Abstract
Background
Erectile dysfunction (ED) is a prevalent side effect of prostate cancer treatment. We hypothesized that the previously reported rates of ED may have improved with the advent of modern technology. The purpose of this project was to evaluate modern external beam radiotherapy and brachytherapy techniques to determine the incidence of radiotherapy (RT) induced ED.
Methods
A systematic review of the literature published between January 2002 and December 2018 was performed to obtain patient reported rates of ED after definitive external beam radiotherapy, ultrafractionated stereotactic radiotherapy, and brachytherapy (BT) to the prostate in men who were potent prior to RT. Univariate and multivariate analyses of radiation dose, treatment strategy, and length of follow-up were analyzed to ascertain their relationship with RT-induced ED.
Results
Of 890 articles reviewed, 24 met inclusion criteria, providing data from 2714 patients. Diminished erectile function status post RT was common and similar across all studies. The median increase in men reporting ED was 17%, 26%, 23%, and 23%, 3DCRT, IMRT, low dose rate BT, and SBRT, respectively, at 2-year median follow-up.
Conclusion
ED is a common side effect of RT. Risk of post-RT ED is similar for both LDR brachytherapy and external beam RT with advanced prostate targeting and penile-bulb sparing techniques utilized in modern RT techniques.
Similar content being viewed by others
Introduction
The diagnosis of prostate cancer is increasingly common with many options for treatment1,2. These treatments are quite effective, as evidenced by the more than three million prostate cancer survivors were living in the United States as of 20143.Of the available management strategies, between 33% and 50% of men undergoing prostate cancer treatment choose radiation therapy (RT). Definitive RT options include low and high dose rate brachytherapy, proton therapy, and standard, moderately or severely hypofractionated photon-based EBRT.
Erectile dysfunction (ED) is a prevalent side effect of prostate cancer treatment, and is certainly not limited to the various types of RT. The development of RT-induced ED directly affects a patient’s overall satisfaction with their treatment. Such effects can have far-reaching impacts on survivors’ health. For example, several studies have found a significant correlation between ED and the presence of depression symptoms4,5. Notably, even men of advanced age report feeling very strongly about the importance of sexual function—a survey of nearly 5000 men found that 71% of men between 70 and 80 years of age reported engaging in sexual activity, with 42% having sex weekly6. Thus, development of RT-induced ED is a major health concern for prostate cancer survivors, and ought to be of concern to clinicians as well. Because of the high likelihood of long-term survival after prostate cancer diagnosis and treatment, modern radiotherapeutic techniques are designed to minimize damage to erectile organs and preserve an individual’s quality of life (QOL). High quality prospective studies of patient such as Sanda et alreport outcomes comparing modalities and provide an understanding of change over time7. However these studies typically report aggregate questionnaire scores, rather than the likelihood of the event on a per person basis. This information is key in counseling patients about risks during treatment decision-making.
Reports describing the incidence of ED after prostate cancer treatment have predominantly reported data from older therapy techniques, before recognition of the association between ED and the RT dose delivered to the penile bulb8. Some studies comparing EBRT, brachytherapy (BT), and surgery have suggested a reduced rate of RT induced ED in patients undergoing BT. This difference in ED rates between RT modalities has been attributed to the physical properties of BT, which minimizes dose to organs outside of the prostate capsule9. Given the relative lack of literature comparing rates of RT induced ED across short, medium, and long-term follow-up periods, the aim of this study was to retrospectively evaluate and compare rates of ED after modern RT techniques (BT versus IMRT/SBRT (+3DCRT)) in order to more completely document and describe the risk/benefit profile of the various PC treatments currently available to clinicians and their patients.
Methods
A systematic review of the literature using PRISMA guidelines was performed to collect data concerning changes in patients’ self-reported erectile function both before and after prostate RT. Articles published between January 2002 and December 2018 were reviewed, with PubMed being the primary database utilized in the analysis. “Erectile radiation prostate” and “sexual function radiation prostate” were used as search terms. Studies were omitted from analysis if: (1) the publication was not available in English, (2) ED was not a patient-reported outcome, (3) baseline and endpoint ED rates were not published within the study, (4) radiation dose and fractionation schedule were not provided, (5) significantly different dose schedules and/or therapies were combined when reporting outcome, and/or (5) patients received androgen deprivation therapy (ADT).
Several variables (patient sample size, mean or median age, type of radiation therapy, total RT dose, fraction size, ED outcome measurement tool used, length of follow-up, and baseline and endpoint ED rates) were extracted from the selected studies and compiled into a single dataset. If a study reported a range of RT doses (e.g. 72–74 Gy), the mean RT dose was calculated and used to represent the radiation dose used in that study.
The authors examined how the type of RT, duration of follow-up, and median total RT dose affected the change in erectile dysfunction rates over time (∆ED) using sample t-tests or univariate regressions, as appropriate. A multivariate logistic regression model (Eq. 1.1) was also fit to the data to identify how these variables jointly affected ∆ED.
Variation in ∆EDi is explained by the baseline mean µ plus the effect αβ of the type of therapy (3DCRT/IMRT versus BT versus SBRT). βt is the effect of time post-irradiation and βd, the effect of centered total radiation dose. The logistic regression model (1.1) was fit by the method of maximum likelihood. The baseline used IMRT data from the med ian dose and age of the entire dataset (follow-up time of 24 months and total dose of 72 Gy). For 3DCRT, SBRT and LDR BT, dose was centered at the median dose for that modality (71.1, 35.6, and 145 Gy, respectively). We also fit a second logistic regression model wherein the effect of age was added (1.1). Statistical analyses were performed using R (3.6.1), a free software environment for statistical computing and graphics.
Results
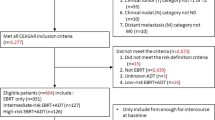
Of 890 articles initially identified, 169 were duplicates. The remaining 721 citations were reviewed first in abstract, then full-text form. Many studies were excluded because they did not state the baseline prevalence of ED or they published only instrument summary change scores for patient cohorts. Twenty-four papers met all eligibility criteria, providing data from 2714 total patients (Fig. 1)10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33. Fourteen unique dose schedules were found within those 24 studies—4 studies utilizing 3DCRT, 5 with IMRT, 6 with SBRT, 11 low dose rate (LDR), 1 proton, and 1 high dose rate (HDR) BT studies were included in analyses. Of note, data from the study describing HDR BT and the study describing proton therapy were included in descriptive statistics, but not in univariate or multivariate analyses. The median follow-up time for the included studies ranged between 12 and 84 months. The compiled data are provided in Table 1 and summarized by modality in Table 2.
The EPIC (Expanded Prostate Index Composite), IIEF (International Index of Erectile Function), and the SHIM (Sexual Health Inventory for Men, a shortened version of IIEF) questionnaires were most commonly utilized to gauge the reported ∆ED pre- and post-RT. ED was prevalent in this patient population prior to RT. The majority of studies did not report response rate for the questionnaires. Of the five which did, two reported rates >80%, two reported rates between 65% and 98% depending on time point, and one reported that full completion was achieved in only 20% of patients. Of the 21 studies which used validated questionnaires, 10 used the full questionnaire summary score. The remainder used one question from the validated questionnaire (most common was item 9 for EPIC).
Of the 2714 patients whose data was included in the analysis, 2326 patients (86%) reported normal erectile function before RT. At the time of follow-up measurement (median: 25 months, range: 12–84 months), 1559 patients (57%) reported having normal erectile function, with studies reporting between 6% and 69% of patients having developed ED during follow-up (Table 1). Figure 2 shows RT-induced ED as relates to study follow-up time and total dose. For patients receiving IMRT, the median increase in ED was 25%. For patients receiving 3DCRT, LDR BT and SBRT, the median increase in men reporting ED was 17%, 26%, 23%, and 23%, respectively. For the one proton study found, the incidence of ED increased 22% at a median follow-up of 5 years.
Incidence of RT-induced ED over time as a function of: a follow-up time and b total radiation dose. Each symbol shows the reported percentage change in ED from beginning to end of each study. The type of RT used is indicated by different symbols. The black line shows the estimated effect of follow-up time and dose, respectively, in the baseline case (IMRT, 72 Gy and 24 months of follow-up) obtained from the multivariate logistic regression fitted to the data. For each modality, dose was centered at median dose for that modality.
Univariate and multivariate analyses explored the role of treatment strategy and duration of follow-up on the development of RT-induced ED. In univariate analyses (Table 3) we found no significant differences between radiation types (p-values > 0.06). Multivariate analysis (Table 4) found a marginally statistically significant increased risk of developing ED with LDR relative to IMRT (p = 0.05). There was no significant difference noted between SBRT and 3DCRT, but the small sample size contributed to a large standard error (p = 0.22 and 0.10, respectively). This is thought to explain the non-significant result. The baseline likelihood of RT induced ED for a 66-year-old patient receiving IMRT with a follow-up time of 24 months and total dose of 72 Gy was 18%. This increased to 26% for a patient of similar age and follow-up receiving LDR (p = 0.06 univariate analysis, 0.05 multivariate analysis). For similar patients receiving SBRT, the risk of ED increased to 23%, but was insignificant (p = 0.34 univariate, = 0.22 multivariate). There was an increased risk of developing ED with higher radiation dose (p < 0.0001, Table 4). The odds of developing ED increased by 2.2% for every 1 Gy increase in dose. Similarly there was a significant increase in ED with increased length of follow-up (p = 0.0001). The odds of developing ED increased by 1.5% for every 1 month increase in follow-up.
Median/mean age was included in a second multivariate analysis, finding similar relationships between follow-up time and total dose and RT induced ED but no significant association with age (Table 4, p = 0.07). Of note, raw age data was not available and so only the median or mean age as reported by each study was included.
Discussion
In this paper, we evaluated the effects of time and radiation modality on the development of ED in men who were potent prior to RT. Unsurprisingly, we found that there is a direct correlation between the incidence of ED and increasing radiation dose, as well as length of follow-up after irradiation. Importantly, our study documents that the rate of IMRT-induced ED is lower than that reported in older studies, and appears to be similar to the risk associated with LDR BT. We attribute this result to the high conformity of modern IMRT techniques. Comparison of SBRT with other techniques will require more studies with high quality ED data and sufficient follow-up.
Our findings suggest that use of modern highly conformal external beam techniques have successfully lowered the risk of ED compared to older studies – these note that the risk of developing ED in a man with normal erectile function prior to RT is 40–60%34. We hypothesize that the avoidance of erectile tissues, be it penile bulb, other nearby vascular structure, or neurovascular bundle, with very conformal RT relates to this risk reduction. This risk compares favorably with surgery. A prospective study comparing type of prostatectomy in the hands of experienced surgeons found that 68% and 74% of men reported ED two years after robotic vs open prostatectomy, respectively 34.
Our multivariate analysis notes a marginally significant trend where LDR BT is associated with a higher rates of ED development. We believe this is due to variability in the incidence of RT-induced ED for the LDR BT studies (range 16–67%) compared to IMRT (20–28% with one outlier of 90%). While more data over time is needed, we propose that the major finding here is the improvement in EBRT risk rather than the comparison between techniques.
There are several limitations to the data. First, in 2011, Alemozaffar et al. established the relationship between patient demographic data (patient-reported pre-treatment erectile function, age, prostate specific antigen levels, race/ethnicity, BMI), treatment modality, and predicted erectile function two years after prostate cancer treatment35. Given the nature of systematic reviews, patient populations across studies likely differ in ED risk factors. In particular, age was included in a secondary MVA analysis but with the caveat that raw age data was not available; thus sensitivity for differences by age is significantly reduced. Given the clear relationship between age and ED generally, we recommend that patients should be counseled on the impact age and various radiation treatment factors have one’s long-term likelihood of developing of ED. We also note that co-morbidities and the use of sexual aids such as PDE-5 inhibitors, which can improve patient-reported sexual function, were rarely commented on in the studies reviewed. Comorbidities such as diabetes and peripheral vascular disease are common causes of ED which could confound the rates reported in these studies. Use of such sexual aids such as prescription medications, over the counter supplements, or physical aids could affect the reporting of erectile function at baseline and post-RT.
Secondly, studies included within this analysis likely varied in planning volume delineation, daily treatment set-up, and dosimetry to the penile bulb region, a structure that some have found to be predictive of ED36. Variation in timing of measurement is perhaps the greatest confounder in this effort. Between studies, median follow-up varied from 12 to 84 months. Because ∆ED is likely dependent on time after the completion of RT, this could have affected the ∆ED measured for study populations. Studies with shorter follow-up may therefore underestimate the incidence of RT induced ED, as it is likely progressive over time.
Finally, the method of ED measurement varied between studies. Currently, no consensus exists regarding the definition of erectile dysfunction, particularly amongst patients. The lack of standardization was reflected in our data, as 11 different instruments were utilized to evaluate changes in erectile function. Moreover, three studies utilized self-developed questionnaires rather than validated instruments to evaluate changes in RT-induced ED. This variability in ED measurement may affect the classification and quantification of erectile function as each patient-reported outcome measurement tool utilizes different scales and follow-up data points. Thus percent changes in erectile function across studies may not correspond well with one another. To negate some of this variability, we choose to measure the decline of erectile function within a study before comparing ∆EDi values across all studies included in the analysis.
Conclusions
This study summarizes and presents real-life experiences of patients receiving radiotherapy alone for previously untreated prostate cancer, and as such, offers insight into and support for common predictors of ED development. We note that RT-induced ED is still a common side effect of modern RT techniques, although at a somewhat lower frequency than has been reported in the past using older non-conformal EBRT. With modern treatment planning and delivery techniques, IMRT has a similar risk of ED development to LDR BT. This study not only highlights the progressive effect of RT on erectile tissues over time, but also a positive association between radiation dose and ED. Future research into RT-induced ED could be strengthened by careful planned reporting of erectile function. Up to 61% of patients in any individual study had ED prior to radiotherapy, thus we feel that attention to pre-existing ED is important in any analysis of treatment effect. By standardizing the methods used to classify and report ED, much more data could become available for future research. Moreover, further reduction in risk of RT-induced ED will be unlikely by technologic precision alone, but may require better understanding of the biologic mechanism of RT-induced ED. Additional information and understanding about how tissues surrounding the prostate respond to RT could possibly provide new approaches to protecting erectile functioning and providing significant improvements in QOL for millions of prostate cancer survivors in the United States and abroad.
References
Gronberg H. Prostate cancer epidemiology. Lancet. 2003;361:859–64.
Peate I. Understanding prostate cancer and PSA testing. Practice. Nursing. 2010;21:154–9.
Cancer Treatment and Survivorship Facts & Figures 2014-2015. Atlanta: American Cancer Society; 2014, p. 48.
Helgason AR, Adolfsson J, Dickman P, Fredrikson M, Arver S, Steineck G. Waning sexual function-the most important disease-specific distress for patients with prostate cancer. Br J Cancer. 1996;73:1417–21.
Nelson CJ, Mulhall JP, Roth AJ. The association between erectile dysfunction and depressive symptoms in men treated for prostate cancer. J Sex Med. 2011;8:560–6.
Braun M, Wassmer G, Klotz T, Reifenrath B, Mathers M, Engelmann U. Epidemiology of erectile dysfunction: results of the ‘Cologne Male Survey’. Int J Impot Res. 2000;12:305–11.
Sanda MG, Dunn RL, Michalski J, Sandler HM, Northouse L, Hembroff L, et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N. Engl J Med. 2008;358:1250–61.
Robinson JW, Moritz S, Fung T. Meta-analysis of rates of erectile function after treatment of localized prostate carcinoma. Int J Radiat Oncol Biol Phys. 2002;54:1063–8.
Wortel RC, Incrocci L, Mulhall JP. Reporting erectile function outcomes after radiation therapy for prostate cancer: challenges in data interpretation. J Sex Med. 2017;14:1260–9.
Valicenti RK, Bissonette EA, Chen C, Theodorescu D. Longitudinal comparison of sexual function after 3-dimensional conformal radiation therapy or prostate brachytherapy. J Urol 2002;168:2499–2504
Yeoh EEK, Fraser RJ, McGowan RE, Botten RJ, Di Matteo AC, Roos DE, et al. Evidence for efficacy without increased toxicity of hypofractionated radiotherapy for prostate carcinoma: early results of a phase III randomized trial. Int J Radiat Oncol Biol Phys. 2003;55:943–55.
van der Wielen GJ, van Putten WL, Incrocci L. Sexual function after three-dimensional conformal radiotherapy for prostate cancer: results from a dose-escalation trial. Int J Radiat Oncol Biol Phys. 2007;68:479–84.
Pinkawa M, Gagel B, Piroth MD, Fischedick K, Asadpour B, Kehl M, et al. Erectile dysfunction after external beam radiotherapy for prostate cancer. Eur Urol. 2009;55:227–34.
Fan KH, Chen YC, Chuang CK, Hsieh ML, Hong JH. Preliminary treatment results of intensity-modulated radiotherapy for prostate cancer. Chang Gung Med J. 2006;29:313–24.
McDonald AM, Baker CB, Shekar K, Popple RA, Clark GM, Yang ES, et al. Reduced radiation tolerance of penile structures associated with dose-escalated hypofractionated prostate radiotherapy. Urology. 2014;84:1383–7.
Chen RC, Basak R, Meyer AM, Kuo TM, Carpenter WR, Agans RP, et al. Association between choice of radical prostatectomy, external beam radiotherapy, brachytherapy, or active surveillance and patient-reported quality of life among men with localized prostate cancer. JAMA. 2017;317:1141–50.
Hoffman KE, Skinner H, Pugh TJ, Voong KR, Levy LB, Choi S, et al. Patient-reported urinary, bowel, and sexual function after hypofractionated intensity-modulated radiation therapy for prostate cancer: results from a randomized trial. Am J Clin Oncol. 2018;41:558–67.
Friedland JL, Freeman DE, Masterson-McGary ME, Spellberg DM. Stereotactic body radiotherapy: an emerging treatment approach for localized prostate cancer. Technol Cancer Res Treat. 2009;8:387–92.
Chen LN, Suy S, Uhm S, Oermann EK, Ju AW, Chen V, et al. Stereotactic body radiation therapy (SBRT) for clinically localized prostate cancer: the Georgetown University experience. Radiat Oncol. 2013;8:58.
Meier R, Kaplan I, Beckman A, Henning G, Woodhouse S, Williamson S, et al. Patient-reported quality of life outcomes in intermediate-risk prostate cancer patients treated with stereotactic body radiation therapy. Int J Radiat Oncol Biol Phys. 2013;87:S25.
Obayomi-Davies O, Chen LN, Bhagat A, Wright HC, Uhm S, Kim JS, et al. Potency preservation following stereotactic body radiation therapy for prostate cancer. Radiat Oncol. 2013;8:256.
Dess RT, Hartman HE, Aghdam N, Jackson WC, Soni PD, Abugharib AE, et al. Erectile function after stereotactic body radiotherapy for localized prostate cancer. BJU Int. 2018;121:61–8.
Fuller DB, Falchook AD, Crabtree T, Kane BL, Medbery CA, Underhill K, et al. Phase 2 multicenter trial of heterogeneous-dosing stereotactic body radiotherapy for low- and intermediate-risk prostate cancer: 5-year outcomes. Eur Urol Oncol. 2018;1:540–7.
Ho CK, Bryant CM, Mendenhall NP, Henderson RH, Mendenhall WM, Nichols RC, et al. Long-term outcomes following proton therapy for prostate cancer in young men with a focus on sexual health. Acta Oncol. 2018;57:582–8.
Feigenberg SJ, Lee WR, Desilvio ML, Winter K, Pisansky TM, Bruner DW, et al. Health-related quality of life in men receiving prostate brachytherapy on RTOG 98-05. Int J Radiat Oncol Biol Phys. 2005;62:956–64.
Nobes JP, Khaksar SJ, Hawkins MA, Cunningham MJ, Langley SE, Laing RW. Novel prostate brachytherapy technique: improved dosimetric and clinical outcome. Radiother Oncol. 2008;88:121–6.
Solan AN, Cesaretti JA, Stone NN, Stock RG. There is no correlation between erectile dysfunction and dose to penile bulb and neurovascular bundles following real-time low-dose-rate prostate brachytherapy. Int J Radiat Oncol Biol Phys. 2009;73:1468–74.
Huyghe E, Delannes M, Wagner F, Delaunay B, Nohra J, Thoulouzan M, et al. Ejaculatory function after permanent 125I prostate brachytherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys. 2009;74:126–32.
Taira AV, Merrick GS, Galbreath RW, Butler WM, Wallner KE, Kurko BS, et al. Erectile function durability following permanent prostate brachytherapy. Int J Radiat Oncol Biol Phys. 2009;75:639–48.
Pinkawa M, Asadpour B, Piroth MD, Gagel B, Nussen S, Kehl M, et al. Health-related quality of life after permanent I-125 brachytherapy and conformal external beam radiotherapy for prostate cancer-a matched-pair comparison. Radiother Oncol. 2009;91:225–31.
Matsushima M, Kikuchi E, Maeda T, Nakashima J, Sugawara A, Ando T, et al. A prospective longitudinal survey of erectile dysfunction in patients with localized prostate cancer treated with permanent prostate brachytherapy. J Urol. 2013;189:1014–8.
Frank SJ, Pugh TJ, Blanchard P, Mahmood U, Graber WJ, Kudchadker RJ, et al. Prospective phase 2 trial of permanent seed implantation prostate brachytherapy for intermediate-risk localized prostate cancer: efficacy, toxicity, and quality of life outcomes. Int J Radiat Oncol Biol Phys. 2018;100:374–82.
Ghadjar P, Oesch SL, Rentsch CA, Isaak B, Cihoric N, Manser P, et al. Late toxicity and five year outcomes after high-dose-rate brachytherapy as a monotherapy for localized prostate cancer. Radiat Oncol. 2014;9:122.
Nyberg M, Hugosson J, Wiklund P, Sjoberg D, Wilderang U, Carlsson SV, et al. Functional and oncologic outcomes between open and robotic radical prostatectomy at 24-month follow-up in the Swedish LAPPRO Trial. Eur Urol Oncol. 2018;1:353–60.
Alemozaffar M, Regan MM, Cooperberg MR, Wei JT, Michalski JM, Sandler HM, et al. Prediction of erectile function following treatment for prostate cancer. JAMA. 2011;306:1205–14.
Wernicke AG, Valicenti R, Dieva K, Houser C, Pequignot E. Radiation dose delivered to the proximal penis as a predictor of the risk of erectile dysfunction after three-dimensional conformal radiotherapy for localized prostate cancer. Int J Radiat Oncol Biol Phys. 2004;60:1357–63.
Acknowledgements
This work was funded by a grant from the Josiah Charles Trent Memorial Foundation Endowment Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hunt, A.A., Choudhury, K.R., Nukala, V. et al. Risk of erectile dysfunction after modern radiotherapy for intact prostate cancer. Prostate Cancer Prostatic Dis 24, 128–134 (2021). https://doi.org/10.1038/s41391-020-0247-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41391-020-0247-x
- Springer Nature Limited