Abstract
Background/Objectives
Bioelectrical impedance is one of the most used clinical techniques to assess body composition; however, it is necessary that the available predictive equations are valid for the evaluated subjects. This study aimed to develop and cross-validate equations for fat-free mass (FFM) and lean soft tissue mass (LSTM) by bioelectrical impedance for Brazilian women, in addition to test the validity of other available equations.
Subjects/Methods
Cross-sectional study with 222 women aged 20–59 years, randomly divided into two groups: development and cross-validation. The standard technique for assessing fat mass, FFM and LSTM was dual energy X-ray absorptiometry. Paired t test, multiple regression, and Bland–Altman plots were used to test the validity of the proposed models, as well as to perform cross-validation of the models.
Results
The equations derived in this study were: FFM = 16.284 + 0.442 × (Height2/Resistance) − 0.13 × age + 0.302 × Weight − 0.121 × Waist Circumference; r2 = 0.86; SEE = 2.32 kg; and LSTM = 14.732 + 0.427 × (Height2/Resistance) − 0.125 × age + 0.291 × Weight − 0.115 × Waist Circumference; r2 = 0.92; SEE = 2.29 kg. In addition, the new equation for FFM showed better agreement when compared to another equation developed for a Brazilian population.
Conclusions
The newly developed equations provide a valid FFM and LSTM estimation and are recommended for Brazilian women with similar characteristics.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Body composition assessment is becoming increasingly important for monitoring weight loss. It is crucial for the prescription of personalized nutritional programs, physical exercise programs, and accurate adjustments of drug dosage for optimizing and preserving health with progressive weight loss [1].
Bioelectrical impedance (BIA) is a commonly used method to assess body composition in clinical practice and field research. Recent studies on BIA technology involve systems that incorporate multiple frequencies and multiple body segments. It is a quick and simple technique, which estimates total body water (TBW) for determining fat-free mass (FFM) and fat mass (FM), assuming a constant hydration of 73.2% in FFM [2, 3]. However, different population groups may present different amounts of FFM hydration, depending on sex, age, and ethnicity, suggesting the need for specific equations addressing these differences [4, 5]. Currently, there are also equations developed based on the BIA to estimate FFM without going through the assumption of hydration [6,7,8]. However, at the molecular level of body composition analysis, models thas provide fat- and free- bone mass models, usually refered as lean-soft tissue mass (LSTM) are absence with several papers providing BIA-appendicular lean mass or appendicular skeletal muscle mass derived equations, as described in a recent systematic review [9]. The prediction of LSTM can provide additional information that may detect changes resulting from malnutrition and sarcopenia as FFM does not discriminate lean tissue from bone quantity and quality. To perform these estimates, BIA measures resistance (R) and reactance (Xc), which, in combination with other variables such as height, body mass, sex, and age, are used in specific predictive equations for the studied population [10, 11]. Thus, when these equations are used to assess the body composition of different populations than the original ones, the results are inconsistent, indicating that they cannot be generalized to several populations [10].
In this way, several studies have proposed BIA predictive equations for different population groups [6, 12,13,14,15] or have tested their validity in different populations [8, 16]. In Brazil, few studies have developed BIA equations to estimate FFM of adult women using dual energy X-ray absorptiometry (DXA) as a reference technique, and there is no evidence of their validity in different regions of the country that present marked morphological differences related to the ethnic miscegenation [8]. Therefore, the objective of the present study was to develop and to cross-validate predictive equations of FFM and LSTM for Brazilian adult women, as well as to test the validity of other commonly used equations.
Materials and methods
This is a descriptive study with a cross-sectional design for the development and cross-validation of regression equations to estimate body composition.
Sample
The convenience sample consisted of 222 women, aged 20–59 years, from the northeast region of Brazil, who were recruited through dissemination among the participants of university extension projects from the Physical Education Department of the Federal University of Rio Grande do Norte (UFRN), nomination by participants, or social media (Fig. 1). After the inclusion in the study, the sample was randomly divided into two groups, development of predictive equation for FFM (111) and cross-validation (111), as previously described [13, 14]. For the sample size calculation, we considered a medium to small effect size (0.12) with four predictors (independent variables), with a type I error of 5% and a power of 82%. Using these parameters, a total of 111 participants would be required.
Inclusion criteria were women aged 20–59 years, with a body mass index (BMI) between 16.5 and 39.9 kg/m2, without any medical condition that could interfere with body composition results. Exclusion criteria were pregnancy; hypo or hypervolemic conditions, including diet, diuretic, or corticoid use; oedema; individuals with any physical disability; or individuals who had prosthesis that could alter the results of the body composition assessment.
Instruments and procedures
All data collections were conducted in a single visit by each participant to the laboratory to perform anthropometric measurements and BIA and DXA assessments. All participants were informed about the study protocol and signed a free and informed consent form (FICF). The research project was approved by the Research Ethics Committee of the University Hospital Onofre Lopes – HUOL/UFRN (protocol n°. 34804414.7.0000.5292).
Anthropometric measurements
Anthropometric measurements were performed by one physical education professional, who was properly trained in accordance with international recommendations [17]. Weight was measured using a digital scale with resolution of 0.1 kg of the brand Sanny®, model BL200PP (American Medical do Brasil, São Bernardo do Campo, Brazil), with the participants barefoot and wearing light clothes. Height was measured using a stadiometer of the brand Sanny® with resolution of 0.1 cm, model Caprice (American Medical do Brasil, São Bernardo do Campo, Brazil), with the participants barefoot and in orthostatic position. Waist circumference (WC) was measured at the midpoint between the iliac crest and the lower border of the last rib, using an anthropometric metal tape measure of the brand Sanny® with resolution of 0.1 cm, with the participants standing and the tape measure over bare skin at the measurement site. BMI was calculated by dividing body mass (kg) by the square of height (m).
BIA
Assessment by BIA, for the determination of resistance (R), reactance (Xc), and phase angle (PhA), was conducted with single frequency tetrapolar equipment (50 kHz) and 800 µA current, of the brand Sanny®, model BIA1010 (American Medical do Brasil, São Bernardo do Campo, Brazil). BIA equipment validation was periodically performed with a calibration device provided by the manufacturer. Validity values were considered normal if the resistance was not higher than 500 ± 5 ohm (Ω), and reactance was not higher than 52 ± 0.5 Ω, according to the manufacturer’s instructions. The choice of the Sanny bioimpedance equipment was made because it is the only one manufactured in Brazil, meaning easy access in the country and relatively low operating cost. In addition, a previous study developed equations to estimate the FFM of 20–59-year-old men, with high validity, using DXA as a reference technique [18].
In order to verify the quality of the measurements obtained by the equipment, reproducibility was calculated for a subgroup of 46 subjects from the population of this study: coefficient of variation (CV) of 0.17% and 0.72% for R and Xc, respectively, and technical error of measurement (TEM) of 0.76 Ω (0.22%) and 0.35 Ω (0.92%) for R and Xc, respectively.
Participants were assessed lying in supine position on a non-conductive stretcher. The skin was cleaned with 70% alcohol before placing the electrodes, which were positioned on the dorsal surface of the wrist, hand, ankle, and foot, in the right hemibody. The evaluated individuals were asked to fast for at least 4 h prior to the assessment, not to perform strenuous physical exercise in the previous 24 h, and not to consume alcohol in the previous 48 h. In addition, they should empty their bladder 30 min before the assessment. Resistance index (Ht2/R) was calculated by dividing the square of height (m) by resistance (Ω).
Total-body water (TBW) was estimated from the BIA model developed by Sun et al. [5].
DXA
DXA was performed with Lunar Prodigy equipment, model NRL 41990 (GE Lunar, Madison, WI, USA). The scan was conducted with the participants lying in supine position along the longitudinal axis of the midline of the table. Feet were positioned together and stuck at the level of the fingers to immobilize the legs, while the hands were held in the prone position within the scanning region of the equipment. The participants remained still during the digitalization process. Measurements were performed following the recommendations proposed by Nanna et al. [19]. Body composition was determined using version 13.6 enCoreTM 2011 software (GE Health Lunar). As described elsewhere, CV for FM, bone mineral content (BMC), and lean soft tissue mass (LSTM) using the current equipment were 0.74%, 0.28%, and 0.26%, respectively. TEM were 0.25 kg, 0.02 kg, and 0.25 kg to FM, BMC, and LSTM, respectively [8]. The FFM was obtained by sum of the BMC and LSTM (FFM = BMC + LSTM).
Statistical analysis
The non-parametric Kolmogorov–Smirnov test was applied to verify normal distribution of data, which was confirmed for all variables. Descriptive analysis consisted of mean and standard deviation of all study variables, and the comparisons between groups were performed by Student’s t test for independent samples. Stepwise multiple regression analysis was used to propose the predictive equations for FFM and LSTM. Stepwise regression analysis was conducted using FFM and LSTM obtained by DXA as dependent variables, and age, weight, height, WC, BMI, resistance, reactance, phase angle, and resistance index as possible independent variables. During model development, normality of residuals and homogeneity of variance were tested. Significance at p < 0.05 was established as the criterion for inclusion of a predictor whereas removal criteria were set at p > 0.1. If more than one variable remained in the model, and to assess multi-collinearity, a variance inflation factor (VIF) and the tolerance (reciprocal of VIF) were calculated for each independent variable [20], and a VIF < 10 or tolerance higher than 0.1 was considered appropriate [21]. To verify the validity of the proposed equations, the estimated mean results were compared to the mean results measured in DXA by paired t test. In addition, Pearson correlation coefficient (r), coefficient of determination (r2), and standard error of the estimate (SEE) were calculated. The approach proposed by Lin [22] was used for the concordance correlation coefficient (CCC) analysis in order to verify validity (Cb) and accuracy (ρ) between estimated and measured FFM and LSTM values. For the cross-validation of the equations proposed in the present study, multiple regression analysis was performed.
Also, the new BIA equations accuracy were evaluated using pure error (PE), which was calculated as the squared root of the sum of squared differences between the measurement and estimate of FFM and LSTM [5]. Bland–Altman plot was used to verify bias and concordance between FFM measurement and estimate, and between LSTM measurement and estimate, in which the limits of agreement (LOA) were defined as the mean of differences ± 1.96 standard deviations, including the analysis of the correlation between the mean and the difference of the methods. In addition, the same procedures were used to test the validity of two other equations, one proposed for Brazilian women, based on a sample from the south of the country [6], and another developed for the Swiss population [23], previously validated for women in the Brazilian population [24]. Analyses were carried out with the statistical package SPSS v.20.0 and MedCalc v.12.5.0. Statistical significance was considered as p < 0.05 for all tests.
Results
Characteristics of both groups, the development group and cross-validation group, are presented in Table 1. None of the variables presented significant difference in the comparison between groups.
Table 2 shows the regression models for the prediction of FFM (kg) and LSTM (kg), respectively. A preliminary model was developed to estimate FFM and another to estimate LSTM including anthropometric variables and BIA—namely, age, weight (W), height (H), WC, BMI, resistance (R), reactance (Xc), phase angle (PhA), and resistance index (Ht2/R). Only the variables that contribute to the estimates using a backward stepwise approach were used in the model. The performance of the models developed can be observed by the high coefficients of determination (FFM, r2 = 0.86; LSTM, r2 = 0.92) and low standard errors of the estimate (FFM, SEE = 2.32 kg; LSTM, SEE = 2.29 kg). The resulting prediction models included are presented as follows:
As an example of using the FFM and LSTM equations, we provided the actual values observed for the youngest woman (age = 20.0 years; Ht2/R = 41.59; weight = 67.5 kg; and WC = 81.3 cm) and for the oldest woman (age = 59.5 years; Ht2/R = 53.69; weight = 94.4 kg; and WC = 103.0 cm) included in this sample, who presented FFM of 42.6 kg (63.1%) and 48.3 kg (51.2%), respectively; and LSTM of 40.3 kg (59.7%) and 45.8 kg (48.6%), respectively. When measured by DXA, these women obtained FFM of 41.6 kg (61.7%) and 46.8 kg (49.6%); LSTM of 39.2 kg (58.0%) and 44.6 kg (47.3%), respectively.
From the results of FFM, it is possible to calculate FM in kilograms by subtracting FFM from body mass (FM = BM − FFM). Then, it is also possible to calculate body fat percentage by the mathematical expression: FM% = (FM × 100)/BM.
Estimated FFM and LSTM by the specific equations developed in the present study did not present significant difference in comparison with the values determined by DXA for both the development and cross-validation groups. All parameters used for proposing and validating the equations confirmed their validity (Table 3). In addition, no association was found between the mean and the difference of the methods (FFM, r = −0.021; p = 0.825; LSTM, r = 0.111; p = 0.098).
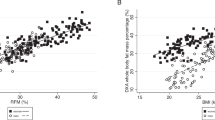
Figure 2 presents the LOA for FFM and LSTM between the standard method (DXA) and the BIA equations derived in the present study. Mean difference in the Bland–Altman plot was not different from zero in the cross-validation group for FFM (p = 0.839) and to LSTM (p = 0.724). The LOA of the FFM model ranged between −4.4 kg and 4.6 kg, and for the LSTM model ranged between −4.0 kg and 4.4 kg indicating acceptable limits of agreement between the developed equations and the reference method. Then, the validity of two other equations commonly used in Brazil, one developed in southern Brazil [6] and the other in Switzerland [23], was tested for the total sample of the present study, which demonstrated that both equations showed a significant difference for FFM when compared to the standard technique, in addition to lower validity parameters than the equation proposed in the present study for FFM (Table 4).
Mean differences in the Bland–Altman analysis were different from zero for both equations: Gonzalez et al. [6]. (p < 0.001) and Kyle et al. [23]. (p = 0.007). The LOA obtained by both equations, through Bland–Altman plot, are presented in Fig. 2 and demonstrate low agreement in FFM estimation. In addition, there was an association between the mean and the difference of the methods for both equations: Gonzalez et al. [6]. (r = 0.447; p < 0.001) and Kyle et al. [23]. (r = 0.198; p = 0.038).
Discussion
In the present study, the BIA equations were developed to estimate FFM and LSTM of Brazilian women aged 20–59 years, using the measurement obtained in DXA as reference. After its proposition, the equations went through a cross-validation process in a group of women of the same age group and with similar characteristics.
Among the variables obtained in the proposed models, resistance index (Ht2/R) proved to be the most relevant predictor, explaining 70% of the variability of both equations, FFM and LSTM. The use of BIA for estimating FFM by the resistance index is based on the electric properties of the human body. Resistance (R) of conductor is expressed by R = ρL2/V, so V = ρL2/R, wherein ρ is the conductor resistivity, L is the length, and V is volume [25, 26]. Therefore, as lean tissue contains a large amount of water, it presents low resistance to the flow of electric current, unlike FM. In this way, TBW allows to estimate FFM.
In this sense, body composition estimates by BIA assume that FFM has a fixed water amount of ~73.2%, when hydrometric techniques are used as standard [2, 3, 27]. However, it has been shown that the amount of body water can vary according to ethnicity, age, gender, physical activity level, and health status, among other factors, suggesting the need for specific equations according to the population to be assessed [28,29,30].
Several studies have demonstrated that the inclusion of age, sex, and anthropometric measurements significantly improves the predictive value of FFM estimation equations [8, 13, 15, 18, 31]. In the present study, the anthropometric variable that entered the model was WC, which means that for the sample studied, the larger this circumference, the lower the FFM and LSTM. This can be explained by the direct relationship between WC with visceral adipose tissue (VAT) and whole-body FM [32]. This anthropometric measure improved the proposed models, increasing the r2 and decreasing the SEE (Table 2).
In Brazil, only two studies were conducted to propose predictive equations of BIA to estimate body composition of women in the same age group of the present study. The first study was conducted with 58 women aged 18–30 years, university students of the state of Pernambuco, in which the authors derived five equations and proposed as the best results an equation that uses resistance, height, and body mass in the regression model [33]. It is important to highlight that the authors adopted hydrostatic weighing as standard, without the direct measurement of pulmonary residual volume, which can significantly compromise the body density values obtained [34]. In addition, the validation was only based on Pearson linear correlation, SEE, and Student’s t test for dependent samples, lacking a more robust analysis to evidence the validity of the referred equations.
More recently, in a second study [6], equations for men and women were derived, in which 155 women aged 20–70 years participated in the female group, from the city of Pelotas, southern Brazil. The equation developed in this study proved to be valid and showed better results when the authors compared it with other validated equations used in Brazil [23, 35]. Considering that the study suggests that this equation would be a good option to estimate FFM of the Brazilian population, we tested its validity in the sample of the present study, from the northeast region of Brazil.
Our results demonstrated that the referred equation did not meet the validity criteria to estimate FFM of the studied sample (Table 4; Fig. 2), which can be explained by the high miscegenation and ethnic differences found in the different regions of the country [8]. The validity of an equation developed for the Swiss population aged 20–94 years was also tested, which included 141 women and used DXA as a reference technique [23]. Although a study previously conducted in Brazil [24] with 120 women aged 60–81 years demonstrated validity of this equation to estimate FFM, in the sample of the present study, the equation did not demonstrate to be valid (Table 4; Fig. 2).
It is important to note that in addition to ethnic characteristics, differences in body size and shape between the populations that originated the equations and different bioimpedance equipment can also generate bias in the results [3, 36]. However, studies usually compare equipment with different evaluation characteristics, such as Foot-to-Foot and Hand-to-Foot [37] or segmental and whole-body [38]. Although the equation developed by Gonzalez et al. [6]. used BIA Quantum (RJL Systems, Detroit, MI, USA) and the equation developed by Kyle et al. [23]. used Xitron 4000B (Xitron Technologies, San Diego, CA, USA), both are tetrapolar devices for whole-body measurement, with the subjects in the supine position, using a frequency of 50 kHz and current of 800 µA. Moreover, Silva et al. [39] observed differences in raw BIA parameters between single frequency and the multifrequency devices at the frequency of 50 kHz. Nevertheless, the same characteristics were reproduced in the present study.
BIA was used to evaluate the amount of body water, which is extrapolated to FFM in some BIA devices assuming a relative constant value for FFM hydration [8]. The relative constancy for FFM hydration was proposed by previous authors [27, 40], showing relative stability in mammals of 73.2%. However, several studies showed that this value may vary according to age [41, 42], ethnicity [35, 43], obesity status [44], and other conditions that affect fluid distribution [26]. Indeed, in both the development and cross-validation groups, mean values of 75.6% and 75.2% of FFM hydration, assessed as the ratio between TBW/FFM based on a BIA prediction model for TBW determination [5] and FFM from DXA is slightly higher than the proposed value of 73.2%. Therefore, the choice of the equation to be used should take into account age, sex, ethnicity, and the health status of the subjects [11, 45].
Another aspect that deserves attention is that many devices do not allow the identification of the equations available in their software, which may compromise a correct identification of the most appropriate equation. If we consider that even among the gold standard techniques, such as DXA, there may be differences between the results obtained by different brands and models [46, 47], we can expect that the same occurs with bioelectrical impedance.
The best solution would be that manufacturers of bioimpedance equipment provide different predictive equations to be chosen according to the characteristics of the evaluated subjects. However, if the equipment does not have such a possibility, the professional can use the results of resistance, reactance, and other necessary variables obtained in a theoretically valid equation for the subject under evaluation. The lack of body composition predictive equations by BIA produced in Brazil, or validated in different regions of the country, justifies the implication of the present study for this area, with regard to research and its application in clinical practice.
A strength of this study included the development of a LSTM equation in addition to FFM considering the usefulness of discriminating FFM from bone mass to better assess and track interventions to manage malnutrition and sarcopenia related conditions. The equations developed in our study showed a high coefficient of determination and good limits of agreement in relation to the reference method, in addition to low VIF and high tolerance values; and all parameters used for the proposition and cross-validation of both models confirmed their validity for the studied population [5, 21, 22, 48], which can be used to monitor changes in FFM and LSTM resulting from dietary programs and physical exercises [1, 9, 49].
To exemplify the use of the equations developed in the present study, in the results we selected the minimum and maximum ages to present the practical implications as age is a variable that has a substantial impact on the biological predictors used in these models and can be useful for illustrative purposes.
However, several limitations should also be addressed. DXA is not considered a gold standard method to determine FFM at the molecular level. The four-compartment model (4 C) is the criterion method for assessing FM and FFM at the molecular level, given that the variability of the main FFM components (water, protein, and minerals) is assessed [50]. In addition, this study included a sample from only one region of the country, and ethnicity was not assessed. Therefore, miscegenation and ethnic differences suggest the need to validate the equation proposed in the present study in other regions of the country and with subjects of different ethnic origins. It should be noted that the new equations are only useful for Brazilian women with similar characteristics. In addition, further research should be conducted to test the accuracy of the new model in tracking FFM and LSTM.
Conclusions
The equations developed in the present study met the validation criteria to estimate FFM and LSTM, while the equations developed in the south of Brazil and in Switzerland for FFM were not considered valid for the studied sample. In this way, these new equations can be considered as a good alternative for body composition assessment by BIA of women with similar characteristics.
References
Becroft L, Ooi G, Forsyth A, King S, Tierney A. Validity of multi-frequency bioelectric impedance methods to measure body composition in obese patients: a systematic review. Int J Obes. 2019;43:1497–507. https://doi.org/10.1038/s41366-018-0285-9. e-pub ahead of print 2018/12/21.
Kushner RF, Schoeller DA. Estimation of total body water by bioelectrical impedance analysis. Am J Clin Nutr. 1986;44:417–24.
Lemos T, Gallagher D. Current body composition measurement techniques. Curr Opin Endocrinol Diabetes Obes. 2017;24:310–4. https://doi.org/10.1097/MED.0000000000000360.
Lohman TG. Applicability of body composition techniques and constants for children and youths. Exerc sport Sci Rev. 1986;14:325–57.
Sun SS, Chumlea WC, Heymsfield SB, Lukaski HC, Schoeller D, Friedl K, et al. Development of bioelectrical impedance analysis prediction equations for body composition with the use of a multicomponent model for use in epidemiologic surveys. Am J Clin Nutr. 2003;77:331–40. https://doi.org/10.1093/ajcn/77.2.331.
Gonzalez MC, Orlandi SP, Santos LP, Barros AJ. Body composition using bioelectrical impedance: development and validation of a predictive equation for fat-free mass in a middle-income country. Clin Nutr. 2019;38:2175–9.
Wang L, Hui SS-C, Wong SH-S. Validity of bioelectrical impedance measurement in predicting fat-free mass of Chinese children and adolescents. Med Sci Monit: Int Med J Exp Clin Res. 2014;20:2298.
Langer RD, Borges JH, Pascoa MA, Cirolini VX, Guerra-Junior G, Goncalves EM. Validity of bioelectrical impedance analysis to estimation fat-free mass in the army cadets. Nutrients. 2016;8:121. https://doi.org/10.3390/nu8030121.
Beaudart C, Bruyère O, Geerinck A, Hajaoui M, Scafoglieri A, Perkisas S, et al. Equation models developed with bioelectric impedance analysis tools to assess muscle mass: a systematic review. Clin Nutr ESPEN. 2020;35:47–62.
Dehghan M, Merchant AT. Is bioelectrical impedance accurate for use in large epidemiological studies?. Nutr J. 2008;7:26. https://doi.org/10.1186/1475-2891-7-26.
Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg P, Elia M, Gomez JM, et al. Bioelectrical impedance analysis–part I: review of principles and methods. Clin Nutr. 2004;23:1226–43. https://doi.org/10.1016/j.clnu.2004.06.004.
Jayawardena R, Wijetunga W, Ranasinghe P, Wanninayake L, Wickramasinghe VP. Assessment of body composition in Sri Lankan adults: development and validation of bioelectrical impedance prediction equation. Eur J Clin Nutr. 2019. https://doi.org/10.1038/s41430-019-0521-y. e-pub ahead of print 2019/11/02.
Aglago KE, Menchawy IE, Kari KE, Hamdouchi AE, Barkat A, Bengueddour R, et al. Development and validation of bioelectrical impedance analysis equations for predicting total body water and fat-free mass in North-African adults. Eur J Clin Nutr. 2013;67:1081–6. https://doi.org/10.1038/ejcn.2013.125.
Hastuti J, Kagawa M, Byrne NM, Hills AP. Proposal of new body composition prediction equations from bioelectrical impedance for Indonesian men. Eur J Clin Nutr. 2016;70:1271–7. https://doi.org/10.1038/ejcn.2016.113.
Hughes JT, Maple-Brown LJ, Piers LS, Meerkin J, O’Dea K, Ward LC. Development of a single-frequency bioimpedance prediction equation for fat-free mass in an adult Indigenous Australian population. Eur J Clin Nutr. 2015;69:28–33. https://doi.org/10.1038/ejcn.2014.54. e-pub ahead of print 2014/04/24.
Hofsteenge GH, Chinapaw MJ, Weijs PJ. Fat-free mass prediction equations for bioelectric impedance analysis compared to dual energy X-ray absorptiometry in obese adolescents: a validation study. BMC Pediatri. 2015;15:158. https://doi.org/10.1186/s12887-015-0476-7.
Lohman TG, Roche AF, Martorell R Anthropometric standardization reference manual, vol. 177. Champaign: Human kinetics books; 1988.
Costa RF, Masset KB, Sousa EC, Cabral BT, Dantas PS. Development and cross-validation of predictive equations of fat-free mass by bioelectrical impedance for Brazilian men aged 20 to 59 years old. Motricidade. 2018;14:26–32.
Nana A, Slater GJ, Stewart AD, Burke LM. Methodology review: using dual-energy X-ray absorptiometry (DXA) for the assessment of body composition in athletes and active people. Int J sport Nutr Exerc Metab. 2015;25:198–215.
Guo S, Chumlea WC. Statistical methods for the development and testing of predictive equations. Hum body composition. 1996;10:191–202.
Ndagire CT, Muyonga JH, Odur B, Nakimbugwe D. Prediction equations for body composition of children and adolescents aged 8–19 years in Uganda using deuterium dilution as the reference technique. Clin Nutr ESPEN. 2018;28:103–9. https://doi.org/10.1016/j.clnesp.2018.09.004. e-pub ahead of print 2018/11/06.
Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics. 1989;45:255–68. e-pub ahead of print 1989/03/01.
Kyle UG, Genton L, Karsegard L, Slosman DO, Pichard C. Single prediction equation for bioelectrical impedance analysis in adults aged 20–94 years. Nutrition. 2001;17:248–53.
Rech CR, Cordeiro BA, Petroski EL, Vasconcelos FA. Validation of bioelectrical impedance for the prediction of fat-free mass in Brazilian elderly subjects. Arq Bras Endocrinol Metabol. 2008;52:1163–71.
Houtkooper LB, Lohman TG, Going SB, Howell WH. Why bioelectrical impedance analysis should be used for estimating adiposity. Am J Clin Nutr. 1996;64:436S–448S. https://doi.org/10.1093/ajcn/64.3.436S.
Khalil SF, Mohktar MS, Ibrahim F. The theory and fundamentals of bioimpedance analysis in clinical status monitoring and diagnosis of diseases. Sensors. 2014;14:10895–928. https://doi.org/10.3390/s140610895. e-pub ahead of print 2014/06/21.
Wang Z, Deurenberg P, Wang W, Pietrobelli A, Baumgartner RN, Heymsfield SB. Hydration of fat-free body mass: new physiological modeling approach. Am J Physiol-Endocrinol Metab. 1999;276:E995–1003.
Aleman-Mateo H, Rush E, Esparza-Romero J, Ferriolli E, Ramirez-Zea M, Bour A, et al. Prediction of fat-free mass by bioelectrical impedance analysis in older adults from developing countries: a cross-validation study using the deuterium dilution method. J Nutr Health Aging. 2010;14:418–26.
Dioum A, Gartner A, Cissé AS, Delpeuch F, Maire B, Wade S, et al. Validity of impedance-based equations for the prediction of total body water as measured by deuterium dilution in African women. Am J Clin Nutr. 2005;81:597–604.
Sergi G, De Rui M, Stubbs B, Veronese N, Manzato E. Measurement of lean body mass using bioelectrical impedance analysis: a consideration of the pros and cons. Aging Clin Exp Res. 2017;29:591–7.
Stevens J, Truesdale KP, Cai J, Ou FS, Reynolds KR, Heymsfield SB. Nationally representative equations that include resistance and reactance for the prediction of percent body fat in Americans. Int J Obes. 2017;41:1669–75. https://doi.org/10.1038/ijo.2017.167. e-pub ahead of print 2017/07/25.
Swainson MG, Batterham AM, Tsakirides C, Rutherford ZH, Hind K. Prediction of whole-body fat percentage and visceral adipose tissue mass from five anthropometric variables. PloS ONE. 2017;12:e0177175. https://doi.org/10.1371/journal.pone.0177175. e-pub ahead of print 2017/05/12.
de Carvalho ABR, Neto CSP. Desenvolvimento e validação de equações para estimativa da massa corporal magra através da impedância bioelétrica em mulheres. Rev Brasileira Ativ FíSci Saúde. 1998;3:14–21.
Demura S, Yamaji S, Kitabayashi T. Residual volume on land and when immersed in water: effect on percent body fat. J Sports Sci. 2006;24:825–33. https://doi.org/10.1080/02640410500128163.
Kotler DP, Burastero S, Wang J, Pierson RN Jr. Prediction of body cell mass, fat-free mass, and total body water with bioelectrical impedance analysis: effects of race, sex, and disease. Am J Clin Nutr. 1996;64:489S–97S.
Marra M, Sammarco R, De Lorenzo A, Iellamo F, Siervo M, Pietrobelli A, et al. Assessment of body composition in health and disease using bioelectrical impedance analysis (BIA) and dual energy x-ray absorptiometry (DXA): a critical overview. Contrast Media Mol Imaging. 2019;2019:3548284. https://doi.org/10.1155/2019/3548284. e-pub ahead of print 2019/07/06.
Gagnon C, Menard J, Bourbonnais A, Ardilouze J-L, Baillargeon J-P, Carpentier AC, et al. Comparison of foot-to-foot and hand-to-foot bioelectrical impedance methods in a population with a wide range of body mass indices. Metab Syndr Relat Disord. 2010;8:437–41.
Guedes DP, Calabrese JC, Pirolli PM. Use of different segmental multi-frequency bioelectrical impedance devices for analysis of body composition in young adults: comparison with bioelectrical spectroscopy. Nutricion hospitalaria. 2019;36:618–25. https://doi.org/10.20960/nh.2376. e-pub ahead of print 2019/06/14.
Silva AM, Matias CN, Nunes CL, Santos DA, Marini E, Lukaski HC, et al. Lack of agreement of in vivo raw bioimpedance measurements obtained from two single and multi-frequency bioelectrical impedance devices. Eur J Clin Nutr. 2019;73:1077–83. https://doi.org/10.1038/s41430-018-0355-z. e-pub ahead of print 2018/10/24.
Pace N, Rathbun EN. Studies on body composition. 3. The body water and chemically combined nitrogen content in relation to fat content. J Biol Chem. 1945;158:685–91.
Silva AM, Wang J, Pierson RN Jr, Wang Z, Heymsfield SB, Sardinha LB, et al. Extracellular water: greater expansion with age in African Americans. J Appl Physiol. 2005;99:261–7.
Wells JC, Williams JE, Chomtho S, Darch T, Grijalva-Eternod C, Kennedy K, et al. Pediatric reference data for lean tissue properties: density and hydration from age 5 to 20 y. Am J Clin Nutr. 2010;91:610–8.
Rush EC, Puniani K, Valencia ME, Davies PS, Plank LD. Estimation of body fatness from body mass index and bioelectrical impedance: comparison of New Zealand European, Maori and Pacific Island children. Eur J Clin Nutr. 2003;57:1394–401. https://doi.org/10.1038/sj.ejcn.1601701. e-pub ahead of print 2003/10/25.
Minderico CS, Silva AM, Keller K, Branco TL, Martins SS, Palmeira AL, et al. Usefulness of different techniques for measuring body composition changes during weight loss in overweight and obese women. Br J Nutr. 2008;99:432–41.
Sluyter JD, Schaaf D, Scragg RK, Plank LD. Prediction of fatness by standing 8‐electrode bioimpedance: a multiethnic adolescent population. Obesity. 2010;18:183–9.
Mason J, Morris C, Long DE, Sanden MN, Flack K. Comparison of body composition estimates among norland elite®, lunar iDXA®, and the bodPod® in overweight to obese adults. Meas Phys Educ Exerc Sci. 2020;24:65–73.
Sakai Y, Ito H, Meno T, Numata M, Jingu S. Comparison of body composition measurements obtained by two fan-beam DXA instruments. J Clin Densitom. 2006;9:191–7. https://doi.org/10.1016/j.jocd.2006.03.008.
Heyward VH, Wagner DR. Applied body composition assessment. Champaigne, IL: Human Kinetics; 2004.
Bosy-Westphal A, Jensen B, Braun W, Pourhassan M, Gallagher D, Müller MJ. Quantification of whole-body and segmental skeletal muscle mass using phase-sensitive 8-electrode medical bioelectrical impedance devices. Eur J Clin Nutr. 2017;71:1061–7. https://doi.org/10.1038/ejcn.2017.27.
Heymsfield SB, Ebbeling CB, Zheng J, Pietrobelli A, Strauss BJ, Silva AM, et al. Multi‐component molecular‐level body composition reference methods: evolving concepts and future directions. Obes Rev. 2015;16:282–94.
Acknowledgements
The authors would like to thank all volunteers who participated in this study and all students and research assistants who made it possible to complete the study.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
RFC devised the conceptual idea and study design. RFC and KVSBM were involved in data collection. RFC, AMS, and KVSBM were involved in statistical analysis and drafting the paper. All authors provided critical feedback on the paper. AMS BGATC, and PMSD revised the paper. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Costa, R.F.d., Masset, K.V.d.S.B., Silva, A.M. et al. Development and cross-validation of predictive equations for fat-free mass and lean soft tissue mass by bioelectrical impedance in Brazilian women. Eur J Clin Nutr 76, 288–296 (2022). https://doi.org/10.1038/s41430-021-00946-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-021-00946-x
- Springer Nature Limited
This article is cited by
-
The bioelectrical impedance analysis (BIA) international database: aims, scope, and call for data
European Journal of Clinical Nutrition (2023)
-
Prognostic prediction by hypermetabolism varies depending on the nutritional status in early amyotrophic lateral sclerosis
Scientific Reports (2021)