Abstract
Carbo- and heterocycles are frequently used as crucial scaffolds in natural products, fine chemicals, and biologically and pharmaceutically active compounds. Transition-metal-catalyzed cyclization of 1,6-enynes has emerged as a powerful strategy for constructing functionalized carbo- and heterocycles. Despite significant progress, the regioselectivity of alkyne functionalization is entirely substrate-dependent. And only exo-cyclization/cross-coupling products can be obtained, while endo-selective cyclization/cross-coupling remains elusive and still poses a formidable challenge. In this study, we disclose a nickel-catalyzed switchable arylation/cyclization of 1,6-enynes in which the nature of the ligand dictates the regioselectivity of alkyne arylation, while the electrophilic trapping reagents determine the selectivity of the cyclization mode. Specifically, using a commercially available 1,10-phenanthroline as a ligand facilitates trans-arylation/cyclization to obtain seven-membered ring products, while a 2-naphthyl-substituted bisbox ligand promotes cis-arylation/cyclization to access six-membered ring products. Diastereoselective cyclizations have also been developed for the synthesis of enantioenriched piperidines and azepanes, which are core structural elements of pharmaceuticals and natural products possessing important biological activities. Furthermore, experimental and density functional theory studies reveal that the regioselectivity of the alkyne arylation process is entirely controlled by the steric hindrance of the ligand; the reaction mechanism involves exo-cyclization followed by Dowd-Beckwith-type ring expansion to form endo-cyclization products.
Similar content being viewed by others
Introduction
The structural diversity of core molecular scaffolds is considered a critical feature of compound libraries, which is decisive for ensuring their success in drug discovery and biological research1,2,3. Compound libraries constructed by traditional synthetic strategies are mainly composed of molecules with similar structures. This is because the structural diversity of the products depends only on the starting scaffolds and building blocks. Therefore, the development of new strategies to efficiently construct structurally distinct and diverse molecular scaffolds, thereby giving rise to compound libraries with differing and selective biological activities is of great siginificance4,5. Ligand-directed divergent synthesis (LDS) is highly sought-after in library synthesis because it allows the construction of structurally diverse molecular scaffolds from common starting materials by simply changing the ligands of metal catalysts6. However, it is undoubtedly the most challenging strategy because ligand design alone requires subversion of the inherent regiopreferences of reactants and metal centers.
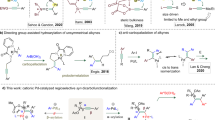
Cyclic scaffolds, especially heterocycles, are ubiquitous in natural products, pharmaceuticals, and numerous functional molecules. The development of effective methods for the rapid construction of various cyclic compounds represents one of the major domains of synthetic organic chemistry. In this regard, transition-metal-catalyzed cycloisomerization7,8,9,10,11 and reductive cyclization12,13,14,15,16 of 1,6-enynes have been established to generate five-membered (hetero)cycles. Advances along this line have revealed some intriguing transition metal-catalyzed functionalizing cyclization reactions, providing efficient and convenient access to a wide array of elusive cyclic systems17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34. The appeal of these cyclizations lies in the streamlining construction of structurally complex and functionalized cyclic scaffolds from readily accessible 1,6-enynes. From a reaction mechanism perspective, these transformations are primarily initiated by the formation of metalacyclic intermediates via oxidative cyclization pathways or the formation of alkenyl metal species via carbometallation processes. The former has been well explored7,8,9,10,11,12,13,14,15,16, but controlling the regioselectivity of the latter is difficult, usually in two ways: α-insertion or β-insertion (Fig. 1a)35,36,37,38. 5-Exo-cyclization of the resulting alkenyl-metal intermediate I (via β-insertion) would provide the conventional five-membered ring product III bearing an exocyclic double bond. Alternatively, α-insertion followed by E/Z isomerization39,40,41,42,43,44 would afford alkenyl-metal intermediate II, which undergoes 6-exo cyclization to afford six-membered ring product V possessing an endocyclic double bond. Despite enormous efforts, these strategies still suffer from many limitations: the regioselectivity of alkynes insertion into organometallic species is entirely substrate-dependent (electronic or steric effects of substituents on alkynes). Furthermore, since exo-cyclizations are anyway kinetically superior to the corresponding endo-cyclizations, 6-endo- or 7-endo-cyclization products are rarely obtained45,46,47,48.
Nickel-catalyzed reductive cross-coupling has been emerged as a powerful and straightforward method for constructing carbon-carbon bonds49,50,51,52,53,54. Furthermore, due to its unique properties, nickel has been used in a range of ligand-controlled divergent transformations55,56,57. To address the major remaining challenges of 1,6-enyne cyclization, we were curious whether changing the structure and/or electronegativity of the ligands in nickel catalysts would enable regioselective and switchable arylation of alkynes. Here, we demonstrate that judicious choice of ligands enables the switchable arylation/ cyclization of 1,6-enynes, in which the nature of the ligand dictates the regioselectivity of alkyne arylation, while the electrophilic trapping reagents determine the selectivity of the cyclization mode. Specifically, in the presence of electron-deficient alkenes, using a commercially available 1,10-phenanthroline as a ligand facilitates trans-arylation/endo-cyclization to obtain seven-membered ring products, while a 2-naphthyl-substituted Bisbox ligand promotes cis-arylation/endo-cyclization to access six-membered ring products (Fig. 1b). Diastereoselective cyclizations have also been developed for the synthesis of enantioenriched piperidines and azepanes, which are core structural elements of pharmaceuticals and natural products possessing important biological activities. This transformation is modular, with the ligand rather than the substrate precisely determining the regioselectivity of alkyne arylation process, and the electrophilic trapping reagents determining the regioselectivity of cyclization. Such ligand-directed divergent synthesis (LDS) remains hitherto elusive despite persistent interest.
Results and discussion
Reaction development
To test our hypothesis, we started our investigation by evaluating the nickel-catalyzed three-component cascade cyclization of 1,6-enyne (1) with PhBr (2) and trifluoromethyl alkene (3) (Table 1). After a series of experiments, a combination of NiBr2(DME), 1,10-phenanthroline L4, Mn, CsF, and DMA at 60 °C afforded gem-difluoroallylated seven-membered ring product 4 in 63% isolated yield with exquisite regioselectivity (4/5 = 20/1) (entry 1). Inferior results were observed when either TBAI or NaI was used instead of CsF (entries 2 and 3). Lowering the reaction temperature to 40 °C resulted in no reaction, while increasing the reaction temperature to 80 °C resulted in lower yield (entries 4 and 5). Traces, if any, of 4 were observed when Zn was used instead of Mn as the terminal reducing agent (entry 6). Notably, the direct reductive coupling of aryl bromides and trifluoromethyl alkenes in this catalytic system is well suppressed.
We evaluated a range of different ligands with the goal of identifying the conditions for switchable access to the seven-membered ring product 4 and the six-membered ring product 5. 2,2′-Bipyridine (L1-L3) and 1,10-phenanthroline (L4) favored the formation of 4, especially the latter providing 4 in higher yield and excellent regioselectivity (entries 7–10). Strikingly, under the same reaction conditions as shown in entry 10, the use of relatively electron-rich Pyrox L5 or Bisbox L6 as ligands resulted in a regioselective switch en route to 5, albeit in lower yields (entries 11 and 12). We further synthesized a series of Bisbox ligands with different substituents on the oxazoline ring and examined their performances. The phenyl-substituted ligand L7 showed slightly higher regioselectivity (4/5 = 1/8, entry 13). The benzyl-substituted ligand L8 provided 5 in 58% yield and improved the regioselectivity to 1:15 (entry 14). Using a 2-naphthyl-substituted Bisbox ligand L9, 5 was obtained in 71% isolated yield and excellent regioselectivity (4/5 = 1:28) (entry 15). These results clearly demonstrate that the backbone of the ligand determines the regioselectivity pattern.
Having identified optimal reaction conditions for the ligand-controlled divergent synthesis of seven- and six-membered ring products, we turned our attention to investigating the scope and limitations of those two protocols. We first explored the trans-arylative/endo cyclization protocol for the selective synthesis of seven-membered ring products using ligand L4 (Fig. 2). In terms of 1,6-enynes, the carbon-tethered and nitrogen-tethered 1,6-enynes were examined, and the corresponding products 6 and 7 were obtained as expected. The molecular structure of 7 was unambiguously confirmed by single-crystal X-ray diffraction analysis. Electron-donating (OMe) or electron-withdrawing groups (CF3, CN) on the aromatic ring at the alkyne terminal (R1) were found to be compatible, leading to the corresponding products 8-10 in 49-69% yields. Notably, chlorine substitution on the aromatic ring also proceeded smoothly, affording 11 in 68% yield, providing an opportunity for further derivatization of the obtained product. Complex molecules such as estrone could also be successfully incorporated into product (12). Nonetheless, alkyl-substituted alkynes and terminal alkynes failed to produce the expected products. Introduction of a phenyl group on the alkene moiety did not preclude the transformation (13).
We then evaluated the scope of aryl (pseudo)halides. We were pleased to find that the presence of electron-donating and electron-withdrawing groups at the para or meta position of the aromatic ring did not affect selectivity and efficiency (14–23). Aryl triflate was also tolerated as shown by the efficient formation of product 15. Heteroaryl bromides such as 5-bromo-2-methoxypyridine, 3-bromothiophene, 2-bromodibenzofuran, 2-bromodibenzothiophene, 5-bromobenzofuran, 5-bromoindole, and 3-bromocarbazole were all efficiently transformed into the corresponding products (24–30) in satisfactory yields.
The generality of the electron-deficient alkenes was also investigated. Trifluoromethyl alkenes with various useful functional groups, such as cyano, ester, amide, sulfonate, trifluoromethoxy, chloro, and alkoxy on the aromatic ring, could be successfully transferred to the corresponding products (31–40). In addition, quinoline, benzothiophene and carbazole-substituted trifluoromethyl alkenes (41, 42 and 43) were also compatible with the reaction conditions. The seven-membered ring product 44 and 45 were also obtained under the same reaction conditions when electron-deficient alkenes such as acrylate or vinyl sulfone were used respectively.
We next examined the scope of the cis-arylative/endo cyclization protocol for the selective synthesis of six-membered ring products using ligand L9 (Fig. 3). Similar to what we observed with the trans-arylative/endo cyclization, C- or N-tethered 1,6-enynes had little effect on the reactivity and selectivity, providing the corresponding products (46-47) in 58–63% yields. 1,6-Enynes with substituents on the alkene moiety such as phenyl and ester were also compatible (48-49). Both electron-withdrawing (Cl) and electron-donating groups (OMe) on the aromatic ring at the alkyne terminal (R1) were found to be compatible (50-51).
Aryl (pseudo)halides with versatile functional groups were then examined. It is noteworthy to mention that substrates with both electron-donating and withdrawing groups proceed smoothly to afford the corresponding products (52-58) in moderate to good yields. In particular, the p-chlorine substituted product 54 and the 3-bromocarbazole substituted product 59 were both isolated in good yields.
In addition, the compatibility of the transformation with various trifluoromethyl alkenes was evaluated in a robustness screening. A series of valuable functional groups such as cyano, ester, amide, sulfonate, ketone, trifluoromethoxy, chloro, and alkoxy were all well-tolerated (60–69). Trifluoromethyl alkenes bearing heterocyclic groups could also be transformed into the desired products 70–73 in moderate yields. In the case of the trifluoromethylalkenes with a strong electron-donating group, the desired defluorinative products 68–72 were isolated in a mixture with the reductive Heck coupling products which retained the trifluoromethyl group.
Enantiomerically pure N-heterocyclic compounds, especially piperidines and azepanes, represent important motifs in pharmaceutically important natural alkaloids58. Consequently, it is of great significant to develop reliable synthetic methods for the synthesis of such heterocycles. We screened a series of chiral ligands in an attempt to render the reaction asymmetric (Supplementary Table 1). Simultaneously controlling the regioselectivity of alkyne migratory insertion and the stereoselectivity of the cyclization process is indeed a very challenging task, and all our attempts resulted in only racemic products.
We turned to developing a solution to obtain enantioenriched piperidines and azepanes by diastereoselective cyclization of substrates with pre-existing stereocenters59,60. Enantioenriched 1,6-enyne 74 (e.r. > 99:1) was prepared employing Ellman’s auxiliary61 to test subsequent ligand-controlled arylative cyclization for the divergent synthesis of chiral piperidines and azepanes (Fig. 4a). Excitingly, by a simple ligand switch, enantioenriched piperidines 75–82 and azepanes 83–86 can be obtained, respectively, from the same starting materials in synthetically useful yields and excellent diastereoselectivities (15:1 to > 20:1 d.r.). The absolute configuration of 76 was determined by single-crystal X-ray diffraction analysis, and that of all other products were assigned accordingly.
We further found that the reaction of 1,6-enyne with aryl bromide underwent trans-arylative exo-cyclization/cross-coupling to give diarylated products when 1,10-phenanthroline L4 was used as a ligand (Fig. 4b). The presence of electron-donating and electron-withdrawing groups at the para- or meta-position of the aryl bromides does not affect selectivity and efficiency (87–95). Diastereoselective diarylative cyclization gave enantioenriched piperidine 96 in excellent diastereoselectivity ( > 20:1 d.r.). Despite extensive attempts, cis-arylation cyclization/cross-coupling products have not yet been obtained using L9 as a ligand.
Mechanistic investigation
To shed light on the reaction mechanism, we designed a series of experiments as outlined in Fig. 5. First, Ph-Ni(II)Br complex 98 and 99 were synthesized and submitted to the reactions with 1,6-enyne 97 and trifluoromethyl alkene 3 under our standard reaction conditions. The corresponding products 7 and 47 were obtained in 31% and 30% yields, respectively (Fig. 5a). These results clearly demonstrate that active aryl-nickel(II) species are generated during the reaction and that the ligand determines the regioselectivity alkyne arylation.
We further synthesized alkenyl iodide 100 and subjected to the reaction with trifluoromethylalkene 3 under standard conditions. The target product 7 was isolated in 65% yield, evidently confirming the formation of the key alkenyl-nickel intermediate and the involvement of cis/trans isomerization of alkenyl-nickel species during the reaction (Fig. 5b). Deuterium labeling experiments were performed. Reaction of 100 with 5 equivalents of D2O afforded the deuterated exo-cyclization product 101-D in 43% yield. Reaction of 100 with trifluoromethylalkene 3 in the presence of 10 equivalents of D2O gave product 101-D and 7 in 24% and 49% yield, respectively (Fig. 5c). These deuterated products should result from deuteration of the corresponding organonickel intermediates, suggesting that both 6-exo-cyclization and 7-endo-cyclization of alkenyl-nickel intermediate appear to be possible.
Three possible reaction pathways are proposed to explain the transformation of six-membered-ring nickel intermediate 103 to seven-membered-ring nickel intermediate 106 in Fig. 5d. Considering that β-carbon elimination from a non-strained metal complex is a high energy process62,63,64, a retro 6-exo cyclization via β-carbon elimination followed by 7-endo cyclization deemed less probable (Path I, Fig. 5d).
To verify 1,2-dyotropic rearrangement mechanism (Path II, Fig. 5d), deuterated 1,6-enyne (Z)-107-D was prepared and subjected to the reaction with trifluoromethylalkene 3 and PhBr under standard conditions. Product 7-D was isolated in 55% yield with 1:1 diastereoselectivity (Fig. 5e). Although this process is non-stereoselective, considering the carbon-nickel bond is easily undergo homolytic cleavage to generate the corresponding carbon radical and then recombine, we cannot rule out the mechanism of 1,2-dyotropic rearrangement65,66,67.
Furthermore, Dowd-Beckwith ring expansion process was also considered (Path III, Fig. 5d)68,69, that is, the primary radical intermediate 104 and the tertiary radical intermediate 105 coexist in the reaction system; the type of electrophilic trapping reagents determine the structure of products. When cross-coupling with aryl bromides via an oxidative addition/reductive elimination mechanism, primary radicals 104 are preferred because the process is more sensitive to steric hindrance of ligands. The nucleophilic tertiary radicals 105 are favored when cross-coupling with less hindered electron-deficient alkenes (acrylates or trifluoromethyl alkenes) via Giese addition.
DFT calculations
To better understand the reaction mechanism and the origin of selectivity-controlled steps of arylative of alkynes with different ligands, density functional theory (DFT) studies were performed (Fig. 6)70. The computed energy profile for the nickel catalytic cycle using ligand L4 is shown in Scheme 8 A, while the influence of ligand L9 is shown in Supplementary Fig. 6. The overall pathway commences with the coordination of bromobenzene 2 to the in-situ generated Ni(0)/ligand species, forming intermediate INT1A. The oxidative addition of the C−Br bond proceeds through transition TS2A and subsequent substrate 1 replaces the Br− ligand at INT2A, resulting in the formation of INT3A. The noncovalent interactions (NCI) between substrates and ligands is more pronounced for the regioselectivity of migratory insertion (Fig. 6b)71,72. In the case of smaller ligand L4, phenyl connected with nickel center and phenyl substituent of alkyne are the most sterically congesting moiety, thus resulting the energy barrier of transition state TS4A-L4 is 4.9 kcal mol−1 higher than that of transition state TS4B-L4 (17.8 vs 12.9 kcal mol−1). The α-migratory insertion to form intermediate INT4B-L4 is kinetically more favorable pathway. For ligand L9, there is a significant attractive interaction between naphthyl group and phenyl unit of alkyne, rendering an increased energy barrier of TS4B-L9 to 21.8 kcal mol−1. This result illustrates intermediate INT3A-L9 favors β-insertion over α-insertion. The resulting intermediate INT4B-L4 undergoes rapid cis/trans isomerization via reversible Ni–C bond homolysis73,74,75 or a η2-coordinated alkenylnickel species (see Supplementary Information for details)76,77,78 to form intermediate INT5B-L4, which facilitates the proximity of the nickel(II) center to the C = C bond for the subsequent cyclization process35,79,80. The competitive cyclization modes with ligand L4 and L9 are further investigated, respectively (Fig. 6c). Compared to the endo-cyclization, the exo-cyclization through TS5B-L4-exo and TS5A-L9-exo is a more favorable process (14.3 vs 11.3 kcal mol−1 for L4 and 19.4 vs 2.8 kcal mol−1 for L9). Upon adding D2O, the generated INT5B-L4-exo can be converted into six-membered skeleton smoothly, consistent with the experimental observation shown in Fig. 6c. Finally, the Ni(II)-alkyl bond of intermediate INT5B-L4-exo and INT5A-L9-exo undergoes homolytic cleavage to generate alkyl radical and Ni(I) species, which was then reduced by the present of Mn to regenerate INT1A, completing nickel catalytic cycle.
Furthermore, extensive computational studies on alkyl-nickel(II) species INT5B-L4-exo and INT5A-L9-exo were performed (Fig. 7a, b). We located a homolytic cleavage pathway of alkyl-nickel(II) to generate radical INT7B (Fig. 7a). By comparison, the alternative pathways involving directly insertion into the double bond of trifluoromethyl alkene 3 (TS6A-L4-exo and TS6A-L9-exo, ∆G≠ = 24.0 and 33.0 kcal mol−1, respectively), 1,2-alkenyl/Ni concerted dyotropic rearrangement (TS6C-L4-exo and TS6C-L9-exo, ∆G≠ = 41.5 and 47.2 kcal mol−1, respectively), and 1,2-alkyl/Ni concerted dyotropic rearrangement (TS6D-L4-exo and TS6D-L9-exo, ∆G≠ = 52.1 and 57.7 kcal mol−1, respectively), were calculated to be kinetically unfavorable.
The subsequent pathway for the generated radical INT7B was shown in Fig. 8. The radical induced Dowd-Beckwith-type ring expansion68,81,82,83,84,85 through transition TS8B and TS9B, leading to the formation of seven membered ring radical INT9B. After that, INT9B captured by substrate 3 leads to the formation of INT10B with an activation free energy barrier of 7.9 kcal mol−1. For the enantiomerically pure N-heterocyclic compound 72, the radical addition preferentially occurred at the less steric hindrance to result excellent diastereoselectivities. Owing to the computed high energy barrier (TS11B, ∆G≠ = 29.6 kcal mol−1), the direct β-F elimination of INT10B is excluded. The single electron transfer (SET) process between INT10B and reductant Mn proceeds to afford anion INT11B, which is followed by β-F elimination via transition state TS12B with an activation energy of 9.9 kcal mol−1, yielding the desired product 4. The energy profile for the radical produced by INT5A-exo-L9 is also calculated to obtain product 5 (for the details of the computational data, see Supplementary Fig. 8). The frontier molecular orbital theory is further used to analysis the reactivity of the generated radicals (Fig. 8c). Compared with radical INT7B, the radical INT9B with higher SOMO energy exhibits more nucleophilicity, which should be thermodynamically more favorable to attack electron-deficient alkenes to generate radical INT10B, delivering the desired cyclization products.
Proposed reaction mechanism
Based on the above experimental and DFT studies, a plausible catalytic cycle is outlined in Fig. 9. Oxidative addition of aryl bromide to Ni(0) species produces aryl-nickel(II) species. The steric interaction between the ligands and substrates precisely controls the regioselectivity of the migratory insertion of aryl-nickel(II) species into alkynes, where L9 promotes β-insertion to form alkenyl-nickel(II) intermediate A and L4 facilitates α-insertion to form alkenyl-nickel(II) intermediate E. Intermediates A and E undergo highly selective exo-cyclization to give alkyl-nickel(II) intermediates B and F, which then undergo homolytic cleavage to afford the corresponding primary alkyl radical intermediates C and G, respectively. Direct cross-coupling of radical G with aryl bromides in the presence of Ni(0) gives diarylated products. Intermediates C and G undergo Dowd-Beckwith ring expansion to form more stable tertiary radical intermediates D and H, respectively. Finally, radical addition followed by fluorine elimination will furnish the corresponding six- and seven-membered ring products, respectively.
The structure of the final products is related to the nature of the electrophilic trapping reagents. The primary radical G may react preferentially because it is formed first and is more kinetically accessible. The tertiary radicals D and H, although more crowded, are more stable and survive longer in the reaction system. When reacting with ArBr via an oxidative addition/reductive elimination mechanism, primary radicals C and G are preferred because the process is more sensitive to steric hindrance of the ligand. More nucleophilic tertiary radicals D and H are favored when reacting with electron-deficient alkenes such as trifluoromethylalkenes or acrylates via Giese addition.
Methods
General procedure for the synthesis of 7-membered ring products
An oven-dried sealed tube equipped with a PTFE-coated stir bar was charged with NiBr2(DME) (10 mol%), L4 (20 mol%), trifluoromethyl alkene (0.1 mmol), 1,6-enyne (0.2 mmol), CsF (1.0 equiv), Mn powder (3.0 equiv), aryl bromide (2.0 equiv) and anhydrous DMA (2 mL). The sealed tube was sealed and removed from the glovebox. Then the reaction was stirred at 60 oC until the reaction was complete (monitored by TLC). The resulting mixture was quenched with saturated NH4Cl solution (5 mL) and further diluted with water (10 mL). The aqueous layer was extracted with EtOAc and the combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated under vacuum. The residue was purified by chromatography on silica gel, eluting with PE/EtOAc (50/1 ~ 5/1) to afford the desired 7-membered ring products.
General procedure for the synthesis of 6-membered ring products
An oven-dried sealed tube equipped with a PTFE-coated stir bar was charged with NiBr2(DME) (10 mol%), L9 (20 mol%), trifluoromethyl alkene (0.1 mmol), 1,6-enyne (0.2 mmol), Mn powder (3.0 equiv), aryl bromide (2.0 equiv) and anhydrous DMA (2 mL). The sealed tube was sealed and removed from the glovebox. Then the reaction was stirred at 60 oC until the reaction was complete (monitored by TLC). The resulting mixture was quenched with saturated NH4Cl solution (5 mL) and further diluted with water (10 mL). The aqueous layer was extracted with EtOAc and the combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated under vacuum. The residue was purified by chromatography on silica gel, eluting with PE/EtOAc (50/1 ~ 5/1) to afford the desired 6-membered ring products.
Data availability
The authors declare that all the data supporting the findings of this work are available within the article and its Supplementary Information files or from the corresponding author upon request. The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition numbers 2220556 (7), 2220555 (46), and 2240774 (74). CIF files and crystal structure data are available in the Supplementary Information and Supplementary Data 1.
References
Shelat, A. A. & Guy, R. K. Scaffold composition and biological relevance of screening libraries. Nat. Chem. Biol. 3, 442–446 (2007).
Frearson, J. A. & Collie, I. T. HTS and hit finding in academia-from chemical genomics to drug discovery. Drug Discov. Today 14, 1150–1158 (2009).
Garcia-Castro, M., Zimmermann, S., Sankar, M. G. & Kumar, K. Scaffold diversity synthesis and its application in probe and drug discovery. Angew. Chem. Int. Ed. 55, 7586–7605 (2016).
Burke, M. D. & Schreiber, S. L. A planning strategy for diversity-oriented synthesis. Angew. Chem. Int. Ed. 43, 46–58 (2004).
O’Connor, C. J., Beckmann, H. S. G. & Spring, D. R. Diversity-oriented synthesis: producing chemical tools for dissecting biology. Chem. Soc. Rev. 41, 4444–4456 (2012).
Lee, Y., Kumar, K. & Waldmann, H. Ligand-directed divergent synthesis of carbo- and heterocyclic ring systems. Angew. Chem. Int. Ed. 57, 5212–5226 (2018).
Trost, B. M. Palladium-catalyzed cycloisomerizations of enynes and related reactions. Acc. Chem. Res. 23, 34–42 (1990).
Aubert, C., Buisine, O. & Malacria, M. The behavior of 1,n-enynes in the presence of transition metals. Chem. Rev. 102, 813–834 (2002).
Jiménez-Núñez, E. & Echavarren, A. M. Gold-catalyzed cycloisomerizations of enynes: a mechanistic perspective. Chem. Rev. 108, 3326–3350 (2008).
Michelet, V., Toullec, P. Y. & Genét, J.-P. Cycloisomerization of 1,n-enynes: challenging metal-catalyzed rearrangements and mechanistic insights. Angew. Chem. Int. Ed. 47, 4268–4315 (2008).
Marinetti, A., Jullien, H. & Voituriez, A. Enantioselective, transition metal catalyzed cycloisomerizations. Chem. Soc. Rev. 41, 4884–4908 (2012).
Trost, B. M. & Rise, F. Reductive cyclization of 1,6- and 1,7-enynes. J. Am. Chem. Soc. 109, 3161–3163 (1987).
Jang, H.-Y. & Krische, M. J. Rhodium-catalyzed reductive cyclization of 1,6-diynes and 1,6-enynes mediated by hydrogen: catalytic C-C bond formation via capture of hydrogenation intermediates. J. Am. Chem. Soc. 126, 7875–7880 (2004).
Jang, H.-Y. et al. Enantioselective reductive cyclization of 1,6-enynes via rhodium-catalyzed asymmetric hydrogenation: C-C bond formation precedes hydrogen activation. J. Am. Chem. Soc. 127, 6174–6175 (2005).
Chen, M., Weng, Y., Guo, M., Zhang, H. & Lei, A. Nickel-catalyzed reductive cyclization of unactivated 1,6-enynes in the presence of organozinc reagents. Angew. Chem. Int. Ed. 47, 4268–4315 (2008).
Sylvester, K. T. & Chirik, P. J. Iron-catalyzed, hydrogen-mediated reductive cyclization of 1,6-enynes and diynes: evidence for bis(imino)pyridine ligand participation. J. Am. Chem. Soc. 131, 8772–8774 (2009).
Miura, T., Sasaki, T., Nakazawa, H. & Murakami, M. Ketone synthesis by intramolecular acylation of organorhodium(I) with ester. J. Am. Chem. Soc. 127, 1390–1391 (2005).
Marco-Martinez, J., Lopez-Carrillo, V., Buñuel, E., Simancas, R. & Cardenas, D. J. Pd-catalyzed borylative cyclization of 1,6-enynes. J. Am. Chem. Soc. 129, 1874–1875 (2007).
Fan, B., Xie, J., Li, S., Wang, L.-X. & Zhou, Q.-L. Highly enantioselective hydrosilylation/cyclization of 1,6-enynes catalyzed by rhodium(I) complexes of spiro diphosphines. Angew. Chem. Int. Ed. 46, 1275–1277 (2007).
Shintani, R., Isobe, S., Takeda, M. & Hayashi, T. Rhodium-catalyzed asymmetric synthesis of spirocarbocycles: arylboron reagents as surrogates of 1,2-dimetalloarenes. Angew. Chem. Int. Ed. 49, 3795–3798 (2010).
Jiang, M., Jiang, T. & Bäckvall, J.-E. Palladium-catalyzed oxidative diarylating carbocyclization of enynes. Org. Lett. 14, 3538–3541 (2012).
Liu, P. et al. Cu-catalyzed asymmetric borylative cyclization of cyclohexadienone-containing 1,6-enynes. J. Am. Chem. Soc. 135, 11700–11703 (2013).
Yang, C. M., Mannathan, S. & Cheng, C. H. Nickel-catalyzed chemo- and stereoselective alkenylative cyclization of 1,6-enynes with alkenyl boronic acids. Chem. Eur. J. 19, 12212–12216 (2013).
Fukui, Y. et al. Tunable arylative cyclization of 1,6-enynes triggered by rhodium(III)-catalyzed C-H activation. J. Am. Chem. Soc. 136, 15607–15614 (2014).
Santhoshkumar, R., Mannathan, S. & Cheng, C. H. Ligand-controlled divergent C-H functionalization of aldehydes with enynes by cobalt catalysts. J. Am. Chem. Soc. 137, 16116–16120 (2015).
Yu, S., Wu, C. & Ge, S. Cobalt-catalyzed asymmetric hydroboration/cyclization of 1,6-enynes with pinacolborane. J. Am. Chem. Soc. 139, 6526–6529 (2017).
Wang, C. & Ge, S. Versatile cobalt-catalyzed enantioselective entry to boryl-functionalized all-carbon quaternary stereogenic centers. J. Am. Chem. Soc. 140, 10687–10690 (2018).
Chen, J. H. & Hayashi, T. Asymmetric synthesis of alkylzincs by rhodium-catalyzed enantioselective arylative cyclization of 1,6-enynes with arylzincs. Angew. Chem. Int. Ed. 59, 18510–18514 (2020).
Whyte, A. et al. Cobalt-catalyzed enantioselective hydroarylation of 1,6-enynes. J. Am. Chem. Soc. 142, 9510–9517 (2020).
Whyte, A., Bajohr, J., Torelli, A. & Lautens, M. Enantioselective cobalt-catalyzed intermolecular hydroacylation of 1,6-enynes. Angew. Chem. Int. Ed. 59, 16409–16413 (2020).
You, Y. & Ge, S. Asymmetric cobalt-catalyzed regioselective hydrosilylation/cyclization of 1,6-enynes. Angew. Chem. Int. Ed. 60, 12046–12052 (2021).
Ren, X. et al. Enantioselective hydroesterificative cyclization of 1,6-enynes to chiral γ-lactams bearing a quaternary carbon stereocenter. Org. Lett. 23, 3561–3566 (2021).
Ding, Z., Wang, Y., Liu, W., Chen, Y. & Kong, W. Diastereo- and enantioselective construction of spirocycles by nickel-catalyzed cascade borrowing hydrogen cyclization. J. Am. Chem. Soc. 143, 53–59 (2021).
Tambe, S. D. et al. Nickel-catalyzed enantioselective synthesis of 2,3,4-trisubstituted 3-pyrrolines. Angew. Chem. Int. Ed. 61, e202203494 (2022).
Bottcher, S. E., Hutchinson, L. E. & Wilger, D. J. Nickel-catalyzed anti-selective alkyne functionalization reactions. Synthesis 52, 2807–2820 (2020).
Liu, W. & Kong, W. Ni-Catalyzed stereoselective difunctionalization of alkynes. Org. Chem. Front. 7, 3941–3955 (2020).
Gillbard, S. M. & Lam, H. W. Nickel-catalyzed arylative cyclizations of alkyne- and allene-tethered electrophiles using arylboron reagents. Chem. Eur. J. 28, e202104230 (2022).
Corpas, J., Mauleón, P., Arrayás, R. G. & Carretero, J. C. Transition-metal-catalyzed functionalization of alkynes with organoboron reagents: new trends, mechanistic insights, and applications. ACS Catal. 11, 7513–7551 (2021).
Petrone, D. A. et al. Palladium-catalyzed hydrohalogenation of 1,6-enynes: hydrogen halide salts and alkyl halides as convenient HX surrogates. J. Am. Chem. Soc. 139, 3546–3557 (2017).
Chen, X. et al. Pd(0)-catalyzed asymmetric carbohalogenation: H‑bonding-driven C(sp3)-halogen reductive elimination under mild conditions. J. Am. Chem. Soc. 143, 1924–1931 (2021).
Huggins, J. M. & Bergman, R. G. Mechanism, regiochemistry, and stereochemistry of the insertion reaction of alkynes with Methyl(2,4-Pentanedionato)(Triphenylphosphine) Nickel. A cis insertion that leads to trans kinetic products. J. Am. Chem. Soc. 103, 3002–3011 (1981).
Clarke, C., Incerti-Pradillos, C. A. & Lam, H. W. Enantioselective nickel-catalyzed anti-carbometallative cyclizations of alkynyl electrophiles enabled by reversible alkenylnickel E/Z isomerization. J. Am. Chem. Soc. 138, 8068–8071 (2016).
Zhang, X., Xie, X. & Liu, Y. Nickel-catalyzed cyclization of alkyne-nitriles with organoboronic acids involving anti-carbometalation of alkynes. Chem. Sci. 7, 5815–5820 (2016).
Yap, C. et al. Enantioselective nickel-catalyzed intramolecular allylic alkenylations enabled by reversible alkenylnickel E/Z isomerization. Angew. Chem. Int. Ed. 56, 8216–8220 (2017).
Mikami, K. & Hatano, M. Palladium-catalyzed carbocyclization of 1,6-enynes leading to six-membered rings or oxidized five membered trifluoroacetates. Proc. Natl Acad. Sci. 101, 5767–5769 (2004).
Ding, L. et al. Palladium-catalyzed divergent cycloisomerization of 1,6-enynes controlled by functional groups for the synthesis of pyrroles, cyclopentenes, and tetrahydropyridines. Org. Chem. Front. 8, 4785–4790 (2021).
Babu, U. S., Reddy Singam, M. K., Kumar, M. N., Nanubolu, J. B. & Reddy, M. S. Palladium-catalyzed carbo-aminative cyclization of 1,6-enynes: access to napthyridinone derivatives. Org. Lett. 24, 1598–1603 (2022).
Dong, M., Qi, L., Qian, J., Yu, S. & Tong, X. Pd(0)-catalyzed asymmetric 7-endo hydroacyloxylative cyclization of 1,6-enyne enabled by an anion ligand-directed strategy. J. Am. Chem. Soc. 145, 1973–1981 (2023).
Tasker, S. Z., Standley, E. A. & Jamison, T. F. Recent advances in homogeneous nickel catalysis. Nature 509, 299–309 (2014).
Weix, D. J. Methods and mechanisms for cross-electrophile coupling of Csp2 halides with alkyl electrophiles. Acc. Chem. Res. 48, 1767–1775 (2015).
Lucas, E. L. & Jarvo, E. R. Stereospecific and stereoconvergent cross-couplings between alkyl electrophiles. Nat. Rev. Chem. 1, 0065 (2017).
Liu, J., Ye, Y., Sessler, J. L. & Gong, H. Cross-electrophile couplings of activated and sterically hindered halides and alcohol derivatives. Acc. Chem. Res. 53, 1833–1845 (2020).
Diccianni, J., Lin, Q. & Diao, T. Mechanisms of nickel-catalyzed coupling reactions and applications in alkene functionalization. Acc. Chem. Res. 53, 906–919 (2020).
Poremba, K. E., Dibrell, S. E. & Reisman, S. E. Nickel-catalyzed enantioselective reductive cross-coupling reactions. ACS Catal. 10, 8237–8246 (2020).
Pan, Q., Ping, Y., Wang, Y., Guo, Y. & Kong, W. Ni-catalyzed ligand-controlled regiodivergent reductive dicarbofunctionalization of alkenes. J. Am. Chem. Soc. 143, 10282–10291 (2021).
Ping, Y., Li, X., Pan, Q. & Kong, W. Ni-catalyzed divergent synthesis of 2-benzazepine derivatives via tunable cyclization and 1,4-acyl transfer triggered by amide N-C bond cleavage. Angew. Chem. Int. Ed. 61, e202201574 (2022).
Ping, Y. et al. Switchable 1,2-rearrangement enables expedient synthesis of structurally diverse fluorine-containing scaffolds. J. Am. Chem. Soc. 144, 11626–11637 (2022).
Buckingham, J., Baggaley, K. H., Roberts, A. D. & Szabó, L. F. In Dictionary of Alkaloids, 2nd ed.; CRC Press/Taylor & Francis Group, Boca Raton, FL, USA, 2010.
Ager, D. J., Prakash, I. & Schaad, D. R. 1,2-Amino alcohols and their heterocyclic derivatives as chiral auxiliaries in asymmetric synthesis. Chem. Rev. 96, 835–876 (1996).
Thaler, T. et al. Highly diastereoselective Csp3-Csp2 Negishi cross-coupling with 1,2-, 1,3- and 1,4-substituted cycloalkylzinc compounds. Nat. Chem. 2, 125–130 (2010).
Petrone, D. A., Yoon, H., Weinstabl, H. & Lautens, M. Additive effects in the palladium-catalyzed carboiodination of chiral N-allyl carboxamides. Angew. Chem. Int. Ed. 53, 7908–7912 (2014).
Ye, J. et al. Remote C-H alkylation and C-C bond cleavage enabled by an in situ generated palladacycle. Nat. Chem. 9, 361–368 (2017).
Marchese, A. D., Mirabi, B., Johnson, C. E. & Lautens, M. Reversible C-C bond formation using palladium catalysis. Nat. Chem. 14, 398–406 (2022).
Xu, B. et al. Palladium/Xu-Phos-catalyzed enantioselective cascade Heck/remote C(sp2)-H alkylation reaction. Chem 8, 836–849 (2022).
Cao, J., Wu, H., Wang, Q. & Zhu, J. C-C bond activation enabled by dyotropic rearrangement of Pd(IV) species. Nat. Chem. 13, 671–676 (2021).
Yang, G. et al. Migrative carbofluorination of saturated amides enabled by Pd-based dyotropic rearrangement. J. Am. Chem. Soc. 144, 14047–14052 (2022).
Gong, J., Wang, Q. & Zhu, J. Apparent 6-endo-trig carbofluorination of alkenes enabled by palladium-based dyotropic rearrangement. Angew. Chem. Int. Ed. 61, e202211470 (2022).
Dowd, P. & Zhang, W. Free radical-mediated ring expansion and related annulations. Chem. Rev. 93, 2091–2115 (1993).
Singha, T., Mondal, A. R. S., Midya, S. & Hari, D. P. The Dowd-Beckwith reaction: history, strategies, and synthetic potential. Chem. Eur. J. 28, e202202025 (2022).
Frisch, M. J. et al. Gaussian 09, revision E.01 (Gaussian, Inc., 2013).
Krenske, E. H. & Houk, K. N. Aromatic interactions as control elements in stereoselective organic reactions. Acc. Chem. Res. 46, 979–989 (2013).
Fanourakis, A., Docherty, P. J., Chuentragool, P. & Phipps, R. J. Recent developments in enantioselective transition metal catalysis featuring attractive noncovalent interactions between ligand and substrate. ACS Catal. 10, 10672–10714 (2020).
Wang, X., Liu, Y. & Martin, R. Ni-catalyzed divergent cyclization/carboxylation of unactivated primary and secondary alkyl halides with CO2. J. Am. Chem. Soc. 137, 6476–6479 (2015).
Börjesson, M., Moragas, T. & Martin, R. Ni-catalyzed carboxylation of unactivated alkyl chlorides with CO2. J. Am. Chem. Soc. 138, 7504–7507 (2016).
Ryan Barber, E. et al. Nickel-catalyzed hydroarylation of alkynes under reductiveconditions with aryl bromides and water. J. Org. Chem. 84, 11612–1162 (2019).
Choi, H., Lyu, X., Kim, D., Seo, S. & Chang, S. Endo-selective intramolecular alkyne hydroamidation enabled by NiH catalysis incorporating alkenylnickel isomerization. J. Am. Chem. Soc. 144, 10064–10074 (2022).
Li, J. et al. DFT study of Ni-catalyzed intramolecular asymmetric anti-hydrometalative cyclization of alkynone: mechanism and origins of selectivity. Org. Chem. Front. 10, 4263–4274 (2023).
Shan, C., He, M., Luo, X., Lia, R. & Zhang, T. Mechanistic insight into anti-carbometalation of an alkyne via η2 -vinyl-nickel type Z/E isomerization. Org. Chem. Front. 10, 4243–4249 (2023).
Huggins, J. M. & Bergman, R. G. Reaction of alkynes with a methylnickel complex: observation of a cis insertion mechanism capable of giving kinetically controlled trans products. J. Am. Chem. Soc. 101, 4410–4441 (1979).
Huggins, J. M. & Bergman, R. G. Mechanism, regiochemistry, and stereochemistry of the insertion reaction of alkynes with methyl(2,4-pentanedionato)(triphenylphosphine)nickel. A cis insertion that leads to trans kinetic products. J. Am. Chem. Soc. 103, 3002–3011 (1981).
Beckwith, A. L. J., Kazlauskas, R. & Syner-Lyons, M. R. beta.-Fission of 9-decalinoxyl radicals: reversible formation of 6-ketocyclodecyl radical. J. Org. Chem. 48, 4718–4722 (1983).
Beckwith, A. L. J., O’Shea, D. M., Gerba, S. & Westwood, S. W. Cyano or acyl group migration by consecutive homolytic addition and β-fission. J. Chem. Soc. Chem. Commun. 1987, 666–667 (1987).
Dowd, P. & Choi, S. C. A new tributyltin hydride-based rearrangement of bromomethyl.beta.-keto esters. A synthetically useful ring expansion to gamma.-keto esters. J. Am. Chem. Soc. 109, 3493–3494 (1987).
Dowd, P. & Choi, S. C. Free radical ring expansion by three and four carbons. J. Am. Chem. Soc. 109, 6548–6549 (1987).
Beckwith, A. L. J., O’Shea, D. M. & Westwood, S. W. Rearrangement of suitably constituted aryl, alkyl, or vinyl radicals by acyl or cyano group migration. J. Am. Chem. Soc. 110, 2565–2575 (1988).
Acknowledgements
Financial support provided by the National Natural Science Foundation of China (22171215 and 22171134), the Hubei Provincial Outstanding Youth Fund (2022CFA092), the National Key R&D Program of China (2022YFA1503200), and GuangDong Basic and Applied Basic Research Foundation (2022A1515010246). The authors also thank the High-Performance Computing Center of Nanjing University for doing the numerical calculations in this paper on its blade cluster system.
Author information
Authors and Affiliations
Contributions
W.K. and WF.L. conceived and designed the project. WF.L. and W.L. conducted the experiments. W.K., WF.L. and W.L. analyzed and interpreted the experimental data. M.W. and W.X. performed the DFT calculations. W.K. and M.W. prepared the manuscript. WF.L. and W.L. prepared the Supplementary Information. All authors contributed to discussions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, W., Li, W., Xu, W. et al. Nickel-catalyzed switchable arylative/endo-cyclization of 1,6-enynes. Nat Commun 15, 2914 (2024). https://doi.org/10.1038/s41467-024-47200-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-47200-z
- Springer Nature Limited