Abstract
Exosomes are a heterogeneous group of cell-derived membranous structures, which mediate crosstalk interaction between cells. Recent studies have revealed a close relationship between exosomes and bone homeostasis. It is suggested that bone cells can spontaneously secret exosomes containing proteins, lipids and nucleic acids, which then to regulate osteoclastogenesis and osteogenesis. However, the network of regulatory activities of exosomes in bone homeostasis as well as their therapeutic potential in bone injury remain largely unknown. This review will detail and discuss the characteristics of exosomes, the regulatory activities of exosomes in bone homeostasis as well as the clinical potential of exosomes in bone injury.
Similar content being viewed by others
Introduction
Bone is a composite tissue, whose matrix consists of proteins and minerals, and which constantly undergoes modelling and remodeling through the coordination of osteoclasts, osteoblasts, and osteocytes. Osteoclasts, derived from mononuclear hematopoietic myeloid lineage cells, are responsible for bone resorption.1 Osteoblasts, accounting for the (4–6)% of total resident cells in the bone, are responsible for bone formation.2 Osteocytes, the most abundant cells in bone, are terminally differentiated from the osteoblasts, and are embedded in mineralized bone matrix. Osteocytes play a critical role in sensing mechanical loading and regulate functions of osteoclasts and osteoblasts.3
The interaction and coordination of these bone cells are important for maintaining bone homeostasis. Bone formation usually begins with the death of osteocytes.3 The apoptotic osteocytes release bioactive molecules, which induce other viable osteocytes to secret receptor activator of nuclear factor κB ligand (RANKL) which is important for osteoclast differentiation.4 Subsequently, osteoclast precursors are recruited by chemokines such as monocyte chemoattractant protein (MCP)-1, -2, and -3.5 The binding of receptor activator of nuclear factor κB (RANK)-RANKL on the surface of monocytes then initiates osteoclastogenesis.6,7 Meanwhile, osteoblasts produce bioactive molecules including macrophage colony-stimulating factor (M-CSF), MCP-1, and RANKL for the further recruitment and differentiation of osteoclast precursors.5,8 While resorbing damaged bone, osteoclasts spontaneously secret “coupling factors”, such as insulin-like growth factor (IGF) I and II and transforming growth factor (TGF)-β, which mediate the refill of resorbed lacunae by osteoblast.9 Finally, bone formation is completed when the newly mineralized-extracellular bone matrix completely replaces the resorbed bone matrix.10
Bone-derived exosomes are considered to be essential for intercellular communication between bone cells. Exosome-mediated transfer of nucleic acid or protein cargos between bone cells can bypass the space barriers between different cells, and plays a vital role in the crosstalk between bone cells regulating bone homeostasis. As the role of exosome is a new mechanism of bone formation and homeostasis, which has only recently emerged, we summarize the characteristics of exosomes, itemise the known functions of exosomes in bone homeostasis, and discuss their potential for clinical applications.
History of the exosome
A general history of the vesicular nature of exosomes
Exosomes,11 microvesicles,12 and secretory autophagosomes13 are three typical extracellular vehicles (EVs) identified recently. However, in early studies, there was no detailed classification or understanding of these extracellular vesicles.
Cellular vesicular components were recognised 140 years ago. Under dark-ground illumination, serum-derived particles were first seen by Edmunds in 1877.14 The main mass of these particles was then proved as fat in 1939.14 Since the function of these particles was unclear, they were just seen as blood dust.14 Clearer structure of cellular vesicles was then seen in microscope in 1962.15 However, the function of cellular vesicular components remains mysterious until 1969, when the finding of crystals of appetite suggested the participation of cartilage-derived matrix vesicles in calcification.16 Five years later, microvesicles in fetal calf serum were detected, which was the last class of EVs detected before exosome was defined.17
In 1981, the term exosome was first used for extracellular vesicles ranging from 50 to 1 000 nm.18 In 1983, the Stahl group and the Johnstone group reported that exosomes derived from reticulocytes could fuse with the plasma membrane and release their contents through exocytosis.19 Then in 1985, the same group provided the electron microscopic evidence for externalization of exosomes.20 In 1987, the formation of exosomes was described, and was the first time that the intraluminal vesicles of multivesicular endosomes (MVEs) were mentioned.21 The analysis of exosomal characteristic developed quickly in first decade after the exosome was defined. However, the function of exosomes remained largely unknown.
A breakthrough in exosomal investigation took place in 1996 when peptide-major histocompatibility complex (MHC) class II complexes-enriched exosomes released from B cells targeting T cells were detected. This finding first described the role of exosome in cell-to-cell communication.22 Following that, dendritic cell (DC)-derived exosomes23 and tumor-derived exosomes24 were investigated one after the other. These two studies showed the interactions and crosstalk between DCs and tumor cells. DC-derived exosomes could suppress the growth of tumors, and tumor cell-derived exosomes which contained tumor-rejection antigens could be carried by DCs for cross-protection from tumors.23,24 These findings were appealing to tumor investigators, and resulted in the generation of numerous reports associated with the tumor-derived exosomes.
The past decade has witnessed an acceleration of exosomal investigations, especially in studies of exosomal function. It is believed that exosomes are the most clearly defined group of secreted membrane vesicles, characteristically containing nucleic acid and proteins for cell signalling.25 Physiologically, they are critical to the immune system as they are involved with both stimulatory and tolerogenic responses.26 Also, it has been postulated that exosomes could be involved in regeneration, reducing tissue injury and improving tissue repair.27 Moreover, they may also be involved in tumor progression28 and delivery of inflammatory mediators.29 Consequently, the investigation of exosomes is becoming increasingly attractive as they are now suggested to be the key regulators of various cellular and physiological functions (Fig. 1).
History of bone-derived exosome
The history of bone-derived exosomes, however, is relatively recent. In 1975, extracellular membrane particles were first found in bone marrows which suggests a possible link between multiple myeloma-derived extracellular vesicles and bone tissue damage.30 Then in 1979, normal bone-derived EVs were first mentioned when alveolar bone-derived extracellular matrix vesicles were detected by microscopy.31 In 1980, osteoblast-derived matrix vesicles were investigated through ultrastructural techniques. While comparing scanning electron microscopy (SEM) with transmission electron microscopy (TEM), researchers theorised that osteoblast-derived vesicles probably serve as the initial locus of calcification32 (Fig. 2).
a Transmission Electron Microscope (TEM) image of osteoblast (OB) sited in bone matrix. The image shows that abundant of matrix vesicles (MV) are released into extracellular bone matrix (EBM). b Insert image demonstrates the diameter of MV from 50 to 150 nm. MN, mineralized nodules; MVB, Multivesicular bodies, late endosomes containing multiple intraluminal vesicles; PM, Plasma membrane
The first mention of bone-derived exosomes was 20 years after the naming of the exosome.33 At the beginning, bone marrow stromal cell-derived exosomes were the focus of bone-derived exosomes. However other bone cell-secreted exosomes were barely mentioned until 2013 when osteoclast precursors were reported to release exosomes.34 This initiated the investigation of exosomes from other bone cells. In 2015, the proteome of osteoblast-derived exosomes was for the first time investigated.35 In 2016, the characteristics and regulatory activities of osteoclast-derive exosomes were demonstrated.36 Then in 2017, osteocyte-derived exosomes and their miRNA contents were demonstrated.37 Now, the emerging data of bone-derived exosomes has established the details of exosome-based cell-to-cell interaction in bone.
Compositions of exosomes
The function and biological characteristic of exosomes are determined by their specific contents. Among the exosomal components, lipids, proteins, and nucleic acids are three main cargos which determine the specificity of exosomes38 and distinguish them from other extracellular vesicles (Fig. 3). A great variety of exosomal cargos have already been identified in exosomes and put together into a database named Exocarta,39 which was subsequently integrated into a broader database, Vesiclepedia.40 Some examples of exosomal cargos are summarized in (Table 1).
The composition of exosomes. Three main components of exosomes are lipids, proteins, and nucleic acids. Lipids are the main components of exosomal skeleton. They are also involved in exosome biogenesis. Among the lipids, sterols, sphingolipids, and phosiphlipids are dominant ones responsible for cargo sorting. Besides lipids, proteins are also enriched on exosomal surface. Tetraspanins, integrins, GTPases, and annexins are considered to be involved in membrane interaction and trafficking after recognition of surface proteins on recipients (Table 1). There are also various nucleic acid cargos including DNAs, RNAs, and miRNAs detected in the exosomes which determine the specificity of exosomal functions
Lipids
Exosomal structure and cargo sorting are largely dependent on lipid composition. Various lipids in exosomes have been investigated in the past decades. In a study of cancer cell-derived exosomes, more than 520 lipids from 36 different classes were identified.41 Lipids generally are enriched in exosomal membranes. The major non-polar lipids in plasma membrane are sterols, which are highly enriched in multivesicular bodies (MVBs) from late endosomes.42 Sphingolipids are also important for exosomal membrane construction, in which sphingomyelin is the dominant component, and is also involved in cargo sorting.43 Among exosomal membrane phospholipids, phosphatidylserine is of importance for being the activator of negative charge and the recruiter of signalling proteins.44,45
Besides contributing to the composition of the exosomal bilayer membranes, lipids in exosomes also play important roles in exosomal trafficking. During the formation of exosomes, the enrichment of sphingomyelin is found in membrane lipid rafts.46 As a result of increased sphingomyelin, down-regulation of ceramide and diacylglycerol occurs and finally reaches a balanced proportion in exosomes.47 Moreover, lipids play multiple roles in the sorting of nucleic acids and protein. In miRNA sorting, neutral sphingomyelinase 2 is the first molecule suggested to be associated with this mechanism.48 Other lipids such as sphingomyelin, ceramide, and sphingosine 1-phosphate have been proved to play important roles in protein sorting mechanisms.49 On the other hand, endosomal sorting complex required for transport (ESCRT)-independent exosome secretion is largely dependent on lipids, which are reported to significantly participate in the release of proteolipid-positive exosomes50 and Aβ-peptide-bearing exosomes.51 Although lipids are not the main participators of exosomal intercellular communication, their roles for maintaining the biological characteristics of exosomes are of importance.
Proteins
Through proteomic analysis, many proteins have been found in all mammalian exosomes, such as cytoskeletal components (tubulin, actin, cofilin, profilin), annexins (annexins I, II, IV, V, and VII), and the small GTPase family members rab7 and rab11. Among all these exosomal proteins, cytosolic exosomal enriched proteins such as Alix and TSG101, tetraspanins like CD9 and CD63 are the markers for distinguishing exosomes from other extracellular particles (Table 1). The detection of the proteins listed above allows researchers to quickly assess the characteristic of exosomes.52 Recent studies have also suggested that the heat-shock proteins (Hsp) are highly prevalent in exosomes. Among them, Hsp40 can improve the protein-folding environment in recipients, and Hsp70 is the up-regulator of pro-inflammatory cytokines.53 Protein composition is also crucially involved in ESCRT-dependent cargo sorting during the formation of exosomes. In ESCRT (-0, -I, -II, -III), recombinant human vacuolar protein sorting proteins (VPS proteins) play a major role functioning as membrane binders and cargo recognisers.54
Besides the various proteins mentioned above, there are also several other proteins in exosomes that reflect the specificity of cell origin and distinct exosomal functions. For example, latent membrane protein 1 (LAMP1) is highly expressed in exosomes released from nasopharyngeal cancer (NPC)-derived malignant epithelial cells.55 Similarly, a specific cell surface proteoglycan, glypican-1 (GPC1), was detected in exosomes from pancreatic cancer.56 Collectively, to maintain the specificity of exosomes derived from different donors, various protein cargos must be sorted into exosomes before their release.
Nucleic acids
Nucleic acids are also enriched in exosomes. Coding RNAs, non-coding RNAs, single-stranded or double-stranded DNAs are all found in exosomes.57,58,59
It is reported that more than 1 600 mRNAs and 700 miRNAs are detected in mammalian cell-derived exosomes. mRNAs contained in exosomes are usually related to cytogenesis, protein synthesis, and RNA posttranscriptional modification.57 Exosomal mRNA have been used as biomarkers since they are specific cargos.60 In patients with kidney diseases, downregulation of exosomal CD2AP mRNA has been detected in Urine, which can be used for early diagnosis.61 Exosomal mRNAs are also suggested to be involved in drug resistance of tumors. Therefore, the detection of exosomal mRNA level may be used to predict optimal treatment options as well as prognosis.62 Another recent report suggests that synthetic exosomal mRNA triggers exogenous protein expression, this may be a novel approach for treatment of genetic protein deficiency-related diseases.63
Exosomes also contain abundant miRNAs. In the immune system, miRNA-enriched exosomes are released from T-lymphocyte cells, B-lymphocyte cells and DCs, and the miRNAs are involved in the interaction between T-lymphocytes and antigen-presenting cells.64,65 In several tumors, exosomal miRNAs participate in tumor growth,66 metastasis, and drug resistance.67 Since specific variation of exosomal miRNAs can be detected in some diseases, exosomal profiling can be used as a tool for disease detection.
Exosomal DNA studies began much later than that of RNA, consequently, there is less information available in the literature. To the best of our knowledge, both single-stranded and double-stranded DNAs are contained in exosomes.58 Evidence has suggested that carrying cytoplasmic DNAs in exosomes protects against cell senescence and cell death caused by DNA injury. Cells can secret exosomes and remove harmful DNAs to extracellular matrix.68,69 However, there is only limited data to elucidate the function of double-stranded DNA in exosomes, and only little is known about the contribution of single-stranded DNAs. In a study of cancer cells, double-stranded DNA is reportedly being used to identify the mutations in cancer cells.58 Intriguingly, the expression of DNA cargo in tumor cell-derived exosomes is much higher than that in normal cell-derived exosomes, suggesting that tumor cells can modify target cells via the transfer of DNAs.70 There is still long way to go for the complete understanding of the role of exosomal DNA since the mechanism of chromosomal DNA sorting within intralumenal vesicles (ILVs) is still largely unknown.71
Exosomal trafficking
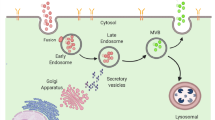
Exosomal trafficking involves three distinct mechanisms: cargo sorting, exosome release and exosome uptake (Fig. 4). During the generation of the endosomal machinery, ILVs, the early stage of exosomes, are formed through inward budding. Together with the sorting of specific proteins, lipids and nucleic acids into ILVs, the formation of MVEs results.72 Subsequently, MVE fuse with the cell membrane leading to the secretion of exosomes. Following that, the surface binding protein activates the uptake of exosomes in the recipient cells.73 Finally, as endocytosis progresses, exosomes release their contents which may influence regulatory processes or they may be degraded in lysosomes.74
Exosome release and uptake in cells. a Exosome formation starts with the formation of early endosome. Subsequently, ESCRT-dependent mechanism (a), which consists of four multiprotein subcomplexes (ESCRT 0, I, II, and III), or ESCRT-independent mechanism (b), which relies on tetraspanins-associated dynamic membrane platform, mediates the maturation of exosomes. After the fusion of late endosome, which contains mature exosomes, to the cell plasma membrane, exosomes are released into extracellular matrix. b Exosome uptake begins with the recognition of specific surface proteins of target cells. Subsequently, they are internalized through several internalization pathways. After that, exosomes can either release their cargos to exert their functions or be directly degraded by lysosome for recycling
Sorting cargos into exosomes
Sorting of protein into exosomes relies on specialized mechanisms, which ensure the specificity of exosomes for various intracellular communication purposes. Here, the ESCRT system, constituted of four multiprotein subcomplexes (ESCRT 0, I, II, and III) appears to be the main mechanism for exosomal formation.75 ESCRT 0, I, and II are responsible for recognizing and sequestering ubiquinated membrane proteins at the endosomal membrane, and ESCRT III is responsible for membrane budding and repartition of intraluminal vesicles.76 Categorizing of exosomes, however, appears to be a part of cargo ubiquination and only specific ESCRT segments are involved.77 The sorting of membrane proteins of the syndecan family into exosomes is regulated by an ESCRT accessory protein Alix through the cytosolic adaptor syntenin.74 Alix then binds to ESCRT III which is in control of ILV formation at the MVEs.78 Lateral involvement of heparin sulfate polysaccharide chains was reported to determine syndecan complex formation, which are degraded into shorter ones by heparinase activity in endosomes, favoring clustering of syndecans.79 Heparinase-induced recruitment is also believed to incite the binding of syndecan cytoplasmic domains to PDZ domains of syntenin, leading the sorting of proteins via Alix-ESCRT pathway.78,80
ESCRT-independent protein sorting is another important pathway for exosomal formation. This process requires the formation of a tetraspanins-associated dynamic membrane platform, where cytosolic and transmembrane proteins exert their ability to accept specific proteins into ILVs.81 Examples can be seen in CD63-induced endosomal sorting in melanocytes,82 and in tetranspanins-dependent recruitment of cholesterol-contained cone-like structures for inward budding.83 Although ESCRT-independent protein sorting is different to its counterpart, they both undergo cargo clustering and membrane budding.
Nucleic acid sorting, however, relies on a different mechanism. While DNA sorting is still largely unknown, RNA sorting is concluded previously. Loading RNA into exosomes begins with the formation of the raft-like region.84 Subsequently, anionic phospholipids are enriched in the raft-like region of exosomes,which then recruits neutral sphingomyelinase 2 to produce ceramide molecules, an indispensable factor for RNA sorting.85,86,87 Binding of RNAs to the raft-like region is dependent on differential affinity of RNA motifs,88 and randomly structured RNAs can bind to rafted domains with a 20-fold higher affinity. Once binding to the budding in the raft-like region, the RNA becomes encapsulated into ILVs and then released into the extracellular space within this vesicle.84
Release of exosomes
The greatest difference in the exocytosis pathway between exosomes and other extracellular vesicles (autophagosomes and microvesicles) is that exosomes are dependent on late endosomes for their release,74 and fusion of the MVBs, the late endosomes containing ILVs, with the plasma membrane is the last step before the exosomes are secreted to extracellular matrix. During this phase SNARE proteins and synaptotagmin family members are the main mediators.89 Exosomal exocytosis requires SNARE complexes, consisting of syntaxin 7, synaptotagmin 7, and VAMP 7. The SNARE complex is activated by upregulation of intercellular calcium which is Rab protein-dependent.90 Subsequently, vesicle (v)-SNAREs and target (t)-SNAREs promote the apposition of budding vesicles and cell membranes.91 After the coupling of v-SNAREs and t-SNAREs, the chaperone ATPase N-ethylmaleimide-sensitive factor (NSF) and soluble NSF attachment proteins (SNAPs) catalyze the disassembling of SNARE complexes, leading to the release of exosomes.91
Another key factor for exosomal release involves Rab proteins. They are a family of more than 60 proteins which participate in vesicle budding, cytoskeleton interaction and tethering of the receptor compartment to the membrane.92 Several examples revealed their participation in exosomal release. In oligodendroglia, Rab 35 was found to participate in PLP (genuine myelin proteins)-bearing exosomal secretion.93 Moreover, Rab 27A and Rab27B have been linked to MVBs interaction with the plasma membrane.94 These Rab proteins are thought to participate in the eventual fusion of the membranes of exosomes and the plasmalemma of donor cells, resulting in the exocytosis of exosomes.95
Uptake of exosomes
The fusion of exosomes with recipient cells relies on the interaction of vesicular ligands with cellular receptors, such as tetraspanins, integrins, and intercellular adhesion molecules (ICAMs), which induce the binding of exosomes to the surface of target cells. The recognition of surface proteins is the first step during exosomal internalization.96 Compelling evidence has proved that exosomal uptake is highly dependent on the signalling status of target cells and of exosomal surface proteins.11,97,98
During exosomal internalization, various pathways, including those of endocytosis, phagocytosis, micropinocytosis, and membrane fusion, are shown to participate.99 Among them, endocytosis seems to be the commonest way for exosomal uptake. This is a quick process occurring within 15 min.100 The most distinctive part of exosomal endocytosis is inward budding of the plasmalemma, which is dependent on the participation of caveolin101 and clathrin.102 By contrast, during macropinocytosis, exosomes are attached to a highly ruffled region on the cell surface and then taken in via the internalization of the whole region.103 This process is similar to phagocytosis.104
Moreover, exosomes can also directly fuse their membrane with the plasma membrane of target cells.105 This is dependent on two steps of intermediates: hemifusion structures and fusion pores.106,107 In most cases hemifusion structures are suggested to be lipid mixture without content mixing which represents the content of outer leaflets but not the inner leaflets of the two bilayers.106 Fusion initiates from the formation of fusion stalk, a point-like membrane protrusion of outer leaflet that establishes an hourglass-like connection between the apposed monolayers.108 Then an immediate contact of proximal leaflets leads to the formation of hemifusion stalk where leaflets fused and distal leaflets unfused. Finally, a fusion pore opens in the hemifusion diagram dependent on the expansion of stalk,109 where a connection between apposed membranes leads to the release of secretions.107
Exosomes in bone homeostasis
Bone homeostasis is of critical importance and relies on the transfer of active molecules between cells, which are summarized in (Table 2). Previous studies have suggested a direct interaction with secretion exchange among bone cells to occur.110,111 Recently, however, compelling evidence has emerged to show the regulatory activities that exosomes exert in bone remodelling. Almost all bone cells have been suggested to secret exosomes, and the relationship between bone remodelling and bone-cell derived exosomes is now well documented. Published reports have suggested that transfer of exosomal specific proteins, mRNA and miRNA is the main mechanism for exosome-mediated bone remodelling. This crosstalk establishes a novel network for cell-to-cell interaction during bone homeostasis.112
Exosome induces osteogenic differentiation of mesenchymal stem cells (MSCs) and osteogenesis
Bone remodelling is a complex process, which is mainly associated with two steps: osteoclastogenesis (for clearance of damaged bone tissues) and osteogenesis (for bone formation). It has been shown that exosomes are crucially involved in these two steps (Fig. 5).
Network of interaction between bone cells via exosomes. a Monocyte is the precursor of osteoclast, the differentiation of which relies on RANK-RANKL binding. While monocyte-derived exosomes and RANK-enriched exosomes released from osteoblast can activate osteoclastogenesis, osteoclast secret RANKL-enriched exosomes competitively inhibit this process. b Osteogenesis is regulated by several genes which can be transferred via crosstalk. Myostatin-modified osteocyte can secrete miRNA-218 enriched-exosomes to activate osteoblast differentiation. Besides, up-regulation loop can be seen between osteoblast and its precursor via the release of exosomes. However, osteoclast-derived exosomes play an inhibitory role in osteogenesis. Collectively, osteogenesis and osteoclastogenesis can be induced by exosomes derived from various bone cells whereas it seems that only osteoclast-secreted exosomes inhibit these two processes indicating their special role in bone homeostasis
During the process of bone formation, exosomes are suggested to involve in osteogenic differentiation of MSCs. Monocyte-derived exosomes are important stimulators for osteoblast differentiation.34 Fusion of these exosomes with MSCs can trigger the up-regulation of two osteogenic markers: RUNX2 and BMP-2.34 Intriguingly, newly formed osteoblasts can also secret exosomes to affect their progenitor cells. A group of researchers113 found that mature osteoblast-derived exosomes could trigger variation of miRNA expression profiles which, in turn, cooperatively inhibit the expression of Axin1, a central component of Wnt signalling pathway. As a result, β-catenin was up-regulated leading to the enhancement of osteogenic differentiation.
Osteogenesis is also dependent on exosomal functions. Before differentiating into osteoblasts, osteoblast precursors secret exosomes to promote osteogenesis.114 During fracture healing, bone marrow stem cell-derived exosomes express MCP-1, MCP-3, SDF-1, angiogenic factors, mRNAs and miRNAs and cooperatively contribute to bone remodelling.114 They probably also enhance osteoblast proliferation and differentiation by upregulating osteogenesis-related proteins (RUNX-2, ALP, OCN, and OPN), as well as several genes (miRNA-196a, miRNA-27a, and miRNA-206.115 Enhancement of osteoblast proliferation induced by MSC-derived exosomes has also been reported and that the MAPK pathway may be a key factor in exosome-mediated osteoblast activity.116
In addition, exosomes derived from osteoblasts and osteoclasts are also involved in osteogenesis. Osteoblasts can secret exosomes to enhance osteogenesis and Let-7-enriched exosomes derived from osteoblasts have been reported to enhance osteogenesis by regulating AT-hook 2 (HMGA2) and AXIN2.113,117 By contrast, osteoclast-derived exosomes act as inhibitors of osteogenesis. Exosomal miR-214-3p was suggested to be involved in the inhibition of osteoblast activity by targeting the 3′-untranslated region (UTR) of ATF4 mRNA. The exosomal transfer of miR-214-3p from osteoclasts to osteoblasts was also detected in vitro and thus triggered the reduction of bone mass in mice models.36,118,119
Exosome induces osteoclastogenesis and bone resorption
It is widely accepted that osteoclastogenesis is the basis for bone resorption. The classical osteoclastogenesis model is based on the direct interaction between different bone cells. However, recent studies have suggested a novel mechanism dependent on crosstalk phenomena. Initially, osteoblasts secret RANKL-enriched exosomes which targets monocytes. The RANKL-RANK binding on the monocyte surface then activates osteoclastogenesis.120 This process can be augmented by MSC-derived exosomes that can upregulate the expression of Nfatc1, Trap, and Ctsk. While osteoclast differentiation is initiated, the mechanism that controls the number of osteoclasts is initiated. This can be mediated either by osteoclast-derived exosomes or osteoblast-derived exosomes.121,122,123 Newly formed osteoclasts release RANK-enriched exosomes, and these exosomes can either directly fuse to osteoblasts or competitively bind RANKL in the extracellular matrix to regulate the formation of osteoclasts121 (Fig. 5b). Additionally, osteoblasts can release exosomes containing miR-503-3p to inhibit osteoclastogenesis by inactivating the RANK-RANKL signalling pathway.113,122,123 Alternately, large numbers of monocytes can secret exosomes to promote osteoclast differentiation.121 The end result is that osteoclasts are rapidly recruited during this phase, even though osteoclastogenesis-inhibiting exosomes are constantly released.
During bone resorption, the resorbing ability of osteoclasts can be also affected by exosomes. For example, exosomes derived from serum of osteoporotic, osteopenic or aged patients enhance bone resorption.124 When bone resorption is close to completion, abundant RANK-enriched exosomes derived from osteoclasts impede osteoclastogenesis. Finally, RANKL-enriched exosomes that are secreted from osteoblasts can inhibit bone resorption via the induction of osteoclast apoptosis.125
Osteocyte-derived exosomes in bone homeostasis
Compared to the investigation of osteoblast and osteoclast-derived exosomes, studies focusing on osteocyte-derived exosomes are few. Available data show that osteocytes also have the ability to release exosomes,37 and there appears to be a link between osteocyte-derived exosomes and bone homeostasis.
A group of researchers have shown that myostatin-modified osteocytic exosomes can regulate osteoblastic differentiation via exosomal miRNA-218, by targeting the wnt/β-catenin-signalling pathway.126 Wnt/β-catenin signaling is of great importance in bone homeostasis, involving both bone formation and bone resorption, and is widely believed to be orchestrated by the osteocyte.127,128 Previous studies have revealed that Sclerostin and DKK1 were the inhibitors of Wnt signalling by binding to the Wnt co-receptors LRP5/6, thereby contributing to bone loss.127 Interestingly, induction of exosomes containing miRNA-218 derived from myostatin-modified osteocyte was also inhibited. These exosomes were then found accepted by osteoblast leading to the up-regulation of sclerostin, DDK1, and RANKL.
Another interesting finding is that osteocytes can secret exosomes in response to mechanical loading. Initially, mechanical stimulation triggers immediate contraction of the actin network which results in Ca2+ transients. Simultaneously, mechanical stimulation induces the secretion of osteocytic exosomes, shown by immunostaining with the secretory vesicle marker, lysosomal-associated membrane protein 1 (LAMP-1). This process can also be enhanced by the upregulation of intercellular Ca2+. Finally, released exosomes which contain sclerostin, RANKL, and osteoprotegerin target osteoblasts to activate osteogenesis.129
Exosomes derived from tumor cells in bone homeostasis
Exosomes can be released from a variety of cell types. The tumor-cell derived exosomes130 can affect bone homeostasis. These effects of cancer cells on bone remodelling provides a new perspective for understanding bone diseases in the course of malignancy.
Tumor cells can spontaneously secret exosomes, and fusion of exosomes to bone cells may trigger either inhibition or abnormal enhancement of bone cell function. Exosomes released from multiple myeloma cells have been proved to support the survival of osteoclast precursors via the down-regulation of TRAP mRNA expression induced by inhibition of caspase-3 activity. Further, enhanced differentiation of osteoclast precursor was observed, which explains the increased bone resorption in myeloma patients.130 Enhanced osteoblast activity has been observed as well. It can be induced by the transfer of exosomal miRNA-214-3p which facilitates osteoblastic metastases.131 As bone is the initial site for tumor metastases,132 Exosomes can also participate in the establishment of bone metastasis, leading to tumor‐induced osteolysis.133 In the process of metastasis, exosomes play an important role as they are carrier of miRNA-192, a pivotal factor in tumor induced angiogenic activity.134 This is likely to influence pathways involved in the generation of proteases, adhesion molecules, and chemokine ligands, contributing to the metastatic spread of the tumors.
Exosomes-based clinical applications in fracture healing
Recent studies have shown the therapeutic potential of exosomes in different stage of fracture healing, suggesting that individualized strategies can be used to promote bone tissue repair. The initial step of fracture repair is the establishment of new vessels and formation of hematoma at the fracture site where inflammatory cells are being recruited.135 Prolonged activation or attenuation of inflammation may lead to excessive bone tissue damage or accumulation of necrotic bone respectively.136,137,138 MSC-derived exosomes are supposed to ideally attenuate inflammation-based delay of fracture healing. By using MSC-derived exosomes, proinflammation factors TNF-α and IL-1β are significantly suppressed, while anti-inflammatory factor TGF-β is increased139 (Fig. 6a). Moreover, exosomes are stable carriers for antiinflammation drug delivery. When encapsulated in exosomes, curcumin, an anti-inflammation drug, is more highly concentrated in blood. Moreover, as the drug is more accurately delivered to inflammatory cells due to target specificity of exosomes, there is obvious reduction of unwanted side effects. However, in a certain period of bone healing, inflammation is suggested to be indispensable. Over inactivation of inflammation may lead to delay of fracture healing or even non-union.140 Thus, timing of intervention is important. Exosomes also play a role in proinflammatory processes. There is evidence to suggest that acrophage-derived exosomes induce the differentiation of naive monocytes into macrophages.141 In this way, recruitment of macrophages, which contain approximately 29 cytokines for tissue repair and inflammation, will relieve an inflammation deficiency-based fracture healing delay.140
Roles of exosomes in fracture healing at different stages. a Mesenchymal stem cell (MSC)-derived exosomes have anti-inflammation property by upregulating anti-inflammation factor TGF-β and suppressing pro-inflammation factors: TNF-α and IL-1β. Thus, exosomes can be used to prevent over reaction of inflammation-induced delay of fracture healing. b In the stage of callus formation, exosomes act as promoters by enhancing proliferation and differentiation of endothelial cells as well as the formation of fibroblasts. As a result, enhanced angiogenesis and callus formation induced by exosomes can be detected at this stage. c Osteoblast (OB) and MSC-derived exosomes are promoters of bone remodelling. MSC-derived exosomes are suggested to boost the proliferation and liability of OBs. Besides, MSC and OB-derived exosomes are shown to enhance osteoclast (OC) differentiation. OB-derived exosomes are also enhancers of osteocyte (OY) formation by regulating AT-hook 2 (HMGA2) and AXIN2
Tissue repair is the second stage of bone healing when exosomes act as promoters of angiogenesis and bone regeneration (Fig. 6b). MSC-derived exosomes have been reported to contain abundant angiogenesis-related proteins.142 The latter enable endothelial cell proliferation and vessel formation.143 Interestingly, pro-angiogenesis effect and tissue repair are detected contemporaneously in vitro.144 In vivo, MSC-derived exosomes are seen to promote angiogenesis and osteogenesis. Eight weeks after implantation of MSC-derived exosomes, strong formation of vessels and bone tissue is detected in osteoporotic rats compared to untreated controls.145 These findings provide a novel approach for enhancing early tissue repair when revascularization and fibroblast proliferation in soft callus occur.135 Also, the wide range of exosomal functions may allow the use of MSC-exosomes throughout the whole period of fracture healing.145
Bone remodelling at its final stage generally is long-lasting (Fig. 6c). It reaches a degree of homeosteosis between different bone cells. Bone-derived exosomes have been proposed to have a regulatory function on each bone cell type. Osteoclast precursors together with osteoblast-derived exosomes have been detected to promote osteoclastogenesis in vivo,121,125 thus, could be used to boost the clearance of damaged tissue. During bone remodelling, MSC-derived exosomes have been shown to promote this process.146 In a femur fracture model of CD9−/− mice, which is suppressed in exosome production, there is obvious delay of callus formation leading to retardation of bone union. By local injection of exosomes, however, this retardation is rescued.114 Enhancement of cell proliferation and protection from cell death, MSC-derived exosomes could then serve as a powerful tool in bone remodelling.147 Such data support the concept that MSC-exosomes-based therapy is ideal for fracture healing for the repair of large bone defects.148
Conclusion
The past decade has witnessed significant progress in the investigation of exosomes as regulators of bone homeostasis, although the function of each of their single molecular species requires further analysis. Whether exosomes are, however, dominant factors in bone homeostasis needs to be further addressed in the future. Such studies will help to better understand the nosogenesis of several exosome-associated bone diseases. Although the introduction of exosomes into clinical practice is not likely to be soon, the perceived power of exosomes in bone homeostasis provides the possibility of novel approaches in the treatment of bone damage and disease.
Change history
25 January 2019
In the original publication of this article [1] there is an error in the formatting of figure 6. The updated figure 6 is published in this correction article. The publisher regrets the error.
References
Cappariello, A., Maurizi, A., Veeriah, V. & Teti, A. The great beauty of the osteoclast. Arch. Biochem. Biophys. 558, 70–78 (2014).
Capulli, M., Paone, R. & Rucci, N. Osteoblast and osteocyte: games without frontiers. Arch. Biochem. Biophys. 561, 3–12 (2014).
Compton, J. T. & Lee, F. Y. A review of osteocyte function and the emerging importance of sclerostin. J. Bone Jt. Surg. Am. 96, 1659–1668 (2014).
Kennedy, O. D. et al. Activation of resorption in fatigue-loaded bone involves both apoptosis and active pro-osteoclastogenic signaling by distinct osteocyte populations. Bone 50, 1115–22 (2012). 1115-cell 1122.
Graves, D. T., Jiang, Y. & Valente, A. J. The expression of monocyte chemoattractant protein-1 and other chemokines by osteoblasts. Front. Biosci. 4, D571–D580 (1999).
Lacey, D. L. et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176 (1998).
Nakagawa, N. et al. RANK is the essential signaling receptor for osteoclast differentiation factor in osteoclastogenesis. Biochem. Biophys. Res. Commun. 253, 395–400 (1998).
Yoshida, H. et al. The murine mutation osteopetrosis is in the coding region of the macrophage colony stimulating factor gene. Nature 345, 442–444 (1990).
Martin, T. J. & Sims, N. A. Osteoclast-derived activity in the coupling of bone formation to resorption. Trends Mol. Med. 11, 76–81 (2005).
Raggatt, L. J. & Partridge, N. C. Cellular and molecular mechanisms of bone remodeling. J. Biol. Chem. 285, 25103–25108 (2010).
Thery, C., Ostrowski, M. & Segura, E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 9, 581–593 (2009).
Tricarico, C., Clancy, J. & D’Souza-Schorey, C. Biology and biogenesis of shed microvesicles. Small GTPases 8, 220–232 (2017).
Ponpuak, M. et al. Secretory autophagy. Curr. Opin. Cell Biol. 35, 106–116 (2015).
Frazer, A. C. & Stewart, H. C. Ultramicroscopic particles in normal human blood. J. Physiol. 90, 18–30 (1937).
Barland, P., Novikoff, A. B. & Hamerman, D. Electron microscopy of the human synovial membrane. J. Cell Biol. 14, 207–220 (1962).
Anderson, H. C. Vesicles associated with calcification in the matrix of epiphyseal cartilage. J. Cell Biol. 41, 59–72 (1969).
Dalton, A. J. Microvesicles and vesicles of multivesicular bodies versus “virus-like” particles. J. Natl Cancer Inst. 54, 1137–1148 (1975).
Trams, E. G., Lauter, C. J., Salem, N. Jr. & Heine, U. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim Biophys. Acta 645, 63–70 (1981).
Harding, C., Heuser, J. & Stahl, P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J. Cell Biol. 97, 329–339 (1983).
Pan, B. T., Teng, K., Wu, C., Adam, M. & Johnstone, R. M. Electron microscopic evidence for externalization of the transferrin receptor in vesicular form in sheep reticulocytes. J. Cell Biol. 101, 942–948 (1985).
Johnstone, R. M., Adam, M., Hammond, J. R., Orr, L. & Turbide, C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem. 262, 9412–9420 (1987).
Raposo, G. et al. B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med. 183, 1161–1172 (1996).
Zitvogel, L. et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 4, 594–600 (1998).
Wolfers, J. et al. Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat. Med. 7, 297–303 (2001).
Lai, R. C., Yeo, R. W., Tan, K. H. & Lim, S. K. Exosomes for drug delivery - a novel application for the mesenchymal stem cell. Biotechnol. Adv. 31, 543–551 (2013).
Bobrie, A., Colombo, M., Raposo, G. & Thery, C. Exosome secretion: molecular mechanisms and roles in immune responses. Traffic 12, 1659–1668 (2011).
Lo Cicero, A., Stahl, P. D. & Raposo, G. Extracellular vesicles shuffling intercellular messages: for good or for bad. Curr. Opin. Cell Biol. 35, 69–77 (2015).
Hoshino, D. et al. Exosome secretion is enhanced by invadopodia and drives invasive behavior. Cell Rep. 5, 1159–1168 (2013).
Gasser, O. & Schifferli, J. A. Activated polymorphonuclear neutrophils disseminate anti-inflammatory microparticles by ectocytosis. Blood 104, 2543–2548 (2004).
Bernier, G. M., Duca, V. D. Jr, Brereton, R. & Graham, R. C. Jr Multiple myeloma with intramedullary masses of M-component. Blood 46, 931–935 (1975).
Bab, I. A., Muhlrad, A. & Sela, J. Ultrastructural and biochemical study of extracellular matrix vesicles in normal alveolar bone of rats. Cell Tissue Res. 202, 1–7 (1979).
Ornoy, A., Atkin, I. & Levy, J. Ultrastructural studies on the origin and structure of matrix vesicles in bone of young rats. Acta Anat. (Basel) 106, 450–461 (1980).
Peche, H., Heslan, M., Usal, C., Amigorena, S. & Cuturi, M. C. Presentation of donor major histocompatibility complex antigens by bone marrow dendritic cell-derived exosomes modulates allograft rejection. Transplantation 76, 1503–1510 (2003).
Ekstrom, K. et al. Monocyte exosomes stimulate the osteogenic gene expression of mesenchymal stem cells. PLoS ONE 8, e75227 (2013).
Ge, M., Ke, R., Cai, T., Yang, J. & Mu, X. Identification and proteomic analysis of osteoblast-derived exosomes. Biochem. Biophys. Res. Commun. 467, 27–32 (2015).
Li, D. et al. Osteoclast-derived exosomal miR-214-3p inhibits osteoblastic bone formation. Nat. Commun. 7, 10872 (2016).
Sato, M., Suzuki, T., Kawano, M. & Tamura, M. Circulating osteocyte-derived exosomes contain miRNAs which are enriched in exosomes from MLO-Y4 cells. Biomed. Rep. 6, 223–231 (2017).
Villarroya-Beltri, C., Baixauli, F., Gutierrez-Vazquez, C., Sanchez-Madrid, F. & Mittelbrunn, M. Sorting it out: regulation of exosome loading. Semin. Cancer Biol. 28, 3–13 (2014).
Mathivanan, S., Ji, H. & Simpson, R. J. Exosomes: extracellular organelles important in intercellular communication. J. Proteom. 73, 1907–1920 (2010).
Kalra, H. et al. Vesiclepedia: a compendium for extracellular vesicles with continuous community annotation. PLoS Biol. 10, e1001450 (2012).
Lydic, T. A. et al. Rapid and comprehensive ‘shotgun’ lipidome profiling of colorectal cancer cell derived exosomes. Methods 87, 83–95 (2015).
Mobius, W. et al. Recycling compartments and the internal vesicles of multivesicular bodies harbor most of the cholesterol found in the endocytic pathway. Traffic 4, 222–231 (2003).
van Meer, G., Voelker, D. R. & Feigenson, G. W. Membrane lipids: where they are and how they behave. Nat. Rev. Mol. Cell Biol. 9, 112–124 (2008).
Moser von Filseck, J. et al. Intracellular transport. Phosphatidylserine transport by ORP/Osh proteins is driven by phosphatidylinositol 4-phosphate. Science 349, 432–436 (2015).
Brouwers, J. F. et al. Distinct lipid compositions of two types of human prostasomes. Proteomics 13, 1660–1666 (2013).
Simons, K. & Toomre, D. Lipid rafts and signal transduction. Nat. Rev. Mol. Cell Biol. 1, 31–39 (2000).
Luberto, C. & Hannun, Y. A. Sphingomyelin synthase, a potential regulator of intracellular levels of ceramide and diacylglycerol during SV40 transformation. Does sphingomyelin synthase account for the putative phosphatidylcholine-specific phospholipase C? J. Biol. Chem. 273, 14550–14559 (1998).
Kosaka, N. et al. Neutral sphingomyelinase 2 (nSMase2)-dependent exosomal transfer of angiogenic microRNAs regulate cancer cell metastasis. J. Biol. Chem. 288, 10849–10859 (2013).
Theos, A. C. et al. A lumenal domain-dependent pathway for sorting to intralumenal vesicles of multivesicular endosomes involved in organelle morphogenesis. Dev. Cell 10, 343–354 (2006).
Trajkovic, K. et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science 319, 1244–1247 (2008).
Yuyama, K., Sun, H., Mitsutake, S. & Igarashi, Y. Sphingolipid-modulated exosome secretion promotes clearance of amyloid-beta by microglia. J. Biol. Chem. 287, 10977–10989 (2012).
Keerthikumar, S. et al. ExoCarta: A Web-Based Compendium of Exosomal Cargo. J. Mol. Biol. 428, 688–692 (2016).
Takeuchi, T. et al. Intercellular chaperone transmission via exosomes contributes to maintenance of protein homeostasis at the organismal level. Proc. Natl Acad. Sci. USA 112, E2497–E2506 (2015).
Henne, W. M., Buchkovich, N. J. & Emr, S. D. The ESCRT pathway. Dev. Cell 21, 77–91 (2011).
Keryer-Bibens, C. et al. Exosomes released by EBV-infected nasopharyngeal carcinoma cells convey the viral latent membrane protein 1 and the immunomodulatory protein galectin 9. BMC Cancer 6, 283 (2006).
Melo, S. A. et al. Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature 523, 177–182 (2015).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9, 654–659 (2007).
Thakur, B. K. et al. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 24, 766–769 (2014).
Li, S. P., Lin, Z. X., Jiang, X. Y. & Yu, X. Y. Exosomal cargo-loading and synthetic exosome-mimics as potential therapeutic tools. Acta Pharmacol. Sin. 39, 542–551 (2018).
Januszyk, K. & Lima, C. D. The eukaryotic RNA exosome. Curr. Opin. Struct. Biol. 24, 132–140 (2014).
Lv, L. L. et al. CD2AP mRNA in urinary exosome as biomarker of kidney disease. Clin. Chim. Acta 428, 26–31 (2014).
Shao, H. et al. Chip-based analysis of exosomal mRNA mediating drug resistance in glioblastoma. Nat. Commun. 6, 6999 (2015).
Liu, R., Liu, J., Ji, X. & Liu, Y. Synthetic nucleic acids delivered by exosomes: a potential therapeutic for generelated metabolic brain diseases. Metab. Brain Dis. 28, 551–562 (2013).
Mittelbrunn, M. et al. Unidirectional transfer of microRNA-loaded exosomes from T cells to antigen-presenting cells. Nat. Commun. 2, 282 (2011).
Luo, S. S. et al. Human villous trophoblasts express and secrete placenta-specific microRNAs into maternal circulation via exosomes. Biol. Reprod. 81, 717–729 (2009).
Liu, H. et al. Tumor-derived exosomes promote tumor self-seeding in hepatocellular carcinoma by transferring miRNA-25-5p to enhance cell motility. Oncogene: https://doi.org/10.1038/s41388-018-0309-x (2018).
Wang, J. et al. Bone marrow stromal cell-derived exosomes as communicators in drug resistance in multiple myeloma cells. Blood 124, 555–566 (2014).
Takahashi, A. et al. Exosomes maintain cellular homeostasis by excreting harmful DNA from cells. Nat. Commun. 8, 15287 (2017).
Hoeijmakers, J. H. DNA damage, aging, and cancer. N. Engl. J. Med. 361, 1475–1485 (2009).
Kahlert, C. et al. Identification of double-stranded genomic DNA spanning all chromosomes with mutated KRAS and p53 DNA in the serum exosomes of patients with pancreatic cancer. J. Biol. Chem. 289, 3869–3875 (2014).
Kalluri, R. & LeBleu, V. S. Discovery of Double-Stranded Genomic DNA in Circulating Exosomes. Cold Spring Harb. Symp. Quant. Biol. 81, 275–280 (2016).
Raiborg, C. & Stenmark, H. The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature 458, 445–452 (2009).
Svensson, K. J. et al. Exosome uptake depends on ERK1/2-heat shock protein 27 signaling and lipid Raft-mediated endocytosis negatively regulated by caveolin-1. J. Biol. Chem. 288, 17713–17724 (2013).
van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19, 213–228 (2018).
Henne, W. M., Stenmark, H. & Emr, S. D. Molecular mechanisms of the membrane sculpting ESCRT pathway. Cold Spring Harb. Perspect. Biol. 5, https://doi.org/10.1101/cshperspect.a016766 (2013).
Stoorvogel, W. Resolving sorting mechanisms into exosomes. Cell Res. 25, 531–532 (2015).
Colombo, M. et al. Analysis of ESCRT functions in exosome biogenesis, composition and secretion highlights the heterogeneity of extracellular vesicles. J. Cell Sci. 126, 5553–5565 (2013).
Baietti, M. F. et al. Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat. Cell Biol. 14, 677–685 (2012).
Ramani, V. C., Pruett, P. S., Thompson, C. A., DeLucas, L. D. & Sanderson, R. D. Heparan sulfate chains of syndecan-1 regulate ectodomain shedding. J. Biol. Chem. 287, 9952–9961 (2012).
Roucourt, B., Meeussen, S., Bao, J., Zimmermann, P. & David, G. Heparanase activates the syndecan-syntenin-ALIX exosome pathway. Cell Res. 25, 412–428 (2015).
Charrin, S., Jouannet, S., Boucheix, C. & Rubinstein, E. Tetraspanins at a glance. J. Cell Sci. 127, 3641–3648 (2014).
van Niel, G. et al. The tetraspanin CD63 regulates ESCRT-independent and -dependent endosomal sorting during melanogenesis. Dev. Cell 21, 708–721 (2011).
Zimmerman, B. et al. Crystal structure of a full-length human tetraspanin reveals a cholesterol-binding pocket. Cell 167, 1041–1051 (2016). e1011.
Janas, T., Janas, M. M., Sapon, K. & Janas, T. Mechanisms of RNA loading into exosomes. FEBS Lett. 589, 1391–1398 (2015).
Wu, B. X. et al. Identification of novel anionic phospholipid binding domains in neutral sphingomyelinase 2 with selective binding preference. J. Biol. Chem. 286, 22362–22371 (2011).
Record, M., Carayon, K., Poirot, M. & Silvente-Poirot, S. Exosomes as new vesicular lipid transporters involved in cell-cell communication and various pathophysiologies. Biochim. Biophys. Acta 1841, 108–120 (2014).
Kosaka, N. et al. Secretory mechanisms and intercellular transfer of microRNAs in living cells. J. Biol. Chem. 285, 17442–17452 (2010).
Janas, T., Janas, T. & Yarus, M. Specific RNA binding to ordered phospholipid bilayers. Nucleic Acids Res. 34, 2128–2136 (2006).
Kozlov, M. M. & Chernomordik, L. V. Membrane tension and membrane fusion. Curr. Opin. Struct. Biol. 33, 61–67 (2015).
Savina, A., Fader, C. M., Damiani, M. T. & Colombo, M. I. Rab11 promotes docking and fusion of multivesicular bodies in a calcium-dependent manner. Traffic 6, 131–143 (2005).
Wei, Y. et al. Pyruvate kinase type M2 promotes tumour cell exosome release via phosphorylating synaptosome-associated protein 23. Nat. Commun. 8, 14041 (2017).
Colombo, M., Raposo, G. & Thery, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell. Dev. Biol. 30, 255–289 (2014).
Fruhbeis, C., Frohlich, D., Kuo, W. P. & Kramer-Albers, E. M. Extracellular vesicles as mediators of neuron-glia communication. Front. Cell. Neurosci. 7, 182 (2013).
Ostrowski, M. et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat. Cell Biol. 12, 19–30 (2010). sup pp 11-13.
Kowal, J., Tkach, M. & Thery, C. Biogenesis and secretion of exosomes. Curr. Opin. Cell Biol. 29, 116–125 (2014).
Christianson, H. C., Svensson, K. J., van Kuppevelt, T. H., Li, J. P. & Belting, M. Cancer cell exosomes depend on cell-surface heparan sulfate proteoglycans for their internalization and functional activity. Proc. Natl Acad. Sci. USA 110, 17380–17385 (2013).
Cocucci, E., Racchetti, G. & Meldolesi, J. Shedding microvesicles: artefacts no more. Trends Cell Biol. 19, 43–51 (2009).
Keller, S. et al. Systemic presence and tumor-growth promoting effect of ovarian carcinoma released exosomes. Cancer Lett. 278, 73–81 (2009).
Mulcahy, L. A., Pink, R. C. & Carter, D. R. Routes and mechanisms of extracellular vesicle uptake. J. Extracell. Vesicles 3, https://doi.org/10.3402/jev.v3.24641 (2014).
Feng, D. et al. Cellular internalization of exosomes occurs through phagocytosis. Traffic 11, 675–687 (2010).
Boscher, C. & Nabi, I. R. Caveolin-1: role in cell signaling. Adv. Exp. Med. Biol. 729, 29–50 (2012).
Kirchhausen, T. Clathrin. Annu. Rev. Biochem. 69, 699–727 (2000).
Tian, T. et al. Exosome uptake through clathrin-mediated endocytosis and macropinocytosis and mediating miR-21 delivery. J. Biol. Chem. 289, 22258–22267 (2014).
Swanson, J. A. Shaping cups into phagosomes and macropinosomes. Nat. Rev. Mol. Cell Biol. 9, 639–649 (2008).
Parolini, I. et al. Microenvironmental pH is a key factor for exosome traffic in tumor cells. J. Biol. Chem. 284, 34211–34222 (2009).
Chernomordik, L. V. & Kozlov, M. M. Mechanics of membrane fusion. Nat. Struct. Mol. Biol. 15, 675–683 (2008).
Chernomordik, L. V. & Kozlov, M. M. Protein-lipid interplay in fusion and fission of biological membranes. Annu. Rev. Biochem. 72, 175–207 (2003).
Kozlov, M. M. & Markin, V. S. [Possible mechanism of membrane fusion]. Biofizika 28, 242–247 (1983).
Kozlov, M. M., Leikin, S. L., Chernomordik, L. V., Markin, V. S. & Chizmadzhev, Y. A. Stalk mechanism of vesicle fusion. Intermixing of aqueous contents. Eur. Biophys. J. 17, 121–129 (1989).
Helfrich, M. H. et al. Beta 1 integrins and osteoclast function: involvement in collagen recognition and bone resorption. Bone 19, 317–328 (1996).
Crockett, J. C., Mellis, D. J., Scott, D. I. & Helfrich, M. H. New knowledge on critical osteoclast formation and activation pathways from study of rare genetic diseases of osteoclasts: focus on the RANK/RANKL axis. Osteoporos. Int. 22, 1–20 (2011).
Li, J. et al. Exosomes mediate the cell-to-cell transmission of IFN-alpha-induced antiviral activity. Nat. Immunol. 14, 793–803 (2013).
Cui, Y., Luan, J., Li, H., Zhou, X. & Han, J. Exosomes derived from mineralizing osteoblasts promote ST2 cell osteogenic differentiation by alteration of microRNA expression. FEBS Lett. 590, 185–192 (2016).
Furuta, T. et al. Mesenchymal stem cell-derived exosomes promote fracture healing in a mouse model. Stem Cells Transl. Med. 5, 1620–1630 (2016).
Qin, Y., Wang, L., Gao, Z., Chen, G. & Zhang, C. Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo. Sci. Rep. 6, 21961 (2016).
Zhao, P., Xiao, L., Peng, J., Qian, Y. Q. & Huang, C. C. Exosomes derived from bone marrow mesenchymal stem cells improve osteoporosis through promoting osteoblast proliferation via MAPK pathway. Eur. Rev. Med. Pharmacol. Sci. 22, 3962–3970 (2018).
Wei, J. et al. let-7 enhances osteogenesis and bone formation while repressing adipogenesis of human stromal/mesenchymal stem cells by regulating HMGA2. Stem. Cells Dev. 23, 1452–1463 (2014).
Wang, X. et al. miR-214 targets ATF4 to inhibit bone formation. Nat. Med. 19, 93–100 (2013).
Sun, W. et al. Osteoclast-derived microRNA-containing exosomes selectively inhibit osteoblast activity. Cell Discov. 2, 16015 (2016).
Deng, L. et al. Osteoblast-derived microvesicles: a novel mechanism for communication between osteoblasts and osteoclasts. Bone 79, 37–42 (2015).
Huynh, N. et al. Characterization of regulatory extracellular vesicles from osteoclasts. J. Dent. Res. 95, 673–679 (2016).
Chen, C. et al. MiR-503 regulates osteoclastogenesis via targeting RANK. J. Bone Miner. Res. 29, 338–347 (2014).
Xie, Y., Chen, Y., Zhang, L., Ge, W. & Tang, P. The roles of bone-derived exosomes and exosomal microRNAs in regulating bone remodelling. J. Cell. Mol. Med. 21, 1033–1041 (2017).
Xie, Y. et al. Involvement of serum-derived exosomes of elderly patients with bone loss in failure of bone remodeling via alteration of exosomal bone-related proteins. Aging Cell 17, e12758 (2018).
Cappariello, A. et al. Osteoblast-derived extracellular vesicles are biological tools for the delivery of active molecules to bone. J. Bone Miner. Res. 33, 517–533 (2018).
Qin, Y. et al. Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA-218: a novel mechanism in muscle-bone communication. J. Biol. Chem. 292, 11021–11033 (2017).
Bonewald, L. F. The amazing osteocyte. J. Bone Miner. Res. 26, 229–238 (2011).
Kramer, I. et al. Osteocyte Wnt/beta-catenin signaling is required for normal bone homeostasis. Mol. Cell. Biol. 30, 3071–3085 (2010).
Lu, X. L., Huo, B., Park, M. & Guo, X. E. Calcium response in osteocytic networks under steady and oscillatory fluid flow. Bone 51, 466–473 (2012).
Raimondi, L. et al. Involvement of multiple myeloma cell-derived exosomes in osteoclast differentiation. Oncotarget 6, 13772–13789 (2015).
Ye, Y. et al. Exosomal miR-141-3p regulates osteoblast activity to promote the osteoblastic metastasis of prostate cancer. Oncotarget 8, 94834–94849 (2017).
Mundy, G. R. Metastasis to bone: causes, consequences and therapeutic opportunities. Nat. Rev. Cancer 2, 584–593 (2002).
Guise, T. A. et al. Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer-mediated osteolysis. J. Clin. Invest. 98, 1544–1549 (1996).
Valencia, K. et al. miRNA cargo within exosome-like vesicle transfer influences metastatic bone colonization. Mol. Oncol. 8, 689–703 (2014).
Claes, L., Recknagel, S. & Ignatius, A. Fracture healing under healthy and inflammatory conditions. Nat. Rev. Rheumatol. 8, 133–143 (2012).
Grundnes, O. & Reikeraas, O. Effects of macrophage activation on bone healing. J. Orthop. Sci. 5, 243–247 (2000).
Yu, B., Zhang, X. & Li, X. Exosomes derived from mesenchymal stem cells. Int. J. Mol. Sci. 15, 4142–4157 (2014).
Schmidt-Bleek, K. et al. Inflammatory phase of bone healing initiates the regenerative healing cascade. Cell Tissue Res. 347, 567–573 (2012).
Chen, W. et al. Immunomodulatory effects of mesenchymal stromal cells-derived exosome. Immunol. Res. 64, 831–840 (2016).
Kolar, P. et al. The early fracture hematoma and its potential role in fracture healing. Tissue Eng. Part B Rev. 16, 427–434 (2010).
Ismail, N. et al. Macrophage microvesicles induce macrophage differentiation and miR-223 transfer. Blood 121, 984–995 (2013).
Anderson, J. D. et al. Comprehensive proteomic analysis of mesenchymal stem cell exosomes reveals modulation of angiogenesis via nuclear factor-kappaB signaling. Stem Cells 34, 601–613 (2016).
Salomon, C. et al. Exosomal signaling during hypoxia mediates microvascular endothelial cell migration and vasculogenesis. PLoS ONE 8, e68451 (2013).
Shabbir, A., Cox, A., Rodriguez-Menocal, L., Salgado, M. & Van Badiavas, E. Mesenchymal stem cell exosomes induce proliferation and migration of normal and chronic wound fibroblasts, and enhance angiogenesis in vitro. Stem. Cells Dev. 24, 1635–1647 (2015).
Qi, X. et al. Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells repair critical-sized bone defects through enhanced angiogenesis and osteogenesis in osteoporotic rats. Int. J. Biol. Sci. 12, 836–849 (2016).
Narayanan, R., Huang, C. C. & Ravindran, S. Hijacking the cellular mail: exosome mediated differentiation of mesenchymal stem cells. Stem Cells Int. 2016, 3808674 (2016).
De Jong, O. G., Van Balkom, B. W., Schiffelers, R. M., Bouten, C. V. & Verhaar, M. C. Extracellular vesicles: potential roles in regenerative medicine. Front. Immunol. 5, 608 (2014).
Ferreira, E. & Porter, R. M. Harnessing extracellular vesicles to direct endochondral repair of large bone defects. Bone Jt. Res. 7, 263–273 (2018).
Nazarenko, I. et al. Cell surface tetraspanin Tspan8 contributes to molecular pathways of exosome-induced endothelial cell activation. Cancer Res. 70, 1668–1678 (2010).
Escola, J. M. et al. Selective enrichment of tetraspan proteins on the internal vesicles of multivesicular endosomes and on exosomes secreted by human B-lymphocytes. J. Biol. Chem. 273, 20121–20127 (1998).
Yanez-Mo, M., Barreiro, O., Gordon-Alonso, M., Sala-Valdes, M. & Sanchez-Madrid, F. Tetraspanin-enriched microdomains: a functional unit in cell plasma membranes. Trends Cell Biol. 19, 434–446 (2009).
Tamai, K. et al. Exosome secretion of dendritic cells is regulated by Hrs, an ESCRT-0 protein. Biochem. Biophys. Res. Commun. 399, 384–390 (2010).
Mathew, A., Bell, A. & Johnstone, R. M. Hsp-70 is closely associated with the transferrin receptor in exosomes from maturing reticulocytes. Biochem. J. 308(Pt 3), 823–830 (1995).
Hsu, C. et al. Regulation of exosome secretion by Rab35 and its GTPase-activating proteins TBC1D10A-C. J. Cell Biol. 189, 223–232 (2010).
Gutierrez-Vazquez, C., Villarroya-Beltri, C., Mittelbrunn, M. & Sanchez-Madrid, F. Transfer of extracellular vesicles during immune cell-cell interactions. Immunol. Rev. 251, 125–142 (2013).
Laulagnier, K. et al. PLD2 is enriched on exosomes and its activity is correlated to the release of exosomes. FEBS Lett. 572, 11–14 (2004).
Shinozaki, K. & Waite, M. A novel phosphatidylglycerol-selective phospholipase A2 from macrophages. Biochemistry 38, 1669–1675 (1999).
Beh, C. T., McMaster, C. R., Kozminski, K. G. & Menon, A. K. A detour for yeast oxysterol binding proteins. J. Biol. Chem. 287, 11481–11488 (2012).
Xu, C. et al. CD82 endocytosis and cholesterol-dependent reorganization of tetraspanin webs and lipid rafts. FASEB J. 23, 3273–3288 (2009).
Carayon, K. et al. Proteolipidic composition of exosomes changes during reticulocyte maturation. J. Biol. Chem. 286, 34426–34439 (2011).
Subra, C., Laulagnier, K., Perret, B. & Record, M. Exosome lipidomics unravels lipid sorting at the level of multivesicular bodies. Biochimie 89, 205–212 (2007).
Falguieres, T. et al. In vitro budding of intralumenal vesicles into late endosomes is regulated by Alix and Tsg101. Mol. Biol. Cell 19, 4942–4955 (2008).
Yang, M. et al. Microvesicles secreted by macrophages shuttle invasion-potentiating microRNAs into breast cancer cells. Mol. Cancer 10, 117 (2011).
Ibrahim, A. G., Cheng, K. & Marban, E. Exosomes as critical agents of cardiac regeneration triggered by cell therapy. Stem Cell Rep. 2, 606–619 (2014).
Fujita, Y. et al. Intercellular communication by extracellular vesicles and their microRNAs in asthma. Clin. Ther. 36, 873–881 (2014).
Liu, S. et al. MSC Transplantation Improves Osteopenia via Epigenetic Regulation of Notch Signaling in Lupus. Cell. Metab. 22, 606–618 (2015).
Zhang, J. et al. Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signaling pathway. Stem Cell Res. Ther. 7, 136 (2016).
Morrell, A. E. et al. Mechanically induced Ca(2+) oscillations in osteocytes release extracellular vesicles and enhance bone formation. Bone Res. 6, 6 (2018).
Martin, P. J. et al. Adipogenic RNAs are transferred in osteoblasts via bone marrow adipocytes-derived extracellular vesicles (EVs). BMC Cell. Biol. 16, 10 (2015).
Acknowledgements
We would like to thank the support from Perron Institute for Neurological and Translational Science, Department of Orthopaedics, The Second Affiliated Hospital and Yuying Children's Hospital of Wenzhou Medical University and Department of Orthopaedics, Shanghai Sixth People’s Hospital of Shanghai Jiaotong University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, M., Gao, W., Papadimitriou, J.M. et al. Exosomes—the enigmatic regulators of bone homeostasis. Bone Res 6, 36 (2018). https://doi.org/10.1038/s41413-018-0039-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41413-018-0039-2
- Springer Nature Limited
This article is cited by
-
Non-bone-derived exosomes: a new perspective on regulators of bone homeostasis
Cell Communication and Signaling (2024)
-
Engineering extracellular vesicles for ROS scavenging and tissue regeneration
Nano Convergence (2024)
-
Exosome-mediated regulation of inflammatory pathway during respiratory viral disease
Virology Journal (2024)
-
Bioengineering extracellular vesicles: smart nanomaterials for bone regeneration
Journal of Nanobiotechnology (2023)
-
Disease-microenvironment modulation by bare- or engineered-exosome for rheumatoid arthritis treatment
Biomaterials Research (2023)