Abstract
Lifestyle factors, especially exercise, impact the manifestation and progression of psychiatric and neurodegenerative disorders such as depression and Alzheimer’s disease, mediated by changes in hippocampal neuroplasticity. The beneficial effects of exercise may be due to its promotion of adult hippocampal neurogenesis (AHN). Gut microbiota has also been showed to be altered in a variety of brain disorders, and disturbances of the microbiota have resulted in alterations in brain and behaviour. However, whether exercise can counteract the negative effects of altered gut microbiota on brain function remains under explored. To this end, chronic disruption of the gut microbiota was achieved using an antibiotic cocktail in rats that were sedentary or allowed voluntary access to running wheels. Sedentary rats with disrupted microbiota displayed impaired performance in hippocampal neurogenesis-dependent tasks: the modified spontaneous location recognition task and the novelty suppressed feeding test. Performance in the elevated plus maze was also impaired due to antibiotics treatment. These behaviours, and an antibiotics-induced reduction in AHN were attenuated by voluntary exercise. The effects were independent of changes in the hippocampal metabolome but were paralleled by caecal metabolomic changes. Taken together these data highlight the importance of the gut microbiota in AHN-dependent behaviours and demonstrate the power of lifestyle factors such as voluntary exercise to attenuate these changes.
Similar content being viewed by others
Introduction
The gut microbiota comprises the trillions of microorganisms inhabiting the gastrointestinal tract and represents an important component in the bidirectional communication between the brain and the gut. Numerous studies have now implicated the gut microbiota in the aetiology of neurological and neuropsychiatric diseases [1] including Alzheimer’s [2], Parkinson’s [3] and depression [4]. The gut microbiota composition changes throughout the lifespan [5], depending on environmental or lifestyle factors, notably diet and exercise [6,7,8].
A growing body of evidence shows that exercise changes the gut microbial composition [9, 10] and related metabolites, that those changes confer benefits on health and may delay disease progression [11]. For example, it has been shown that exercise impacts gut microbiota diversity and increases taxa that produce health-promoting metabolites such as short chain fatty acids (SCFA) [12]. A sedentary lifestyle represents a risk factor for cognitive impairments [13], and many studies now report that physical activity has beneficial effect on brain health [14, 15]. Exercise increases level of growth factors such as brain-derived neurotrophic factor (BDNF) [16], vascular endothelial growth factor [17] or insulin growth factor-1 (IGF-1) [18] in the brain. Interestingly, the gut microbiota has been shown to modulate some of these factors [19]. Thus, the ability of exercise to shape the microbiome and subsequent gut-brain-associated pathways may have crucial implications for the development of strategies to treat neurological and psychiatric disorders.
The main brain structure affected by exercise is the hippocampus [20], which plays a role in learning, memory, and in mood-related behaviours. The hippocampus is also capable of generating new neurons across the lifespan in a process called adult hippocampal neurogenesis (AHN) [21]. This form of brain plasticity is involved in cognitive tasks which require spatial and contextual memory [22] and in anxiety-related behaviours [23]. There is consensus that AHN is predominantly required for pattern separation [24, 25], which is the ability to discriminate between memories of similar experiences or environments. Notably, rodent studies have shown that exercise is a robust enhancer of certain types of cognitive function through its ability to increase AHN [26, 27].
While the gut microbiome regulates cognitive behaviours [7, 28, 29], neuroplasticity [30, 31] and AHN [4, 32, 33], the interaction between the gut microbiota and exercise on AHN and associated function remains unresolved. In this study, we examined the ability of exercise to ameliorate the potential negative effects of gut microbiota disruption using a cocktail of antibiotics on AHN, cognitive function, and mood related behaviours in adult male rats.
Methods
Animals and experimental design
Adult (9 week old) male Sprague‐Dawley rats were purchased from Envigo, UK, and maintained on a 12‐h:12‐h light:dark cycle (lights on at 07:00 h) at 22 ± 1 °C, with access to standard chow (Teklad 2018S) and water ad libitum. Rats were pair-housed and randomly assigned to an experimental group as follows: sedentary control “Sed”, sedentary and antibiotics cocktail “Sed+ABX”, voluntary running “Ex”, and voluntary running and antibiotics cocktail “Ex+ABX” (n = 10; Fig. 1a). Antibiotics were administered in the drinking water and bottles were changed every second day. Rats were pair-housed in either standard housing cages (Cage size 560 × 380 × 170 mm, with hollow paper tubes and shredded paper) or in cages with continuous and free access to running wheels (Cage size 425 × 266 × 185 mm; Activity wheel 33 cm Ø, Tecniplast, UK) for the duration of the experiment. Animals were pair-housed in order to avoid any potential social isolation stress effects of single housing on hippocampal neurogenesis [34, 35]. We have previously shown that social isolation impacts upon exercise-induced changes in neurogenesis in mice [36]. Rats were placed in cages with running wheels 2 weeks after the start of antibiotic administration and were intraperitoneally injected with 5-Bromo-2’-deoxyuridine (BrdU; 150 mg/kg of body weight; #B5002, Sigma) once a day for five consecutive days. Wheel revolutions were continuously monitored and were consistent across running groups (Fig. 1c). All animal procedures were performed under authorisations issued by the Health Products Regulatory Authority (HPRA, Ireland), in accordance with the European Communities Council Directive (2010/63/EU) and approved by the Animal Experimentation Ethics Committee of University College Cork.
a Experimental timeline. All rats were pair housed and received antibiotics (ABX) in drinking water or not throughout the study. Two weeks after the beginning of ABX treatment rats were separated in 4 experimental groups, sedentary (Sed), sedentary with Abx (Sed+ABX) or had continuous access to a running wheel (Ex) or exercise with ABX (Ex+ABX). All rats received BrdU (150 mg/kg/day for 5 days) and behavioural testing commenced after 3 weeks of exercise. Exercise continued throughout testing for a total of 8 weeks. b Body weight of rats and (c) Running wheel activity (average km/24 h per rat) (n = 5; Repeated Measures ANOVA; main effect of time [F(7, 63) = 20.38; p < 0.0001]) throughout the experiment. d Relative mRNA expression of PGC1α in skeletal muscle (n = 6–7; two-way ANOVA; main effect of exercise; a priori comparisons €€ p < 0.01). e Corticosterone levels (ng/ml) in plasma (n = 9–10; two-way ANOVA; main effect of exercise: € p < 0.05 and €€ p < 0.01). Data are graphed as means + SEM. See supplementary Table 4 for details of statistical analysis.
Behaviours
All rats underwent behavioural testing (Pattern separation; Y-maze; Novelty suppressed feeding; Elevated plus maze (EPM) and Forced swim test) starting three weeks following initiation of exercise and BrdU injection. Rats were singly housed and acclimated to the testing room at least 45 min prior to each test. All behavioural tests were conducted during the light phase (8:30 am to 5:30 pm). All apparatus and objects were cleaned with a 70% ethanol solution between exposures of each animal to remove odour cues. Where possible, behaviours were scored in a blinded fashion or by using automated Ethovision video tracking software (XT 7.0; Noldus, The Netherlands) and AnyMaze (Stoelting, Europe). See SI Materials and Methods for details.
Tissue collection and post-mortem analysis
At week 10, five rats per group were euthanized with an injection of pentobarbital (90 mg/kg; i.p.), blood was collected by cardiac puncture, gut and muscle was dissected, and rats were transcardially perfused with saline followed by 4% (v/v) paraformaldehyde (#30525–89–4; Fisher) for immunohistochemical analysis. Brains were postfixed, cryoprotected in 30% sucrose and subsequently flash frozen. Brains were coronally sectioned (40 µm) in a 1:12 series through the hippocampus and preserved in an anti-freeze solution at –20 °C before processing (see supplementary information). The remaining five rats per group were euthanized by rapid decapitation. Hippocampus, colon, caecum content, and muscle were dissected and immediately snap frozen in liquid nitrogen. Blood samples were collected into ethylene-diamine tetra-acetic acid (EDTA) tubes and centrifuged (15 min, 5000 rpm). The resulting plasma was stored at −80 °C until further analysis (see supplementary information for details).
Metabolomics analysis
Metabolomics screening was performed on caecum content and hippocampus by MS-OMICS (Denmark) as previously described (https://www.msomics.com/metabolomics-methods/technical-information/). See SI Materials and Methods.
Statistical analysis
All datasets were assessed for normal distribution and equality of variances using Shapiro-Wilk test. Datasets that were normally distributed were statistically compared using two-way ANOVA followed by Tukey’s multiple comparison test. Non-parametric datasets were statistically compared using Kruskal–Wallis test followed by post-hoc Dunn’s test. A p value of less than 0.05 was considered significant. For metabolomics differential expression analysis, metabolite features were considered statistically significant at a false discovery rate (FDR)-corrected p < 0.05. For Spearman correlation analyses of metabolites and biochemical, immunohistochemical, and behavioural data, results were considered nominally significant at p < 0.05, and FDR-based correction for multiple comparisons was applied, with a significance threshold of 5%. Details of all statistical analysis are in Supplementary Tables 4–8.
Results
Gut microbiota disruption did not change body weight and running activity
Neither voluntary exercise nor antibiotics treatment affected rat body weight (Fig. 1b). This is similar to previous reports showing that rats that start to exercise in adulthood did not lose weight when compared to their sedentary counterparts [37, 38], and that voluntary running-induced body weight loss is mainly observed when exercise is initiated during adolescence [27]. The average distance run over the course of the experiment was comparable to previous published reports [39, 40] and similar in the exercise (Ex) and exercise+antibiotics (Ex+ABX) group (Fig. 1c). Proliferator-activated receptor gamma coactivator-1a (PGC1a), which has previously been shown to mediate an exercise-induced increase in hippocampal BDNF [41] was significantly increased in skeletal muscle by exercise (Fig. 1d). It has been previously shown that the lack of gut microbiota in germ-free mice increased the corticosterone response to stress [42], while voluntary exercise reduced the corticosterone response to stress [43], suggesting that both can regulate the HPA axis. Two-way ANOVA revealed that exercise decreased plasma corticosterone but there was no effect of antibiotics nor the exercise x antibiotics interaction. Rats that exercised had lower level of plasma corticosterone compared to their sedentary control regardless of antibiotics treatment (Sed vs Ex p = 0.0327; Sed+ABX vs Ex+ABX p = 0.0084) (Fig. 1e).
Gut microbiota disruption-induced low-grade peripheral inflammation and changes in gut physiology
There was a significant main effect of antibiotics on colonic TNFα mRNA expression while colonic IL-6 mRNA was not significantly affected by either intervention. We found that exercise and antibiotics independently increased the mRNA expression of IL-10 (p = 0.0002, Kruskal–Wallis), while the combination of both significantly reduced its expression. Two-way ANOVA showed a main effect of antibiotics on the expression of the chemokine CCL2 in the colon while neither exercise nor the exercise x antibiotics interaction affected its expression (Fig. 2a). We next assessed if the modest increase in gut inflammation was reflected in the blood. Two-way ANOVA analysis of plasma levels of inflammatory cytokines showed only a main effect of antibiotics on the concentration of TNFα. Subsequent planned comparison showed that antibiotics increased levels of TNFα in sedentary rats only (Sed vs Sed+ABX p = 0.0475) (Fig. 2b). Similarly, IL-6 levels were not affected by exercise but by antibiotics and by the exercise x antibiotics interaction. Post hoc analysis revealed that antibiotics increased IL-6 plasma level only in the exercise group (Ex vs Ex+ABX p = 0.0063) (Fig. 2c). We observed that there was a trend towards an effect of antibiotics on faecal output (p = 0.0609, Kurskal–Wallis; Fig. 2d). There was a main effect of antibiotics on the colon length (Fig. 2e), and caecum weight (Fig. S1a, b).
Effect of antibiotics and exercise on (a) relative mRNA expression of TNFɑ (n = 7; two-way ANOVA; main effect of Abx), IL-6 (n = 7; Kruskal–Wallis test), IL-10 (n = 7; Kruskal–Wallis test; p = 0,0002; Abx effect: $$p < 0.01; Exercise effect: €p < 0.05), and CCL2 (n = 7; two-way ANOVA; main effect of Abx) in the colon. Plasma concentration of (b) TNFɑ (pg/ml) and (c) IL-6 (pg/ml) (n = 9; two-way ANOVA; main effect of Abx: a priori comparisons $p < 0.05; interaction Abx x Ex **p < 0.01). d fecal output (n = 9; Kruskal–Wallis test) (e) colon length at the end of the study (n = 10; two-way ANOVA; main effect of Abx); f Volcano plot of caecal metabolites for Sed+ABX and Sed rats. Top ten most significant metabolites are highlighted. g Principal Component Analysis (PCA) of caecal metabolomics (h) Volcano plot of caecal metabolites for Sed and Ex rats. i Normalised peak areas for the metabolite Ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate; ***p < 0.001 (FDR-adjusted; Limma p value). Ex vs Sed: glog2 fold change = −4.8, pFDR-adjusted = 2.8E-8; Ex+ABX vs Sed+ABX: glog2 fold change = −6.2, pFDR-adjusted = 1.9E-11; Sed+ABX vs Sed comparison glog2 fold change = 2.6, pFDR-adjusted = 3.2E-5. Data are graphed as means + SEM. Abbreviation: FC fold change, glog2 generalised logarithm base 2. See Supplementary Table 5 for details of statistical analysis.
Gut microbiota disruption impacted the caecal but not hippocampal metabolome
Because the long-term disruption of gut microbiota induced physiological changes in the gut, we next performed mass spectrometry-based untargeted metabolomics analysis of caecal content and hippocampal tissue (See SI Materials and Methods). The antibiotic treatment caused a large shift in the caecal metabolome (Fig. 2f–i; Fig S1; Fig S2). Of the 610 caecal metabolites quantified, 527 were differentially regulated (pFDR-adjusted < 0.05) between Sed+ABX and Sed rats (Fig. 2f, g). Pathway analysis on differentially expressed metabolites revealed an enrichment in aminoacyl-tRNA synthesis (18 metabolites implicated), the citric acid cycle (7 metabolites implicated), and several amino acid-related pathways (Supplementary Table 3). Conversely, exercise had few effects on the caecal metabolome, as only six metabolites were differentially regulated between Ex and Sed rats (Fig. 2h) and only one in the Ex+ABX vs Sed+ABX comparison (Fig. S1b). The metabolite ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate was consistently downregulated by exercise (Sed vs Ex: pFDR-adjusted = 2.8E-8; ABX vs ABX+Ex pFDR-adjusted = 1.9E-11) but was also increased by antibiotics in the Sed vs Abx comparison (pFDR-adjusted = 3.2E-5; Fig. 2i). Of the 210 hippocampal metabolites quantified and annotated, two were differentially abundant as a function of antibiotics treatment in the Sed+ABX vs Sed comparison (Fig. S2a, b). The metabolite trimethylamine-N-oxide (TMAO) was decreased between Sed+ABX and Sed rats (pFDR-adjusted = 1.5E-5) and below the limit of the detection in Ex+ABX rats (Fig. S2e). This is consistent with the gut microbiota’s role in the biosynthesis of TMAO [44]. Interestingly, ergothioneine, a precursor of TMAO, was identified as significantly increased in the hippocampus (pFDR-adjusted = 0.045) following antibiotic treatment (Fig. S2b–g), but was not detected in caecal content. Several other TMAO precursors, such as carnitine metabolites and choline, were altered in the caecum, but not the hippocampus (Fig. S1c S2g; Supplementary Table 2), in the Sed+ABX vs Sed comparison. The metabolite ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate was not differentially expressed in the hippocampus (all comparisons, p > 0.05; Fig S2f). These results suggest loss of TMAO in the hippocampus by antibiotics-induced disruption of gut bacteria. No metabolites were differentially regulated as a function of exercise in the hippocampus in the Ex vs Sed and Ex+ABX vs Sed+ABX comparisons (all pFDR-adjusted > 0.05); though, 9 and 17 features, respectively, reached nominal significance (p < 0.05).
Exercise prevented a gut microbiota disruption-induced decrease in pattern separation
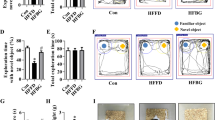
Changes in gut inflammation and the gut metabolome can impact upon hippocampal-dependent cognitive behaviours [45, 46] thus, given our observations, and the established exercise-induced enhancement of hippocampal function [47, 48] we examined the effects of exercise and antibiotics on hippocampal-dependent memory. Two-way ANOVA revealed that antibiotics but not exercise nor their interaction affected rats performance in the large separation paradigm of the MSLRT, a task of location discrimination in conditions of low contextual overlap (Fig. 3a). In the small separation, a condition of high contextual overlap (pattern separation) that requires AHN [24, 49], two-way ANOVA revealed a trend toward the effect of exercise on rats ability to pattern separate [p = 0.0742]. Both antibiotics and the interaction exercise x antibiotics significantly affected rats performance in this task (Fig. 3b). Subsequent post hoc analysis showed that antibiotics decreased rats performance in the small separation task (Sed vs Sed+ABX; p = 0.0003) and that exercise reversed this impairment (Sed+ABX vs Ex+ABX p = 0.045). In the two-trial Y-maze, a test of spatial reference memory, two-way ANOVA revealed no effect of exercise or exercise x antibiotics interaction but a main effect of antibiotics on the discrimination ratio (Fig. 3c), which was independent of rats’ locomotor activity in both trials on the Y-maze (Supplementary Fig. 3a).
a Schematic representation and discrimination ratio in the large separation task (n = 9; two-way ANOVA; main effect of Abx) and (b) in the small separation task (pattern separation) (n = 9; two-way ANOVA; interaction Abx x Ex; *p < 0.05;***p < 0.001); c Schematic representation and discrimination ratio between the exploration of the novel vs familiar arm during the first 2.5 min of the 2-trial Y-Maze test (n = 8–9; two-way ANOVA; main effect of Abx); d Schematic representation; latency to eat (n = 10; two-way ANOVA; main effect of Ex) and latency to first approach the centre (n = 10; two-way ANOVA; interaction Abx x Ex; *p < 0.05;**p < 0.01) in the novelty suppressed feeding test (NSFT) ; e Schematic representation and % of time spent in the open arms of the elevated plus maze (EPM) (n = 8–10; two-way ANOVA; interaction Abx x Ex; *p < 0.05;**p < 0.01); f Schematic representation and immobility, swimming (n = 10; two-way ANOVA; main effect Ex; a priori comparisons €p < 0.05) and climbing score in the Forced Swim Test (FST). Data are graphed as means + SEM. See Supplementary Table 6 for details of statistical analysis.
Exercise prevented gut microbiota disruption-induced changes in anxiety-like behaviours
Because the gut microbiota is involved in the regulation of mood-related behaviours [7, 28]. We assessed anxiety-like and despair-like behaviours in the novelty suppressed feeding test (NSFT), elevated plus maze (EPM) and Forced Swim test (FST; Fig. 3). A decreased latency to eat in the NSFT has been associated with increased AHN induced by antidepressant drugs [50, 51]. Two-way ANOVA revealed a main effect of exercise on the latency to eat (Fig. 3d). However, we cannot rule out that results from rats who exercised may have been driven by hunger as we found a significant weight loss induced by exercise (Fig S3b) as well as a significant correlation between the latency to eat and % body weight loss in rats in the exercise group, but not in rats in the sedentary, antibiotics nor exercise+antibiotics groups (Fig. S3c). The latency to approach the centre was significantly affected by the interaction between antibiotics and exercise. Post hoc analysis showed that there was an increased latency to reach the centre in antibiotics-treated rats (Sed vs Sed+ABX p = 0.0192), which was attenuated in rats that were also exposed to exercise (Sed+ABX vs Ex+ABX p = 0.0051). This result is independent of body weight loss as no significant correlations were observed between these parameters in all experimental groups (Fig. 3d). In addition, there was no difference in food consumption 30 min post-test (Fig. 3d). In the EPM (Fig. 3e), two-way ANOVA showed that antibiotics and the antibiotics x exercise interaction had an overall effect on percentage of time spent in the open arms. Post hoc analysis showed that antibiotics-treated rats spent less time in the open arms (Sed vs Sed+ABX p = 0.0096), indicative of an anxiogenic effect, which was attenuated in rats that were also exposed to exercise (Sed+ABX vs Ex+ABX p = 0.028) (Fig. 3e). Finally, we assessed despair-like behaviour in the FST (Fig. 3f). Climbing and immobility behaviours were unaffected by either exercise or antibiotics. Swimming behaviour was significantly affected by exercise. Antibiotics-treated rats who were exposed to exercise swam more than sedentary antibiotics-treated rats (Sed+ABX vs Ex+ABX p = 0.0212) (Fig. 3f). Overall, the data suggest that antibiotics increased anxiety-like behaviour and induce a slight enhancement in depressive-like behaviour which was attenuated by exercise.
Exercise mitigated the negative impact of gut microbiota disruption on adult hippocampal neurogenesis
Previous studies have demonstrated that pattern separation requires AHN [24, 49] and that AHN can be regulated by both exercise and the microbiome [32, 47]. We assessed AHN by determining the number BrdU + /NeuN+ cells in the dentate gyrus (DG) of the hippocampus (Fig. 4a–d). Two-way ANOVA revealed a main effect of exercise and antibiotics on AHN in the whole hippocampus, but not their interaction (Fig. 4a). Subdivision of regions across the longitudinal axis revealed that again, only exercise and antibiotics alone affected AHN in the dorsal region. Planned comparison revealed that the pro-neurogenic effect of exercise was specific to the dorsal region (Sed vs Ex p = 0.05) (Fig. 4b). In the ventral hippocampus, neither antibiotics or exercise significantly affected AHN (Fig. 4c). In line with these findings, we observed that there was no signification interaction between antibiotics and exercise on the total number of DCX-positive cells in the DG (Fig. 4e, f). However, both factors independently affected the number of DCX+ cells such that antibiotics decreased and exercise increased the number of DCX-positive cells in the DG (Fig. 4f, left panel). We next categorised the DCX+ cells as described by Plümpe and colleagues [52]; AB were cells with very short or no processes, CD cells had intermediate processes and immature morphology, and EF cells were more mature with multi-branched dendrites (Fig. 4e). Two-way ANOVA revealed that antibiotics had a main effect on the three categories of developing neurons. Similarly, exercise had a main effect on the maturation status of neurons but there was no significant interaction between Abx and Ex. Post hoc analysis showed that the antibiotics-induced reduction in DCX-positive cells was specific to CD-type cells (Sed vs Sed+Abx p = 0.03; Ex vs Ex+Abx p = 0.002), while the exercise-induced increase was limited to CD-type (Sed vs Ex p < 0.001 ; Abx vs Ex+Abx p = 0.009) and EF-type (Sed vs Ex p = 0.0096; Abx vs Ex+Abx p = 0.0091) cells. (Fig. 4e). Because BDNF mediates the neurogenic effect of exercise [53] we measured BDNF in hippocampus and plasma and found that exercise significantly increased concentrations of BDNF in both hippocampus (Fig. 4g) and plasma (Fig. 4h) regardless of the antibiotics intervention. Subsequent post hoc test showed that rats that exercised had higher levels of plasma BDNF than their sedentary counterparts (Sed vs Ex p = 0.0226; Fig. 4h). Because microglia influence the neurogenic niche [54, 55], and antibiotics activate microglia in mice [56], we assessed microglia status in the hippocampus of rats exposed to exercise and antibiotics (Fig S4). Our results show that long-term antibiotics trigger a low-grade inflammatory state in the hippocampus that is not fully restored by exercise. (See supplementary information and Fig S3). Overall, these results suggest that while long-term microbiota disruption induced low grade inflammation but did not impact upon BDNF, exercise attenuated the negative impact of microbiota disruption on AHN.
Immunohistochemical measurement of the survival of new neurons (BrdU and NeuN positive cells) in brain sections through (a) the whole (n = 5; two-way ANOVA; main effect of Abx; main effect of Ex) (b) dorsal (n = 5; two-way ANOVA; main effect of Abx; main effect of exercise; a priori comparisons €p < 0.05) and (c) ventral hippocampus (n = 5; two-way ANOVA). d representative images of BrdU/NeuN at X20 magnification (n = 5). e representative images of DCX+ cells at X40 magnification (f) DCX counts following classification based on their dendritic tree morphology and total count of all DCX+ cells (n = 5; two-way ANOVA; main effect of exercise: a priori comparisons €€p < 0.01; €€€p < 0.001 and main effect of Abx; a priori comparisons $p < 0.05; $$p < 0.01). g BDNF in hippocampus (pg/mg tissue) (n = 5; two-way ANOVA; main effect of exercise) (h) BDNF (ng/ml) in plasma (n = 10; two-way ANOVA; main effect of exercise: a priori comparisons €p < 0.05). Data are graphed as means + SEM. See Supplementary Table 7 for details of statistical analysis.
The metabolite ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl) acetate correlates with physiological and behavioural changes induced by gut microbiota disruption
After adjusting for multiple comparisons, the expression level of ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate was positively correlated with antibiotics-induced increase in plasma corticosterone (rspearman = 0.55, pFDR-adjusted = 0.0025) and antibiotics-induced increase of anxiety-like behaviour in the NSF (Latency to eat; rspearman = 0.44, pFDR-adjusted = 0.050). Interestingly, this metabolite was negatively correlated with hippocampal BNDF protein levels (rspearman = −0.66, pFDR-adjusted = 0.012), the number of surviving BrdU + /NeuN+ cells in the whole (rspearman = −0.60, pFDR-adjusted = 0.021) and dorsal (rspearman = −0.64, pFDR-adjusted = 0.018) hippocampus, the number of DCX+ cells in the whole hippocampus (rspearman = −0.77, pFDR-adjusted = 0.00061), and the swimming score in the FST (rspearman = −0.46, pFDR-adjusted = 0.050). We also found positive nominal correlation of the metabolite with plasma TNFa (rspearman = 0.34, p = 0.043), microglial soma size (rspearman = 0.50, p = 0.026) and antibiotics-induced anxiety-like behaviour in the NSF (latency to first approach to centre; rspearman = 0.34, p = 0.030), and in the EPM (time spent in the open arms; rspearman = −0.39, p = 0.021). In addition, ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl) acetate was negatively correlated with plasma BDNF (rspearman = −0.33, p = 0.036) and pattern separation performance in the small separation MSLR test (rspearman = −0.37, p = 0.028) (Fig. 5a, b).
a Spearman correlation heatmap for caecal ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate normalised expression, plasma BDNF, IL-6, TNFa, corticosterone, hippocampal BDNF protein levels, number of BrdU+/NeuN+ and DCX+ cells per mm2 in the whole, dorsal, and ventral hippocampus, and microglia soma size in the whole hippocampus. b Spearman correlation heatmap for caecal ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate normalised expression, performance in the MSLR (large and small separation), NSF, EPM and FST (swimming behaviour); *p < 0.5, **p < 0.01, ***p < 0.001; unadjusted.
Discussion
This study shows that selective peripheral and central changes induced by long term disruption of gut microbiota were partially reversed by exercise in adult male rats. We observed that antibiotic treatment led to impairments in pattern separation and anxiety-like behaviours, coupled with a decrease in AHN. We show that antibiotics induced low grade inflammation in the hippocampus by increasing the number of activated microglia, which could be linked to the decreased AHN. Increased low grade neuroinflammation was concomitant with an increase in peripheral and colonic inflammation. Moreover, we found that antibiotics but not exercise induced profound changes in the caecal metabolome and that these were mostly not reflected in the hippocampal metabolome. Exercise partially reversed the anxiety-like behaviours and pattern separation impairment induced by antibiotics by maintaining AHN and BDNF levels and reducing the activation state of the microglia and peripheral inflammatory status.
The association between the gut microbiota and peripheral inflammation has been already established [57, 58]. Microbiota manipulation with fecal microbiota transplant (FMT) regulates inflammation in the colon and plasma in male mice [59]. Here, we found that antibiotics increased plasma levels and colonic expression of TNF-α and that exercise prevented these changes. These results reflect previous findings showing that exercise reduced colonic inflammation induced by a high fat diet in male mice [60], which is known to affect gut microbiota composition [61]. IL-6 signalling is known to be altered by exercise [62] and the microbiome [63]. Here we found that while exercise alone maintained low level of IL-6, the combination of exercise and antibiotics induced an increase in plasma levels of IL-6 while the changes in colonic expression of IL-6 mRNA induced by antibiotics and exercise were more subtle. In addition, IL-6 can regulate the expression of anti-inflammatory cytokines such as IL-10 in the colon [64], which is in line with what we observed.
As a result of long-term microbiota disruption, the number and soma size of microglial cells was increased in both sedentary and exercised male rats. Elegant mixed-gender studies in mice have confirmed that the gut microbiota regulates microglia development and maturation [65]. Antibiotics administration to male mice increased microglia size and number [56] supporting the observations in our study. Another study found that neither antibiotics nor exercise affected microglia status in the whole brain but impacted Ly6Chi monocytes only, in female mice [33] suggesting a possible sex difference in response to exercise and antibiotics. In this regard, recent evidence points toward structural and functional sex differences in rodent microglia [66]. In addition, in the study from Möhle and colleagues, the possible region-specific effects may have been diluted, in contrast to our study, where analysis of microglia was restricted to the hippocampus. Moreover, another factor to consider when comparing our results with previously published observations is that we did not use metronidazole in the antibiotic cocktail. Metronidazole can cross the blood brain barrier (BBB) [67] and is used as a model of neurotoxicity in rats [68]. Mass spectrometry analysis allowed us to confirm that none of the antibiotics used in this study reached the hippocampus after 10 weeks of antibiotics treatment (Fig. S5). This supports previous findings [19] suggesting that our observations are mainly a consequence of the disruption of the gut microbiota without a direct effect of antibiotics on the brain per se. However, we cannot rule out potential off-target actions that may contribute to the observed phenotype. We found that exercise failed to reduce the antibiotics-induced neuroinflammatory state. It has been shown that exercise reduced microglia proliferation in aged mice [69] but had no effect in a mouse model of depression [70]. Another study showed that exercise mainly targeted astrocytes in a mouse model of Alzheimer’s disease [71], thus inconsistent reports on the effect of exercise on microglia suggest that further investigation is needed.
To date, the impact of voluntary exercise on the caecal metabolome in a model of gut microbiota disruption remains unexplored, while it has been reported that the main caecal metabolites affected by long term forced exercise in female rats are amino acids [72]. We found enrichment in several amino acid-related pathways among metabolites differentially expressed following antibiotics treatment in sedentary animals but not in exercised male rats. Indeed, the effects of voluntary exercise on the caecal metabolome were few and all but absent in the hippocampal metabolome. Moreover, the dramatic effects of antibiotics treatment on the caecal metabolome are likely to have masked the more subtle effects of voluntary exercise. The only metabolite to be robustly differentially expressed as a function of exercise but also antibiotics treatment was ethyl 2-(4-oxo-4,5-dihydro-1,3-thiazol-2-yl)acetate (PubChem ID: 658099). To date and to the best of the authors’ knowledge, only one publication mentions this compound (by a synonymous name), wherein it is used as an active methylene reagent in the production of azo dyes (colourant), with possible antimicrobial activity [73]. Xenometabolites as a general class have previously been found to be decreased in human body fluids after exercise [74]. It is interesting to note, that its expression correlated with plasma corticosterone (positively), hippocampal BDNF protein (negatively), surviving new-born neurons and newly born neurons in the hippocampus (negatively) as well as with behavioural tasks, including the AHN-dependent MSLR (nominally significant). Our results show that 86% of all quantified metabolites were differentially expressed in the Sed+ABX vs Sed comparison, implicating aminoacyl-tRNA biosynthesis, the citric acid cycle, and several amino acid-related pathways. This is unsurprising given the established link between gut microbiota species and levels of individual circulating metabolites belonging to these pathways and compound classes [75]. Despite the considerable effect of gut microbiota disruption on the caecal metabolome, few metabolites in the hippocampus were altered by antibiotics suggesting that the hippocampus may largely be protected from drastic peripheral changes. Previously, it has been shown that there were 38 differentially expressed metabolites in the cerebrum of germ-free male mice compared to wild-type mice suggesting an interaction between microbiota and brain metabolome [76]. This is in contrast to our focus on the hippocampus only, thus there may be region-specific effects of gut microbiota depletion and exercise or other communication pathways such as blood-brain barrier integrity [77, 78], the vagus nerve [79], SCFA or neuropeptides [1, 80].
An exception was TMAO, a hepatic metabolite produced from gut microbiota catabolism. TMAO is implicated in inflammatory states [81], BBB function [82], age-related cognitive impairment [83] and could also contribute to Alzheimer’s disease pathology by accelerating amyloid aggregation [84] and impairing synaptic plasticity [85]. Indeed, TMAO is present in human cerebrospinal fluid and associated with Alzheimer’s biomarkers and neurodegeneration [86, 87]. However here, the loss of TMAO as a function of antibiotics treatment was not accompanied by cognitive improvements or reduced inflammation, suggesting the relationship is more complex. Moreover, the antioxidant ergothioneine, a TMAO precursor, is known to increase AHN via TrkB signalling [88, 89] and could improve hippocampus-dependent memory in a neurotrophin-dependent manner [90]. Overall, despite antibiotics broadly impacting the gut metabolome, we could not identify any direct association between the gut metabolome and hippocampal metabolome that could explain the behavioural findings. However, it is possible that natural variations in microbiota composition, along with exercise, could have measurable cognitive effects through these metabolites.
The beneficial effect of exercise on cognition has been widely explored in healthy rodents [16] and in models of cognitive decline [91,92,93]. However, the interaction between exercise and gut microbiota on behaviour remains poorly documented. Previous reports reveal that gut microbiota is involved in hippocampal-dependent behaviours. For example, long term antibiotics exposure induced impairment in the Morris water maze (MWM) in male mice [94] and male rats [28]. To our knowledge we are the first to report that microbiota depletion reduced pattern separation, which is reliant on AHN. FMT approaches have bolstered the notion that gut microbiota are instrumental in hippocampal-dependent cognitive function reliant on AHN [2, 95]. For instance, transfer of a microbiome from Alzheimer’s patients into healthy young male rats impaired their performance in a pattern separation and MWM tasks [2]. The fact that we did not observe an increase in pattern separation by exercise alone is in line with a recent report by the developers of this behavioural task who have highlighted its sensitivity only to interventions that reduce AHN [49]. It is notable however, that exercise rescued the antibiotics-induced impairment in pattern separation, suggesting that the ability of exercise to enhance pattern separation may be a function of gut microbiota integrity.
In relation to anxiety-related behaviours, antibiotics increased the latency to first approach the centre in the NSFT which is reliant on AHN [51, 96]. Similar results were observed in the EPM where a decrease in time spent in the open arms as a result of microbiota depletion was prevented by exercise. These results are in line with previous findings showing that 3 weeks of antibiotics treatment increased anxiety behaviours in the openfield in adult male mice [97]. Exercise alone did not affect anxiety-like behaviours. These results are not surprising as most of the anxiolytic effect of exercise have been described in rat models of pathological states such as post-traumatic stress disorder [98]; restraint stress [99]; or in ageing [100]. Finally, we found that exercise increased the swimming time in the FST in rats treated with antibiotics. Previous study showed that microbiota depletion with antibiotics decreased swimming behaviour in male rats [28] and that a change in microbial diversity is associated with an increased immobility score in FST in male rats [101]. It is interesting to note that the antibiotics cocktail did not induce any body weight change, suggesting that this model did not alter the general health of the animal, similar to previous findings [19, 56].
The relationship between the gut microbiota and AHN has been increasingly explored in recent years [33, 102, 103]. Increased [32] or decreased [104] AHN has been observed in germ-free mice, dependent on sex and age, suggesting a complex interaction between the microbiome and AHN. Interestingly, aberrant AHN coupled with inflammatory changes in the hippocampus was reported in male mice of a mouse model of inflammatory bowel disease [105], a condition in which defective regulation of gut microbiota is evident [106]. Similar to our findings, antibiotics has been shown to decrease AHN in female mice, which was reversed by exercise [33]. The capacity for exercise to enhance AHN is well established [26]. Under baseline conditions, we did not observe a robust effect of exercise on the survival of new neurons (BrdU/NeuN), but the number of immature neurons (DCX) was significantly increased by exercise. Interestingly, we found that the antibiotics-induced reduction of BrdU/NeuN cells was specific to the dorsal hippocampus and that exercise restored this antibiotics-induced decrease in AHN. The dorsal hippocampus plays a role in cognitive behaviours [107] and it has been showed that a neuroinflammatory-induced decrease in neurogenesis in the dorsal hippocampus impaired pattern separation in male rats [108]. We also observed that exercise increased BDNF levels in both plasma and hippocampus irrespective of antibiotics, which could be an important mechanism by which exercise maintains AHN. A previous study has shown that antibiotics decreased mRNA expression of BDNF in mouse hippocampus [109]. We did not observe an effect of antibiotics on BDNF at protein level, which implicates the translation to protein expression as a potential mechanism which negates the effect of microbiota depletion on BDNF. Interestingly, germ-free mice displayed lower protein [110] and mRNA [111] levels of BDNF in the hippocampus. Thus, it is likely that that this reduction in BDNF is due to developmental issues due to the complete lack of microbiome in germ-free mice from birth, in contrast to the changes in the gut microbiome in adulthood that we have induced using antibiotics in the current study. It is important to note that exercise is known to regulate other peripheral factors that cannot be ruled out in the understanding of the gut-brain axis communication as they have an effect of AHN such as circulating hormones (leptin and adiponectin [53]), metabolites (β-hydroxybutyrate [112]) and serum lactate [17].
Our study has certain limitations that must be acknowledged. The pattern separation test (MSLR) employed in our study may not adequately capture the exercise-induced increase in AHN, because measuring improvements in pattern separation in rodents can be challenging [49]. Furthermore, it is crucial for future research to explore the interaction between exercise and the gut microbiota in female rodents considering the sex differences in response to exercise in rats [113, 114] and sex differences in AHN in germ-free mice [104].
Neurogenesis in the adult human hippocampus is still debated [115, 116] and many questions related to its function and regulation remain unanswered [117]. It is not possible to measure the effects of both exercise and a disrupted microbiome on AHN in live human subjects, thus rodent studies are necessary to expand our understanding of the role of gut microbiota in AHN and related behavioural changes. In line with this, a recent study from our laboratory showed that an Alzheimer’s phenotype, including decreased AHN and pattern separation, could be transferred from human Alzheimer’s patients to rats through the gut microbiota [2], confirming that the gut microbiota composition can impact upon cognitive function. Our study contributes to knowledge of the impact of exercise on gut-mediated changes in brain function by showing that it can mitigate cognitive impairment caused by antibiotics-induced gut dysbiosis. Antibiotics are widely prescribed and while their overuse and misuse contribute to the development of antibiotic-resistant bacteria [118], their long-term or repeated usage could also induce cognitive impairment by disrupting the microbiome. In addition, we show that despite the strongly disrupted microbiome, exercise could still exert beneficial effects, suggesting that some of the effects of exercise are independent of the microbiome, and that the use of exercise as a therapy or exercise mimetic [119] may be possible even in the context of disease with disrupted microbiome.
To conclude, our data demonstrate that long-term antibiotics treatment recapitulates the impairments of a disrupted gut microbiota, similar to that of germ-free rodents but without the neurodevelopmental impact of the absence of microbiota during the perinatal period [120]. We observed that exercise partially reversed behavioural and neurogenic changes induced by gut microbiota disruption and that these impairments occurred without significant changes in the hippocampal metabolome, despite drastic shifts in the caecal metabolome. Taken together, these data highlight the importance of the gut microbiota in AHN-dependent behaviours and demonstrate the power of lifestyle factors such as voluntary exercise to attenuate these changes.
Data availability
All code and original data supporting the current study are available upon request.
References
Cryan JF, O’Riordan KJ, Cowan CSM, Sandhu KV, Bastiaanssen TFS, Boehme M, et al. The microbiota-gut-brain axis. Physiol Rev. 2019;99:1877–2013.
Grabrucker S, Marizzoni M, Silajdžić E, Lopizzo N, Mombelli E, Nicolas S, et al. Microbiota from Alzheimer’s patients induce deficits in cognition and hippocampal neurogenesis. Brain. 2023;146:4916–34.
Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE, et al. Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s Disease. Cell. 2016;167:1469–80.e12.
Cruz-Pereira JS, Rea K, Nolan YM, O’Leary OF, Dinan TG, Cryan JF. Depression’s unholy trinity: dysregulated stress, immunity, and the microbiome. Annu Rev Psychol. 2020;71:49–78.
O’Toole PW, Claesson MJ. Gut microbiota: changes throughout the lifespan from infancy to elderly. Int Dairy J. 2010;20:281–91.
Provensi G, Schmidt SD, Boehme M, Bastiaanssen TFS, Rani B, Costa A, et al. Preventing adolescent stress-induced cognitive and microbiome changes by diet. Proc Natl Acad Sci USA. 2019;116:9644–51.
Donoso F, Cryan JF, Olavarría-Ramírez L, Nolan YM, Clarke G. Inflammation, lifestyle factors, and the microbiome-gut-brain axis: relevance to depression and antidepressant action. Clin Pharm Ther. 2022;113:246–59.
Gubert C, Kong G, Renoir T, Hannan AJ. Exercise, diet and stress as modulators of gut microbiota: implications for neurodegenerative diseases. Neurobiol Dis. 2020;134:104621.
Mitchell CM, Davy BM, Hulver MW, Neilson AP, Bennett BJ, Davy KP. Does exercise alter gut microbial composition? A systematic review. Med Sci Sports Exerc. 2019;51:160–7.
Lai Z, Shan W, Li J, Min J, Zeng X, Zuo Z. Appropriate exercise level attenuates gut dysbiosis and valeric acid increase to improve neuroplasticity and cognitive function after surgery in mice. Mol Psychiatry. 2021;26:7167–87.
Monda V, Villano I, Messina A, Valenzano A, Esposito T, Moscatelli F, et al. Exercise modifies the gut microbiota with positive health effects. Oxid Med Cell Longev. 2017;2017:3831972.
Matsumoto M, Inoue R, Tsukahara T, Ushida K, Chiji H, Matsubara N, et al. Voluntary running exercise alters microbiota composition and increases n-butyrate concentration in the rat cecum. Biosci Biotechnol Biochem. 2008;72:572–6.
Vancampfort D, Stubbs B, Lara E, Vandenbulcke M, Swinnen N, Smith L, et al. Mild cognitive impairment and sedentary behavior: a multinational study. Exp Gerontol. 2018;108:174–80.
Hillman CH, Erickson KI, Kramer AF. Be smart, exercise your heart: exercise effects on brain and cognition. Nat Rev Neurosci. 2008;9:58–65.
Nota MHC, Nicolas S, O’Leary OF, Nolan YM. Outrunning a bad diet: interactions between exercise and a Western-style diet for adolescent mental health, metabolism and microbes. Neurosci Biobehav Rev. 2023;149:105147.
Gomes da Silva S, Unsain N, Mascó DH, Toscano-Silva M, De Amorim HA, Silva, et al. Early exercise promotes positive hippocampal plasticity and improves spatial memory in the adult life of rats. Hippocampus 2012;22:347–58.
Morland C, Andersson KA, Haugen ØP, Hadzic A, Kleppa L, Gille A, et al. Exercise induces cerebral VEGF and angiogenesis via the lactate receptor HCAR1. Nat Commun. 2017;8:1–9.
Llorens-Martín M, Torres-Alemán I, Trejo JL. Mechanisms mediating brain plasticity: IGF1 and adult hippocampal neurogenesis. Neuroscientist. 2009;15:134–48.
Fröhlich EE, Farzi A, Mayerhofer R, Reichmann F, Jačan A, Wagner B, et al. Cognitive impairment by antibiotic-induced gut dysbiosis: analysis of gut microbiota-brain communication. Brain Behav Immun. 2016;56:140–55.
Rendeiro C, Rhodes JS. A new perspective of the hippocampus in the origin of exercise–brain interactions. Brain Struct Funct. 2018;223:2527–45.
Kozareva DA, Cryan JF, Nolan YM. Born this way: hippocampal neurogenesis across the lifespan. Aging Cell. 2019;18:e13007.
Anacker C, Hen R. Adult hippocampal neurogenesis and cognitive flexibility-linking memory and mood. Nat Rev Neurosci. 2017;18:335–46.
Revest JM, Dupret D, Koehl M, Funk-Reiter C, Grosjean N, Piazza PV, et al. Adult hippocampal neurogenesis is involved in anxiety-related behaviors. Mol Psychiatry. 2009;14:959–67.
Clelland CD, Choi M, Romberg C, Clemenson GD, Fragniere A, Tyers P, et al. A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science. 2009;325:210–3.
Nakashiba T, Cushman JD, Pelkey KA, Renaudineau S, Buhl DL, McHugh TJ, et al. Young dentate granule cells mediate pattern separation, whereas old granule cells facilitate pattern completion. Cell. 2012;149:188–201.
Van Praag H, Christie BR, Sejnowski TJ, Gage FH. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc Natl Acad Sci USA. 1999;96:13427–31.
O’Leary JD, Hoban AE, Murphy A, O’Leary OF, Cryan JF, Nolan YM, et al. Differential effects of adolescent and adult-initiated voluntary exercise on context and cued fear conditioning. Neuropharmacology. 2019;145:49–58.
Hoban AE, Moloney RD, Golubeva AV, McVey Neufeld KA, O’Sullivan O, Patterson E, et al. Behavioural and neurochemical consequences of chronic gut microbiota depletion during adulthood in the rat. Neuroscience. 2016;339:463–77.
Rei D, Saha S, Haddad M, Haider Rubio A, Perlaza BL, et al. Age-associated gut microbiota impairs hippocampus-dependent memory in a vagus-dependent manner. JCI Insight. 2022;7:e147700.
Lupori L, Cornuti S, Mazziotti R, Borghi E, Ottaviano E, Cas MD, et al. The gut microbiota of environmentally enriched mice regulates visual cortical plasticity. Cell Rep. 2022;38:110212.
Darch HT, Collins MK, O’Riordan KJ, Cryan JF. Microbial memories: sex-dependent impact of the gut microbiome on hippocampal plasticity. Eur J Neurosci. 2021;54:5235–44.
Ogbonnaya ES, Clarke G, Shanahan F, Dinan TG, Cryan JF, O’Leary OF. Adult hippocampal neurogenesis is regulated by the microbiome. Biol Psychiatry. 2015;78:e7–9.
Möhle L, Mattei D, Heimesaat MM, Bereswill S, Fischer A, Alutis M, et al. Ly6Chi monocytes provide a link between antibiotic-induced changes in gut microbiota and adult hippocampal neurogenesis. Cell Rep. 2016;15:1945–56.
Stranahan AM, Khalil D, Gould E. Social isolation delays the positive effects of running on adult neurogenesis. Nat Neurosci. 2006;9:526–33.
Leasure JL, Decker L. Social isolation prevents exercise-induced proliferation of hippocampal progenitor cells in female rats. Hippocampus. 2009;19:907–12.
Hueston CM, Cryan JF, Nolan YM. Adolescent social isolation stress unmasks the combined effects of adolescent exercise and adult inflammation on hippocampal neurogenesis and behavior. Neuroscience. 2017;365:226–36.
Lee JCDi, Yau SY, Lee TMC, Lau BWM, So KF. Voluntary wheel running reverses the decrease in subventricular zone neurogenesis caused by corticosterone. Cell Transpl. 2016;25:1979–86.
Lee MC, Inoue K, Okamoto M, Liu YF, Matsui T, Yook JS, et al. Voluntary resistance running induces increased hippocampal neurogenesis in rats comparable to load-free running. Neurosci Lett. 2013;537:6–10.
Matiello R, Fukui RT, Silva ME, Rocha DM, Wajchenberg BL, Azhar S, et al. Differential regulation of PGC-1 expression in rat liver and skeletal muscle in response to voluntary running. Nutr Metab. 2010;7:1–8.
Borzykh AA, Gaynullina DK, Shvetsova AA, Kiryukhina OO, Kuzmin IV, Selivanova EK, et al. Voluntary wheel exercise training affects locomotor muscle, but not the diaphragm in the rat. Front Physiol. 2022;13:1–14.
Wrann CD, White JP, Salogiannnis J, Laznik-Bogoslavski D, Wu J, Ma D, et al. Exercise induces hippocampal BDNF through a PGC-1α/FNDC5 pathway. Cell Metab. 2013;18:649–59.
Luo Y, Zeng B, Zeng L, Du X, Li B, Huo R, et al. Gut microbiota regulates mouse behaviors through glucocorticoid receptor pathway genes in the hippocampus. Transl Psychiatry. 2018;8:187.
Zolfaghari FS, Pirri F, Gauvin E, Peeri M, Amiri S. Exercise and fluoxetine treatment during adolescence protect against early life stress-induced behavioral abnormalities in adult rats. Pharm Biochem Behav. 2021;205:173190.
Zhu Y, Jameson E, Crosatti M, Schäfer H, Rajakumar K, Bugg TDH, et al. Carnitine metabolism to trimethylamine by an unusual Rieske-type oxygenase from human microbiota. Proc Natl Acad Sci USA. 2014;111:4268–73.
Zhan G, Yang N, Li S, Huang N, Fang X, Zhang J, et al. Abnormal gut microbiota composition contributes to cognitive dysfunction in SAMP8 mice. Aging. 2018;10:1257–67.
Guzzetta KE, Cryan JF, O’Leary OF. Microbiota-gut-brain axis regulation of adult hippocampal neurogenesis. Brain Plast. 2022;8:97–119.
van Praag H, Kempermann G, Gage FH. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci. 1999;2:266–70.
Van Praag H, Shubert T, Zhao C, Gage FH. Exercise enhances learning and hippocampal neurogenesis in aged mice. J Neurosci. 2005;25:8680–5.
Reichelt AC, Kramar CP, Ghosh-Swaby OR, Sheppard PAS, Kent BA, Bekinschtein P, et al. The spontaneous location recognition task for assessing spatial pattern separation and memory across a delay in rats and mice. Nat Protoc. 2021;16:5616–33.
Santarelli L, Saxe M, Gross C, Surget A, Battaglia F, Dulawa S, et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science. 2003;301:805–9.
David DJ, Samuels BA, Rainer Q, Wang JW, Marsteller D, Mendez I, et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron. 2009;62:479–93.
Plümpe T, Ehninger D, Steiner B, Klempin F, Jessberger S, Brandt M, et al. Variability of doublecortin-associated dendrite maturation in adult hippocampal neurogenesis is independent of the regulation of precursor cell proliferation. BMC Neurosci. 2006;7:77.
Yau SY, Li A, Hoo RLC, Ching YP, Christie BR, Lee TMC, et al. Physical exercise-induced hippocampal neurogenesis and antidepressant effects are mediated by the adipocyte hormone adiponectin. Proc Natl Acad Sci USA. 2014;111:15810–5.
Sato K. Effects of microglia on neurogenesis. Glia. 2015;63:1394–405.
Kozareva DA, Moloney GM, Hoban AE, Rossini V, Nally K, Cryan JF, et al. A role for the orphan nuclear receptor TLX in the interaction between neural precursor cells and microglia. Neuronal Signal. 2019;3:NS20180177.
Çalışkan G, French T, Enrile Lacalle S, del Angel M, Steffen J, Heimesaat MM, et al. Antibiotic-induced gut dysbiosis leads to activation of microglia and impairment of cholinergic gamma oscillations in the hippocampus. Brain Behav Immun. 2021;99:203–17.
Rooks MG, Garrett WS. Gut microbiota, metabolites and host immunity. Nat Rev Immunol. 2016;16:341–52.
van de Wouw M, Lyte JM, Boehme M, Sichetti M, Moloney G, Goodson MS, et al. The role of the microbiota in acute stress-induced myeloid immune cell trafficking. Brain Behav Immun. 2020;84:209–17.
Kim N, Jeon SH, Ju IG, Gee MS, Do J, Oh MS, et al. Transplantation of gut microbiota derived from Alzheimer’s disease mouse model impairs memory function and neurogenesis in C57BL/6 mice. Brain Behav Immun. 2021;98:357–65.
Liu WX, Wang T, Zhou F, Wang Y, Xing JW, Zhang S, et al. Voluntary exercise prevents colonic inflammation in high-fat diet-induced obese mice by up-regulating PPAR-γ activity. Biochem Biophys Res Commun. 2015;459:475–80.
McKim DB, Niraula A, Tarr AJ, Wohleb ES, Sheridan JF, Godbout JP. Neuroinflammatory dynamics underlie memory impairments after repeated social defeat. J Neurosci. 2016;36:2590–604.
Pedersen BK. IL-6 signalling in exercise and disease. Biochem Soc Trans. 2007;35:1295–7.
Wang C, Li W, Wang H, Ma Y, Zhao X, Zhang X, et al. Saccharomyces boulardii alleviates ulcerative colitis carcinogenesis in mice by reducing TNF-α and IL-6 levels and functions and by rebalancing intestinal microbiota. BMC Microbiol. 2019;19:1–12.
Ye M, Joosse ME, Liu L, Sun Y, Dong Y, Cai C, et al. Deletion of IL-6 exacerbates colitis and induces systemic inflammation in IL-10-deficient mice. J Crohn’s Colitis. 2020;14:831–40.
Erny D, Hrabě de Angelis AL, Jaitin D, Wieghofer P, Staszewski O, David E, et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci. 2015;18:965–77.
Han J, Fan Y, Zhou K, Blomgren K, Harris RA. Uncovering sex differences of rodent microglia. J Neuroinflamm. 2021;18:1–11.
Ogbodo E, Ogbonye E, Ejimofor O, Ezeugwunne I, Madukwe D. The effect of metronidazole on the histology of the cerebellum and pituitary gland in female wistar rats. IP Indian J Neurosci. 2020;6:67–72.
Chaturvedi S, Malik MY, Rashid M, Singh S, Tiwari V, Gupta P, et al. Mechanistic exploration of quercetin against metronidazole induced neurotoxicity in rats: possible role of nitric oxide isoforms and inflammatory cytokines. Neurotoxicology. 2020;79:1–10.
Kohman RA, DeYoung EK, Bhattacharya TK, Peterson LN, Rhodes JS. Wheel running attenuates microglia proliferation and increases expression of a proneurogenic phenotype in the hippocampus of aged mice. Brain Behav Immun. 2012;26:803–10.
Svensson M, Andersson E, Manouchehrian O, Yang Y, Deierborg T. Voluntary running does not reduce neuroinflammation or improve non-cognitive behavior in the 5xFAD mouse model of Alzheimer’s disease. Sci Rep. 2020;10:1–10.
Belaya I, Ivanova M, Sorvari A, Ilicic M, Loppi S, Koivisto H, et al. Astrocyte remodeling in the beneficial effects of long-term voluntary exercise in Alzheimer’s disease. J Neuroinflamm. 2020;17:1–19.
Deda O, Gika H, Panagoulis T, Taitzoglou I, Raikos N, Theodoridis G. Impact of exercise on fecal and cecal metabolome over aging: a longitudinal study in rats. Bioanalysis. 2017;9:21–36.
Hassan EA, Mashaly HM, Hashem ZM, Zayed SE, Abo-Bakr AM. Eco-friendly synthesis of new polyfunctional Azo Dyes using shrimp chitin as a catalyst: application on polyester fabrics and their biological activities. Fibers Polym. 2022;23:2373–83.
Schranner D, Kastenmüller G, Schönfelder M, Römisch-Margl W, Wackerhage H. Metabolite concentration changes in humans after a bout of exercise: a systematic review of exercise metabolomics studies. Sport Med - open. 2020;6:11.
Dekkers KF, Sayols-Baixeras S, Baldanzi G, Nowak C, Hammar U, Nguyen D, et al. An online atlas of human plasma metabolite signatures of gut microbiome composition. Nat Commun.2023;14:2971.
Matsumoto M, Kibe R, Ooga T, Aiba Y, Sawaki E, Koga Y, et al. Cerebral low-molecular metabolites influenced by intestinal microbiota: a pilot study. Front Syst Neurosci. 2013;7:9.
Parker A, Fonseca S, Carding SR. Gut microbes and metabolites as modulators of blood-brain barrier integrity and brain health. Gut Microbes. 2020;11:135–57.
Celorrio M, Abellanas MA, Rhodes J, Goodwin V, Moritz J, Vadivelu S, et al. Gut microbial dysbiosis after traumatic brain injury modulates the immune response and impairs neurogenesis. Acta Neuropathol Commun. 2021;9:40.
Fülling C, Dinan TG, Cryan JF. Gut microbe to brain signaling: what happens in Vagus…. Neuron. 2019;101:998–1002.
Morais LH, Schreiber HL 4th, Mazmanian SK. The gut microbiota-brain axis in behaviour and brain disorders. Nat Rev Microbiol. 2021;19:241–55.
Janeiro MH, Ramírez MJ, Milagro FI, Martínez JA, Solas M. Implication of Trimethylamine N-Oxide (TMAO) in disease: potential biomarker or new therapeutic target. Nutrients. 2018;10:1398.
Knox EG, Aburto MR, Clarke G, Cryan JF, O’Driscoll CM. The blood-brain barrier in aging and neurodegeneration. Mol Psychiatry. 2022;27:2659–73.
Li D, Ke Y, Zhan R, Liu C, Zhao M, Zeng A, et al. Trimethylamine-N-oxide promotes brain aging and cognitive impairment in mice. Aging Cell. 2018;17:e12768.
Yang DS, Yip CM, Huang TH, Chakrabartty A, Fraser PE. Manipulating the amyloid-beta aggregation pathway with chemical chaperones. J Biol Chem. 1999;274:32970–4.
Govindarajulu M, Pinky PD, Steinke I, Bloemer J, Ramesh S, Kariharan T, et al. Gut metabolite TMAO induces synaptic plasticity deficits by promoting endoplasmic reticulum stress. Front Mol Neurosci. 2020;13:138.
Del Rio D, Zimetti F, Caffarra P, Tassotti M, Bernini F, Brighenti F, et al. The gut microbial metabolite trimethylamine-N-oxide is present in human cerebrospinal fluid. Nutrients. 2017;9:2–5.
Vogt NM, Romano KA, Darst BF, Engelman CD, Johnson SC, Carlsson CM, et al. The gut microbiota-derived metabolite trimethylamine N-oxide is elevated in Alzheimer’s disease. Alzheimer’s Res Ther. 2018;10:1–8.
Ishimoto T, Masuo Y, Kato Y, Nakamichi N. Ergothioneine-induced neuronal differentiation is mediated through activation of S6K1 and neurotrophin 4/5-TrkB signaling in murine neural stem cells. Cell Signal. 2019;53:269–80.
Nakamichi N, Nakayama K, Ishimoto T, Masuo Y, Wakayama T, Sekiguchi H, et al. Food-derived hydrophilic antioxidant ergothioneine is distributed to the brain and exerts antidepressant effect in mice. Brain Behav. 2016;6:e00477.
Nakamichi N, Nakao S, Nishiyama M, Takeda Y, Ishimoto T, Masuo Y, et al. Oral administration of the food-derived hydrophilic antioxidant ergothioneine enhances object recognition memory in mice. Curr Mol Pharm. 2021;14:220–33.
Crowley EK, Nolan YM, Sullivan AM. Exercise as a therapeutic intervention for motor and non-motor symptoms in Parkinson’s disease: evidence from rodent models. Prog Neurobiol. 2019;172:2–22.
Horowitz AM, Fan X, Bieri G, Smith LK, Sanchez-Diaz CI, Schroer AB, et al. Blood factors transfer beneficial effects of exercise on neurogenesis and cognition to the aged brain. Science. 2020;369:167–73.
Choi SH, Bylykbashi E, Chatila ZK, Lee SW, Pulli B, Clemenson GD, et al. Combined adult neurogenesis and BDNF mimic exercise effects on cognition in an Alzheimer’s mouse model. Science. 2018;361:eaan8821.
Mosaferi B, Jand Y, Salari AA. Antibiotic-induced gut microbiota depletion from early adolescence exacerbates spatial but not recognition memory impairment in adult male C57BL/6 mice with Alzheimer-like disease. Brain Res Bull. 2021;176:8–17.
Lee J, Venna VR, Durgan DJ, Shi H, Hudobenko J, Putluri N, et al. Young versus aged microbiota transplants to germ-free mice: increased short-chain fatty acids and improved cognitive performance. Gut Microbes. 2020;12:1–14.
Nicolas S, Veyssière J, Gandin C, Zsürger N, Pietri M, Heurteaux C, et al. Neurogenesis-independent antidepressant-like effects of enriched environment is dependent on adiponectin. Psychoneuroendocrinology. 2015;57:eaan8821.
Li J, Pu F, Peng C, Wang Y, Zhang Y, Wu S, et al. Antibiotic cocktail-induced gut microbiota depletion in different stages could cause host cognitive impairment and emotional disorders in adulthood in different manners. Neurobiol Dis. 2022;170:105757.
Yakhkeshi R, Roshani F, Akhoundzadeh K, Shafia S. Effect of treadmill exercise on serum corticosterone, serum and hippocampal BDNF, hippocampal apoptosis and anxiety behavior in an ovariectomized rat model of post-traumatic stress disorder (PTSD). Physiol Behav. 2022;243:113629.
Lapmanee S, Charoenphandhu J, Teerapornpuntakit J, Krishnamra N, Charoenphandhu N. Agomelatine, venlafaxine, and running exercise effectively prevent anxiety- and depression-like behaviors and memory impairment in restraint stressed rats. PLoS One. 2017;12:1–23.
Pietrelli A, Di Nardo M, Masucci A, Brusco A, Basso N, Matkovic L. Lifelong aerobic exercise reduces the stress response in rats. Neuroscience. 2018;376:94–107.
Forouzan S, Hoffman KL, Kosten TA. Methamphetamine exposure and its cessation alter gut microbiota and induce depressive-like behavioral effects on rats. Psychopharmacology. 2021;238:281–92.
Chevalier G, Siopi E, Guenin-Macé L, Pascal M, Laval T, Rifflet A, et al. Effect of gut microbiota on depressive-like behaviors in mice is mediated by the endocannabinoid system. Nat Commun. 2020;11:6363.
Dohm-Hansen S, Donoso F, Lucassen PJ, Clarke G, Nolan YM. The gut microbiome and adult hippocampal neurogenesis: a new focal point for epilepsy? Neurobiol Dis. 2022;170:105746.
Scott GA, Terstege DJ, Vu AP, Law S, Evans A, Epp JR. Disrupted neurogenesis in germ-free mice: effects of age and sex. Front Cell Dev Biol. 2020;8:407.
Gampierakis IA, Koutmani Y, Semitekolou M, Morianos I, Polissidis A, Katsouda A, et al. Hippocampal neural stem cells and microglia response to experimental inflammatory bowel disease (IBD). Mol Psychiatry. 2021;26:1248–63.
Sheehan D, Shanahan F. The gut microbiota in inflammatory bowel disease. Gastroenterol Clin North Am. 2017;46:143–54.
Fanselow MS, Dong HW. Are the dorsal and ventral hippocampus functionally distinct structures? Neuron. 2010;65:7–19.
Hueston CM, O’Leary JD, Hoban AE, Kozareva DA, Pawley LC, O’Leary OF, et al. Chronic interleukin-1β in the dorsal hippocampus impairs behavioural pattern separation. Brain Behav Immun. 2018;74:252–64.
Desbonnet L, Clarke G, Traplin A, O’Sullivan O, Crispie F, Moloney RD, et al. Gut microbiota depletion from early adolescence in mice: Implications for brain and behaviour. Brain Behav Immun. 2015;48:165–73.
Sudo N, Chida Y, Aiba Y, Sonoda J, Oyama N, Yu XN, et al. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J Physiol. 2004;558:263–75.
Heijtz RD, Wang S, Anuar F, Qian Y, Björkholm B, Samuelsson A, et al. Normal gut microbiota modulates brain development and behavior. Proc Natl Acad Sci USA. 2011;108:3047–52.
Sleiman SF, Henry J, Al-Haddad R, El Hayek L, Abou Haidar E, Stringer T, et al. Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body β-hydroxybutyrate. Elife. 2016;5:e15092.
Boakes RA, Mills KJ, Single JP. Sex differences in the relationship between activity and weight loss in the rat. Behav Neurosci. 1999;113:1080–9.
Purohit DC, Mandyam AD, Terranova MJ, Mandyam CD. Voluntary wheel running during adolescence distinctly alters running output in adulthood in male and female rats. Behav Brain Res. 2020;377:112235.
Moreno-Jiménez EP, Flor-García M, Terreros-Roncal J, Rábano A, Cafini F, Pallas-Bazarra N, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25:554–60.
Sorrells SF, Paredes MF, Cebrian-Silla A, Sandoval K, Qi D, Kelley KW, et al. Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults. Nature. 2018;555:377–81.
Kempermann G, Gage FH, Aigner L, Song H, Curtis MA, Thuret S, et al. Human adult neurogenesis: evidence and remaining questions. Cell Stem Cell. 2018;23:25–30.
Aggarwal R, Mahajan P, Pandiya S, Bajaj A, Verma SK, Yadav P, et al. Antibiotic resistance: a global crisis, problems and solutions. Crit Rev Microbio. 1–26. Available from: https://doi.org/10.1080/1040841X.2024.2313024.
Guerrieri D, Moon HY, van Praag H. Exercise in a pill: the latest on exercise-mimetics. Brain Plast. 2017;2:153–69.
Gareau MG. Cognitive function and the microbiome. Int Rev Neurobiol. 2016;131:227–46.
Acknowledgements
We thank Dr. Stefanie Grabrucker, Dr. Kieran Rea, Tara Foley and Patrick Fitzgerald for technical assistance. This work was funded by Science Foundation Ireland (SFI) under Grant Number SFI/FFP/6820. SN was a recipient of an Irish Research Council Postdoctoral Fellowship (GOIPD/2018/550). YMN and AL are funded investigators and JFC a principal investigator of the APC Microbiome Ireland, which is a research centre funded by SFI, through the Irish Government’s National Development Plan (Grant number 12/RC/2273_P2).
Author information
Authors and Affiliations
Contributions
SN: conceptualisation, data acquisition and analysis, interpretation, writing – original draft preparation, editing. SD-H: analysis, interpretation, and writing of metabolomics data. AL and TFSB: analysis and interpretation of metabolomics data. JAE: supervision, interpretation of metabolomics data. JFC: conceptualisation, supervision, review and editing. YN: conceptualisation, funding acquisition, supervision, interpretation, writing, review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nicolas, S., Dohm-Hansen, S., Lavelle, A. et al. Exercise mitigates a gut microbiota-mediated reduction in adult hippocampal neurogenesis and associated behaviours in rats. Transl Psychiatry 14, 195 (2024). https://doi.org/10.1038/s41398-024-02904-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-024-02904-0
- Springer Nature Limited
This article is cited by
-
Can exercise benefits be harnessed with drugs? A new way to combat neurodegenerative diseases by boosting neurogenesis
Translational Neurodegeneration (2024)