Mutation of Arabidopsis BARD1 causes meristem defects by failing to confine WUSCHEL expression to the organizing center. Plant Cell 20, 1482–1493 (2008).
Zhang, Y., Jiao, Y., Liu, Z. & Zhu, Y. X. ROW1 maintains quiescent centre identity by confining WOX5 expression to specific cells. Nat. Commun. 6, 6003 (2015).
Zhai, H. et al. SEUSS integrates transcriptional and epigenetic control of root stem cell organizer specification. EMBO J. 39, e105047 (2020).
Tang, X. et al. Ubiquitinated DA1 negatively regulates vascular cambium activity through modulating the stability of WOX4 in Populus. Plant Cell 34, 3364–3382 (2022).
Wu, X. et al. ERECTA regulates seed size independently of its intracellular domain via MAPK-DA1-UBP15 signaling. Plant Cell 34, 3773–3789 (2022).
Pfeiffer, A. et al. Integration of light and metabolic signals for stem cell activation at the shoot apical meristem. eLife https://doi.org/10.7554/eLife.17023 (2016).
Liu, H. et al. TaDA1, a conserved negative regulator of kernel size, has an additive effect with TaGW2 in common wheat (Triticum aestivum L).Plant Biotechnol J 18, 1330–1342 (2020).
Xie, G., Li, Z., Ran, Q., Wang, H. & Zhang, J. Over-expression of mutated ZmDA1 or ZmDAR1 gene improves maize kernel yield by enhancing starch synthesis. Plant Biotechnol. J. 16, 234–244 (2018).
Wang, J. L. et al. Down-regulation of BnDA1, whose gene locus is associated with the seeds weight, improves the seeds weight and organ size in Brassica napus. Plant Biotechnol. J. 15, 1024–1033 (2017).
Wu, H. et al. WUSCHEL triggers innate antiviral immunity in plant stem cells. Science 370, 227–231 (2020).
Ma, Y. et al. WUSCHEL acts as an auxin response rheostat to maintain apical stem cells in Arabidopsis. Nat. Commun. 10, 5093 (2019).
Ding, Z. & Friml, J. Auxin regulates distal stem cell differentiation in Arabidopsis roots. Proc. Natl Acad. Sci. USA 107, 12046–12051 (2010).
Xia, T. et al. The ubiquitin receptor DA1 interacts with the E3 ubiquitin ligase DA2 to regulate seed and organ size in Arabidopsis. Plant Cell 25, 3347–3359 (2013).
Acknowledgements
We would like to thank Drs. Thomas Laux, Chuanyou Li, Yuxin Hu, Zhong Zhao, Zhaojun Ding, and Xigang Liu for mutant and transgenic seeds. We also thank NASC for wus-1, clv1-1, clv2-1 and clv3-2 seeds. We thank Dr. Yuling Jiao for helping WUS-GFP observation. This work was supported by the grants from the National Natural Science Foundation of China (32370357, 31872663 and 31961133001) and the strategic priority research program of the Chinese Academy of Sciences (XDB27010102).
Author information
Authors and Affiliations
Contributions
YH.Li conceived and designed this project. GC.Cui and YU.Li performed most experiments. C.S performed the cleavage of WUS. LY.Zheng, YU.Li, GC.Cui, M.B, and YH.Li analyzed data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cui, G., Li, Y., Zheng, L. et al. The peptidase DA1 cleaves and destabilizes WUSCHEL to control shoot apical meristem size. Nat Commun 15, 4627 (2024). https://doi.org/10.1038/s41467-024-48361-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-48361-7
- Springer Nature Limited