Abstract
Dinitrogen is an abundant and promising material for valuable organonitrogen compounds containing carbon–nitrogen bonds. Direct synthetic methods for preparing organonitrogen compounds from dinitrogen as a starting reagent under mild reaction conditions give insight into the sustainable production of valuable organonitrogen compounds with reduced fossil fuel consumption. Here we report the catalytic reaction for the formation of cyanate anion (NCO−) from dinitrogen under ambient reaction conditions. A molybdenum–carbamate complex bearing a pyridine-based 2,6-bis(di-tert-butylphosphinomethyl)pyridine (PNP)-pincer ligand is synthesized from the reaction of a molybdenum–nitride complex with phenyl chloroformate. The conversion between the molybdenum–carbamate complex and the molybdenum–nitride complex under ambient reaction conditions is achieved. The use of samarium diiodide (SmI2) as a reductant promotes the formation of NCO− from the molybdenum–carbamate complex as a key step. As a result, we demonstrate a synthetic cycle for NCO− from dinitrogen mediated by the molybdenum–PNP complexes in two steps. Based on this synthetic cycle, we achieve the catalytic synthesis of NCO− from dinitrogen under ambient reaction conditions.
Similar content being viewed by others
Introduction
Dinitrogen is an abundant and promising material for valuable organonitrogen compounds containing carbon–nitrogen bonds. In general, it is considerably difficult to convert dinitrogen into valuable organonitrogen compounds because of the thermodynamic and kinetic stability of dinitrogen1. To use dinitrogen as a nitrogen source, dinitrogen is converted into ammonia by the Haber–Bosch process, which requires harsh reaction conditions and consumes over 1% of world fossil fuel2. In addition, some steps are necessary to produce valuable organonitrogen compounds from ammonia. Therefore, direct synthetic methods for preparing organonitrogen compounds from dinitrogen as a starting reagent under mild reaction conditions give insight into the sustainable production of valuable organonitrogen compounds with reduced fossil fuel consumption.
To overcome the inertness of dinitrogen, the direct conversion of dinitrogen into organonitrogen compounds mediated by transition metal complexes has been conducted for the last half-century3,4,5,6. In particular, organonitrogen compounds can be prepared from the reactions of transition metal–dinitrogen complexes with carbon-centred electrophiles to functionalise the coordinated dinitrogen on the metal atoms under mild reaction conditions5,6,7,8,9,10,11. Since the milestone work for the cleavage of dinitrogen to synthesise the corresponding nitride complex reported by the Cummins’ group12,13, organonitrogen compounds can be synthesised from the reactions of transition metal–nitride complexes. This is an alternative method to prepare organonitrogen compounds from dinitrogen. The transition metal–nitride complexes, which are generated via the direct cleavage of a nitrogen–nitrogen triple bond, reacted with carbon-centred electrophiles to functionalise the nitride ligand under mild reaction conditions14,15,16,17,18,19,20,21,22,23,24,25,26. Based on the experimental results of stoichiometric reactions with transition metal complexes, several research groups have constructed synthetic cycles to prepare organonitrogen compounds from dinitrogen by step-by-step reactions requiring a stoichiometric amount of transition metal complexes9,10,11,14,15,16,17,18,19,22,23,25,26,27. However, the catalytic and direct transformations of dinitrogen into valuable organonitrogen compounds with transition metal complexes under mild reaction conditions have not been reported until now.
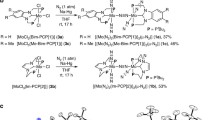
As the first investigation for the catalytic and direct preparation of organonitrogen compounds from dinitrogen, we have focused on isocyanates (R–N=C=O) and their derivatives using CO and its derivatives because isocyanate derivatives are attractive synthetic compounds from the viewpoint of thermodynamics and usefulness in organic synthesis28. In particular, sodium and potassium salts of isocyanate are commercially important with the world demand of 8000–10000 tonnes per year. The isocyanate salts are applied in steel hardening and fine chemical and pharmaceutical synthesis as well as in the agrochemical industry. Currently, the isocyanate salts are produced from the reaction of the corresponding carbonate salts with urea at over 400 °C, which overall requires two moles of NH3 for each mole of cyanate29. Recently, some research groups have achieved synthetic cycles to prepare isocyanate derivatives from dinitrogen via stepwise procedures22,23,25,26. Kawaguchi’s group and Schneider’s group have reported synthetic cycles for isocyanate derivatives in five and seven steps, respectively (Fig. 1a, b)22,25. Both synthetic cycles comprise the formation of the corresponding isocyanate complexes from the reactions of the nitride complexes, which are formed via the direct cleavage of a nitrogen–nitrogen triple bond of dinitrogen, with carbon monoxide (CO) as key reaction steps. As a related work, Sita’s group has reported a synthetic cycle for trimethylsilylisocyanate in five steps, where the reaction of a molybdenum–silylimide complex, which is formed via the direct cleavage of a nitrogen–nitrogen triple bond of dinitrogen and silylation, with CO2 is a key reaction step (Fig. 1c)23. In these cases, it is considerably difficult to expand the synthetic cycles into the corresponding catalytic cycles. This is because of the multiple (five and seven) steps under different reaction conditions and the use of CO and CO2 to inhibit the generation of the metal–nitride complexes via the cleavage of dinitrogen. Quite recently, Hou’s group has reported a synthetic cycle for trimethylsilylisocyanate in four steps, where the reaction of titanium–dinitrogen complex with CO2 is a key step (Fig. 1d)26. However, for the same reason, it is also difficult to achieve the corresponding catalytic reaction.
Recently, we have found effective catalytic ammonia formation from dinitrogen under ambient reaction conditions. A molybdenum–nitride complex bearing a pyridine-based PNP-type pincer ligand [Mo(N)I(PNP)] (1) (PNP = 2,6-bis(di-tert-butylphosphinomethyl)pyridine), which is formed via the direct cleavage of a nitrogen–nitrogen triple bond of dinitrogen, works as a key reactive intermediate (Fig. 2a)30,31,32. In addition, we have investigated the reactions of 1 with various carbon-centred electrophiles towards preparing organonitrogen compounds33. The stoichiometric amount of organic amide is successfully synthesised from the reaction of 1 with acyl chloride and water.
In this work, we report the synthesis of cyanate anion (NCO−) from dinitrogen using chloroformate ester mediated by molybdenum complexes under ambient reaction conditions (Fig. 2b). Some research groups previously used chloroformate esters as a carbon monoxide source to obtain the corresponding ketones from transition metal-catalysed reactions34,35. The synthetic cycle reported herein comprises the carbamylation of the molybdenum–nitride complex and the reduction of the molybdenum–carbamate complex followed by the direct cleavage of a nitrogen–nitrogen triple bond of dinitrogen in two steps. This methodology enables the catalytic production of NCO− from dinitrogen under ambient reaction conditions.
Results
Synthetic cycle for NCO− from dinitrogen
The reaction of 1 with 1.1 equiv of phenyl chloroformate in tetrahydrofuran (THF) at room temperature for 2 h afforded the corresponding carbamate complex [Mo(NCO2Ph)ICl(PNP)] (2) in 83% yield (Fig. 3a). This carbamate complex 2 was characterised by 1H and 31P{1H} nuclear magnetic resonance (NMR). The presence of a carbamate ligand was supported by infrared (IR) measurement (1690 cm−1 (νC=O) and 642 cm−1 (νMo=N)). According to the same method, the corresponding 15N-labelled carbamate complex [Mo(15NCO2Ph)ICl(PNP)] (2-15N) was obtained from the reaction of the 15N-labelled nitride complex [Mo(15N)I(PNP)] (1-15N), which was prepared from the reaction of [MoI3(PNP)] with 2.2 equiv of KC8 under 1 atm of 15N2, with phenyl chloroformate. This complex 2-15N was characterised by 1H, 31P{1H}, and 15N{1H} NMR. The 15N{1H} NMR of 2-15N showed a resonance at −51.6 ppm (t, JN–P = 4.9 Hz). Interestingly, the reaction of 1 with 1.1 equiv of phenyl iodoformate under similar reaction conditions did not afford the corresponding carbamate complex but the corresponding cationic nitride complex [Mo(N)I(PNP)][I] ([1][I]) quantitatively (see Supplementary Information). This result indicates that the use of chloroformate as a carbamylation reagent is necessary to obtain the corresponding carbamate complex.
a Synthesis of carbamate complex 2. b, c ORTEP drawings of 2 (b) and 4a (c). Thermal ellipsoids are shown at the 50% probability level. All hydrogen atoms and solvent molecules are omitted for clarity. d Reduction of 2 with an excess amount of SmI2 under 1 atm of N2. e Reduction of 2-15N with SmI2 under 1 atm of 14N2. f Two-step synthetic cycle for cyanate anion. g Synthesis of isocyanate complex 4a. h Reduction of 4a using an excess amount of SmI2 under 1 atm of N2.
The detailed molecular structure of carbamate complex 2 was confirmed by X-ray crystallography. An ORTEP drawing of 2 is shown in Fig. 3b. Complex 2 has a distorted octahedral geometry around the molybdenum atom, where a carbamate ligand is coordinated to the molybdenum centre with the Mo1-N2 bond distance of 1.766(3) Å and the Mo1-N2-C24 angle of 173.4 (2)° in the apical position to the nitrogen atom of the pyridine ring. An iodo ligand and a chloro ligand are presented in cis and trans positions to the carbamate ligand, respectively. The bond distances and angles of 2 are similar to that of an acylimide complex [Mo(NCOCH2Ph)ICl(PNP)]33.
The reduction of carbamate complex 2 with 5 equiv of SmI2 in THF at room temperature for 2 h under an atmospheric pressure of dinitrogen afforded nitride complex 1 and samarium phenoxy complex [SmI2(OPh)] (3a) in 74 and 93% NMR yields, respectively. Further, samarium isocyanate species, such as [SmI2(NCO)], were formed as a formal composition formula (Fig. 3d). Samarium complex 3a was separately prepared from the reaction of SmI3 by ligand exchange with 1 equiv of KOPh, and the molecular structure of 3a was confirmed by preliminary X-ray crystallography (see Supplementary Information). Unfortunately, the molecular structure of the samarium isocyanate species has not been characterised until now. The amount of the formed NCO− after the hydrolysis of the reaction mixture was determined by ion chromatography (IC), where 0.75 equiv of NCO− were obtained based on 2 (75% IC yield). The formation of 1 and NCO− was characterised by electrospray ionisation–mass spectroscopy (ESI − MS) (m/z = 634.18) and IR (ν14NCO = 2170 cm−1), respectively.
Instead of the characterisation of the samarium isocyanate species, such as [SmI2(NCO)], a samarium isocyanate complex {[SmI(NCO)(18-crown-6)][I]}n (3b) (18-crown-6 = 1,4,7,10,13,16-hexaoxacyclooctadecane) was separately prepared in 60% yield from the reaction of SmI2 with 1 equiv of AgNCO as an oxidant with 1 equiv of 18-crown-6. The molecular structure of 3b was confirmed by preliminary X-ray crystallography (see Supplementary Information). The hydrolysis of 3b quantitatively afforded NCO−. Therefore, we suppose that the samarium isocyanate species [SmI2(NCO)] also quantitatively released NCO− after hydrolysis.
The transformation of carbamate complex 2 into nitride complex 1 and NCO−, as shown in Fig. 3d, is an unexpected and interesting result. To trace the origin of the N-atom in 1 and NCO− in this reaction, an isotope labelling experiment was conducted (Fig. 3e). The reduction of 15N-labelled carbamate complex [Mo(15NCO2Ph)ICl(PNP)] 2-15N with 5 equiv of SmI2 in THF at room temperature for 2 h under an atmospheric pressure of 14N2 gas afforded nitride complex [Mo(14N)I(PNP)] 1 and [SmI2(OPh)] 3a in 41 and 97% NMR yields, respectively, with 15N-labelled cyanate anion (15NCO−) in 62% IC yield. The formation of [Mo(14N)I(PNP)] 1 and 15NCO− was confirmed by ESI − MS (m/z = 634.18) and IR (ν15NCO = 2153 cm−1), respectively. This isotope labelling experiment suggests that, in this reaction system, [{MoI(PNP)}2(μ-N2)] (A, shown in Fig. 2a) was formed after the dissociation of 15N-labelled cyanate anion 15NCO− from the corresponding molybdenum complex, followed by the reaction with nitrogen gas (14N2) to form the nitride complex via the direct cleavage of the bridging dinitrogen on the dinitrogen-bridged dimolybdenum complex.
The experimental results shown in Fig. 3a, d, e suggest that the conversion of dinitrogen into cyanate anion under ambient reaction conditions is possible via the interconversion between the nitride and carbamate complexes (Fig. 3f).
To get more information on the reaction pathway, the one-electron reduction of carbamate complex 2 was conducted. The reduction of 2 with only 1.0 equiv of SmI2 in THF at room temperature for 10 min under an atmospheric pressure of Ar afforded the corresponding isocyanate complex [Mo(NCO)ICl(PNP)] (4a) in 68% yield (Fig. 3g). In the IR spectrum, an isocyanate (NCO) stretching vibration was observed at 2209 cm−1. A redshift (Δν15N = 14 cm−1) was observed in the 15N-labelled isocyanate complex [Mo(15NCO)ICl(PNP)](4a-15N). This result indicates that the isocyanate complex was formed from the carbamate complex via carbon–oxygen bond cleavage in the carbamate ligand during the reductive conversion. In contrast, the reduction of 2 with 1.1 equiv of CoCp*2 (Cp* = η5-C5Me5) in THF at room temperature for 16 h under an atmospheric pressure of Ar afforded another isocyanate complex [Mo(NCO)(OPh)Cl(PNP)] (4b) in 49% yield (see Supplementary Information). These results indicate that the high oxophilicity of samarium species plays an essential role to remove the phenoxy ligand from the molybdenum complex.
An ORTEP drawing of 4a is shown in Fig. 3c. Complex 4a exhibited a distorted octahedral geometry around the molybdenum atom, where an isocyanate ligand was coordinated to the molybdenum centre in the apical position to the nitrogen atom of the pyridine ring. An iodo ligand and a chloro ligand are presented in cis and trans positions to the isocyanate ligand, respectively. A detailed discussion of bond lengths and bond angles around the isocyanate ligand is difficult because of a slight disorder between the isocyanate and the chloro ligands.
The reduction of 4a with 5 equiv of SmI2 in THF at room temperature for 2 h under an atmospheric pressure of dinitrogen afforded the nitride complex [Mo(N)I(PNP)] 1 in 66% NMR yield, with cyanate anion NCO− in 75% IC yield (Fig. 3h). This result indicates that isocyanate complex 4a is an intermediate in the transformation of carbamate complex 2 into nitride complex 1 and cyanate anion, as shown in Fig. 3d. Contrarily, the reduction of 4b using 3 equiv of CoCp*2 with 3 equiv of NaI in THF at room temperature under an atmospheric pressure of dinitrogen did not afford nitride complex 1 but some paramagnetic species (see Supplementary Information). These experimental results indicate that the use of SmI2 as a reductant plays an essential role to transfer the isocyanate and chloro ligands from the molybdenum atom into the samarium atom. The removal of the isocyanate, phenoxy, and chloro moieties from 2 was necessary to regenerate nitride complex 1 via the direct cleavage of the bridging dinitrogen ligand on the dinitrogen-bridged dimolybdenum complex.
Based on these stoichiometric reactions shown in Fig. 3a, d–h, a detailed reaction pathway of the synthetic cycle for NCO− from dinitrogen mediated by molybdenum complexes can be proposed (Fig. 4). First, the carbamylation of nitride complex 1 with phenyl chloroformate affords the corresponding carbamate complex 2. Next, the reduction of 2 with an excess amount of SmI2 produces the dinitrogen-bridged dimolybdenum complex A and samarium complex, such as [SmI2(OPh)] 3a, [SmI2(NCO)], and [SmI2Cl], via the formation of the corresponding isocyanate complex 4a. Finally, the dinitrogen-bridged dimolybdenum complex A is converted into the starting nitride complex 1 via the direct cleavage of the bridging dinitrogen ligand of A.
(i) The carbamylation of nitride complex 1 with phenyl chloroformate affords carbamate complex 2. (ii) Reductive carbon−oxygen bond cleavage in the carbamate ligand of 2 produces isocyanate complex 4a. (iii) After one-electron reduction, the dissociation of the chloride ligand of 4a affords a five-coordinate isocyanate complex. A pair of the isocyanate complex sandwiches a dinitrogen molecule to form a dinitrogen-bridged dimolybdenum complex with a Mo–N≡N–Mo core, which is a precursor of [{MoI(PNP)}2(μ-N2)] A. (iv) The bridging N2 ligand of A undergoes a direct N≡N bond cleavage to regenerate 1.
Thus, this synthetic cycle for the formation of NCO− from dinitrogen mediated by the molybdenum–PNP complexes was closed in two steps, carbamylation and reduction (Fig. 3f). The overall yield of NCO− in the reaction of dinitrogen with phenyl chloroformate and SmI2 was 62% yield based on the molybdenum complex. This synthetic cycle provides a pathway for producing organonitrogen compounds from dinitrogen under mild reaction conditions with only two steps. The two steps are the fewest number of steps in the so-far reported synthetic cycles that consist of the combination of various stoichiometric reactions22,23,25,26. In addition, the formation of the isocyanate complexes from the reaction of the corresponding nitride complexes with CO was a key step for previously reported isocyanate derivatives synthesis from dinitrogen22,25. In contrast to these reports, the reaction of nitride complex 1 with CO did not afford an isocyanate complex but a cationic nitride carbonyl complex33. Therefore, this stoichiometric reaction is noteworthy as an alternative synthetic method for isocyanate derivatives from dinitrogen.
DFT calculations to support the proposed pathway
Here, we have performed density-functional theory calculations at the B3LYP-D3 level to theoretically examine the proposed synthetic cycle shown in Fig. 4. This synthetic cycle can be divided into four reaction steps (i)–(iv): (i) The carbamylation of nitride complex 1 with phenyl chloroformate affords carbamate complex 2. (ii) Reductive carbon−oxygen bond cleavage in the carbamate ligand of 2 produces isocyanate complex 4a. (iii) After one-electron reduction, the dissociation of the chloride ligand of 4a affords a five-coordinate isocyanate complex. A pair of the isocyanate complex sandwiches a dinitrogen molecule to form a dinitrogen-bridged dimolybdenum complex with a Mo–N≡N–Mo core, which is a precursor of [{MoI(PNP)}2(μ-N2)] A. (iv) The bridging N2 ligand of A undergoes a direct N≡N bond cleavage to regenerate 1. We experimentally and computationally demonstrated that the direct N≡N bond cleavage of A is a key reaction step in the catalytic transformation of dinitrogen into ammonia using Mo–PNP complexes30,32. Therefore, we would like to propose that steps (iii) and (iv) should be involved in the stoichiometric transformation of dinitrogen into NCO−. In this paper, we will focus on steps (i)–(iii), since step (iv) has been thoroughly investigated30.
Figure 5a presents a free energy profile at 298 K (ΔG298) in THF calculated for step (i). The carbonyl carbon atom of phenyl chloroformate attacked the nitride ligand of 1. The formation of the C–N bond was linked to the cleavage of the C–Cl bond (RC → TS → PC), where RC, TS and PC represent reactant complex, transition state and product complex, respectively. The released Cl– anion occupied the vacant coordination site of the molybdenum centre to afford 2. The formation of 2 was highly exergonic by 36.6 kcal/mol, with a very low activation free energy of ΔG298‡ = 5.3 kcal/mol.
a Free energy profile at 298 K (ΔG298) calculated for the reaction of nitride complex 1 with phenyl chloroformate yielding carbamate complex 2 in the closed-shell singlet state. Selected interatomic distances are presented in Å. b Free energy profile at 298 K (ΔG298) calculated for the formation of 4a from I in the ground spin state (see more details in Supplementary Information). Selected interatomic distances are presented in Å.
Next, we move to step (ii) in Fig. 4. At this step, the reduction of 2 was followed by the C–O bond cleavage to yield isocyanate complex 4a and a phenolate (PhO−). Cyclic voltammetry of 2 showed an irreversible reduction wave at Epc = −1.76 V vs. FeCp20/+ (Cp = η5-C5H5), which was assignable to the one-electron reduction of 2 (Supplementary Fig. 6). Therefore, our theoretical consideration started with the reduced species of 2 (complex I in Fig. 5b). The C–O bond cleavage of I smoothly occurred with a spin inversion between the doublet and quartet states. The formation of 4a (quartet) from I (doublet) went through TSI/4a (quartet), with a considerably low activation free energy of 1.6 kcal/mol, and proceeded in a highly exergonic way by 30.4 kcal/mol. During this reaction step, the Mulliken spin density was localised at the Mo centre, and no spin density was assigned to the leaving phenolate group (Supplementary Fig. 27). These results suggest that step (ii) can be regarded as the heterolysis of the C–OPh bond.
In step (iii), dimolybdenum complex A with a MoI–N≡N–MoI core was formed from MoIII complex 4a via two-electron reduction, the dissociation of both NCO and Cl ligands and the coordination of dinitrogen. First, the NCO or Cl ligand was dissociated from the six-coordinate Mo centre of 4a because a vacant coordination site must be prepared for incoming dinitrogen. As presented in Fig. 6a, a MoII complex II, the reduced species of 4a, undergoes a facile dissociation of Cl− (II → III + Cl−; ΔG298 = 1.1 kcal/mol). Cyclic voltammetry of 4a showed a quasi-reversible reduction wave at Epc = −1.86 V vs. FeCp20/+, assignable to the one-electron reduction of 4a (Supplementary Fig. 7). The NCO ligand should remain coordinated because the ΔG298 value for the dissociation of NCO− from II (II → MoICl(PNP) + NCO−) was calculated to be 11.8 kcal/mol. As described later, the release of the NCO group from the Mo centre will be accomplished by forming a dimolybdenum Mo–N≡N–Mo structure. Afterward, an incoming dinitrogen molecule is coordinated to a five-coordinate Mo–NCO complex III. The coordination of N2 smoothly proceeded in an exergonic way by 9.4 kcal/mol (III + N2 → IV). The generated complex IV reacted with another molecule of III to afford a six-coordinate dinitrogen-bridged dimolybdenum complex V, [{MoII(NCO)I(PNP)}2(μ-N2)]. The bridging dinitrogen strongly united two of the MoII–NCO units (III + IV → V; ΔG298 = − 19.8 kcal/mol). We here assume a stepwise reduction of V towards the generation of the five-coordinate dimolybdenum complex A, as presented in Fig. 6b. The one-electron reduction of V caused a facile dissociation of one of the NCO ligands occupying the trans positions of the bridging dinitrogen (VI → VII + NCO−; ΔG298 = −0.8 kcal/mol). The generated MoI–MoII complex VII readily liberated the remaining NCO ligand after the one-electron reduction (VIII → A + NCO−; ΔG298 = + 0.3 kcal/mol). The calculated results indicate that the formation of the Mo–N≡N–Mo core efficiently weakened the MoI–NCO bonds. We previously reported a similar situation in the reaction pathway for the formation of A from a dinitrogen-bridged MoI–MoI complex C, [{MoI(NH3)I(PNP)}2(μ-N2)], as shown in Fig. 2a32. In conclusion, from a theoretical point of view, the proposed synthetic cycle shown in Fig. 4 is reasonable if 4a can undergo two-electron reduction.
Here we would like to discuss the role of samarium complexes in the proposed synthetic cycle. As described in the experimental section, SmI2 may play a role to transfer the phenoxy and/or the isocyanate groups from the molybdenum atom into the samarium atom. We computationally examined the possibility of two transfer reactions from a thermodynamic viewpoint; (a) the transfer of PhO− of [4a···−OPh] to a cationic Sm(III) species [SmIIII2(THF)5]+, and (b) the transfer of NCO− of VI to [SmIIII2(THF)5]+. In the present calculations, we chose [SmIIII2(THF)5]+ as an acceptor of the leaving groups because the release of both PhO− and NCO− groups is preceded by the reduction of Mo complexes by SmI2. The structure of the Sm(III) species in THF was taken from the crystal structure of SmI2, [SmIII2(THF)5]36. The ΔG298 values for the transfer reactions were calculated to be −26.2 kcal/mol for reaction [4a···−OPh] + [SmI2(THF)5]+ → 4a + SmI2(OPh)(THF)4 + THF, and −10.5 kcal/mol for reaction VI + [SmI2(THF)5]+ → VII + SmI2(NCO)(THF)4 + THF, both of which are highly exergonic. These results could support that the presence of the Sm(III) species inhibits undesired side reactions such as formation of Mo-OPh intermediates and promotes subsequent reactions.
Catalytic formation of NCO− from dinitrogen
The two stoichiometric reactions in the synthetic cycle achieved above, carbamylation and reduction, can be carried out under almost the same reaction conditions. This result prompts us to investigate the catalytic reaction based on the stoichiometric reaction. Typical results are shown in Table 1. The reaction of the atmospheric pressure of dinitrogen with 36 equiv of SmI2 as a reductant and 12 equiv of phenyl chloroformate as a carbon-centred electrophile with a catalytic amount of 1 in THF at room temperature for 16 h afforded 0.45 equiv of NCO− based on the molybdenum atom of the catalyst (Table 1, run 1). Next, we examined other molybdenum–nitride complexes bearing different pincer ligands. The use of a molybdenum–nitride complex bearing a PPP-type pincer ligand [Mo(N)Cl(PPP)] (5)37 (PPP = bis(di-tert-butylphosphinoethyl)phenylphosphine) afforded a relatively low amount of NCO− (0.24 equiv based on the molybdenum atom) than that of 1. However, the use of a molybdenum–nitride complex bearing a carbene-based PCP-type pincer ligand [Mo(N)I(PCP)] (6)38 (PCP = 1,3-bis(di-tert-butylphosphinomethyl)benzimidazol-2-ylidene) as a catalyst slightly increased the amount of NCO− formed (0.74 equiv based on molybdenum atom of the catalyst; Table 1, runs 2 and 3). These results indicate that only a stoichiometric amount of NCO− was formed under these reaction conditions.
We envisaged that a direct reaction between SmI2 and phenyl chloroformate may suppress the catalytic formation of NCO−. Therefore, the slow addition of a THF solution containing phenyl chloroformate using a syringe pump to the mixture of 6 and SmI2 in a THF solution was investigated as a next step. This modification of the procedure substantially increased the amount of NCO− to 1.43 equiv based on the molybdenum atom of the catalyst (Table 1, run 4). Furthermore, when methyl chloroformate was used instead of phenyl chloroformate to reduce the reactivity of chloroformate ester towards SmI2, 2.51 equiv of NCO− were obtained based on the molybdenum atom of the catalyst (Table 1, run 5). This result indicates that a superstoichiometric amount of NCO− was formed from dinitrogen under ambient reaction conditions.
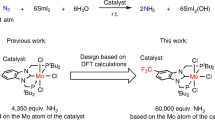
Contrarily, the use of ethyl chloroformate and isopropyl chloroformate was less effective than that of methyl chloroformate in this system (Table 1, runs 6 and 7). These results indicate that the bulkiness of the substituent on the chloroformate ester was critical for the present reaction system. We confirmed that catalyst, reductant, chloroformate ester and dinitrogen were essential factors to form a superstoichiometiric amount of NCO− based on the molybdenum atom of the catalyst (Table 1, runs 8–11). Finally, the slow addition of 24 equiv of methyl chloroformate for an increased period, such as 12 h, yielded 8.99 equiv of NCO− based on the molybdenum atom of the catalyst (75% yield based on SmI2; Table 1, run 12). This experimental result indicates that the catalytic formation of NCO− from dinitrogen was achieved under ambient reaction conditions. Thus, the amount of NCO− from the catalytic reaction is 9 times greater than the theoretical amount of NCO− produced from the stoichiometric reaction. As a result, we believe that this is the first successful example of the direct conversion of dinitrogen into organonitrogen compounds, such as NCO−, with transition metal complexes as catalysts under ambient reaction conditions.
Discussion
We demonstrated a two-step synthetic cycle of cyanate anion NCO− from dinitrogen with molybdenum complexes bearing a pyridine-based PNP-type pincer ligand under ambient reaction conditions. The reductive transformation of molybdenum–carbamate complex into molybdenum–isocyanate complex is a key step to promote the synthetic cycle. The overall yield of the formation of cyanate anion from the reaction of dinitrogen with SmI2 as a reductant and phenyl chloroformate as a carbon-centred electrophile was 62% based on the molybdenum atom of the complex. This synthetic cycle is the fewest number of steps in the so-far reported stoichiometric reactions. Based on this synthetic cycle, we achieved the catalytic reaction for the formation of NCO− from dinitrogen under ambient reaction conditions. We believe that the results described in this manuscript provide valuable information to achieve the catalytic and direct transformations of dinitrogen into more valuable organonitrogen compounds under ambient reaction conditions.
Methods
General information
Detailed experimental procedures, characterization of compounds and the computational details can be found in the Supplementary Methods, Supplementary Figs. 1–30, and Supplementary Tables 1–5. Cartesian coordinates are available in Supplementary Data 1.
Catalytic reaction
A typical experimental procedure for the catalytic reactions is described below. To a mixture of [Mo(N)I(PNP)] (1) (6.3 mg, 0.010 mmol) and SmI2 (198 mg, 0.36 mmol) was added THF (3 mL) under N2. Then, a THF solution (2 mL) containing phenyl chloroformate (18.8 mg, 0.12 mmol) was added, and the mixture was stirred at room temperature for 16 h. After the reaction, volatiles were removed in vacuo. A KOH aqueous solution (0.1 M, 25 mL) was added to the residual solid, and the mixture was stirred at room temperature for 30 min. The supernatant of the suspension was filtered through OnGuard II Na. The yield of NCO– in the residual solution was determined by ion chromatography.
Data availability
The X-ray crystallographic coordinates for structures reported in this article have been deposited at the Cambridge Crystallographic Data Centre (CCDC), under deposition number CCDC 2159843 ([1][I]), 2159844 (2), 2159845 (3a), 2159846 (3b•nCH3CN), 2159847 (4a), 2159848 (4b) and 2159849 ([Mo(NCO)Cl(PNP)]2(μ-N2)). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/ data_request/cif. Cartesian coordinates are available in Supplementary Data 1. All other data are available from the authors upon request.
References
Jia, H.-P. & Quadrelli, E. A. Mechanistic aspects of dinitrogen cleavage and hydrogenation to produce ammonia in catalysis and organometallic chemistry: relevance of metal hydride bonds and dihydrogen. Chem. Soc. Rev. 43, 547–564 (2014).
Boerner, L. K. Industrial ammonia production emits more CO2 than any other chemical-making reaction. Chemists want to change that. Chem. Eng. N. 97, 18–23 (2019).
Forrest, S. J. K., Schluschaß, B., Yuzik-Klimova, E. Y. & Schneider, S. Nitrogen fixation via splitting into nitrido complexes. Chem. Rev. 121, 6522–6587 (2021).
Masero, F., Perrin, M. A., Dey, S. & Mougel, V. Dinitrogen fixation: rationalizing strategies utilizing molecular complexes. Chem. – Eur. J. 27, 3892–3928 (2021).
Kim, S., Loose, F. & Chirik, P. J. Beyond ammonia: nitrogen–element bond forming reactions with coordinated dinitrogen. Chem. Rev. 120, 5637–5681 (2020).
Lv, Z.-J. et al. Direct transformation of dinitrogen: synthesis of N-containing organic compounds via N − C bond formation. Natl. Sci. Rev. 7, 1564–1583 (2020).
Hidai, M. Chemical nitrogen fixation by molybdenum and tungsten complexes. Coord. Chem. Rev. 185–186, 99–108 (1999).
Hidai, M. & Mizobe, Y. Recent advances in the chemistry of dinitrogen complexes. Chem. Rev. 95, 1115–1133 (1995).
Seino, H., Ishii, Y., Sasagawa, T. & Hidai, M. Synthesis and reactivities of pyrrolylimido complexes of molybdenum and tungsten: formation of pyrrole and N-aminopyrrole from molecular nitrogen. J. Am. Chem. Soc. 117, 12181–12193 (1995).
Lv, Z.-J., Huang, Z., Zhang, W.-X. & Xi, Z. Scandium-promoted direct conversion of dinitrogen into hydrazine derivatives via N–C bond formation. J. Am. Chem. Soc. 141, 8773–8777 (2019).
McWilliams, S. F. et al. Coupling dinitrogen and hydrocarbons through aryl migration. Nature 584, 221–226 (2020).
Laplaza, C. E. & Cummins, C. C. Dinitrogen cleavage by a three-coordinate molybdenum(III) complex. Science 268, 861–863 (1995).
Laplaza, C. E. et al. Dinitrogen cleavage by three-coordinate molybdenum(III) complexes: mechanistic and structural data. J. Am. Chem. Soc. 118, 8623–8638 (1996).
Curley, J. J., Sceats, E. L. & Cummins, C. C. A cycle for organic nitrile synthesis via dinitrogen cleavage. J. Am. Chem. Soc. 128, 14036–14037 (2006).
Figueroa, J. S., Piro, N. A., Clough, C. R. & Cummins, C. C. A nitridoniobium(V) reagent that effects acid chloride to organic nitrile conversion: synthesis via heterodinuclear (Nb/Mo) dinitrogen cleavage, mechanistic insights, and recycling. J. Am. Chem. Soc. 128, 940–950 (2006).
Guru, M. M., Shima, T. & Hou, Z. Conversion of dinitrogen to nitriles at a multinuclear titanium framework. Angew. Chem. Int. Ed. 55, 12316–12320 (2016).
Klopsch, I., Kinauer, M., Finger, M., Würtele, C. & Schneider, S. Conversion of dinitrogen into acetonitrile under ambient conditions. Angew. Chem. Int. Ed. 55, 4786–4789 (2016).
Klopsch, I., Schendzielorz, F., Volkmann, C., Würtele, C. & Schneider, S. Synthesis of benzonitrile from dinitrogen. Z. Anorg. Allg. Chem. 644, 916–919 (2018).
Schendzielorz, F. et al. Metal‐ligand cooperative synthesis of benzonitrile by electrochemical reduction and photolytic splitting of dinitrogen. Angew. Chem. Int. Ed. 58, 830–834 (2019).
Song, J., Liao, Q., Hong, X., Jin, L. & Mézailles, N. Conversion of dinitrogen into nitrile: cross-metathesis of N2-derived molybdenum nitride with alkynes. Angew. Chem. Int. Ed. 60, 12242–12247 (2021).
Knobloch, D. J., Lobkovsky, E. & Chirik, P. J. Dinitrogen cleavage and functionalization by carbon monoxide promoted by a hafnium complex. Nat. Chem. 2, 30–35 (2010).
Ishida, Y. & Kawaguchi, H. Nitrogen atom transfer from a dinitrogen-derived vanadium nitride complex to carbon monoxide and isocyanide. J. Am. Chem. Soc. 136, 16990–16993 (2014).
Keane, A. J., Farrell, W. S., Yonke, B. L., Zavalij, P. Y. & Sita, L. R. Metal-mediated production of isocyanates, R3EN = C = O from dinitrogen, carbon dioxide, and R3ECl. Angew. Chem. Int. Ed. 54, 10220–10224 (2015).
Falcone, M., Chatelain, L., Scopelliti, R., Živković, I. & Mazzanti, M. Nitrogen reduction and functionalization by a multimetallic uranium nitride complex. Nature 547, 332–335 (2017).
Schluschaß, B. et al. Cyanate formation via photolytic splitting of dinitrogen. JACS Au 1, 879–894 (2021).
Zhuo, Q. et al. Dinitrogen cleavage and functionalization with carbon dioxide in a dititanium dihydride framework. J. Am. Chem. Soc. 144, 6972–6980 (2022).
Fritz, M. et al. Photoelectrochemical conversion of dinitrogen to benzonitrile: selectivity control by electrophile- versus proton-coupled electron transfer. Angew. Chem. Int. Ed. 61, e202205922 (2022).
Bezdek, M. J. & Chirik, P. J. Expanding boundaries: N2 cleavage and functionalization beyond early transition metals. Angew. Chem. Int. Ed. 55, 7892–7896 (2016).
Schalke, P. M. in Ullmann’s Encyclopedia of Industrial Chemistry; pp. 669–672 (Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2000).
Arashiba, K. et al. Catalytic nitrogen fixation via direct cleavage of nitrogen–nitrogen triple bond of molecular dinitrogen under ambient reaction conditions. Bull. Chem. Soc. Jpn. 90, 1111–1118 (2017).
Ashida, Y., Arashiba, K., Nakajima, K. & Nishibayashi, Y. Molybdenum-catalysed ammonia production with samarium diiodide and alcohols or water. Nature 568, 536–540 (2019).
Arashiba, K., Tanaka, H., Yoshizawa, K. & Nishibayashi, Y. Cycling between molybdenum-dinitrogen and -nitride complexes to support the reaction pathway for catalytic formation of ammonia from dinitrogen. Chem. – Eur. J. 26, 13383–13389 (2020).
Itabashi, T., Arashiba, K., Kuriyama, S. & Nishibayashi, Y. Reactivity of molybdenum–nitride complex bearing pyridine-based PNP-type pincer ligand toward carbon-centered electrophiles. Dalton Trans. 51, 1946–1954 (2022).
Rérat, A., Michon, C., Agbossou-Niedercorn, F. & Gosmini, C. Synthesis of symmetrical diaryl ketones by cobalt-catalyzed reaction of arylzinc reagents with ethyl chloroformate. Eur. J. Org. Chem. 2016, 4554–4560 (2016).
Shi, R. & Hu, X. From alkyl halides to ketones: nickel-catalyzed reductive carbonylation utilizing ethyl chloroformate as the carbonyl source. Angew. Chem. Int. Ed. 58, 7454–7458 (2019).
Evans, W. J., Gummersheimer, T. S. & Ziller, J. W. Coordination chemistry of samarium diiodide with ethers including the crystal structure of tetrahydrofuran-solvated samarium diiodide, SmI2(THF)5. J. Am. Chem. Soc. 117, 8999–9002 (1995).
Arashiba, K. et al. Catalytic reduction of dinitrogen to ammonia by use of molybdenum–nitride complexes bearing a tridentate triphosphine as catalysts. J. Am. Chem. Soc. 137, 5666–5669 (2015).
Ashida, Y. et al. Catalytic production of ammonia from dinitrogen employing molybdenum complexes bearing N-heterocyclic carbene-based PCP-type pincer ligands. Chem. Rxiv https://doi.org/10.26434/chemrxiv-2022-jp6hz (2022).
Acknowledgements
The present project is supported by CREST, JST (JPMJCR1541). We thank Grants-in-Aids for Scientific Research (Nos. JP20H05671, 20K21203, and 22K19041) from JSPS. T.I. is a recipient of the JSPS Predoctoral Fellowships for Young Scientists. This paper is based on results obtained from a project, JPNP21020, commissioned by the New Energy and Industrial Technology Development Organization (NEDO).
Author information
Authors and Affiliations
Contributions
Y.N. and K.Y. conceived and designed this project. T.I., K.A., K.S., S.S. and S.K. conducted the experimental work, including the X-ray analysis. A.E. and H.T. conducted the theoretical studies. All authors discussed the results and drafted the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Itabashi, T., Arashiba, K., Egi, A. et al. Direct synthesis of cyanate anion from dinitrogen catalysed by molybdenum complexes bearing pincer-type ligand. Nat Commun 13, 6161 (2022). https://doi.org/10.1038/s41467-022-33809-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-33809-5
- Springer Nature Limited