Abstract
Lipid-rich drusen are the sine qua non of age-related macular degeneration (AMD), the leading cause of blindness in older adults in the developed world. Efforts directed at uncovering effective therapeutic strategies have led to the hypothesis that altered lipid metabolism may play a pathogenic role in AMD. This hypothesis is supported by the fact that: (1) drusen, the hallmark histopathologic feature of AMD, are composed of lipids, (2) polymorphisms of genes involved in lipid homeostasis are associated with increased odds of AMD, (3) metabolomics studies show that patients with AMD have alterations in metabolites from lipid pathways, and (4) alterations in serum lipid profiles as a reflection of systemic dyslipidemia are associated with AMD. There is strong evidence that statins, which are well described for treating dyslipidemia and reducing risk associated with cardiovascular disease, may have a role for treating certain cohorts of AMD patients, but this has yet to be conclusively proven. Of interest, the specific changes in serum lipoprotein profiles associated with decreased cardiovascular risk (i.e., high HDL levels) have been shown in some studies to be associated with increased risk of AMD. In this review, we highlight the evidence that supports a role for altered lipid metabolism in AMD and provide our perspective regarding the remaining questions that need to be addressed before lipid-based therapies can emerge for specific cohorts of AMD patients.
摘要
富含脂质的玻璃疣是年龄相关性黄斑变性 (AMD) 的重要特征, AMD是发达国家老年人失明的主要原因。为发现有效的治疗策略提出了一个假说, 即脂质代谢的改变可能在AMD中起致病作用。这一假说目前有以下事实支持: (1) 作为AMD标志性组织病理学特征的玻璃疣是由脂质组成, (2) 参与脂质稳态的基因多态性与AMD的患病几率增加有关, (3) 代谢组学研究表明AMD患者的脂质代谢产物发生了改变, 以及4) 反映全身性血脂异常的血脂谱变化与AMD有关。有强有力的证据表明, 他汀类药物可能在治疗某些AMD患者的队列中发挥作用, 但这一观点尚未得到确凿的证明, 且他汀类药物在治疗血脂异常和降低与心血管疾病相关的风险方面得到了证实。有趣的是, 一些研究表明, 血清脂蛋白谱中与心血管风险降低 (即高密度脂蛋白水平) 相关的特定变化与AMD风险增加有关。在这篇综述中, 我们强调了支持脂质代谢改变在AMD中作用的证据, 并就AMD患者特定队列中在脂质治疗前需要解决的问题提出了我们的观点。
Similar content being viewed by others
Introduction
Age-related macular degeneration (AMD) is a leading cause of blindness in older adults [1]. Early AMD is characterized by focal accumulation of lipids and proteins between Bruch’s membrane and the retinal pigment epithelium (RPE) known as drusen. The presence of multiple large drusen is a risk factor for progression to either advanced nonexudative/dry AMD, characterized by photoreceptor and RPE cell death known as geographic atrophy (GA), or advanced exudative/wet AMD, characterized by abnormal blood vessel growth beneath the eye known as choroidal neovascularization (CNV). Both forms of advanced disease cause devastating vision loss.
Despite the significant morbidity associated with AMD, there are limited treatment options for nonexudative AMD, which affects the vast majority of patients. Nutritional supplements in the form of vitamins and antioxidants may have a small role in slowing progression to advanced neovascular AMD but do not have a definitive role in treating nonexudative AMD [2, 3]. Anti-vascular endothelial growth factor agents have revolutionized the treatment landscape for patients with exudative AMD, but there is no equivalent option for patients with the nonexudative form of the disease.
This lack of treatment options for most AMD patients has led to significant efforts to identify cellular and molecular pathways involved in AMD pathogenesis. Numerous lines of evidence suggest that altered lipid homeostasis may contribute to AMD pathogenesis, including biochemical studies revealing that drusen are predominately composed of lipids [4,5,6], genetic studies that have identified polymorphisms in genes involved in lipid metabolism that are associated with AMD [7,8,9,10,11], and studies reporting that patients with AMD have altered serum lipoprotein profiles [12,13,14,15]. Nonetheless, since not all studies have yielded the same results, there is still not clear consensus regarding the specific role of dyslipidemia in AMD pathogenesis. In this review, we summarize the evidence that supports a role of altered lipid homeostasis in AMD pathogenesis and provide our perspective regarding important remaining research questions. This research may lead to much-needed, novel lipid-based therapies for early to intermediate AMD to prevent progression to advanced disease and blindness.
Biochemical evidence of altered lipid homeostasis in AMD
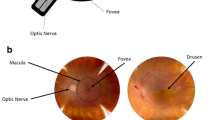
One of the most prominent clinical and histopathologic features of AMD is the focal accumulation of extracellular material between the inner collagenous layer of Bruch’s membrane and the basal lamina of the RPE. When present in a thin linear pattern, these accumulations are referred to as basal linear deposits (BLinD) [16, 17]. In contrast, when they are present in a focal, dome-shaped lesion, these accumulations are known as drusen (Fig. 1). BLinD and drusen are considered to be alternative forms of the same entity, and patients with AMD are orders of magnitude more likely to have BLinD and large drusen (>125 μm) compared with age-matched controls [16, 18]. Classical drusen are typically found within the fovea in association with cone photoreceptors and Müller cells. This is in contrast with another type of lipid-rich, AMD-associated lesion known as subretinal drusenoid deposits, which are located between the photoreceptors and the RPE and mainly in rod photoreceptor-rich regions of the macula [6, 19, 20]. There is speculation that these distinct lipid-rich lesions of the retina may reflect different underlying pathobiological processes of AMD.
The presence of a few small (less than 63 µm in diameter) drusen, also called drupelets, is considered a part of the normal aging process of the eye. Small drusen are associated with a rather small risk (i.e., <1%) of developing late AMD [21]. However, the presence of numerous medium (between 63 and 125 µm in diameter) or large (>125 µm in diameter) drusen, especially with indistinct borders, defines the transition from normal aging to early/intermediate AMD by multiple disease classification criteria, including that of the Age-Related Eye Disease Study and the Beckman Initiative for Macular Research Classification Committee (Table 1) [21, 22]. The reason why medium and large drusen define the transition from normal aging to disease is because they are associated with increased risk of progression to vision-threatening advanced AMD, including exudative AMD and GA [23, 24]. The estimated rate of progression to advanced AMD within 5 years approaches 50% for patients with large drusen and pigmentary abnormalities in both eyes [21].
The specific composition of drusen has been investigated in numerous biochemical and histological studies. Cumulatively, these studies reveal that drusen are highly heterogeneous but have significant lipid composition, supporting that altered lipid metabolism contributes to AMD pathogenesis [4]. Lipoprotein-derived particles that contain the physical forms of cholesterol seen in the core of atherosclerotic plaques with apolipoprotein B100, as well as various immunologic effectors, such as complement and molecules that promote macrophage migration, are seen in drusen and thought to contribute to the progression from nonexudative to exudative AMD. Almost a decade ago, Curcio et al. proposed the “oil spill” model to characterize the process by which lipids accumulate within the retina during physiologic aging and in the transition to AMD, culminating in drusen formation and progression to advanced AMD (Fig. 2) [5, 6]. Although the specific mechanism by which drusen biogenesis leads to GA and CNV remains an area of active investigation, there is a clear link between altered lipid homeostasis in the retina, formation of pathologic drusen, and the development of early/intermediate AMD.
Identification of specific proteins within drusen also support a role for altered lipid homeostasis in AMD pathogenesis [25]. Bergen et al. recently combined multiple published studies to curate a list of 89 drusen proteins [26]. Pathway analysis performed on this curated list of drusen proteins revealed that three of the five most significantly enriched canonical pathways involve lipid and cholesterol metabolism: liver X receptor/retinoid X receptor (LXR/RXR) activation, farnesoid X receptor (FXR)/RXR activation, and atherosclerosis signaling (Table 2).
Genetic evidence of altered lipid homeostasis in AMD
Near the turn of the millennium, polymorphisms in the apolipoprotein E gene (APOE) were found to be associated with AMD [7, 8]. Specifically, the APOE-ɛ4 allele was associated with decreased risk of AMD, while the APOE-ɛ2 allele was associated with increased risk of AMD, although one recent study challenged this idea by identifying a protective role for APOE-ɛ2 within a Northern Spanish population [27]. Interestingly, these risk profiles are opposite to those that have been identified for Alzheimer disease (AD), wherein the ɛ4 allele is associated with increased risk of AD and the ɛ2 allele is associated with decreased risk of AD. Two companion genome-wide association studies (GWAS) provided further associations between AMD and variants located near or within genes for hepatic lipase gene (LIPC), cholesterol ester transfer protein (CETP), ATP binding cassette subfamily A member 1 (ABCA1), and lipoprotein lipase (LPL) [9, 10]. A more recent, large GWAS of 16,144 patients and 17,832 non-AMD controls confirmed these associations between AMD and variants located near or in the ABCA1, APOE, CETP, and LIPC genes [11]. Table 3 summarizes the function of the proteins encoded by these genes. Recent meta-analysis of multiple GWAS confirmed that many AMD-associated loci were located near or within genes involved in high-density lipoprotein particle remodeling, cholesterol transporter activity, and macrophage foam cell differentiation, among other processes [28]. Although the specific cellular and molecular consequences of these variants remain under active investigation, these associations suggest that altered lipid homeostasis may contribute to AMD pathogenesis. Notably, the magnitude of altered risk associated with these gene variants is rather small, suggesting that these changes may occur in tandem with other biological processes that remain to be discovered.
Plasma metabolome
Untargeted plasma metabolomics studies also support the hypothesis that lipid homeostasis is altered in patients with AMD. In one cross-sectional, case–control study of 90 patients with AMD compared with 30 non-AMD controls, 87 plasma metabolites were found to be significantly different between patients with AMD versus controls. Of these 87 metabolites, the majority (82.8%) were lipid species, especially from the glycerophospholipid pathway [29]. A more recent and larger study of 2267 patients with AMD compared with 4266 non-AMD controls also identified 60 plasma metabolites that were significantly associated with AMD [30]. These metabolites included various lipoproteins, such as large and extra-large high-density lipoprotein (HDL) subclasses and very low-density lipoprotein (VLDL), which facilitate circulation of hydrophobic lipid species within the hydrophilic bloodstream.
Serum lipoprotein profiles in AMD
Given the many shared risk factors for patients with cardiovascular disease and AMD, many studies have focused on examining serum lipoprotein profiles in patients with AMD compared with non-AMD controls. Despite numerous studies that have examined this question, there is no clear consensus, though most recent studies tend to report an association between increased serum HDL and increased risk of AMD (Table 4).
To our knowledge, the first study to examine the association between serum cholesterol and AMD was a small study by Albrink and Fasanella, which found that patients with AMD (N = 11) had similar serum total cholesterol (257 ± 52 mg/dL) compared with young healthy controls (210 ± 32 mg/dL) and older healthy controls (238 ± 43 mg/dL) [31]. Twenty years later, Vidaurri et al. examined the association between cardiovascular risk factors and drusen in Jewish patients with AMD (Israel) [32]. When comparing patients with and without drusen, they found a statistically significant difference only in systolic and diastolic blood pressure (P = 0.011, P = 0.025, respectively). Interestingly, this was the first study to note a tendency towards a negative association between HDL, LDL, and drusen area.
The first large population-based studies to examine serum lipid levels in AMD were based on the National Health and Nutrition Examination Survey (NHANES) [33] and the Framingham Eye Study [34]. Although both failed to find any significant association between serum cholesterol and AMD, they found that blood pressure may be associated with AMD. Interestingly, the NHANES data suggested that there may be a trend towards higher cholesterol having a protective effect against AMD [33]. In contrast to these two studies, a subsequent large population-based study, the Eye Disease Case–Control Study, reported a significant association between elevated cholesterol and AMD 35. The next large study to look into serum lipoprotein levels and AMD was the Beaver Dam Eye Study, which did not find strong associations between cardiovascular risk factors and AMD but did identify an association between elevated serum HDL and incidence of GA [36, 37]. This association between elevated serum HDL and AMD was also identified by the Age-Related Macular Degeneration Risk Factors Study Group [38].
Given that both the Beaver Dam Eye Study and the Age-Related Macular Degeneration Risk Factors Study Group identified elevated serum HDL as a risk factor for AMD, van Leeuwen et al. were the first to examine the relationship between serum lipoprotein profiles and APOE genotype and the risk of early and late AMD. In the Rotterdam Study, they found that elevated serum HDL, but not total cholesterol, was indeed associated with increased risk of AMD and that this association remained even after controlling for APOE genotype [39]. A pooled analysis of the largest studies at the time from three continents was unable to provide a clear hypothesis to explain disparate conclusions regarding the association between serum cholesterol and AMD [40]. Dashti et al. suggested that these contradictory findings may stem from the fact that cholesterol and apolipoproteins found in drusen may arise from intraocular rather than systemic origin [41].
In the subsequent decade, emerging genetic evidence of variants in lipid-related genes conferring increased risk of AMD continued to fuel research into the association between serum lipids and AMD. A meta-analysis of three large, population-based cohorts, including data from the Blue Mountains Eye Study, the Beaver Dam Eye Study, and the Rotterdam Study found no associations between cholesterol measures and the incidence or progression of AMD when controlling for known risk alleles in lipid pathway genes, further muddling an already confusing picture regarding how systemic lipid profiles may influence AMD pathogenesis [42].
In 2016, Wang et al. performed a systematic review and meta-analysis examining the association between serum lipoprotein levels and AMD, analyzing 82,966 participants from 19 studies published at the time of their writing [12]. They identified a significantly increased risk of AMD associated with higher serum HDL (relative risk [RR]: 1.18 per 1 mmol/L increase; 95% confidence interval [CI]: 1.01–1.35). In contrast, higher serum total cholesterol (TC), LDL, and triglycerides (TG), were associated with a decreased risk of AMD (RR: 0.92–0.95). These findings challenge the paradigm that LDL is “bad“ cholesterol and that HDL is “good” cholesterol, as is case in the context of cardiovascular disease, although an earlier meta-analysis reported no clear associations [43].
Some recent studies that were published after this meta-analysis have yielded similar results. For example, a multi-center European cross-sectional study of 32,483 subjects found that higher serum HDL was associated with increased risk of AMD (odds ratio [OR]: 1.21 per 1 mmol/L increase; 95% CI: 1.14–1.29), whereas higher serum TG was associated with decreased risk of AMD (OR: 0.94 per 1 mmol/L increase; 95% CI: 0.91–0.97) [13]. Similarly, the Tsuruoka Metabolomics Cohort Study in Japan, which included 3,988 participants, reported a significant association between AMD and increased serum HDL (OR: 1.61 per 1 mmol/L increase; 95% CI: 1.17–2.23) and between AMD and decreased serum TG (OR: 0.78 per 1 mmol/L increase; 95% 0.64–0.96) in male participants [44]. In female participants, however, there was an association between early AMD and elevated TC (OR: 1.21; 95% CI: 1.01–1.44) and elevated serum LDL (OR: 1.26; 95% CI: 1.03–1.53) [44].
Other studies have corroborated the association between AMD and elevated serum HDL but not with other serum lipoproteins. A prospective cohort study of 5,344 participants from the United Kingdom found a similar association with elevated serum HDL conferring a higher risk of AMD (OR: 1.15 per 0.5 mmol/L increase, 95% CI 1.01–1.30) but no similar association with serum LDL and AMD [14]. Similarly, a Mendelian randomization investigation using published data from the International Age-Related Macular Degeneration Genomics Consortium with 33,526 participants of predominately European ancestry found evidence that serum HDL is a causal risk factor for AMD (OR: 1.22 per 1 standard deviation increase; 95% CI: 1.03–1.44) but no evidence of a causal association of serum LDL or TG with AMD [15]. Other Mendelian randomization studies in both European and Asian populations have further corroborated that serum HDL may be a causal factor in AMD pathogenesis [45, 46].
Notably, other recent cross-sectional and cohort studies have reported no significant associations between serum lipoprotein profiles and AMD [47, 48]. It remains unknown whether these discrepancies are due to differences in underlying study design, lack of appropriate sample size for sufficient statistical power, differences in case definition, or due to uncontrolled confounding. Taken together, although not all studies have yielded the same results, there is some evidence that there may be a link between systemic dyslipidemia and AMD, especially with HDL. This link between HDL and AMD is supported by mechanistic studies in disease models that suggest that targeting HDL interactions with Bruch’s membrane may slow the pathologic processes that leads to lipid accumulation, drusen biogenesis, and ultimately, development of AMD [49].
Cholesterol modulation as a therapeutic strategy in AMD
Given these possible associations between dyslipidemia and AMD, some have proposed the use of cholesterol modulation to treat early AMD. Observational studies in the last few decades have yielded inconsistent results. Some studies have reported a protective effect of statins on AMD, while others have reported a null result or even an adverse result whereby statins cause increased risk of exudative AMD; there is insufficient high-quality evidence to make definitive conclusions [50,51,52,53]. These discrepancies are likely due to underlying methodological limitations and bias. Moreover, there are numerous types of statins with differing lipophilicity, bioactivity, and dosage schemes. Since animal studies have confirmed that not all statins are able to penetrate the blood-retina barrier to the same extent [54], it is possible that the statin type and dose must be optimized to achieve the desired clinical outcome.
Our recent open-label, prospective clinical study reported that the use of high-intensity atorvastatin (80 mg daily) may lead to regression of drusen in some patients with associated improvement in visual acuity and no progression to advanced exudative AMD [55]. The patients in our study had, on average, a baseline cholesterol level of 208 ± 35 mg/dL with an average reduction of 62 ± 35 mg/dL. Of note, those who had regression of drusen volume after high-intensity atorvastatin (N = 10) showed a trend towards less reduction in total cholesterol after statin therapy compared to those who did not show change in drusen volume (N = 13) (−49 ± 31 mg/dL versus −71 ± 36 mg/dL; P = 0.14). In comparison, an earlier study by Guymer et al. reported that patients randomized to moderate-intensity simvastatin (40 mg daily; equivalent to atorvastatin 20 mg) had significantly reduced odds of AMD progression after adjusting for age, sex, smoking, and unilateral AMD status at baseline (P = 0.05), especially among patients who were homozygous for the AMD risk-conferring Y402H genotype of the complement factor H (CFH) gene [56]. Large randomized controlled trials and prospective cohort studies are necessary to definitively answer the question of what role statins and other lipid-lowering medications such as fibrates [57] may have in treating AMD. These future studies must consider the numerous heterogeneous subtypes of AMD, patients’ genetic profiles that contribute to differing levels of underlying risk, and lipid subspecies that may play disparate roles in disease pathogenesis. High-intensity statin therapy may be necessary to achieve regression of drusen burden and slow AMD progression. This would align with work in the cardiovascular field, which has suggested that intensive as opposed to moderate statin therapy may be necessary to stop atheroma growth and to yield the greatest protection against further cardiovascular events [58, 59].
Remaining questions
Taken together, there are multiple lines of evidence that support the idea that altered lipid homeostasis contributes to drusen biogenesis, which ultimately leads to the transition from physiologic aging to AMD. Although they have relatively small effect sizes, polymorphisms in genes involved with lipid metabolism perturb lipid and cholesterol homeostasis and thereby predispose individuals to developing AMD. Finally, although not all studies have reported consistent results, these perturbations in lipid homeostasis can be measured systemically based on serum lipoprotein profiles. Of interest, this evidence supports the idea that therapies targeted at restoring lipid homeostasis may have efficacy in reducing drusen biogenesis, preventing development of early AMD, or slowing development of advanced disease. However, there are numerous issues that need to be addressed before lipid therapies become a reality.
Challenging the paradigm of HDL as “good” cholesterol and LDL as “bad” cholesterol
Serum HDL is classically considered “good” cholesterol, as it is responsible for cholesterol transport from tissues to the liver, where it can be removed from the circulation. In contrast, serum LDL is considered ‘bad’ cholesterol, as it circulates cholesterol and can contribute to pathologic build-up of atherosclerotic plaques in the vasculature. Nonetheless, although not all studies have yielded the same results, this dogma has been challenged in studies of patients with AMD. In direct contrast with the pattern observed for cardiovascular disease, there is significant, yet not conclusive, evidence for elevated HDL as a risk factor for AMD. This notion of HDL as “good” cholesterol is also being challenged in the cardiovascular field as well. Most recently, the cardiovascular field has been moving toward the analysis of protein-defined subspecies of HDLs [60]. HDL particles contain small amounts of functional proteins that are used to define single subspecies. It appears that subspecies containing proteins involved in inflammation, immune response, thrombosis, and lipid metabolism are associated with higher risk of cardiovascular disease, whereas subspecies containing apolipoprotein C1 or E are associated with lower risk. Thus, risk determination should be made not by the total HDL level alone but by determining the balance between beneficial versus detrimental HDL subspecies and their local distribution. Future studies that measure lipids should take this into account. Increased efforts should also be made to identify and determine risk-conferring characteristics of HDL subspecies within the framework of AMD.
AMD: a disease of altered local or systemic lipid homeostasis or both?
Further studies are also necessary to determine the extent to which systemic modulation of lipid homeostasis may affect lipid homeostasis within the retina. This is especially important in the context of studies suggesting that local biosynthesis is the primary source of cholesterol within the retina [61]. Although systemic statin therapy has been well established as a therapy for dyslipidemia and can produce measurable improvements in serum lipid levels, it has not been definitively established that systemic statin therapy can alter local lipid profiles within the retina in humans. Moreover, local therapies may be necessary not only for improved bioavailability but also for the ability the optimize both systemic and local lipid profiles for patients with concomitant cardiovascular disease and AMD. Systemic lipid-lowering therapies may still be needed for AMD to create a permissive state for lipid efflux from the tissues, as well as for improving the function of circulating scavenger macrophages that are otherwise impaired by the accumulation of lipid material [62]. Targeting dyslipidemia may also have broad applicability for other neurodegenerative disorders beyond the retina, such as in AD, where dyslipidemia may also play a pathogenic role [63, 64].
Drusen: an innocent bystander or causative factor?
There is no doubt that the presence of multiple large drusen defines the transition from normal aging to early/intermediate AMD and increases risk of progression to advanced AMD. However, to date, it remains unclear whether drusen are a by-product of the underlying pathologic process or causative in the disease pathway or both. Future studies that carefully characterize the natural history of AMD and explore the effect of lipid-based therapies in reducing not only the quantity and size of drusen but also the risk of progression to advanced disease are essential to provide further insight into this issue.
Conclusions
Multiple lines of evidence support a role for altered lipid metabolism in AMD pathogenesis, from AMD lesion composition to systemic lipid changes and genetic risks associations. The exact association is far more complex than initially hypothesized with the most notable example being the association of elevated HDL with increased AMD risk being complicated by the importance of local lipid production and dyshomeostasis, as well as identification of HDL subspecies with potentially different risk profiles. Key takeaways are highlighted in Table 5. Although further research is necessary, the development of therapies that target alterations in lipid homeostasis and thereby address the underlying cellular and molecular pathophysiology of AMD would be revolutionary for many AMD patients.
References
Wong WL, Su X, Li X, Cheung CMG, Klein R, Cheng C-Y, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014;2:e106–16.
Vavvas DG, Small KW, Awh CC, Zanke BW, Tibshirani RJ, Kustra R. CFH and ARMS2 genetic risk determines progression to neovascular age-related macular degeneration after antioxidant and zinc supplementation. Proc Natl Acad Sci USA. 2018;115:E696–704.
Chew EY, Clemons TE, Agrón E, Sperduto RD, Sangiovanni JP, Kurinij N, et al. Long-term effects of vitamins C and E, β-carotene, and zinc on age-related macular degeneration: AREDS report no. 35. Ophthalmology. 2013;120:1604–11.e4.
Wang L, Clark ME, Crossman DK, Kojima K, Messinger JD, Mobley JA, et al. Abundant lipid and protein components of drusen. PLoS ONE. 2010;5:e10329.
Curcio CA, Johnson M, Rudolf M, Huang J-D. The oil spill in ageing Bruch membrane. Br J Ophthalmol. 2011;95:1638–45.
Curcio CA. Soft Drusen in Age-Related Macular Degeneration: Biology and Targeting Via the Oil Spill Strategies. Investig Ophthalmol Vis Sci. 2018;59:AMD160–81.
Schmidt S, Klaver C, Saunders A, Postel E, De La Paz M, Agarwal A, et al. A pooled case-control study of the apolipoprotein E (APOE) gene in age-related maculopathy. Ophthalmic Genet. 2002;23:209–23.
Klaver CC, Kliffen M, van Duijn CM, Hofman A, Cruts M, Grobbee DE, et al. Genetic association of apolipoprotein E with age-related macular degeneration. Am J Hum Genet. 1998;63:200–6.
Neale BM, Fagerness J, Reynolds R, Sobrin L, Parker M, Raychaudhuri S, et al. Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (LIPC). Proc Natl Acad Sci USA. 2010;107:7395–400.
Chen W, Stambolian D, Edwards AO, Branham KE, Othman M, Jakobsdottir J, et al. Genetic variants near TIMP3 and high-density lipoprotein-associated loci influence susceptibility to age-related macular degeneration. Proc Natl Acad Sci USA. 2010;107:7401–6.
Fritsche LG, Igl W, Bailey JNC, Grassmann F, Sengupta S, Bragg-Gresham JL, et al. A large genome-wide association study of age-related macular degeneration highlights contributions of rare and common variants. Nat Genet. 2016;48:134–43.
Wang Y, Wang M, Zhang X, Zhang Q, Nie J, Zhang M, et al. The association between the lipids levels in blood and risk of age-related macular degeneration. Nutrients. 2016;8:663.
Colijn JM, den Hollander AI, Demirkan A, Cougnard-Grégoire A, Verzijden T, Kersten E, et al. Increased high-density lipoprotein levels associated with age-related macular degeneration: evidence from the EYE-RISK and European Eye Epidemiology Consortia. Ophthalmology. 2019;126:393–406.
Yip JLY, Khawaja AP, Chan MPY, Broadway DC, Peto T, Tufail A, et al. Cross sectional and longitudinal associations between cardiovascular risk factors and age related macular degeneration in the EPIC-Norfolk Eye Study. PLoS ONE. 2015;10:e0132565.
Burgess S, Davey, Smith G. Mendelian randomization implicates high-density lipoprotein cholesterol-associated mechanisms in etiology of age-related macular degeneration. Ophthalmology. 2017;124:1165–74.
Curcio CA, Millican CL. Basal linear deposit and large drusen are specific for early age-related maculopathy. Arch Ophthalmol. 1999;117:329–39.
Sarks S, Cherepanoff S, Killingsworth M, Sarks J. Relationship of Basal laminar deposit and membranous debris to the clinical presentation of early age-related macular degeneration. Investig Ophthalmol Vis Sci. 2007;48:968–77.
Bressler NM, Silva JC, Bressler SB, Fine SL, Green WR. Clinicopathologic correlation of drusen and retinal pigment epithelial abnormalities in age-related macular degeneration. Retina. 1994;14:130–42.
Curcio CA. Antecedents of soft drusen, the specific deposits of age-related macular degeneration, in the biology of human macula. Investig Ophthalmol Vis Sci. 2018;59:AMD182–94.
Chen L, Messinger JD, Kar D, Duncan JL, Curcio CA. Biometrics, impact, and significance of basal linear deposit and subretinal drusenoid deposit in age-related macular degeneration. Investig Ophthalmol Vis Sci. 2021;62:33.
Ferris FL, Wilkinson CP, Bird A, Chakravarthy U, Chew E, Csaky K, et al. Clinical classification of age-related macular degeneration. Ophthalmology. 2013;120:844–51.
Ferris FL, Davis MD, Clemons TE, Lee L-Y, Chew EY, Lindblad AS, et al. A simplified severity scale for age-related macular degeneration: AREDS Report No. 18. Arch Ophthalmol. 2005;123:1570–4.
Nathoo NA, Or C, Young M, Chui L, Fallah N, Kirker AW, et al. Optical coherence tomography-based measurement of drusen load predicts development of advanced age-related macular degeneration. Am J Ophthalmol. 2014;158:757–61.e1.
Folgar FA, Yuan EL, Sevilla MB, Chiu SJ, Farsiu S, Chew EY, et al. Drusen volume and retinal pigment epithelium abnormal thinning volume predict 2-year progression of age-related macular degeneration. Ophthalmology. 2016;123:39–50.e1.
Crabb JW, Miyagi M, Gu X, Shadrach K, West KA, Sakaguchi H, et al. Drusen proteome analysis: an approach to the etiology of age-related macular degeneration. Proc Natl Acad Sci USA. 2002;99:14682–7.
Bergen AA, Arya S, Koster C, Pilgrim MG, Wiatrek-Moumoulidis D, van der Spek PJ, et al. On the origin of proteins in human drusen: the meet, greet and stick hypothesis. Prog Retin Eye Res. 2019;70:55–84.
Fernández-Vega B, García M, Olivares L, Álvarez L, González-Fernández A, Artime E, et al. The association study of lipid metabolism gene polymorphisms with AMD identifies a protective role for APOE-E2 allele in the wet form in a Northern Spanish population. Acta Ophthalmol. 2020;98:e282–91.
Han X, Gharahkhani P, Mitchell P, Liew G, Hewitt AW, MacGregor S. Genome-wide meta-analysis identifies novel loci associated with age-related macular degeneration. J Hum Genet. 2020;65:657–65.
Laíns I, Kelly RS, Miller JB, Silva R, Vavvas DG, Kim IK, et al. Human plasma metabolomics study across all stages of age-related macular degeneration identifies potential lipid biomarkers. Ophthalmology. 2018;125:245–54.
Acar İE, Lores-Motta L, Colijn JM, Meester-Smoor MA, Verzijden T, Cougnard-Gregoire A, et al. Integrating metabolomics, genomics, and disease pathways in age-related macular degeneration: the EYE-RISK consortium. Ophthalmology. 2020;127:1693–709.
Albrink MJ, Fasanella RM. Serum lipids in patients with senile macular degeneration. Am J Ophthalmol. 1963;55:709–13.
Vidaurri JS, Pe’er J, Halfon ST, Halperin G, Zauberman H. Association between drusen and some of the risk factors for coronary artery disease. Ophthalmologica. 1984;188:243–7.
Goldberg J, Flowerdew G, Smith E, Brody JA, Tso MO. Factors associated with age-related macular degeneration. An analysis of data from the first National Health and Nutrition Examination Survey. Am J Epidemiol. 1988;128:700–10.
Ferris FL. Senile macular degeneration: review of epidemiologic features. Am J Epidemiol. 1983;118:132–51.
Anon. Risk factors for neovascular age-related macular degeneration. The eye disease case-control study group. Arch Ophthalmol. 1992;110:1701–8.
Klein R, Klein BE, Franke T. The relationship of cardiovascular disease and its risk factors to age-related maculopathy. The Beaver Dam Eye Study. Ophthalmology. 1993;100:406–14.
Klein R, Klein BEK, Tomany SC, Cruickshanks KJ. The association of cardiovascular disease with the long-term incidence of age-related maculopathy: the Beaver Dam Eye Study. Ophthalmology. 2003;110:1273–80.
Hyman L, Schachat AP, He Q, Leske MC. Hypertension, cardiovascular disease, and age-related macular degeneration. Age-related macular degeneration risk factors study group. Arch Ophthalmol. 2000;118:351–8.
van Leeuwen R, Klaver CCW, Vingerling JR, Hofman A, van Duijn CM, Stricker BHC, et al. Cholesterol and age-related macular degeneration: is there a link? Am J Ophthalmol. 2004;137:750–2.
Tomany SC, Wang JJ, Van Leeuwen R, Klein R, Mitchell P, Vingerling JR, et al. Risk factors for incident age-related macular degeneration: pooled findings from 3 continents. Ophthalmology. 2004;111:1280–7.
Dashti N, McGwin G, Owsley C, Curcio CA. Plasma apolipoproteins and risk for age related maculopathy. Br J Ophthalmol. 2006;90:1028–33.
Klein R, Myers CE, Buitendijk GHS, Rochtchina E, Gao X, de Jong PTVM, et al. Lipids, lipid genes, and incident age-related macular degeneration: the three continent age-related macular degeneration consortium. Am J Ophthalmol. 2014;158:513–24.e3.
Chakravarthy U, Wong TY, Fletcher A, Piault E, Evans C, Zlateva G, et al. Clinical risk factors for age-related macular degeneration: a systematic review and meta-analysis. BMC Ophthalmol. 2010;10:31.
Sasaki M, Harada S, Kawasaki Y, Watanabe M, Ito H, Tanaka H, et al. Gender-specific association of early age-related macular degeneration with systemic and genetic factors in a Japanese population. Sci Rep. 2018;8:785.
Fan Q, Maranville JC, Fritsche L, Sim X, Cheung CMG, Chen LJ, et al. HDL-cholesterol levels and risk of age-related macular degeneration: a multiethnic genetic study using Mendelian randomization. Int J Epidemiol. 2017;46:1891–902.
Han X, Ong J-S, Hewitt AW, Gharahkhani P, MacGregor S. The effects of eight serum lipid biomarkers on age-related macular degeneration risk: a Mendelian randomization study. Int J Epidemiol. 2021;50:325–36.
Lüdtke L, Jürgens C, Ittermann T, Völzke H, Tost F. Age-related macular degeneration and associated risk factors in the population-based study of health in pomerania (SHIP-Trend). Med Sci Monit. 2019;25:6383–90.
Mao F, Yang X, Yang K, Cao X, Cao K, Hao J, et al. Six-year incidence and risk factors for age-related macular degeneration in a rural Chinese population: the Handan Eye study. Investig Ophthalmol Vis Sci. 2019;60:4966–71.
Kelly UL, Grigsby D, Cady MA, Landowski M, Skiba NP, Liu J, et al. High-density lipoproteins are a potential therapeutic target for age-related macular degeneration. J Biol Chem. 2020;295:13601–16.
Gehlbach P, Li T, Hatef E. Statins for age-related macular degeneration. Cochrane Database Syst Rev. 2016;2016:CD006927.
van Leeuwen R, Vingerling JR, Hofman A, de Jong PTVM, Stricker BHC. Cholesterol lowering drugs and risk of age related maculopathy: prospective cohort study with cumulative exposure measurement. BMJ. 2003;326:255–6.
Wilson HL, Schwartz DM, Bhatt HRF, McCulloch CE, Duncan JL. Statin and aspirin therapy are associated with decreased rates of choroidal neovascularization among patients with age-related macular degeneration. Am J Ophthalmol. 2004;137:615–24.
VanderBeek BL, Zacks DN, Talwar N, Nan B, Stein JD. Role of statins in the development and progression of age-related macular degeneration. Retina. 2013;33:414–22.
Mast N, Bederman IR, Pikuleva IA. Retinal Cholesterol Content Is Reduced In Simvastatin-treated Mice Due To Inhibited Local Biosynthesis Albeit Increased Uptake Of Serum Cholesterol. Drug Metab Dispos. 2018;46:1528–37.
Vavvas DG, Daniels AB, Kapsala ZG, Goldfarb JW, Ganotakis E, Loewenstein JI, et al. Regression of some high-risk features of age-related macular degeneration (AMD) in patients receiving intensive statin treatment. EBioMedicine. 2016;5:198–203.
Guymer RH, Baird PN, Varsamidis M, Busija L, Dimitrov PN, Aung KZ, et al. Proof of concept, randomized, placebo-controlled study of the effect of simvastatin on the course of age-related macular degeneration. PLoS ONE. 2013;8:e83759.
Wang K, Hsieh M-J, Chien H-W, Lee C-Y, Yeh C-B, Huang J-Y, et al. Medical compliance of fibrate and the decreased risk of age-related macular degeneration in dyslipidemia-related diseases: a population-based cohort study. Int J Environ Res Public Health. 2021;18:E301.
Nissen SE, Tuzcu EM, Schoenhagen P, Brown BG, Ganz P, Vogel RA, et al. Effect of intensive compared with moderate lipid-lowering therapy on progression of coronary atherosclerosis: a randomized controlled trial. JAMA. 2004;291:1071–80.
Cannon CP, Braunwald E, McCabe CH, Rader DJ, Rouleau JL, Belder R, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med. 2004;350:1495–504.
Sacks FM, Liang L, Furtado JD, Cai T, Davidson WS, He Z, et al. Protein-defined subspecies of HDLs (High-density lipoproteins) and differential risk of coronary heart disease in 4 prospective studies. Arterioscler Thromb Vasc Biol. 2020;40:2714–27.
Lin JB, Mast N, Bederman IR, Li Y, Brunengraber H, Björkhem I, et al. Cholesterol in mouse retina originates primarily from in situ de novo biosynthesis. J Lipid Res. 2016;57:258–64.
Sene A, Khan AA, Cox D, Nakamura REI, Santeford A, Kim BM, et al. Impaired cholesterol efflux in senescent macrophages promotes age-related macular degeneration. Cell Metab. 2013;17:549–61.
van der Kant R, Langness VF, Herrera CM, Williams DA, Fong LK, Leestemaker Y, et al. Cholesterol metabolism is a druggable axis that independently regulates tau and amyloid-β in iPSC-derived Alzheimer’s disease neurons. Cell Stem Cell. 2019;24:363–75.e9.
Kosowski M, Smolarczyk-Kosowska J, Hachuła M, Maligłówka M, Basiak M, Machnik G, et al. The effects of statins on neurotransmission and their neuroprotective role in neurological and psychiatric disorders. Molecules. 2021;26:2838.
Acknowledgements
The authors thank Danyel Cavazos for assistance with the figures.
Funding
DGV was supported by the Monte J. Wallace Chair in Retina, the Ines and Fred Yeatts Retina Research lab fund, the MLS Foundation, and the American Macular Degeneration Foundation. The funders played no role in study design, data collection and analysis, decision to publish, or preparation of the paper.
Author information
Authors and Affiliations
Contributions
Conceptualization: JBL, DGV. Writing—original draft: JBL, OAH. Writing—review and editing: JBL, OAH, DH, JWM, and DVG. Supervision: JBL, DGV. Funding acquisition: DGV.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lin, J.B., Halawa, O.A., Husain, D. et al. Dyslipidemia in age-related macular degeneration. Eye 36, 312–318 (2022). https://doi.org/10.1038/s41433-021-01780-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01780-y
- Springer Nature Limited
This article is cited by
-
APOB100 transgenic mice exemplify how the systemic circulation content may affect the retina without altering retinal cholesterol input
Cellular and Molecular Life Sciences (2024)
-
Identify risk factors affecting participation of Turkish women in mammography screening for breast cancer prevention
Breast Cancer Research and Treatment (2024)
-
New insight of metabolomics in ocular diseases in the context of 3P medicine
EPMA Journal (2023)