Abstract
The question of whether immune dysfunction contributes to risk of psychiatric disorders has long been a subject of interest. To assert this hypothesis a plethora of correlative evidence has been accumulated from the past decades; however, a variety of technical and practical obstacles impeded on a cause-effect interpretation of these data. With the advent of large-scale omics technology and advanced statistical models, particularly Mendelian randomization, new studies testing this old hypothesis are accruing. Here we synthesize these new findings from genomics and genetic causal inference studies on the role of immune dysfunction in major psychiatric disorders and reconcile these new data with pre-omics findings. By reconciling these evidences, we aim to identify key gaps and propose directions for future studies in the field.
Similar content being viewed by others
Introduction
Psychiatric disorders have surpassed physical disorders to become the leading causes of disability worldwide [1] but, still, our understanding of their underlying pathophysiology remains incomplete. One of the contemporary hypotheses is the “two-hits” or “multi-hits” model—in the context of genetic predispositions to these psychiatric disorders (the first hit; e.g., schizophrenia, SCZ), the accumulating effects of a second or more adverse exposures (the second hit) may lead to the diagnoses [2,3,4]. Perturbations to the immune system by these adverse exposures, such as infection [5,6,7,8] and stress [9,10,11,12], are an integral part of this hypothesis [2,3,4].
The immune system is the key machinery in protecting the body from exogenous and endogenous threats [13]. It, generally, comprises two interconnected arms, the innate and adaptive arm. The innate immunity is the first line of defense when the body faces threats: it responds swiftly and intensely but not specific to antigens. On the contrary, the adaptive arm responds by the coordination of a range of T cells and B cells that are targeted to the specific antigens in an elegant way, but may require days to fully take in action. Proper functioning of the immune system requires responses from both arms in concert, involving harmonized expression of a large network of molecules, e.g., the signaling molecules—cytokines, within a proper context [14]. In addition, the immune system is dynamic, and both its composition and responses change with development and aging [15]. Unfortunately, this complex nature of the immune system is predisposed to dysregulations which underlie many human diseases [16,17,18,19].
The relationship between immune dysfunction and psychiatric disorders have been extensively explored in the literature. Findings from epidemiological or clinical studies have recently been synthesized in several excellent meta-analyses and reviews [20,21,22,23,24,25,26,27,28]. Thus, we will not review them in depth here; instead, we will, after briefly summarizing the key findings from these studies, focus on recent evidence reported from large-scale genomic studies. Specifically, we highlight the use of Mendelian randomization (MR) [29] for causal inference in psychiatric disorders, which have not been thoroughly reviewed yet. Due to the requirement of large-scale data, MR studies to date have been performed on data shared by few consortia, and resulted in a significant data overlap, making it difficult to conduct a comprehensive meta-analysis. We will employ a narrative approach to summarize these findings and discuss their implications for future research.
Epidemiological and clinical findings
The initial evidence for the involvement of immune dysfunction in psychiatric disorders came from studies examining the relationships between infections and psychiatric diagnosis [5, 7, 8, 30,31,32,33]. Maternal infections or exposures to pathogens have long been suggested as risk factors to psychiatric illness in offspring, for example, attention-deficit/hyperactivity disorders (ADHD) [34], autism spectrum disorder (ASD) [35], schizophrenia (SCZ) [5] and others [6, 33]. Postnatal early life infections have also been extensively reported to associate with later diagnosis of SCZ [5, 36], bipolar disorder (BIP) [31] and ASD [35]. As infection events preceded disorder diagnoses, these findings seemingly dictate potential causal relations. But their limitations are also apparent. Among these studies, there was considerable variability in pathogen types, i.e., from virus (e.g., cytomegalovirus), bacterium to parasite (e.g., toxoplasma gondii), in the infected body systems (e.g., respiratory, gastrointestinal), in the time of infection (the trimester or age of infected) and in severity of the infections examined. Therefore, these findings have not yet reached a consensus on the kind of infections that causes mental health problems later in life. In addition, other forms of confounding have not been carefully considered or controlled for. For example, a recent study has argued that the association of maternal infections during pregnancy with ADHD in offspring could be fully explained by the unmeasured familial confounding when applying a sibling comparison design [37]; another large-scale epidemiological study has reported that the diagnosis of SCZ was associated with increased risk of infection later in life, suggesting a causation in the opposite direction [38].
Chronic inflammation, as indexed by inflammatory marker levels, has been extensively studied as a potential risk factor to psychiatric disorders. Individuals diagnosed with these disorders frequently also develop immune and inflammation related conditions [39,40,41]. For instance, SCZ patients were reported having a 45% higher risk for developing autoimmune disorders compared to the general population. In addition, it has been shown that in most psychiatric patients the pro-inflammatory cytokine levels (e.g., interleukin 6 (IL6), IL1b, C-reactive protein (CRP)) were higher than in healthy controls [22, 23]. Prospective studies had indicated that elevated baseline levels of pro-inflammatory markers were predictive of psychiatric diagnoses many years later [20, 42,43,44]. In clinical settings, levels of these markers were also shown to decrease after treatment in patients [45]. There is evidence for efficacy of some agents with anti-inflammatory properties on first episode psychosis and schizophrenia [46], and reports of anti-inflammatory effects of anti-psychotic drugs are suggesting that their effectiveness is partially due to a modulating effect on the immune system [47]. Patients who respond to antidepressants have lower neuroinflammatory markers compared to non-respondents [48]. Selective and nonselective cytokine inhibitors like non-steroidal anti-inflammatory drugs were effective in improving depression in meta-analysis despite significant heterogeneity of individual studies [49]. A scoping review of anti-inflammatory medications for the treatment of mental disorders suggested different underlying mechanisms for their treatment success in BIP/MDD and SCZ [50].
Despite being promising, epidemiological findings are correlational in nature. The observed immune-psychiatric relationships can be interpreted in multiple non-exclusive ways: (1) Immune dysfunction leads to an increased risk of psychiatric disorders; (2) Immune dysfunction is the consequence of psychiatric disorders or medication for chronic illness; or, (3) unknown/unmeasured variables cause both—confounding effects. While prospective studies seemingly meet the Temporality criterion in Hill’s causal criteria [51], inflammatory markers were typically measured at baseline, thereby not fully capturing chronic immune process in a timely manner. Further, developing psychiatric disorders may take years, thus, even the compliance with this temporality assumption may be questioned.
Genomic association studies’ findings
In the last decade, genome-wide association studies (GWAS) have become a popular design in interrogating genetic associations with psychiatric disorders. In this design, millions of genetic variants are tested for association with a disorder without predefined biological hypotheses. Because germline variants are stable after conception, associations identified by this design are unaffected by reverse causation, a major problem in epidemiological studies. In the last 15 years, the psychiatric genomic consortium (PGC) has analyzed data from tens of thousands patients and healthy controls identifying a large number of genetic associations [52,53,54,55,56], for example, 287 loci for SCZ, 64 for BIP, 27 ADHD and more than 44 for major depression disorders (MDD), many of which were previously unknown. Within these identified loci, immune-related genes have been annotated (Fig. 1): for example, one of the first annotated immune gene is the HLA region gene for SCZ [57, 58].
A Genes reported by the psychiatric genomic consortium for attention-deficit/hyperactive disorder (ADHD), autism spectrum disorder (ASD), bipolar disorder (BIP), major depression disorder or depression (MDD) and schizophrenia (SCZ) are annotated to genes involved in innate immune response (InnateDB, https://www.innatedb.com/) and ImmPort (https://www.immport.org/). Proportions of reported genes annotated to each immune gene source are shown on y axis. The number of annotated genes and reported genes are shown on the top of each bar. WordCloud plots for reported genes annotated to ImmPort (C) and InnateDB (B). Text font sizes indicate the number of disorders associated with the gene.
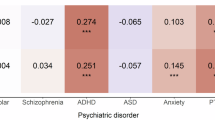
Leveraging massive summary statistics from large-scale GWAS, polygenic genetic overlaps between immune-related and psychiatric disorders have been examined by genetic correlation estimates (gr) which quantifies the degrees of genetic sharing between traits. Significant positive genetic correlations between SCZ and Crohn’s disease (CD; gr = 0.097), inflammatory bowel disease (IBD; gr = 0.117), ulcerative colitis (UC; gr = 0.106), primary biliary cirrhosis (PBC; gr = 0.131) and psoriasis (PSO; gr = 0.182) have been reported [59,60,61]. Among these immune-related disorders, CD (gr = 0.22), UC (gr = 0.23), PSO (gr = 0.29), along with Celiac disease (gr = 0.34) were genetically correlated with BIP; rheumatoid arthritis (RA; gr = 0.16), type 1 diabetes (gr = −0.14), PSO (gr = 0.23) were genetically correlated with ADHD [61]. Moreover, in a recent study based on the UK Biobank (UKBB) data, CRP was shown genetically correlated with MDD (gr = 0.154), ADHD (gr = 0.326), obsessive-compulsive disorder (gr = −0.201), anorexia nervosa (gr = −0.268), post-traumatic stress disorder (gr = 0.238) and a trend (i.e., p > 0,05) was observed with SCZ (gr = −0.058) [62]. Another study reported genetic correlation of smaller magnitude (gr = 0.098) using summary GWAS from PGC (MDD) and CHARGE consortium (CRP) [63].
To assess the immune-psychiatry relations at gene level, we extracted genes reported by the most recent PGC studies for the five disorders (SCZ, BIP, MDD, ADHD and ASD) with largest sample size so far. We compared these genes with those included in two curated immune databases: (1) the InnateDB [64] which includes genes involved in the innate immunity arm and (2) the ImmPort [65] which collects genes involved in general immune functioning (Fig. 1A). As anticipated, all the five disorders exhibit numerous genes associated with immune functioning. The WordClouds generated from the genes associated with these disorders and in the two immune databases highlight that both the innate and the adaptive immune arms were involved in risk of psychiatric disorders (Fig. 2B, C) and that several immune genes are associated with more than one disorder.
A Genes identified by differential expression in brain tissue by Gandal et al. [66] for autism spectrum disorder (ASD), bipolar disorder (BIP), and schizophrenia (SCZ) are annotated to genes involved in innate immune response (InnateDB, https://www.innatedb.com/) and ImmPort (https://www.immport.org/). Proportions of reported genes annotated to each immune gene source are shown on y axis. The number of annotated genes and reported genes are shown on the top of each bar. WordCloud plots for reported genes annotated to ImmPort (C) and InnateDB (B). Wordcloud plots for reported genes identified by brain tissue expression, GWAS and EWAS annotated to ImmPort (D) and InnateDB (E). Text font sizes indicate the number of disorders associated with the gene.
Gene expressions vary in human tissues and are partially regulated by DNA methylations. We curated genes implicated by DNA methylation association studies (epigenome wide association studies (EWAS)) performed on blood samples from the EWAS Catalog and genes differentially expressed in brain tissues from psychiatric disorders and healthy controls reported by Gandal et al. [66]. We aligned these genes with those from the two immune gene databases noted above (Figs. 2 and 3). Similar to the observations from GWAS data, both the innate and adaptive immune systems were indicated (Figs. 2B, C and 3B, C). Together, 83 genes implicated in associations with SCZ were identified in the three types of studies; 16 genes for BIP by the two available studies, and two for ADHD (FOXP1 and ST3GAL3). WordCloud plots created from these overlapping genes demonstrated that two immune genes, OSBPL3 and TRIM27, were associated with both SCZ and BIP (Fig. 2D, E).
A Genes identified by epigenomic wide association studies included in the EWAS Catalog (http://ewascatalog.org/; accessed at January 2023) for attention-deficiency/hyperactive disorder (ADHD), autism spectrum disorder (ASD), depression or depressive symptoms (Dep) and schizophrenia (SCZ) are annotated to genes involved in innate immune response (InnateDB, https://www.innatedb.com/) and ImmPort (https://www.immport.org/). Proportions of reported genes annotated to each immune gene source are shown on y axis. The number of annotated genes and reported genes are shown on the top of each bar. Wordcloud plots for reported genes annotated to ImmPort (C) and InnateDB (B). Text font sizes indicate the number of disorders associated with the gene.
In sum, results from large-scale hypothesis-free genetic studies support the involvement of immune dysfunction in psychiatric disorders. The slightly divergent overlapping patterns among the three types of data sources may be due to differences in study sample sizes, biological nature of DNA variation, methylation and expression, where the latter two vary in age [67] and tissues. It is of note that measuring DNA methylation or gene expressions after diagnosis may represent a consequence of the disorders instead of a cause, and thus cause-effect interpretations for such studies need additional evidence. Another key observation is that, for the three types of studies, the number of genes involved in the overall immune system is not statistically enriched in psychiatric disorders: Assuming 20,000 genes in the genome [68] while there are 1376 and 1509 genes included in the two databases, on average 6–8% of identified psychiatric genes should be involved in the immune system at random; with this threshold, barely any psychiatric disorder would show statistically significant enrichment signal; but, this does not rule out that certain sub-immune response pathways, for example the NF-kb system [66] may be enriched.
Mendelian randomization findings
Mendelian randomization (MR) analysis has been designed aiming to interrogate the cause-effect relations between exposures (here, immune functioning) and outcomes (here, psychiatric disorders) [29, 69]. In MR, genetic variants (e.g., SNPs) are used as proxies to the exposure of interest in a way to examine the causal effect of the exposures on outcomes. As per the Mendelian inheritance laws, genetic variations are randomly inherited from one generation to the next. MR design, under certain conditions such as random mating, can be regarded as a natural randomized trial. In addition, as germline genetic variants are stable, they are less likely affected by environmental factors during lifetime. Therefore, MR estimates are unbiased by reverse causation and confounders for exposures and outcomes (Box 1), in distinction to traditional observational studies. MR analysis can be performed either using the individual-level data (one-sample MR) or summary statistics from GWAS of two independent samples (two-sample MR), one for exposure and the other for outcome; with certain assumptions, the underlying models for these two designs are equivalent.
As publicly sharing of GWAS summary statistics is becoming the norm in the field, MR turns into the most cost-effective causal inference model for examining the relations between the immune dysfunction and psychiatric disorders. As such, the choices of exposures and outcomes in MR are intrinsically constrained by the data that is currently available. We have searched PubMed database for MR studies published in the past 10 years focusing on immune function and major psychiatric disorders (Box 2 and Table 1). In total, we have found 15 studies that investigated the relationships between inflammatory markers and psychiatric disorders (up to nine different diagnoses). Among these 15 studies, 13 used GWAS results from PGC, and for exposures, only a few common GWAS results were used (Table 1).
Among the studied inflammatory markers, CRP and the IL6 signaling components (IL6, sIL6R, sgp130) are the most studied exposures; other markers have only been examined by few studies. While the statistical models of MR and the strategies for selecting instrumental SNPs varied, CRP was consistently shown to have a protective effect on SCZ. This consistency held even when the CRP GWAS sample size increased from ~80,000 [70] to 200,000 [71] and in an analysis from an independent study [72], and when the size of GWAS for SCZ increased from about 80,000 [73] to 320,000 [52]. The only null finding was from a Danish population cohort with about 78,000 individuals using a one-sample MR design [74]. The protective effect of CRP was also demonstrated for ASD [62] and sub-symptoms of depression [44, 75] but not to major depression disorder (MDD). Another relatively consistent finding is the detrimental effects of increased IL6 signaling to SCZ, MDD, depressive sub-symptoms and depression [44, 75,76,77,78]. Significant causal effects for other markers (sIL2Ra and FGFBasic) to psychiatric disorders have been reported by only a single study [79, 80]. In general, there was no causal effects of psychiatric disorders on inflammatory marker levels reported, except by Chen et al. [79] (Table 1). In summary, while sample overlapped, the extremely large sample sizes of the underlying GWAS studies lend some credibility to these causal findings, particularly for CRP and IL6 signaling which have been reported in multiple studies.
Owing to ever increasing samples for GWAS studies, well-powered MR studies are keeping up, discovering novel causal effects of inflammatory markers on psychiatric disorders. However, major challenges remain. Because associations identified by GWAS are typically located in non-coding regions, carefully selecting valid instrumental SNPs is imperative. In the context of studying inflammatory markers as exposures, two popular selection strategies have been used (Table 1): either selecting independent genome-wide significant SNPs (p < 5 × 10−8) disregarding genomic locations (GW-SNPs), or, among these SNPs, selecting only those that are located to the corresponding coding genes (cis-SNPs). Although the latter seems better in avoiding pleiotropic instruments, these selected cis-SNPs may still be in linkage disequilibrium with SNPs nearby but outside of the coding regions. Another challenge is the highly pleiotropic nature of inflammatory markers, e.g., CRP and IL6 [62]. This phenomenon very likely generates the so-called correlated horizontal pleiotropy which can bias MR estimates (Box 1). To date, most of published studies have not specifically handled this bias [81].
Discussion and perspectives
We briefly reviewed the evidence of immune dysfunction influence on psychiatric disorders from epidemiological or clinical studies. We added to the growing body of evidence provided by recent genomic and epigenomic findings. We showed that evidence from the fifteen Mendelian randomization studies, which directly tested for causal effects of inflammatory markers on psychiatric disorders, supports a causal interpretation. However, are we able to claim a causal relation between the two?
Synthesizing evidence from these studies leads to several notable points. Epidemiological studies generally support the notion that elevated inflammatory responses may potentially cause psychiatric disorders. The low-level inflammation may stem from either chronic infection history or other illness indexed by sub-clinical but elevated inflammatory marker levels. These data have been most often interpreted as the involvement of the innate immunity arm. However, both genomic and epigenomic studies implied the involvement of adaptive arm as well, even to a large extent reported by a recent study [82] (Figs. 1–3). This is not contradictory to findings from epidemiological studies. The frequently studied inflammatory markers, such as IL6, have well-known functions in modulating the adaptive immune responses, immune cell differentiation, and other cellular processes [83]. The generally small effect sizes in epidemiological and genetic studies indicate that immune dysfunction may be secondary in causing these diseases or it is relevant only for a subset of patients.
Most MR studies have investigated whether perturbations of the IL-6 signaling pathway may lead to increased disease risk. These findings support the epidemiological observations, i.e., increased activity of this pathway plays a causal role in these disorders. But the consistently reported protective effect of CRP is in a startling contrast to prior epidemiological and clinical observations. CRP, an acute-phase protein, responds to acute infections or inflammations by sharply increasing expression levels, up to thousandfold within few hours [84]. But whether common genetic variations identified by GWAS capture these acute response or basal expression levels is uncertain [85,86,87]. Thus, interpreting elevated CRP levels (<10 mg/l) as indicator of chronic inflammation needs more support: chronic inflammation can lead to elevated CRP levels, but elevated CRP levels may not indicate ongoing chronic inflammation. Moreover, epidemiological studies frequently reported the effects of immune dysfunction within a narrower sampling time window; MR studies, or in general genomic association studies, can only estimate the lifetime average effects. Therefore, this important distinction may lead to divergent conclusions.
The protective effect of CRP is biologically plausible. CRP exists in two isoforms—soluble pentamer and insoluble monomer. In face of infection/inflammation the pentameric form of CRP, mainly produced in the liver, dissociates irreversibly into monomeric form that acts locally and has a pro-inflammatory effect, i.e., by activation of the classical pathway of the complement system [88]. However, CRP-induced complement activation does not lead to the C5-C9 activation, which amplifies pro-inflammatory processes [88]. Thus, the pro-inflammatory effect of CRP is highly regulated and potentially plays a beneficial role for health. In addition, both in vitro and in vivo studies have shown that CRP can opsonize endogenous and exogeneous antigens to facilitate cellular clearance by other immune cells, thereby playing anti-inflammatory and tissue repairing functions [88, 89].
In conclusion, while great progresses have been made in investigating the causal effects of immune dysfunction in psychiatric disorders, we still do not have the full picture. To approach this goal, triangulation of findings from different fields is critical [90]. Currently, most epidemiological or clinical studies have measured inflammatory markers only at baseline in longitudinal studies. As such this design cannot distinguish random fluctuations from true chronic inflammation, which needs multi-timepoints measures of many immune parameters [86, 87]. Although omics technology has brought about novel findings, we are still lacking well-powered cell type specific data to link genetic variations to cellular functions. Lastly, as of today, MR studies are highly biased toward a few, sufficiently sized, datasets, and future work should strive to replicate these findings with independent data sources.
References
GBD 2016 Disease and Injury Incidence and Prevalence Collaborators, Carapetis JR, Dadi AF. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390:1211–59.
Picci G, Scherf KS. A two-hit model of autism: adolescence as the second hit. Clin Psychol Sci. 2014;3:349–71.
Feigenson KA, Kusnecov AW, Silverstein SM. Inflammation and the two-hit hypothesis of schizophrenia. Neurosci Biobehav Rev. 2014;38:72–93.
Rosenblat JD, McIntyre RS. Bipolar disorder and immune dysfunction: epidemiological findings, proposed pathophysiology and clinical implications. Brain Sci. 2017;7:144.
Khandaker GM, Zimbron J, Lewis G, Jones PB. Prenatal maternal infection, neurodevelopment and adult schizophrenia: a systematic review of population-based studies. Psychol Med. 2013;43:239–57.
al-Haddad BJS, Jacobsson B, Chabra S, Modzelewska D, Olson EM, Bernier R, et al. Long-term risk of neuropsychiatric disease after exposure to infection in utero. JAMA Psychiatry. 2019;76:594–602.
Tioleco N, Silberman AE, Stratigos K, Banerjee-Basu S, Spann MN, Whitaker AH, et al. Prenatal maternal infection and risk for autism in offspring: a meta-analysis. Autism Res. 2021;14:1296–316.
Brown AS, Derkits EJ. Prenatal infection and schizophrenia: a review of epidemiologic and translational studies. Am J Psychiatry. 2010;167:261–80.
Solberg DK, Refsum H, Andreassen OA, Bentsen H. A five-year follow-up study of antioxidants, oxidative stress and polyunsaturated fatty acids in schizophrenia. Acta Neuropsychiatr. 2019;31:202–12.
Fraguas D, Díaz-Caneja CM, Ayora M, Hernández-Álvarez F, Rodríguez-Quiroga A, Recio S, et al. Oxidative stress and inflammation in first-episode psychosis: a systematic review and meta-analysis. Schizophr Bull. 2018;45:742–51.
Rantala MJ, Luoto S, Borráz-León JI, Krams I. Bipolar disorder: an evolutionary psychoneuroimmunological approach. Neurosci Biobehav Rev. 2021;122:28–37.
Chen L, Shi X-J, Liu H, Mao X, Gui L-N, Wang H, et al. Oxidative stress marker aberrations in children with autism spectrum disorder: a systematic review and meta-analysis of 87 studies (N = 9109). Transl Psychiatry. 2021;11:15.
Parkin J, Cohen B. An overview of the immune system. Lancet. 2001;357:1777–89.
Shilts J, Severin Y, Galaway F, Müller-Sienerth N, Chong Z-S, Pritchard S, et al. A physical wiring diagram for the human immune system. Nature. 2022;608:397–404.
Simon AK, Hollander GA, McMichael A. Evolution of the immune system in humans from infancy to old age. Proc R Soc B Biol Sci. 2015;282:20143085.
Kotas ME, Medzhitov R. Homeostasis, inflammation, and disease susceptibility. Cell. 2015;160:816–27.
Fernández-Ruiz I. Immune system and cardiovascular disease. Nat Rev Cardiol. 2016;13:503.
Hiam-Galvez KJ, Allen BM, Spitzer MH. Systemic immunity in cancer. Nat Rev Cancer. 2021;21:345–59.
Mondelli V, Dazzan P, Pariante CM. Immune abnormalities across psychiatric disorders: clinical relevance. BJPsych Adv. 2015;21:150–6.
Mac Giollabhui N, Ng TH, Ellman LM, Alloy LB. The longitudinal associations of inflammatory biomarkers and depression revisited: systematic review, meta-analysis, and meta-regression. Mol Psychiatry. 2021;26:3302–14.
Leighton SP, Nerurkar L, Krishnadas R, Johnman C, Graham GJ, Cavanagh J. Chemokines in depression in health and in inflammatory illness: a systematic review and meta-analysis. Mol Psychiatry. 2018;23:48–58.
Carvalho AF, Solmi M, Sanches M, Machado MO, Stubbs B, Ajnakina O, et al. Evidence-based umbrella review of 162 peripheral biomarkers for major mental disorders. Transl Psychiatry. 2020;10:152.
Yuan N, Chen Y, Xia Y, Dai J, Liu C. Inflammation-related biomarkers in major psychiatric disorders: a cross-disorder assessment of reproducibility and specificity in 43 meta-analyses. Transl Psychiatry. 2019;9:233.
Goldsmith DR, Rapaport MH, Miller BJ. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol Psychiatry. 2016;21:1696–709.
Goldsmith DR, Bekhbat M, Mehta ND, Felger JC. Inflammation-related functional and structural dysconnectivity as a pathway to psychopathology. Biol Psychiatry. 2023;93:405–18.
Haapakoski R, Mathieu J, Ebmeier KP, Alenius H, Kivimäki M. Cumulative meta-analysis of interleukins 6 and 1β, tumour necrosis factor α and C-reactive protein in patients with major depressive disorder. Brain Behav Immun. 2015;49:206–15.
Miola A, Dal Porto V, Tadmor T, Croatto G, Scocco P, Manchia M, et al. Increased C-reactive protein concentration and suicidal behavior in people with psychiatric disorders: a systematic review and meta-analysis. Acta Psychiatr Scand. 2021;144:537–52.
Frank P, Jokela M, Batty GD, Cadar D, Steptoe A, Kivimäki M. Association between systemic inflammation and individual symptoms of depression: a pooled analysis of 15 population-based cohort studies. Am J Psychiatry. 2021;178:1107–18.
Sanderson E, Glymour MM, Holmes MV, Kang H, Morrison J, Munafò MR, et al. Mendelian randomization. Nat Rev Methods Prim. 2022;2:6.
Buka SL, Tsuang MT, Torrey EF, Klebanoff MA, Bernstein D, Yolken RH. Maternal infections and subsequent psychosis among offspring. Arch Gen Psychiatry. 2001;58:1032–7.
Oliveira J, Oliveira-Maia AJ, Tamouza R, Brown AS, Leboyer M. Infectious and immunogenetic factors in bipolar disorder. Acta Psychiatr Scand. 2017;136:409–23.
Merzon E, Israel A, Ashkenazi S, Rotem A, Schneider T, Faraone SV, et al. Attention-deficit/hyperactivity disorder is associated with increased rates of childhood infectious diseases: a population-based case-control study. J Am Acad Child Adolesc Psychiatry. 2023;62:253–60.e1.
Lydholm CN, Köhler-Forsberg O, Nordentoft M, Yolken RH, Mortensen PB, Petersen L, et al. Parental infections before, during, and after pregnancy as risk factors for mental disorders in childhood and adolescence: a nationwide Danish study. Biol Psychiatry. 2019;85:317–25.
Walle KM, Askeland RB, Gustavson K, Mjaaland S, Ystrom E, Lipkin WI, et al. Risk of attention-deficit hyperactivity disorder in offspring of mothers with infections during pregnancy. JCPP Adv. 2022;2:e12070.
Meltzer A, Van de Water J. The role of the immune system in autism spectrum disorder. Neuropsychopharmacology. 2017;42:284–98.
Nielsen PR, Benros ME, Mortensen PB. Hospital contacts with infection and risk of schizophrenia: a population-based cohort study with linkage of Danish national registers. Schizophr Bull. 2014;40:1526–32.
Ginsberg Y, D’Onofrio BM, Rickert ME, Class QA, Rosenqvist MA, Almqvist C, et al. Maternal infection requiring hospitalization during pregnancy and attention-deficit hyperactivity disorder in offspring: a quasi-experimental family-based study. J Child Psychol Psychiatry. 2019;60:160–8.
Nielsen PR, Laursen TM, Agerbo E. Comorbidity of schizophrenia and infection: a population-based cohort study. Soc Psychiatry Psychiatr Epidemiol. 2016;51:1581–9.
Benros ME, Waltoft BL, Nordentoft M, Østergaard SD, Eaton WW, Krogh J, et al. Autoimmune diseases and severe infections as risk factors for mood disorders: a nationwide study. JAMA Psychiatry. 2013;70:812–20.
Jeppesen R, Benros ME. Autoimmune diseases and psychotic disorders. Front Psychiatry. 2019;10:131.
Eaton WW, Byrne M, Ewald H, Mors O, Chen C-Y, Agerbo E, et al. Association of schizophrenia and autoimmune diseases: linkage of Danish national registers. Am J Psychiatry. 2006;163:521–8.
Osimo EF, Baxter L, Stochl J, Perry BI, Metcalf SA, Kunutsor SK, et al. Longitudinal association between CRP levels and risk of psychosis: a meta-analysis of population-based cohort studies. npj Schizophr. 2021;7:31.
Chu AL, Stochl J, Lewis G, Zammit S, Jones PB, Khandaker GM. Longitudinal association between inflammatory markers and specific symptoms of depression in a prospective birth cohort. Brain Behav Immun. 2019;76:74–81.
Milaneschi Y, Kappelmann N, Ye Z, Lamers F, Moser S, Jones PB, et al. Association of inflammation with depression and anxiety: evidence for symptom-specificity and potential causality from UK Biobank and NESDA cohorts. Mol Psychiatry. 2021;26:7393–402.
Patlola SR, Donohoe G, McKernan DP. Anti-inflammatory effects of 2nd generation antipsychotics in patients with schizophrenia: a systematic review and meta-analysis. J Psychiatr Res. 2023;160:126–36.
Cakici N, van Beveren NJM, Judge-Hundal G, Koola MM, Sommer IEC. An update on the efficacy of anti-inflammatory agents for patients with schizophrenia: a meta-analysis. Psychol Med. 2019;49:2307–19.
Juncal-Ruiz M, Riesco-Davila L, Ortiz-Garcia de la Foz V, Martinez-Garcia O, Ramirez-Bonilla M, Ocejo-Vinals JG, et al. Comparison of the anti-inflammatory effect of aripiprazole and risperidone in 75 drug-naive first episode psychosis individuals: a 3 months randomized study. Schizophr Res. 2018;202:226–33.
Liu JJ, Wei YB, Strawbridge R, Bao Y, Chang S, Shi L, et al. Peripheral cytokine levels and response to antidepressant treatment in depression: a systematic review and meta-analysis. Mol Psychiatry. 2020;25:339–50.
Kohler O, Benros ME, Nordentoft M, Farkouh ME, Iyengar RL, Mors O, et al. Effect of anti-inflammatory treatment on depression, depressive symptoms, and adverse effects: a systematic review and meta-analysis of randomized clinical trials. JAMA Psychiatry. 2014;71:1381–91.
Fitton R, Sweetman J, Heseltine-Carp W, van der Feltz-Cornelis C. Anti-inflammatory medications for the treatment of mental disorders: a scoping review. Brain Behav Immun Health. 2022;26:100518.
Fedak KM, Bernal A, Capshaw ZA, Gross S. Applying the Bradford Hill criteria in the 21st century: how data integration has changed causal inference in molecular epidemiology. Emerg Themes Epidemiol. 2015;12:14.
Trubetskoy V, Pardiñas AF, Qi T, Panagiotaropoulou G, Awasthi S, Bigdeli TB, et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature. 2022;604:502–8.
Mullins N, Forstner AJ, O’Connell KS, Coombes B, Coleman JRI, Qiao Z, et al. Genome-wide association study of more than 40,000 bipolar disorder cases provides new insights into the underlying biology. Nat Genet. 2021;53:817–29.
Wray NR, Ripke S, Mattheisen M, Trzaskowski M, Byrne EM, Abdellaoui A, et al. Genome-wide association analyses identify 44 risk variants and refine the genetic architecture of major depression. Nat Genet. 2018;50:668–81.
Grove J, Ripke S, Als TD, Mattheisen M, Walters RK, Won H, et al. Identification of common genetic risk variants for autism spectrum disorder. Nat Genet. 2019;51:431–44.
Demontis D, Walters GB, Athanasiadis G, Walters R, Therrien K, Nielsen TT, et al. Genome-wide analyses of ADHD identify 27 risk loci, refine the genetic architecture and implicate several cognitive domains. Nat Genet. 2023;55:198–208.
Purcell SM, Wray NR, Stone JL, Visscher PM, O’Donovan MC, Sullivan PF, et al. Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature. 2009;460:748–52.
Andreassen O, Harbo H, Wang Y, Thompson W, Schork A, Mattingsdal M, et al. Genetic pleiotropy between multiple sclerosis and schizophrenia but not bipolar disorder: differential involvement of immune-related gene loci. Mol Psychiatry. 2015;20:1–8.
Pouget JG, Schizophrenia Working Group of the Psychiatric Genomics Consortium, Han B, Wu Y, Mignot E, Ollila HM, et al. Cross-disorder analysis of schizophrenia and 19 immune-mediated diseases identifies shared genetic risk. Hum Mol Genet. 2019;28:3498–513.
Stringer S, Kahn RS, de Witte LD, Ophoff RA, Derks EM. Genetic liability for schizophrenia predicts risk of immune disorders. Schizophr Res. 2014;159:347–52.
Tylee DS, Sun J, Hess JL, Tahir MA, Sharma E, Malik R, et al. Genetic correlations among psychiatric and immune-related phenotypes based on genome-wide association data. Am J Med Genet Part B Neuropsychiatr Genet. 2018;177:641–57.
Koskeridis F, Evangelou E, Said S, Boyle JJ, Elliott P, Dehghan A, et al. Pleiotropic genetic architecture and novel loci for C-reactive protein levels. Nat Commun. 2022;13:6939.
Pitharouli MC, Hagenaars SP, Glanville KP, Coleman JR, Hotopf M, Lewis CM, et al. Elevated C-reactive protein in patients with depression, independent of genetic, health, and psychosocial factors: results from the UK Biobank. Am J Psychiatry. 2021;178:522–9.
Breuer K, Foroushani AK, Laird MR, Chen C, Sribnaia A, Lo R, et al. InnateDB: systems biology of innate immunity and beyond-recent updates and continuing curation. Nucleic Acids Res. 2013;41:D1228–33.
Bhattacharya S, Dunn P, Thomas CG, Smith B, Schaefer H, Chen J, et al. ImmPort, toward repurposing of open access immunological assay data for translational and clinical research. Sci Data. 2018;5:180015.
Gandal MJ, Haney JR, Parikshak NN, Leppa V, Ramaswami G, Hartl C, et al. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science. 2018;359:693–7.
Horvath S. DNA methylation age of human tissues and cell types. Genome Biol. 2013;14:3156.
Frankish A, Diekhans M, Ferreira A-M, Johnson R, Jungreis I, Loveland J, et al. GENCODE reference annotation for the human and mouse genomes. Nucleic Acids Res. 2018;47:D766–73.
Smith GD, Ebrahim S. Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol. 2003;32:1–22.
Dehghan A, Dupuis J, Barbalic M, Bis JC, Eiriksdottir G, Lu C, et al. Meta-analysis of genome-wide association studies in> 80 000 subjects identifies multiple loci for C-reactive protein levels. Circulation. 2011;123:731–8.
Ligthart S, Vaez A, Võsa U, Stathopoulou MG, de Vries PS, Prins BP, et al. Genome analyses of >200,000 individuals identify 58 loci for chronic inflammation and highlight pathways that link inflammation and complex disorders. Am J Hum Genet. 2018;103:691–706.
Said S, Pazoki R, Karhunen V, Võsa U, Ligthart S, Bodinier B, et al. Genetic analysis of over half a million people characterises C-reactive protein loci. Nat Commun. 2022;13:2198.
Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–7.
Wium-Andersen MK, Ørsted DD, Nordestgaard BG. Elevated C-reactive protein associated with late- and very-late-onset schizophrenia in the general population: a prospective study. Schizophr Bull. 2014;40:1117–27.
Ye Z, Kappelmann N, Moser S, Davey Smith G, Burgess S, Jones PB, et al. Role of inflammation in depression and anxiety: tests for disorder specificity, linearity and potential causality of association in the UK Biobank. eClinicalMedicine. 2021;38:100992.
Kelly KM, Smith JA, Mezuk B. Depression and interleukin-6 signaling: a Mendelian randomization study. Brain Behav Immun. 2021;95:106–14.
Khandaker GM, Zuber V, Rees JMB, Carvalho L, Mason AM, Foley CN, et al. Shared mechanisms between coronary heart disease and depression: findings from a large UK general population-based cohort. Mol Psychiatry. 2020;25:1477–86.
Hartwig FP, Davies NM, Hemani G, Davey Smith G. Two-sample Mendelian randomization: avoiding the downsides of a powerful, widely applicable but potentially fallible technique. Int J Epidemiol. 2017;45:1717–26.
Chen X, Yao T, Cai J, Fu X, Li H, Wu J. Systemic inflammatory regulators and 7 major psychiatric disorders: a two-sample Mendelian randomization study. Prog Neuro Psychopharmacol Biol Psychiatry. 2022;116:110534.
Perry BI, Upthegrove R, Kappelmann N, Jones PB, Burgess S, Khandaker GM. Associations of immunological proteins/traits with schizophrenia, major depression and bipolar disorder: a bi-directional two-sample Mendelian randomization study. Brain Behav Immun. 2021;97:176–85.
Morrison J, Knoblauch N, Marcus JH, Stephens M, He X. Mendelian randomization accounting for correlated and uncorrelated pleiotropic effects using genome-wide summary statistics. Nat Genet. 2020;52:740–7.
Lynall M-E, Soskic B, Hayhurst J, Schwartzentruber J, Levey DF, Pathak GA, et al. Genetic variants associated with psychiatric disorders are enriched at epigenetically active sites in lymphoid cells. Nat Commun. 2022;13:6102.
Garbers C, Heink S, Korn T, Rose-John S. Interleukin-6: designing specific therapeutics for a complex cytokine. Nat Rev Drug Discov. 2018;17:395–412.
Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Investig. 2003;111:1805–12.
Wang Y, Grydeland H, Roe JM, Pan M, Magnussen F, Amlien IK, et al. Associations of circulating C-reactive proteins, APOE ε4, and brain markers for Alzheimer’s disease in healthy samples across the lifespan. Brain Behav Immun. 2022;100:243–53.
Medzhitov R. The spectrum of inflammatory responses. Science. 2021;374:1070–5.
Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019;25:1822–32.
Du Clos TW, Mold C. C-reactive protein. Immunol Res. 2004;30:261–77.
Black S, Kushner I, Samols D. C-reactive protein*. J Biol Chem. 2004;279:48487–90.
Ohlsson H, Kendler KS. Applying causal inference methods in psychiatric epidemiology: a review. JAMA Psychiatry. 2020;77:637–44.
Demontis D, Walters RK, Martin J, Mattheisen M, Als TD, Agerbo E, et al. Discovery of the first genome-wide significant risk loci for attention deficit/hyperactivity disorder. Nat Genet. 2019;51:63–75.
Watson HJ, Yilmaz Z, Thornton LM, Hübel C, Coleman JRI, Gaspar HA, et al. Genome-wide association study identifies eight risk loci and implicates metabo-psychiatric origins for anorexia nervosa. Nat Genet. 2019;51:1207–14.
Stahl EA, Breen G, Forstner AJ, McQuillin A, Ripke S, Trubetskoy V, et al. Genome-wide association study identifies 30 loci associated with bipolar disorder. Nat Genet. 2019;51:793–803.
Arnold PD, Askland KD, Barlassina C, Bellodi L, Bienvenu OJ, Black D, et al. Revealing the complex genetic architecture of obsessive–compulsive disorder using meta-analysis. Mol Psychiatry. 2018;23:1181–8.
Nievergelt CM, Maihofer AX, Klengel T, Atkinson EG, Chen C-Y, Choi KW, et al. International meta-analysis of PTSD genome-wide association studies identifies sex- and ancestry-specific genetic risk loci. Nat Commun. 2019;10:4558.
Yu D, Sul JH, Tsetsos F, Nawaz MS, Huang AY, Zelaya I, et al. Interrogating the genetic determinants of Tourette’s syndrome and other tic disorders through genome-wide association studies. Am J Psychiatry. 2019;176:217–27.
Folkersen L, Fauman E, Sabater-Lleal M, Strawbridge RJ, Franberg M, Sennblad B, et al. Mapping of 79 loci for 83 plasma protein biomarkers in cardiovascular disease. PLoS Genet. 2017;13:e1006706.
Reay WR, Kiltschewskij DJ, Geaghan MP, Atkins JR, Carr VJ, Green MJ, et al. Genetic estimates of correlation and causality between blood-based biomarkers and psychiatric disorders. Sci Adv. 2022;8:8969.
Sudlow C, Gallacher J, Allen N, Beral V, Burton P, Danesh J, et al. UK Biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12:e1001779.
Duncan L, Yilmaz Z, Gaspar H, Walters R, Goldstein J, Anttila V, et al. Significant locus and metabolic genetic correlations revealed in genome-wide association study of anorexia nervosa. Am J Psychiatry. 2017;174:850–8.
Howard DM, Adams MJ, Clarke T-K, Hafferty JD, Gibson J, Shirali M, et al. Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nat Neurosci. 2019;22:343–52.
Ahola-Olli AV, Wurtz P, Havulinna AS, Aalto K, Pitkanen N, Lehtimaki T, et al. Genome-wide association study identifies 27 loci influencing concentrations of circulating cytokines and growth factors. Am J Hum Genet. 2017;100:40–50.
Pardiñas AF, Holmans P, Pocklington AJ, Escott-Price V, Ripke S, Carrera N, et al. Common schizophrenia alleles are enriched in mutation-intolerant genes and in regions under strong background selection. Nat Genet. 2018;50:1–389.
The Interleukin-6 Receptor Mendelian Randomisation Analysis (IL6R MR) Consortium. The interleukin-6 receptor as a target for prevention of coronary heart disease: a Mendelian randomisation analysis. Lancet. 2012;379:1214–24.
Sarwar N, Butterworth AS, Freitag DF, Gregson J, Willeit P, Gorman DN, et al. Interleukin-6 receptor pathways in coronary heart disease: a collaborative meta-analysis of 82 studies. Lancet. 2012;379:1205–13.
Astle WJ, Elding H, Jiang T, Allen D, Ruklisa D, Mann AL, et al. The allelic landscape of human blood cell trait variation and links to common complex disease. Cell. 2016;167:1415–29.e19.
Sun BB, Maranville JC, Peters JE, Stacey D, Staley JR, Blackshaw J, et al. Genomic atlas of the human plasma proteome. Nature. 2018;558:73–9.
Kappelmann N, Arloth J, Georgakis MK, Czamara D, Rost N, Ligthart S, et al. Dissecting the association between inflammation, metabolic dysregulation, and specific depressive symptoms: a genetic correlation and 2-sample Mendelian randomization study. JAMA Psychiatry. 2021;78:161–70.
Penninx BWJH, Beekman ATF, Smit JH, Zitman FG, Nolen WA, Spinhoven P, et al. The Netherlands Study of Depression and Anxiety (NESDA): rationale, objectives and methods. Int J Methods Psychiatr Res. 2008;17:121–40.
de Rojas I, Moreno-Grau S, Tesi N, Grenier-Boley B, Andrade V, Jansen IE, et al. Common variants in Alzheimer’s disease and risk stratification by polygenic risk scores. Nat Commun. 2021;12:3417.
Suhre K, Arnold M, Bhagwat AM, Cotton RJ, Engelke R, Raffler J, et al. Connecting genetic risk to disease end points through the human blood plasma proteome. Nat Commun. 2017;8:14357.
Yao C, Chen G, Song C, Keefe J, Mendelson M, Huan T, et al. Genome‐wide mapping of plasma protein QTLs identifies putatively causal genes and pathways for cardiovascular disease. Nat Commun. 2018;9:3268.
C Reactive Protein Coronary Heart Disease Genetics Collaboration. Association between C reactive protein and coronary heart disease: mendelian randomisation analysis based on individual participant data. BMJ. 2011;342:d548.
Interleukin 1 Genetics Consortium. Cardiometabolic effects of genetic upregulation of the interleukin 1 receptor antagonist: a Mendelian randomisation analysis. Lancet Diabetes Endocrinol 2015;3:243–53.
de Vries PS, Chasman DI, Sabater-Lleal M, Chen M-H, Huffman JE, Steri M, et al. A meta-analysis of 120 246 individuals identifies 18 new loci for fibrinogen concentration. Hum Mol Genet. 2016;25:358–70.
Paré G, Ridker PM, Rose L, Barbalic M, Dupuis J, Dehghan A, et al. Genome-wide association analysis of soluble ICAM-1 concentration reveals novel associations at the NFKBIK, PNPLA3, RELA, and SH2B3 Loci. PLoS Genet. 2011;7:e1001374.
Lin BD, Alkema A, Peters T, Zinkstok J, Libuda L, Hebebrand J, et al. Assessing causal links between metabolic traits, inflammation and schizophrenia: a univariable and multivariable, bidirectional Mendelian-randomization study. Int J Epidemiol. 2019;48:1505–14.
Matteini AM, Li J, Lange EM, Tanaka T, Lange LA, Tracy RP, et al. Novel gene variants predict serum levels of the cytokines IL-18 and IL-1ra in older adults. Cytokine. 2014;65:10–6.
Wium-Andersen MK, Ørsted DD, Nordestgaard BG. Elevated C-reactive protein and late-onset bipolar disorder in 78 809 individuals from the general population. Br J Psychiatry. 2016;208:138–45.
Weiss LA, Arking DE, Daly MJ, Chakravarti A, Arking DE, Brune CW, et al. A genome-wide linkage and association scan reveals novel loci for autism. Nature. 2009;461:802–8.
Sklar P, Ripke S, Scott LJ, Andreassen OA, Cichon S, Craddock N, et al. Large-scale genome-wide association analysis of bipolar disorder identifies a new susceptibility locus near ODZ4. Nat Genet. 2011;43:977–83.
Ripke S, Wray NR, Lewis CM, Hamilton SP, Weissman MM, Breen G, et al. A mega-analysis of genome-wide association studies for major depressive disorder. Mol Psychiatry. 2013;18:497–511.
Prins BP, Abbasi A, Wong A, Vaez A, Nolte I, Franceschini N, et al. Investigating the causal relationship of C-reactive protein with 32 complex somatic and psychiatric outcomes: a large-scale cross-consortium Mendelian randomization study. PLoS Med. 2016;13:e1001976.
Uffelmann E, Huang QQ, Munung NS, de Vries J, Okada Y, Martin AR, et al. Genome-wide association studies. Nat Rev Methods Prim. 2021;1:59.
Yavorska OO, Burgess S. Mendelian randomization: an R package for performing Mendelian randomization analyses using summarized data. Int J Epidemiol. 2017;46:1734–9.
Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015;44:512–25.
Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent estimation in Mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol. 2016;40:304–14.
Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50:693–8.
Zhao Q, Wang J, Hemani G, Bowden J, Small DS. Statistical inference in two-sample summary-data Mendelian randomization using robust adjusted profile score. Ann Stat. 2020;48:1742–69.
Acknowledgements
This work is supported by the Norwegian Research Council (No. 302854), the UiO:Life Science Convergence environment (4MENT), the computational resources provided by UNINETT Sigma2—the National Infrastructure for High Performance Computing and Data Storage in Norway (No. nn9769k/ns9769k).
Author information
Authors and Affiliations
Contributions
OI did the database search, reviewed eligible studies, extracted the data and summarized them, and reviewed the manuscript draft. EHL assessed quality of the chosen studies and reviewed the manuscript draft, YW reviewed eligible studies, independently examined the extracted data, and drafted the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Iakunchykova, O., Leonardsen, E.H. & Wang, Y. Genetic evidence for causal effects of immune dysfunction in psychiatric disorders: where are we?. Transl Psychiatry 14, 63 (2024). https://doi.org/10.1038/s41398-024-02778-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-024-02778-2
- Springer Nature Limited