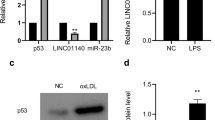
Abstract
Critical roles of several microRNAs have been implicated in atherosclerosis (AS). In this study, we studied the functional role of miR-140-5p in AS. An AS model was constructed in THP-1 macrophages challenged with oxidized low-density lipoprotein (ox-LDL). The expression of miR-140-5p was up- or downregulated with corresponding mimic or inhibitor regents. Our experiments showed that the levels of cell apoptosis and fatty acid accumulation were decreased in THP-1 macrophages treated with miR-140-5p mimic, whereas increased in those treated with miR-140-5p inhibitor. The levels of ROS (reactive oxygen species), MDA (malondialdehyde), TC (Triglyceride), and TG (total cholesterol) were reduced and the level of SOD (superoxide dismutase) was improved in miR-140-5p overexpressed THP-1 macrophages, which can be reversed with miR-140-5p depletion. Moreover, through bioinformatics analysis, we found toll-like receptor 4 (TLR4) was a potential target of miR-140-5p. Luciferase reporter assay demonstrated that miR-140-5p regulated TLR4 expression via binding 3′UTR of TLR4 in THP-1 macrophages. In ox-LDL challenged THP-1 macrophages, the expression of TLR4 was decreased after miR-140-5p mimic transfection, whereas improved after treatment with miR-140-5p inhibitors. As a conclusion, miR-140-5p can participate in inhibiting ox-LDL-induced oxidative stress and cell apoptosis via targeting TLR4 in macrophage-mediated ox-LDL induced AS.
Similar content being viewed by others
Introduction
Atherosclerosis (AS) accounts for various adverse vascular events, such as coronary artery disease, stroke, and peripheral artery disease. AS has become the leading cause of the most cardiovascular diseases (CVD) and is responsible for the most of CVD-related mortality world widely [1, 2]. Previous studies have well-established AS as an inflammation-associated disease, which appears as a persistent and chronic state [3, 4]. Generally, during the genesis of AS, the destructive incidents mediated damage of arterial wall launch the pathophysiologic process [5, 6]. These incidents may involve smoking, hypertension, and most commonly oxidized low-density lipoprotein (ox-LDL) [7,8,9,10]. The oxidation LDL occurs when the LDL cholesterol particles react with free radicals. Oxidized LDL could damage the vascular endothelium tissue, which may activate the immune responses and promote monocytes differentiate into macrophages with unregulated phagocytic activity [11, 12]. Ox-LDL activated macrophages may swallow abundant lipid and result in the formation of foam cells. Macrophages-derived foam cells is one of the hallmark events of AS [13, 14].
Generally, overloaded ox-LDL in blood flow could be uptake by macrophages through scavenger receptors, such as CD36, which contributes to the macrophages activation and increases reactive oxygen species (ROS) production [15, 16]. Excessive ROS can cause a series of cellular damage in macrophages, such as the structural or functional modification of diverse vital macromolecules, like RNA, DNA, lipids and protein, which may finally cause cell metabolic disorder and apoptosis [17,18,19]. As the pivotal role of ROS in the sequentially emerging events initiated by impaired macrophages, therapies designed to reduce the level of ROS may be of vital significance for the maintain of macrophage functions, which may attenuate the damage of ox-LDL thus eases the progression of AS.
Previous studies have well documented that toll-like receptor 4 (TLR4) as a significant participator in regulating innate immune and adaptive immune responses. TLR4 is abundantly expressed on various types of cells, particularly macrophages. TLR4 was also well studied in ox-LDL-induced AS as a participator in macrophage lipid accumulation and cell apoptosis [20,21,22].
MicroRNA is a kind of noncoding RNA no longer than 22 nucleotides, which has been reported to be generally involved in the development of AS [23,24,25]. MiR-140-5p was previously studied as an inflammatory factor in cancer and autoimmune diseases [26,27,28,29], however, its role in AS has not been clarified.
In this study, we reported the inhibitory role of miR-140-5p in the occurrence of ox-LDL induced macrophage-mediated AS, which in combination with further mechanism exploration, may provide new therapeutic targets for AS.
Materials and methods
Cell cultivation and transfection
THP-1 macrophages were maintained routinely in basic RPMI Media 1640 (Gibco, Grand Island, NY) and incubated at 37 °C with 5% CO2. Cells were differentiated into macrophages by stimulation of phorbol ester (PMA, 100 ng/ml; Sigma, St. Louis, USA). THP-1 macrophages (human macrophage cell line) were obtained from the Shanghai Institution of Biochemistry and Cell Biology.
RNA oligonucleotides, transfection Synthetic miR-140-5p mimics and inhibitor, are synthetic (Realgene, Nanjing, China). According to the manufacturer’s manual, using lipofectmin 2000 (Lifetec., MA, USA) transiently transfected for 48 h. After transfection for 48 h, cells were collected.
Co-culture of cells and proliferation assay
The THP-1 macrophages and the smooth muscle cells (HUVSMC) were co-cultured in an disconnect-type co-culture system [30, 31]. Totally, THP-1 macrophages (2 × 105) cells were challenged with ox-LDL (50 mg/L) for 24 h and then were transferred into the upper layer of the Transwell units. Similarly, 2 × 105 HUVSMC cells were transferred to the lower layer of the well (In Vitro Scientific, CA, USA). After incubation for 24 h in medium supplemented with FBS (10%), HUVSMC cells were harvested and then treated using an MTT assay Kit (Thermo Scientific, MA, USA) as the instruction descripts. Cell samples were examined using a Thermo Scientific Microplate Reader (Multiskan MK3, MA, USA).
Real-time polymerase chain reaction (PCR)
TRIzol reagent (Invitrogen, MA, USA) was used for the extraction of total RNAs from cells and then total RNAs were quantized. The RNA integrity was examined by NanoDrop 2000/2000c (Thermo Scientific, MA, USA). For obtaining cDNA, a kit of reverse transcriptase (TaKaRa, Tokyo, Japan) was accordingly used for reverse transcription of total RNAs (500 ng). Finally, real-time PCR was started to run with a Mastermix kit (Takara, Tokyo, Japan) on Step one plus machine (Applied Biosystems, Darmstadt, Germany) and the results were analyzed in three independent assays. Primer sequences: Human GAPDH:
Forward: 5′- GCCTCGTCCCGTAGACAAAA -3′ and
Reverse: 5′- GATGGGCTTCCCGTTGATGA -3′.
Flow cytometry analysis
Cells were cultured with ox-LDL (50 mg/L) or not for 24 h. Then, cell apoptosis was evaluated using Annexin V-FITC/PI Apoptosis Detection Kit (Biosciences, NY, USA) according to the reagent manual. In short, the cells were collected, and then washed with PBS. The cell pellet was resuspended in 100 μL binding buffer and then PE (5 μL) and Annexin APC (5 μL) were added. The cells were slightly vortexed, followed by incubation at room temperature for 10 minu in the dark. The FACS Calibur flow cytometer was performed to detect the treated cells with CellQuest software (BD Biosciences, NY, USA).
Oil-red staining
Cells were incubated for 24 h with ox-LDL (50 mg/L) (Unionbiol, Beijing, China) and immobilized with paraformaldehyde (4%) for 10 min. The cells were washed twice with PBS and then dyed with Oil-Red O solution which was prepared with 60% Oil-Red O dye (Sigma, MO, USA) and 40% water, for 15 min at room temperature, followed by washing cells twice with PBS. The microscope (Olympus, Tokyo, Japan) was used to observe the oil-red staining, and the intensity was detected with Image-Pro Plus 6.0.
Total cholesterol (TC) and triglyceride (TG) assay
According to the instructions, TG and TC were determined in cell lysis using a TG Quantification Kit (Abcam, MA, USA) and a Cholesterol Assay Kit (Abcam, MA, USA), respectively.
Evaluation of ROS and malondialdehyde (MDA) and superoxide dismutase (SOD)
The cells were gathered, washed twice with PBS and then suspended in 10 mM DCFHDA solution. Next, the samples were incubated for 20 min at 37 °C. The fluorescence intensity was detected using a fluorescence spectrophotometer (Hitachi, Tokyo, Japan) with an ROS Assay Kit (Beyotime, Shanghai, China). The cell lysates were gathered and centrifuged, and the levels of MDA and SOD in the supernatants were measured using a kit for Lipid Peroxidation MDA Assay (Beyotime, Shanghai, China) and a kit for SOD Activity Assay (BioVision, CA, USA) in accordance with the instructions. Finally, the 450 nm absorbance was measured using a Benchmark Microplate Reader (Bio-Rad, Hercules, CA, USA).
Western blot analysis
Total proteins of cultured cells extracting using RIPA buffer added with protease and phosphatase inhibitors and then were quantified by the method of Bradford assay (Bio-Rad Laboratories, Hercules, CA). The same amount of protein samples (40–50 μg) was isolated with SDS-PAGE, followed by transfer to the PVDF membrane. Immunoblotting was performed using antibodies for TLR4 (Santa Cruz, CA, USA), CD36 (CST, Ala, USA), and GAPDH (CST, Ala, USA). ECL kit (Pierce, Rockford, IL, USA) was used to visualize the bands, and the ImageJ software (NIH, Bethesda, MD, USA) was used to quantify the integration density of the bands. Antibody used are listed in Supplementary Table 1.
Statistical analysis
Statistical analysis was conducted with GraphPad and SPSS software (SPSS Inc., Chicago, IL, USA) and presented in the way of mean ± SEM. The evaluation of statistical differences between the different groups were assessed using Student’s t test and χ2 test. If not specified, all data was used for two-tailed tests. Single factor analysis of variance and LSD were conducted with the aim of making multiple comparisons. All experiments were conducted for three times. Finally, P < 0.05 was recognized statistically significant.
Results
Ox-LDL induces cell apoptosis and miR-140-5p expression
To evaluate the dynamic changes emerging in ox-LDL challenging macrophages, we established a serial of AS cell model in THP-1 macrophages treated with different concentration of ox-LDL for diverse time periods (Fig. 1). We observed that when challenging with ox-LDL for 24 h under different concentrations (0, 25, 50, and 100 mg/L), a dose-dependent matter presented in the promotion of cell apoptosis and the decrease of miR-140-5p expression (Fig. 1a, c). Following challenge with ox-LDL at 50 mg/L for different time periods (0, 12, 24, and 48 h), it indicated a time-dependent way in the improvement of cell apoptosis and the depression of miR-140-5p expression (Fig. 1b, d). These prompted that miR-140-5p may be involved as a potential participator in ox-LDL-induced macrophage apoptosis.
a, c THP-1 cells were treated with ox-LDL for 24 h. b, d THP-1 macrophages were challenged with 50 mg/L ox-LDL. Flow cytometry was carried out to assay the cell apoptosis level. (NC indicates negative control) (n = 4) Data were from independent experiments performed for triple times. *P < 0.05, compared to control group.
MiR-140-5p promotes the development of ox-LDL-induced macrophage-derived foam cells
As the formation of foam cells is one of the hallmarks in AS, we designed to investigate the potential role of miR-140-5p in the development of ox-LDL-induced macrophage-derived foam cells. Mimics and inhibitor of miR-140-5p were used to increase or decrease the level of miR-140-5p in THP-1 macrophages, respectively (Fig. 2a). THP-1 macrophages with miR-140-5p overexpression or downregulation were treated with ox-LDL (50 mg/L) for 24 h. It showed that ox-LDL-induced CD36 expression was significantly improved in miR-140-5p overexpressing cells and decreased in miR-140-5p knockdown cells (Fig. 2b). Lower levels of oil-red staining (Fig. 2c, d), TC and TG were observed when miR-140-5p was overexpressed, whereas miR-140-5p depletion increased the accumulation of oil and reduced the production of TC and TG (Fig. 2e, f).
a Expression of miR-140-5p were detected in THP-1 macrophages. b The expression levels of CD36 were assayed in THP-1 macrophages after ox-LDL (50 mg/L) treatment for 24 h (n = 4). c, d Oil-red staining results in THP-1 macrophages (Scale bar presented as 50 μm). e, f The levels of TC and TG in THP-1 macrophages were measured as indicated. (NC indicates negative control) (n = 4) Data were from independent experiments performed for triple times. #P < 0.05 vs. untreated group; *P < 0.05 vs. control group.
MiR-140-5p promotes ox-LDL-induced oxidative stress and macrophage apoptosis
In order to study the underlying mechanism of miR-140-5p in regulating the damage of macrophage under ox-LDL treatment, we evaluated the production of ROS, MDA, and SOD in THP-1 macrophages with miR-140-5p overexpression or downregulation. As it showed, significantly improved levels of ROS and MDA, but decreased SOD was found in miR-140-5p overexpressing cells (Fig. 3a–c). However, in miR-140-5p depletion cells, increased SOD, but depressed ROS production and MDA were observed (Fig. 3a–c). Flow cytometry assay indicated that cell apoptosis was promoted when miR-140-5p was upregulated, but attenuated after miR-140-5p downregulation (Fig. 3d, e). Moreover, to delineate the AS events, we performed the co-culture of macrophage and smooth muscle cells to explore their underlying crosstalk. As we found, in THP-1 macrophages challenged with ox-LDL (50 mg/L) for 24 h, miR-140-5p upregulation significantly promoted the proliferation of smooth muscle cells, HUVSMC (Fig. 4a, b). These indicated miR-140-5p as an inhibitor in AS events via enhancing the development of ox-LDL-induced macrophage-derived foam cells and improving the function of smooth muscle cells.
Cells were treated with ox-LDL at 50 mg/L for 24 h. a, b, c The levels of ROS, MDA and SOD, were evaluated. d The ratios of cell apoptosis were assayed by flow cytometry. PE: nucleus staining; APC: cell membrane staining; NC indicates negative control. (n = 4) Data were from independent experiments performed for triple times. #P < 0.05 vs. untreated group; *P < 0.05 vs. control group.
a, b The cell proliferation was evaluated with MTT assay. NC indicates negative control (n = 4). #P < 0.05, compared to co-cultured with THP-1 macrophages-NC group; *P < 0.05, compared to not co-cultured group. c The target regions of miR-140-5p on the 3′UTRs of TLR4. The seed sequence also exists in the recombinant luciferase mRNA transcribed from the wild-type plasmid TLR4-WT, but the seven nucleotides in these regions were deleted in the mutant sequence of plasmid TLR4-MT. d Dual luciferase reporter assay results. Luciferase activity of control group was normalized to 100%. e, f TLR4 and GAPDH expression were assayed by western blot and present in column. (NC indicates negative control) Data were from independent experiments performed for triple times. #P < 0.05, compared to untreated group; *P < 0.05, compared to control group.
MiR-140-5p functions in ox-LDL-treated macrophages via targeting TLR4
To further explore the molecular mechanisms of miR-140-5p involved in ox-LDL-induced AS, we retrieved the well-known database, miRanda, TargetScan and PicTar, as a result, TLR4 was predicted as a potential target of miR-140-5p. In addition, we found that miR-140-5p contained a seed complementary sequence to TLR4 3′UTR (Fig. 4c). As a validation, luciferase reporter assay was performed. The pGL3-TLR4-WT-3′UTR and pGL3–TLR4-MT-3′UTR were designed, and consequently, miR-140-5p could regulated TLR4 expression through binding 3′UTR of TLR4 in THP-1 macrophages (Fig. 4d). Further, we also observed that TLR4 expression was increased in miR-140-5p depletion THP-1 macrophages and reduced when miR-140-5p was overexpressed (Fig. 4e, f). In addition, the mechanism of miR-140-5p involved in AS is shown in Fig. 5.
Discussion
In our present study, we designed to investigate the potential role of miR-140-5p in the development of AS. With an AS in vitro model established in ox-LDL challenging macrophages, we found miR-140-5p could interact with TLR4, thus inhibited the production of several AS associated molecules and protected macrophages from apoptosis.
AS is a chronic inflammation associated disease characterized by injured vascular endothelium, enrolled inflammatory cell infiltration, along with lipid deposition and abundantly uptake by macrophages and smooth muscle cells [32, 33]. Functional impaired macrophages is one of the key points involved in AS, which may, as a result, lead to the disordered metabolism and aggrieved cell viability [12, 14].
When treating THP-1 macrophages with diverse density of ox-LDL for several time periods, we observed that the level of miR-140-5p was depressed in a dose-dependent and time-dependent way, with the cell apoptosis appearing in the similar manner. This provided miR-140-5p as a possible participator in the regulation of cell apoptosis and the occurrence of AS. For the subsequent experiments, we use THP-1 macrophages challenged with the density of ox-LDL (50 mg/L) for 24 h to study the functional participation of miR-140-5p in AS. Macrophage-derived foam cells are one of the symbolic features of AS, so we investigated the role of miR-140-5p in the development of foam cells. As it showed in miR-140-5p overexpressing cells, lipid accumulation, and the production of TC and TG were markedly decreased. Conversely, increased levels of oil-red staining, TC and TG were showed in miR-140-5p-inhibited cells. After challenging with 50 mg/L ox-LDL for 24 h, miR-140-5p overexpressing cells presented higher level of CD36, whereas in miR-140-5p depletion cells, CD36 expression was reduced. These demonstrated that miR-140-5p was involved in ox-LDL induced formation of macrophage-derived foam cells.
Previous studies have well demonstrated that cell apoptosis and oxidative stress of macrophages is a primary mechanism in ox-LDL induced AS [20, 34, 35]. In our study, we found that the apoptotic rates and oxidative stress level of macrophages were improved in miR-140-5p overexpressing cells, but decreased in miR-140-5p knockdown cells. We also found that the production of MDA was increased, but SOD was decreased in miR-140-5p overexpressing cells. However, in miR-140-5p depletion cells, increased level of SOD, but depressed level of MDA was observed.
To further probe into the potential targets of miR-140-5p in AS, we retrieved several databases, miRanda, TargetScan and PicTar. As a result, we found that miR-140-5p may bind to 3′UTR of TLR4 in THP-1 macrophages, which was subsequently demonstrated by luciferase report assay. We additionally evaluated the expression of TLR4 in ox-LDL-treated macrophages, which indicated that TLR4 was a downstream target of miR-140-5p in ox-LDL induced macrophage-mediated AS.
TLR4 is one of the most well-studied TLRs in immune responses. TLR4 has been reported in AS through interacting with several important proteins, such as PTPRO [20], PCSK9 [21], PKC-epsilon [22], SIRT1 [36], and so forth. Mutations and polymorphisms of TLR4 was also found in AS [37,38,39]. Therefore, TLR4 can be a potential therapeutic target for AS prevention.
In our present study, we found a novel miRNA, miR-140-5p, which was involved in the development of AS. In an ox-LDL challenging THP-1 macrophages cell model, we demonstrated the role of miR-140-5p in the inhibition of the formation of macrophage-derived foam cells through attenuating cell apoptosis and production of ROS. Moreover, we found the functional mechanism of miR-140-5p in preventing the genesis of AS was through targeting the 3′UTR region in the mRNA sequence of TLR4, thus decreasing the level of TLR4.
There are also several limitations in our study. First, in vivo study to validate the function of miR-140-5p in AS, and the interaction of miR-140-5p and TLR4 was not performed. Second, due to the limited number of AS samples we currently have, the diagnostic value of miR-140-5p was not investigated in this study. Third, the potential involvement of miR-140-5p in regulating other functions of macrophages, such as polarization, the production of inflammatory cytokines and so forth was not investigated in the present study. These will be well addressed in our further study.
In conclusion, we for the first time reported the protective roles of miR-140-5p in the development of AS, and further demonstrated a miR-140-5p/TLR4 pathway in macrophage-associated AS. With the addition of previous studies, our findings further provided miR-140-5p and TLR4 as promising therapeutic targets for AS.
Data availability
The data sets used in this study are available from the corresponding author on reasonable request.
References
Aronow HD, Beckman JA. Parsing atherosclerosis: the unnatural history of peripheral artery disease. Circulation. 2016;134:438–40.
Willeit P, Skroblin P, Kiechl S, Fernandez-Hernando C, Mayr M. Liver microRNAs: potential mediators and biomarkers for metabolic and cardiovascular disease? Eur Heart J. 2016;37:3260–6.
Damen M, Popa CD, Netea MG, Dinarello CA, Joosten LAB. Interleukin-32 in chronic inflammatory conditions is associated with a higher risk of cardiovascular diseases. Atherosclerosis. 2017;264:83–91.
Li J, Lin S, Vanhoutte PM, Woo CW, Xu A. Akkermansia muciniphila protects against atherosclerosis by preventing metabolic endotoxemia-induced inflammation in Apoe-/-Mice. Circulation. 2016;133:2434–46.
Pedra SR, Pedra CA, Abizaid AA, Braga SL, Staico R, Arrieta R, et al. Intracoronary ultrasound assessment late after the arterial switch operation for transposition of the great arteries. J Am Coll Cardiol. 2005;45:2061–8.
Tounian P, Aggoun Y, Dubern B, Varille V, Guy-Grand B, Sidi D, et al. Presence of increased stiffness of the common carotid artery and endothelial dysfunction in severely obese children: a prospective study. Lancet. 2001;358:1400–4.
Matsumoto T, Takashima H, Ohira N, Tarutani Y, Yasuda Y, Yamane T, et al. Plasma level of oxidized low-density lipoprotein is an independent determinant of coronary macrovasomotor and microvasomotor responses induced by bradykinin. J Am Coll Cardiol. 2004;44:451–7.
Mertens A, Holvoet P. Oxidized LDL and HDL: antagonists in atherothrombosis. FASEB. 2001;15:2073–84.
Rietzschel ER, Langlois M, De Buyzere ML, Segers P, De Bacquer D, Bekaert S, et al. Oxidized low-density lipoprotein cholesterol is associated with decreases in cardiac function independent of vascular alterations. Hypertension. 2008;52:535–41.
Niemann B, Rohrbach S, Miller MR, Newby DE, Fuster V, Kovacic JC. Oxidative stress and cardiovascular risk: obesity, diabetes, smoking, and pollution: Part 3 of a 3-Part series. J Am Coll Cardiol. 2017;70:230–51.
Binder CJ, Horkko S, Dewan A, Chang MK, Kieu EP, Goodyear CS, et al. Pneumococcal vaccination decreases atherosclerotic lesion formation: molecular mimicry between Streptococcus pneumoniae and oxidized LDL. Nature Med. 2003;9:736–43.
Nagy L, Tontonoz P, Alvarez JG, Chen H, Evans RM. Oxidized LDL regulates macrophage gene expression through ligand activation of PPARgamma. Cell. 1998;93:229–40.
Luo Y, Duan H, Qian Y, Feng L, Wu Z, Wang F, et al. Macrophagic CD146 promotes foam cell formation and retention during atherosclerosis. Cell Res. 2017;27:352–72.
Yang L, Yang JB, Chen J, Yu GY, Zhou P, Lei L, et al. Enhancement of human ACAT1 gene expression to promote the macrophage-derived foam cell formation by dexamethasone. Cell Res. 2004;14:315–23.
Rahaman SO, Lennon DJ, Febbraio M, Podrez EA, Hazen SL, Silverstein RL. A CD36-dependent signaling cascade is necessary for macrophage foam cell formation. Cell Metab. 2006;4:211–21.
Stewart CR, Stuart LM, Wilkinson K, van Gils JM, Deng J, Halle A, et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nature Immunol. 2010;11:155–61.
Forstermann U, Xia N, Li H. Roles of vascular oxidative stress and nitric oxide in the pathogenesis of atherosclerosis. Circulation Res. 2017;120:713–35.
Kim YW, Byzova TV. Oxidative stress in angiogenesis and vascular disease. Blood. 2014;123:625–31.
Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res. 2016;118:1808–29.
Liang C, Wang X, Hu J, Lian X, Zhu T, Zhang H, et al. PTPRO promotes oxidized low-density lipoprotein induced oxidative stress and cell apoptosis through toll-like receptor 4/nuclear factor kappaB pathway. Cell Physiol Biochem. 2017;42:495–505.
Tang ZH, Peng J, Ren Z, Yang J, Li TT, Li TH, et al. New role of PCSK9 in atherosclerotic inflammation promotion involving the TLR4/NF-kappaB pathway. Atherosclerosis. 2017;262:113–22.
Zuniga MC, Raghuraman G, Hitchner E, Weyand C, Robinson W, Zhou W. PKC-epsilon and TLR4 synergistically regulate resistin-mediated inflammation in human macrophages. Atherosclerosis. 2017;259:51–59.
Heumuller AW, Dimmeler S. Circular RNA Control of vascular smooth muscle cell functions. Circ Res. 2019;124:456–8.
Kong P, Yu Y, Wang L, Dou YQ, Zhang XH, Cui Y, et al. circ-Sirt1 controls NF-kappaB activation via sequence-specific interaction and enhancement of SIRT1 expression by binding to miR-132/212 in vascular smooth muscle cells. Nucleic Acids Res. 2019;47:3580–93.
Lucas T, Bonauer A, Dimmeler S. RNA therapeutics in cardiovascular disease. Circ Res. 2018;123:205–20.
Al-Modawi RN, Brinchmann JE, Karlsen TA. Multi-pathway protective effects of microRNAs on human chondrocytes in an in vitro model of osteoarthritis. Mol Ther Nucleic Acids. 2019;17:776–90.
Lindsay SL, Johnstone SA, McGrath MA, Mallinson D, Barnett SC. Comparative miRNA-based fingerprinting reveals biological differences in human olfactory mucosa- and bone-marrow-derived mesenchymal stromal cells. Stem Cell Rep. 2016;6:729–42.
Nagy B,Jr, Nagy B, Fila L, Clarke LA, Gonczy F, Bede O, et al. Human epididymis protein 4: a novel serum inflammatory biomarker in cystic fibrosis. Chest. 2016;150:661–72.
Papathanasiou I, Balis C, Trachana V, Mourmoura E, Tsezou A. The synergistic function of miR-140-5p and miR-146a on TLR4-mediated cytokine secretion in osteoarthritic chondrocytes. Biochem Biophys Res Commun. 2020;522:783–91.
Halloran BG, Grange JJ, So BJ, Baxter BT. Macrophage products inhibit human aortic smooth muscle cell proliferation and alter 1 alpha (I) procollagen expression. Ann Vasc Surg. 1997;11:80–84.
Tintut Y, Patel J, Territo M, Saini T, Parhami F, Demer LL. Monocyte/macrophage regulation of vascular calcification in vitro. Circulation. 2002;105:650–5.
Freigang S, Ampenberger F, Weiss A, Kanneganti TD, Iwakura Y, Hersberger M, et al. Fatty acid-induced mitochondrial uncoupling elicits inflammasome-independent IL-1alpha and sterile vascular inflammation in atherosclerosis. Nature Immunol. 2013;14:1045–53.
Moore KJ, Sheedy FJ, Fisher EA. Macrophages in atherosclerosis: a dynamic balance. Nature Rev Immunol. 2013;13:709–21.
Liao X, Sluimer JC, Wang Y, Subramanian M, Brown K, Pattison JS, et al. Macrophage autophagy plays a protective role in advanced atherosclerosis. Cell Metab. 2012;15:545–53.
Subramanian M, Thorp E, Tabas I. Identification of a non-growth factor role for GM-CSF in advanced atherosclerosis: promotion of macrophage apoptosis and plaque necrosis through IL-23 signaling. Circulation Res. 2015;116:e13–24.
Han J, Tong HQ, Cheng SY, Yang L, Chen HY, Chen JD, et al. Effect of Kangshuanyihao formula on the inflammatory reaction and SIRT1/TLR4/NF-kappaB signaling pathway in endothelial injury. Evid Based Complement Alternat Med. 2017;2017:9019765.
Kiechl S, Lorenz E, Reindl M, Wiedermann CJ, Oberhollenzer F, Bonora E, et al. Toll-like receptor 4 polymorphisms and atherogenesis. N Engl J Med. 2002;347:185–92.
Xie X, Shi X, Liu M. The roles of TLR gene polymorphisms in atherosclerosis: a systematic review and meta-analysis of 35,317 subjects. Scand J Immunol. 2017;86:50–58.
Yang IA, Holloway JW, Ye S, Southampton Atherosclerosis Study G. TLR4 Asp299Gly polymorphism is not associated with coronary artery stenosis. Atherosclerosis. 2003;170:187–90.
Funding
This work was supported by the Outstanding Youth Training Project from Shanghai Ninth People’s Hospital [Grant No. jyyq 08201607], the National Natural Science Foundation of China [Grant Nos. 81400802 and 81670735], and Clinical Research Project of Multi-Disciplinary Team, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine [Grant No. 201701022].
Author information
Authors and Affiliations
Contributions
FC designed and supervised the study; HL, ZM, JZ and MS performed the experiments; HL and ZM collected and analyzed the data; JZ and MS supported administration, technique, and materials; HL prepared the paper; FC revised the paper; All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, H., Mao, Z., Zhu, J. et al. MiR-140-5p inhibits oxidized low-density lipoprotein-induced oxidative stress and cell apoptosis via targeting toll-like receptor 4. Gene Ther 28, 413–421 (2021). https://doi.org/10.1038/s41434-020-0139-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41434-020-0139-7
- Springer Nature Limited
This article is cited by
-
Childhood obesity, metabolic syndrome, and oxidative stress: microRNAs go on stage
Reviews in Endocrine and Metabolic Disorders (2023)