Abstract
Highly regio- and enantioselective intermolecular hydroamination of alkenes is a challenging process potentially leading to valuable chiral amines. Hydroamination of alkenes via metal-catalyzed hydrogen atom transfer (HAT) with good regioselectivity and functional group tolerance has been reported, however, high enantioselectivity has not been achieved due to the lack of suitable ligands. Here we report a ligand-promoted cobalt-catalyzed Markovnikov-type selective radical hydroamination of alkenes with diazo compounds. This operationally simple protocol uses unsymmetric NNN-tridentate (UNT) ligand, readily available alkenes and hydrosilanes to construct hydrazones with good functional group tolerance. The hydrazones can undergo nitrogen–nitrogen bond cleavage smoothly to deliver valuable amine derivatives. Additionally, asymmetric intermolecular hydroamination of unactivated aliphatic terminal alkenes using chiral N-imidazolinylphenyl 8-aminoquinoline (IPAQ) ligands has also been achieved to afford chiral amine derivatives with good enantioselectivities.
Similar content being viewed by others
Introduction
Amine and its derivatives are significant in natural products and pharmaceutical chemistry (Fig. 1)1,2,3, (https://www.pharmacy-tech-test.com/top-200-drugs.html). So development of novel methodologies for the synthesis of various amines and their derivatives is highly desirable, particularly from simple and readily available starting materials.
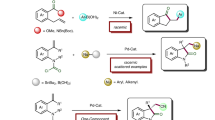
Metal-catalyzed hydrofunctionalization of readily available alkenes with nitrogen sources is one of the most efficient methods for the synthesis of nitrogen-containing molecules; however, to achieve the high regio- and enantioselectivities of this transformation is still a challenge (Fig. 2a)4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19. Among several activation strategies for alkene hydroamination, metal-catalyzed hydrogen atom transfer (HAT) reaction exhibits great Markovnikov selectivity and chemoselectivity (Fig. 2b)20. Okamoto and coworkers reported an early example of cobalt-catalyzed hydronitrosation of styrenes with nitric oxide using a hydroborane salt as a hydrogen source to form oximes21. Since Mukaiyama and coworkers22 reported the iron-catalyzed hydroamination of unactivated alkenes via HAT, using phenyl silane as a reductant and butyl nitrite as an aminating reagent, various aminating reagents, such as azo compounds23,24,25,26, nitro compounds27,28, diazo compounds29, azides24,30, and amides31,32 have been explored by many research groups, which offers a great opportunity for retrosynthetic possibility of new transformations. Although simple 1,3-dicarbonyl metal complexes could promote the reactions, stoichiometric amount or high loading of these complexes used to be necessary20,33. Additionally, due to the generation of radical intermediates, asymmetric intermolecular hydroamination of alkenes via radical HAT process has not been explored. So the development of new types of ligands for efficient radical hydroamination of alkenes via a HAT process is still highly desirable.
It should be noted that the additional ligands used to promote the formation of metal hydride species that could undergo classic alkene insertion34,35. Due to the inhibition of generation of the alkyl radical, the reactivity and selectivity of transformation might decrease dramatically. Otherwise, the tetra- or more dentate ligand-based metal complexes could promote the generation of radical; however, no highly enantioselective examples have been reported so far due to the weak coordination of alkyl radical with metal complexes. So the discovery of suitable ligand scaffolds for metal-catalyzed radical hydroamination of alkenes is a challenge and also has great potential (Fig. 2c).
Continuing our pursuit of efficient earth-abundant transition metal catalysis via ligand design36,37,38,39,40,41,42,43,44,45; here, we report the use of unsymmetric NNN tridentate ligands to promote the cobalt-catalyzed radical hydroamination of alkenes via HAT (Fig. 2d). Meanwhile, asymmetric reaction of unactivated aliphatic terminal alkenes using chiral N-imidazolinylphenyl 8-aminoquinoline (IPAQ) ligand has also been achieved to afford chiral amine derivatives with good enantioselectivities.
Results
Reaction optimization
The reaction of styrene 1a with ethyl 2-diazo-2-phenylacetate 2a in the presence of phenyl silane as a hydrogen donor in a solution of tetrahydrofura (THF) was selected as a model reaction. The reaction using Co(acac)2 afforded the hydroamination product 3a in a poor yield (entry 1, Table 1). The use of Co(OAc)2 did not promote this transformation (entry 2). The known ligands for HAT, such as tpp, dmg, dppe, salen, and tridentate half salen L1 have been tested; however, no reactivity or poor yields were observed (entry 3 and also see in Supplementary Table 1). NN-bidentate ligands such as 2,2′-bipyridine (bpy) and 1,10-phenanthroline (phen) could accelerate this reaction to give 3a in 44 and 37% yields, respectively (entries 4 and 5). The reaction using NNN tridentate N-oxazolinylphenyl picolinamide (OPPA) ligands42,46 L2 and L3 afforded 3a in 88 and 92% yields, respectively (entries 6 and 7). Then, 8-aminoquinoline group was used as a directing group on the ligand instead of 2-picolinamide. The reaction using N-oxazolinylphenyl 8-aminoquinoline (OPAQ) L4 as a ligand delivered 3a in 96% yield (entry 8). So, the standard conditions were identified as 0.36 mmol of alkene 1, 0.3 mmol of α-diazo ester compound 2, 5 mol% of Co(OAc)2, 6 mol% of L4, and 1.2 equiv. of PhSiH3 in a solution of THF (0.25 M) at room temperature (r.t.) for 12 h.
Substrate scope
With the optimized conditions in hand, the substrate scope was shown in Table 2. A variety of styrenes bearing electron-donating or electron-withdrawing substituents at para-, meta-, or ortho-position on the phenyl ring underwent the reactions to afford the corresponding benzylamine derivatives (3b–3t) in 50–95% yields. For more broad synthetic interests, various functional groups, such as halide, ether, aniline, organoboronate, sulfide, nitrile, aldehyde, ester, and free alcohol were well tolerated. Meanwhile, 2-naphthyl (1u), pyridine derivated alkenes (1v and 1w) could be delivered to the corresponding products in 71–92% yields. The reaction of alkyl-substituted terminal alkenes could undergo smoothly. Simple alkenes, such as 1-octene and 1-butene could be delivered to aliphatic amines in 77 and 72% yields, respectively. Alkenes, bearing phenyl, ether, tosyl, free alcohol, amine, amide, as well as nitrogen containing heterocycles, could be converted to the corresponding products in 49–84% yields. Notably, allylic alcohol and allylic amine derivatives could be reacted to deliver amino alcohol 3ae and diamine 3af in 49 and 84% yields, respectively. 1,1- and 1,2-disubstituted alkenes were suitable substrates and provided 3aj–3al in 79–86% yields. Natural products, such as amino acid, menthol, uracil, and estrone bearing a terminal alkene moiety could be employed to deliver products (3am–3ap) in 59–89% yields, which also demonstrated that this method could be suitable for late-stage functionalization of complicated molecules. Terminal alkene bearing anti-inflammatory drug naproxen was converted to 3aq in 61% yield. Configuration was confirmed by the X-ray diffraction of 3m, and the other products were then assigned by analogy to 3m.
Further applications
The gram-scale reaction could be smoothly performed to afford 3a in 94% yield (Fig. 3a). The reaction of 3m with 1,3-diketone afforded the pyrazole 4 in 86% yield (Fig. 3b). The three-step reactions containing alkene hydroamination, zinc-promoted reductive cleavage of N–N bond, and acyl protection of amine could be realized smoothly with once flash column chromatography purification process to deliver the corresponding amides 6 and 7 in 53–75% yields (Fig. 3c). The pharmaceutical substances and biologically active amines 8, such as appetite-suppressant drug clobenzorex, antianginal drug fendiline, and anti-hyperparathyroidism active NPS R568, could be efficiently synthesized in 32–45% yields from simple alkenes using a three-step synthetic protocol (Fig. 3c).
Substrate scope of asymmetric hydroamination
It should be noted that metal-catalyzed highly enantioselective hydroamination of unactivated aliphatic terminal alkenes is still an unsolved problem47. A primary study on asymmetric reaction using chiral unsymmetric NNN-tridentate ligand was explored. A chiral N-imidazolinylphenyl 8-aminoquinoline (IPAQ) ligand was designed and synthesized for asymmetric hydroamination of unactivated aliphatic terminal alkenes followed by cleavage of N–N bond and benzyl protection to afford chiral amine derivatives with up to 92.7 : 7.3 er (Table 3)48. The scope of substrate was quite broad. Various functional groups, such as halide, ether, and indole, could be tolerated. Alkene-bearing free alcohol moiety could underdo this transformation to afford the corresponding amide with a decreased yield. Chiral products with up to 98.6 : 1.4 er could be obtained after recrystallization. Particularly, the reaction of 1-butene afforded corresponding amide in 67% yield and 89.0 : 11.0 er (97.3 : 2.7 er after recrystallization). The chiral antihypertensive drug labetalol could be obtained from 14 via a known procedure49.
Mechanistic studies
Control experiments were conducted to illustrate the possible mechanism. The reaction in the presence of TEMPO did not occur, which indicated a possible radical pathway (Fig. 4a). The reaction of vinyl cyclopropane 28 afforded the ring-opening product 29 in 62% yield (Fig. 4b) via cleavage of the more substituted carbon–carbon bond50, which supported the radical promoted ring-opening pathway51. A deuterium-labeled experimental reaction of indene using PhSiD3 was conducted to afford 30 in 89% yield with 94% D and 1/1 dr (Fig. 4c) which demonstrated that the hydrogen came from hydrosilane and the carbon-centered radical formed as an intermediate.
Based on the experimental studies and previously reported literatures24,27,52,53,54,55,56, a possible mechanism was shown in Fig. 5. The cobalt hydride species A obtained from the reaction of Co(OAc)2 with ligand and silane could undergo a metal hydride HAT process to deliver the carbon radical intermediate and cobalt species B. Due to the possible redox non-innocent property of the ligand, the chemical valence of cobalt was not consistent. The cobalt species B might coordinate with diazo compound and undergo one electron oxidation with the carbon radical intermediate to afford the cobalt–carbon species C, which could undergo alkyl group migration from cobalt to nitrogen atom to generate cobalt oxide species D. The possibility that carbon radical directly attacked the cobalt coordinated diazo compound could not be ruled out. The cobalt species D could react with hydrosilane to regenerate the cobalt hydride species A and afford the vinyl silyl ether intermediate, which could undergo hydrolysis and isomerization to give the product. Further studies are undergoing in our laboratory to gain an accurate understanding of the mechanism.
Discussion
In summary, we reported the use of unsymmetric NNN-tridentate OPAQ ligand to promote the cobalt-catalyzed radical hydroamination of alkenes via HAT. The protocol uses simple and commercially available alkenes to deliver the amination products with good functional group tolerance and high Markovnikov selectivity. The hydrazone compounds could undergo nitrogen–nitrogen bond cleavage smoothly to afford a series of biologically active molecules. In particularly, asymmetric reaction of unactivated aliphatic terminal alkenes using newly developed chiral IPAQ ligand has also been achieved to afford chiral hydroamination products with good enantioselectivity. Further studies on asymmetric hydrofunctionalization of simple alkenes are undergoing in our laboratory.
Methods
Materials
For the optimization of reaction conditions and control experiments of alkene 1a (Supplementary Table 1), and for the experimental procedures and analytic data of compounds synthesized (Supplementary Methods). For nuclear magnetic resonance (NMR) spectra of compounds in this manuscript (Supplementary Fig. 1–166). For high-performance liquid chromatography (HPLC) spectra of compounds in this manuscript (Supplementary Fig. 167–185).
General procedure A for hydroamination of alkenes
A 25 mL Schlenk flask equipped with a magnetic stirrer and a flanging rubber plug was dried with flame under vacuum. When cooled to ambient temperature, it was vacuumed and flushed with N2 and repeated for three times. To the flask, Co(OAc)2 (0.015 mmol), L3 or L4 (0.018 mol), and THF (1.2 mL) were added. The flask was degassed and stirred for 30 min at r.t. Then, PhSiH3 (0.36 mol), diazo compound (0.3 mmol), and alkene (0.36 mmol) were added in sequence. After 12 h, the reaction was quenched with 10 ml of petroleum ether (PE) and the mixture was filtered through a pad of silica gel and washed with PE/EtOAc (5/1, 50 mL). The combined filtrates were concentrated and purified by flash column chromatography using PE/EtOAc as the eluent to afford the corresponding product.
General procedure B for asymmetric hydroamination of alkenes
A 25 mL Schlenk flask equipped with a magnetic stirrer and a flanging rubber plug was dried with flame under vacuum. When cooled to ambient temperature, it was vacuumed and flushed with N2 and repeated for three times. To the flask, Co(OAc)2 (0.015 mmol), IPAQ (0.018 mol), ethyl acetate (1.2 mL), and 2-ethylethanol (100 μL, 0.93 g/mL, 0.9 mmol) were added. The flask was degassed and cooled down to −10 °C and stirred for 30 min. Then, PhSiH3 (0.36 mol), diazo compound (0.3 mmol), and alkene (0.36 mmol) were added in sequence. After 24 h, the reaction was warmed up to r.t. and quenched with 10 ml of PE. The mixture was filtered through a pad of silica gel and washed with PE/EtOAc (5/1, 50 mL). The combined filtrates were concentrated to afford a yellow oil that was used for the next step without further purification.
Cleavage of N–N bond
To the above suspension, AcOH–THF–H2O (3:1:1 v/v/v, 3 mL) was added, followed by addition of activated Zn powder (0.5 g, 7.5 mmol) in several portions at r.t. After that, the mixed solution was warmed up to 60 °C and stirred until completion monitored by thin-layer chromatography (usually 3 h). Then, the reaction mixture was cooled down to r.t. and quenched with water (20 mL). The reaction mixture was basified with a solution of NaOH (6 N) until the solution turned clear (pH > 10) and then extracted with Et2O (20 mL × 4). The combined organic layers were dried over anhydrous Na2SO4, filtered, concentrated to give a yellow oil that was used for the next step without further purification.
Protection of free amines with Bz group
To the above oil, 3 mL (0.1 M) of THF, 55 μL (1.211 g/mL, 0.45 mmol) of BzCl, and 84 μL (0.728 g/mL, 0.6 mmol) of Et3N were added, followed by 2 h stirring. The mixture was quenched with water (20 mL) and then extracted with Et2O (20 mL × 4). The combined organic layers were dried over anhydrous Na2SO4, filtered, concentrated, and purified by flash column chromatography using PE/EtOAc as the eluent to give the corresponding amide.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information file. The X-ray crystallographic coordinates for structures of 3m has been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers CCDC 194446. The data can be obtained free of charge from the Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif. The experimental procedures and characterization of all new compounds are provided in Supplementary Information.
References
Funayama, S. & Cordell, G. A. (eds) Alkaloids: A Treasury of Poisons and Medicines (Waltham, MA, 2014).
Brown, B. R. The Organic Chemistry of Aliphatic Nitrogen Compounds. (Oxford University, New York, 1994).
Nugent, T. C. Chiral Amine Synthesis: Methods, Developments and Applications (Wiley-VCH, 2010).
Reznichenko, A. L. & Hultzsch, K. C. in Organic Reactions, Vol. 88 (eds Denmark, S. E.) Ch 1 (John Wiley & Sons Inc., 2006).
Huang, L.-B., Arndt, M., Gooßen, K., Heydt, H. & Gooßen, L. J. Late transition metal-catalyzed hydroamination and hydroamidation. Chem. Rev. 115, 2596–2697 (2015).
Leporl, C. & Hannedouche, J. First-row late transition metals for catalytic (formal) hydroamination of unactivated alkenes. Synthesis 49, 1158–1167 (2017).
Hannedouche, J. & Schulz, E. Hydroamination and hydroaminoalkylation of alkenes by group 3−5 elements: recent developments and comparison with late transition metals. Organometallics 37, 4313–4326 (2018).
Pirnot, M. T., Wang, Y.-M. & Buchwald, S. L. Copper hydride catalyzed hydroamination of alkenes and alkynes. Angew. Chem. Int. Ed. 55, 48–57 (2016).
Zhang, Z.-B., Lee, S. D. & Widenhoefer, R. A. Intermolecular hydroamination of ethylene and 1-alkenes with cyclic ureas catalyzed by achiral and chiral gold(i) complexes. J. Am. Chem. Soc. 131, 5372–5373 (2009).
Zhu, S.-L., Niljianskul, N. & Buchwald, S. L. Enantio- and regioselective cuh-catalyzed hydroamination of alkenes. J. Am. Chem. Soc. 135, 15746–15749 (2013).
Sevov, C. S., Zhou, J.-R. & Hartwig, J. F. Iridium-catalyzed, intermolecular hydroamination of unactivated alkenes with indoles. J. Am. Chem. Soc. 136, 3200–3207 (2014).
Yang, Y., Shi, S.-L., Niu, D.-W., Liu, P. & Buchwald, S. L. Catalytic asymmetric hydroamination of unactivated internal olefins to aliphatic amines. Science 349, 62–66 (2015).
Nishikawa, D., Hirano, K. & Miura, M. Asymmetric synthesis of α-aminoboronic acid derivatives by copper-catalyzed enantioselective hydroamination. J. Am. Chem. Soc. 137, 15620–15623 (2015).
Yu., F., Chen, P. & Liu, G.-S. Pd(II)-catalyzed intermolecular enantioselective hydroamination of styrenes. Org. Chem. Front. 2, 819–822 (2015).
Gurak, J. A. Jr, Yang, K. S., Liu, Z. & Engle, K. M. Directed, regiocontrolled hydroamination of unactivated alkenes via protodepalladation. J. Am. Chem. Soc. 138, 5805–5808 (2016).
Nakafuku, K. M., Fosu, S. C. & Nagib, D. A. Catalytic alkene difunctionalization via imidate radicals. J. Am. Chem. Soc. 140, 11202–11205 (2018).
Vanable, E. P. et al. Rhodium-catalyzed asymmetric hydroamination of allyl amines. J. Am. Chem. Soc. 141, 739–742 (2019).
Bathamonde, A., Rifaie, B. A., Martín-Heras, V., Allen, J. R. & Sigman, M. S. Enantioselective Markovnikov addition of carbamates to allylic alcohols for the construction of α-secondary and α-tertiary amines. J. Am. Chem. Soc. 141, 8708–8711 (2019).
Li, H.-Z., Shen, S.-J., Zhu, C.-L. & Xu, H. Direct intermolecular anti-Markovnikov hydroazidation of unactivated olefins. J. Am. Chem. Soc. 141, 9415–9421 (2019).
Crossley, S. W. M., Obradors, C., Martinez, R. M. & Shenvi, R. A. Mn-, Fe-, and co-catalyzed radical hydrofunctionalizations of olefins. Chem. Rev. 116, 8912–9000 (2016).
Okamoto, T., Kobayashi, K., Oka, S. & Tanimoto, S. Cobalt-catalyzed reaction of nitric oxide with aryl-substituted olefins in the presence of tetrahydroborate ion. J. Org. Chem. 52, 5089–5092 (1987).
Kato, K. & Mukaiyama, T. Iron(III) complex catalyzed nitrosation of terminal and 1,2-disubstituted olefins with butyl nitrite and phenylsilane. Chem. Lett. 21, 1137–1140 (1992).
Waser, J. & Carreira, E. M. Convenient synthesis of alkylhydrazides by the cobalt-catalyzed hydrohydrazination reaction of olefins and azodicarboxylates. J. Am. Chem. Soc. 126, 5676–5677 (2004).
Waser, J., Gaspar, B., Nambu, H. & Carreira, E. M. Hydrazines and azides via the metal-catalyzed hydrohydrazination and hydroazidation of olefins. J. Am. Chem. Soc. 128, 11693–11712 (2006).
Waser, J. & Carreira, E. M. Catalytic hydrohydrazination of a wide range of alkenes with a simple Mn complex. Angew. Chem. Int. Ed. 43, 4099–4102 (2004).
Zhang, Y. et al. Modular synthesis of alkylarylazo compounds via iron(III)-catalyzed olefin hydroamination. Org. Lett. 21, 2261–2264 (2019).
Gui, J.-H. et al. Practical olefin hydroamination with nitroarenes. Science 348, 886–891 (2015).
Zhu, K.-L., Shaver, M. P. & Thomas, S. P. Amine-bis(phenolate) iron(III)-catalyzed formal hydroamination of olefins. Chem. Asian J. 11, 977–980 (2016).
Zheng, J., Qi, J.-F. & Cui, S.-L. Fe-catalyzed olefin hydroamination with diazo compounds for hydrazone synthesis. Org. Lett. 18, 128–131 (2016).
Waser, J., Nambu, H. & Carreira, E. M. Cobalt-catalyzed hydroazidation of olefins: convenient access to alkyl azides. J. Am. Chem. Soc. 127, 8294–8295 (2005).
Shigehisa, H. et al. Catalytic hydroamination of unactivated olefins using a co catalyst for complex molecule synthesis. J. Am. Chem. Soc. 136, 13534–13537 (2014).
Zhou, X.-L. et al. Cobalt-catalyzed intermolecular hydrofunctionalization of alkenes: evidence for a bimetallic pathway. J. Am. Chem. Soc. 141, 7250–7255 (2019).
Ishikawa, H. et al. Total synthesis of vinblastine, vincristine, related natural products, and key structural analogues. J. Am. Chem. Soc. 131, 4904–4916 (2009).
Chen, J.-H., Guo, J. & Lu, Z. Recent advances in hydrometallation of alkenes and alkynes via the first row transition metal catalysis. Chin. J. Chem. 36, 1075–1109 (2018).
Ai, W.-Y., Zhong, R., Liu, X.-F. & Liu, Q. Hydride transfer reactions catalyzed by cobalt complexes. Chem. Rev. 119, 2876–2953 (2019).
Chen, J.-H., Cheng, B., Cao, M.-Y. & Lu, Z. Iron-catalyzed asymmetric hydrosilylation of 1,1-disubstituted alkenes. Angew. Chem. Int. Ed. 54, 4661–4664 (2015).
Guo, J. & Lu, Z. Highly chemo-, regio-, and stereoselective cobalt-catalyzed Markovnikov hydrosilylation of alkynes. Angew. Chem. Int. Ed. 55, 10835–10838 (2016).
Guo, J., Shen, X.-Z. & Lu, Z. Regio- and enantioselective cobalt-catalyzed sequential hydrosilylation/hydrogenation of terminal alkynes. Angew. Chem. Int. Ed. 56, 615–618 (2017).
Cheng, B., Lu, P., Zhang, H.-Y., Cheng, X.-P. & Lu, Z. Highly enantioselective cobalt-catalyzed hydrosilylation of alkenes. J. Am. Chem. Soc. 139, 9439–9442 (2017).
Guo, J., Cheng, B., Shen, X.-Z. & Lu, Z. Cobalt-catalyzed asymmetric sequential hydroboration/hydrogenation of internal alkynes. J. Am. Chem. Soc. 139, 15316–15319 (2017).
Cheng, B., Liu, W.-B. & Lu, Z. Iron-catalyzed highly enantioselective hydrosilylation of unactivated terminal alkenes. J. Am. Chem. Soc. 140, 5014–5017 (2018).
Chen, X., Cheng, Z.-Y., Guo, J. & Lu, Z. Asymmetric remote C-H borylation of internal alkenes via alkene isomerization. Nat. Commun. 9, 3939 (2018).
Cheng, Z.-Y. et al. Highly regioselective sequential 1,1-dihydrosilylation of terminal aliphatic alkynes with primary silanes. Chin. J. Chem. 37, 457–461 (2019).
Guo, J., Wang, H.-L., Xing, S.-P., Hong, X. & Lu, Z. Cobalt-catalyzed asymmetric synthesis of gem-bis(silyl)alkanes by double hydrosilylation of aliphatic terminal alkynes. Chem 5, 881–895 (2019).
Cheng, X.-K., Lu, H.-Z. & Lu, Z. Enantioselective benzylic C–H arylation via photoredox and nickel dual catalysis. Nat. Commun. 10, 3549 (2019).
Chen, X., Cheng, Z.-Y. & Lu, Z. Iron-catalyzed, Markovnikov-selective hydroboration of styrenes. Org. Lett. 19, 969–971 (2017).
Coombs, J. R. & Morken, J. P. Catalytic enantioselective functionalization of unactivated terminal alkenes. Angew. Chem. Int. Ed. 55, 2636–2649 (2016).
Das, S. et al. Asymmetric catalysis of the carbonyl-amine condensation: kinetic resolution of primary amines. J. Am. Chem. Soc. 139, 1357–1359 (2017).
Sanfilippo, C., Paternò, A. A. & Patti, A. Resolution of racemic ̀amines via lipase-catalyzed benzoylation: chemoenzymatic synthesis of the pharmacologically active isomers of labetalol. Mol. Catal. 449, 79–84 (2018).
O’Reilly, M. E., Dutta, S. & Veige, A. S. β-Alkyl elimination: fundamental principles and some applications. Chem. Rev. 116, 8105–8145 (2016).
Chen, C.-H., Shen, X.-Z., Chen, J.-H., Hong, X. & Lu, Z. Iron-catalyzed hydroboration of vinylcyclopropanes. Org. Lett. 19, 5422–5425 (2017).
Green, S. A. et al. The high chemofidelity of metal-catalyzed hydrogen atom transfer. Acc. Chem. Res. 51, 2628–2640 (2018).
Li, W. et al. New electrophilic addition of α-diazoesters with ketones for enantioselective C–N bond formation. J. Am. Chem. Soc. 133, 15268–15271 (2011).
Discolo, C. A., Touney, E. E. & Pronin, S. V. Catalytic asymmetric radical−polar crossover hydroalkoxylation. J. Am. Chem. Soc. 141, 17527–17532 (2019).
Dong, X.-Y. et al. A general asymmetric copper-catalysed Sonogashira C(sp3)–C(sp) coupling. Nat. Chem. 11, 1158–1166 (2019).
Gu, Q.-S., Li, Z.-L. & Liu, X.-Y. Copper(I)-catalyzed asymmetric reactions involving radicals. Acc. Chem. Res. https://doi.org/10.1021/acs.accounts.9b00381 (2019).
Acknowledgements
Financial support was provided by NSFC (21922107 and 21772171), Zhejiang Provincial Natural Science Foundation of China (LR19B020001), Center of Chemistry for Frontier Technologies, and the Fundamental Research Funds for the Central Universities (2019QNA3008).
Author information
Authors and Affiliations
Contributions
Z. L. designed the experiments. X. S., J. C. and Y. S. performed the experiments. Z. L. and X. C. designed the racemic ligands. Z. L. and Z. C. designed the chiral ligands. Z. L. and X. S. prepared this manuscript. X. S., J. C., Y. S. and Z. L. prepared the supplementary information.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shen, X., Chen, X., Chen, J. et al. Ligand-promoted cobalt-catalyzed radical hydroamination of alkenes. Nat Commun 11, 783 (2020). https://doi.org/10.1038/s41467-020-14459-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-14459-x
- Springer Nature Limited
This article is cited by
-
Regioselective hydroamination of unactivated olefins with diazirines as a diversifiable nitrogen source
Nature Communications (2024)
-
Catalytic enantioselective intramolecular hydroamination of alkenes using chiral aprotic cyclic urea ligand on manganese (II)
Nature Communications (2024)
-
Cobalt(III) hydride HAT mediated enantioselective intramolecular hydroamination access to chiral pyrrolidines
Science China Chemistry (2024)
-
Recent synthetic strategies for N-arylation of pyrrolidines: a potential template for biologically active molecules
Molecular Diversity (2024)
-
Intramolecular hydroamidation of alkenes enabling asymmetric synthesis of β-lactams via transposed NiH catalysis
Nature Catalysis (2023)