Abstract
Background
Intrauterine exposure to hypertensive disorders of pregnancy, including gestational hypertension (GH) and preeclampsia (PE), may influence infant growth and have long-term health implications. This study aimed to compare growth outcomes of infants exposed to a normotensive pregnancy (NTP), GH, or PE from birth to 2 years.
Methods
Infants were children of women enroled in the prospective Postpartum Physiology, Psychology and Paediatric (P4) cohort study who had NTP, GH or PE. Birth, 6-month (age-corrected) and 2-year (age-corrected) weight z-scores, change in weight z-scores, rapid weight gain (≥0.67 increase in weight z-score) and conditional weight gain z-scores were calculated to assess infant growth (NTP = 240, GH = 19, PE = 66).
Results
Infants exposed to PE compared to NTP or GH had significantly lower birth weight and length z-scores, but there were no differences in growth outcomes at 6 months or 2 years. GH and PE-exposed infants had significantly greater weight z-score gain [95% CI] (PE = 0.93 [0.66–1.18], GH = 1.03 [0.37–1.68], NTP = 0.45 [0.31–0.58], p < 0.01) and rapid weight gain (GH = 63%, PE = 59%, NTP = 42%, p = 0.02) from birth to 2 years, which remained significant for PE-exposed infants after confounder adjustment.
Conclusion
In this cohort, GH and PE were associated with accelerated infant weight gain that may increase future cardiometabolic disease risk.
Impact
-
Preeclampsia exposed infants were smaller at birth, compared with normotensive pregnancy and gestational hypertension exposed infants, but caught up in growth by 2 years of age.
-
Both preeclampsia and gestational hypertension exposed infants had significantly accelerated weight gain from birth to 2 years, which remained significant for preeclampsia exposed infants after adjustment for confounders including small for gestational age.
-
Monitoring of growth patterns in infants born following exposure to a hypertensive disorder of pregnancy may be indicated to prevent accelerated weight gain trajectories and obesity.
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Approximately 5–10% of pregnancies worldwide are complicated by hypertensive disorders of pregnancy (HDP).1 This includes 3–5% gestational hypertension (GH), characterised by new onset hypertension ≥20 weeks gestation, and 2–5% preeclampsia (PE), where new onset hypertension is associated with maternal organ dysfunction or foetal compromise.1,2,3,4,5 For affected mothers, GH and PE carry an increased lifetime risk of cardiometabolic diseases including hypertension, ischaemic heart disease, stroke, and type 2 diabetes.3,5,6,7,8,9,10
PE is also associated with adverse foetal outcomes such as foetal growth restriction (FGR), placental abruption, stillbirth and neonatal mortality. Approximately 12–33% of PE-exposed neonates are born small for gestational age (SGA).11,12 As delivery is the only definitive treatment for PE,13 many neonates are born preterm (<37 weeks gestation), with associated complications including respiratory distress syndrome and sepsis,6,14,15,16,17 and poorer outcomes are more likely for neonates exposed to early-onset PE (diagnosis <34 weeks gestation).18,19 While GH alone is associated with less severe perinatal outcomes,20 25% of GH cases develop into PE, with a higher rate in early onset.13,21 In the long term, intrauterine exposure to HDP has been associated with an increased incidence of cardiometabolic,22,23,24,25 immunological26,27,28,29 and neurodevelopmental30,31,32,33,34,35,36,37 morbidities in children.
The Developmental Origins of Health and Disease (DOHaD) hypothesis provides a plausible explanation for the long-term complications seen in children exposed to HDP. This hypothesis suggests that foetal adaptations to an adverse intrauterine environment, as experienced in HDP, may cause ‘developmental programming’ that increases future risk of morbidity compared to children of normotensive pregnancies (NTPs).38,39,40 Rapid growth trajectories during infancy may also occur in response to an adverse intrauterine environment, typically nutrient insufficiency, a phenomenon known as the ‘thrifty phenotype’.41 Such rapid weight gain has been associated with later neurological, cardiovascular, renal and respiratory morbidity,42,43 as well as an increased risk of obesity and later cardiometabolic dysfunction.44,45,46
Our recent narrative review identified 11 studies assessing growth from birth to 2-years in infants exposed to PE versus NTP.47 Overall, studies reported that PE-exposed infants either had lower weight, length and BMI at 2 years than normotensive controls, or that they instead experienced accelerated weight gain to catch up in growth by 2 years.47 Combined with these inconsistent findings is the fact that the role of SGA and prematurity status within these studies was inconsistently explored. For instance, several prior studies were limited to HDP-exposed infants born premature or of very-low-birthweight (VLBW),48,49,50,51 which themselves are independent risk factors for impaired infant growth.52,53 Further research adjusting for these confounders is required and no prior study has investigated the difference in growth trajectories in a cohort comprising NTP, GH and PE-exposed infants in the first two years of life.
Our previous study54 reported lower absolute weight and weight z-scores in PE exposed compared with NTP infants at 6 months of age. The present study extends this research by comparing anthropometric growth measures and growth trajectories, including rapid weight gain and conditional weight gain, in this cohort at 2-years of age following exposure to NTP, GH or PE. Furthermore, we aim to elucidate impacts on anthropometric outcomes independent of SGA and prematurity status.
Methods
Study design
This is a sub-study of the prospective, single-centre cohort study, the P4 (Postpartum Physiology, Psychology and Paediatric) follow-up study being conducted at St. George Hospital, a metropolitan teaching hospital in Sydney, Australia. The study was approved by the Prince of Wales Hospital Human Research Ethics Committee (HREC/12/POWH395). A detailed study protocol has been published,55 and this study follows published papers reporting maternal outcomes6 and infant growth outcomes54 6 months postpartum.
Study population
Participants in this study were infants of P4-participating mothers. Mothers were eligible if they gave birth to a live singleton infant without major congenital abnormalities between January 2013 and December 2018 and had a good understanding of written and spoken English. Women were excluded from participating in the P4 study if they were pregnant again at the time of the 6 month postpartum assessment, or if they had pre-existing diabetes, hypertension, renal or any other serious maternal disease prior to the index pregnancy. Written informed consent for the mother and infant was obtained at study enrolment, which occurred by 6 months postpartum.
The P4 study initially recruited 415 women who were grouped based on their final diagnosis as either NTP (n = 302), GH (n = 23) or PE (n = 90) according to the International Society for the Study of Hypertension in Pregnancy (ISSHP) Guidelines.21 GH was defined as persistent, new onset hypertension (Blood Pressure (BP) ≥140 mmHg systolic or ≥90 mmHg diastolic) at or after 20 weeks gestation, while PE was new onset hypertension accompanied by evidence of maternal organ dysfunction including proteinuria, acute kidney injury, liver dysfunction, neurological features, haematological complications, and/or uteroplacental dysfunction.21
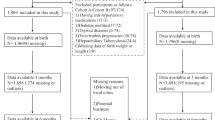
This study includes infants exposed to NTP, GH or PE that had weight recorded at 2 years (Fig. 1). Prematurity status was defined as birth before 37 weeks gestation,21 and SGA status as a birthweight z-score corrected for sex and gestational age less than −1.28 (corresponding to below the 10th percentile).56,57
Data collection
Birthweight to the nearest gram and length to the nearest 0.5 cm were retrieved retrospectively from the mother’s electronic medical record. Weight, length and head circumference were measured at 6 months and weight and head circumference measured at 2 years, corrected for gestational age at birth, by a paediatrician.58 Weight in kilograms to the nearest one decimal place was measured with the infant lightly clothed without shoes using digital scales. Recumbent length at 6 months was measured with a measuring tape to the nearest 0.5 cm. Head circumference around the occipitofrontal diameter was measured with a measuring tape to the nearest millimetre.
Maternal sociodemographic information, medical history, birth details and all other details considered covariates, including total months breastfed, were collected both retrospectively from the mother’s electronic medical record, and prospectively from P4 surveys and study visits at 6 months and 2 years postpartum. Maternal BP, body composition, and metabolic markers were also collected at these study visits.
Outcomes
As previously described (Gow et al. (2021)54), birth and 6-month weight and length z-scores were calculated for term infants using the World Health Organisation (WHO) Child Growth Standards,59,60 and for preterm infants using the INTERGROWTH-21st Preterm Postnatal Growth Standards,61 which allow for the differing pattern of postnatal growth to 6 months that preterm infants experience.61 The INTERGROWTH-21st standards converge with the WHO Child Growth Standards at 6 months, and thus the WHO standards were used for all infants from 6 months corrected age.59,61 Two-year weight z-scores were calculated for all infants using the WHO Child Growth Standards.59,60
Secondary measures of longitudinal infant growth calculated from birth to 2 years, and from 6 months to 2 years, included change in weight z-score, rapid weight gain, defined as an increase in weight z-score ≥0.67,62 and conditional weight gain, which considers the potential confounding of age, sex, birthweight, and regression to the mean on z-score change. Conditional weight gain is calculated using the standardised residuals from the linear regression of the 2-year versus 6-month, and 2-year versus birthweight z-scores with age and sex included as covariates. A positive value indicates a faster (and negative value a slower) rate of weight gain compared to the age and sex-adjusted population mean, adjusted for by the previous measurement to account for possible regression to the mean.54,63,64
Covariates
Maternal, infant and birth data considered covariates in relation to GH or PE exposure and infant growth outcomes included:
-
Maternal data: age, parity, smoking history, gestational diabetes mellitus status, maternal and paternal ethnicity and education, and first trimester, 6-month and 2-year weight, BMI, average systolic and diastolic BPs, Homoeostatic Model Assessment for Insulin Resistance (HOMA-IR) scores and Edinburgh Postnatal Depression Scale (EPDS) scores.
-
Birth data: labour onset and mode of birth.
-
Infant data: sex, birth gestation, prematurity status, SGA status, length of any neonatal intensive care unit or special care nursery (NICU/SCN) stay, and total months breastfed in the first 2 years.
Statistical analysis
Descriptive statistics were used to summarise infant growth outcomes and covariates for the three exposure groups (NTP, GH or PE). One-way Analysis of Variance tests (parametric continuous data) and Kruskal-Wallis tests (non-parametric continuous data) with Tukey Post-Hoc analysis, and Chi-Square or Fisher’s Exact tests (categorical data) were used to estimate whether infant growth outcomes and covariates differed between groups. Associations between covariates and infant growth outcomes (i.e., weight, weight z-scores, z-score changes, rapid-weight-gain and conditional weight gain z-scores) were explored using simple linear regression. Significant associations were explored in multivariable linear regression, including adjustment for covariates and confounders such as SGA and prematurity status. Subgroup analyses stratified by SGA and prematurity status were conducted for selected growth outcomes significant in simple regression. Statistical analysis was performed using IBM SPSS Statistics, version 26.0 (Chicago, IL). A p value of <0.05 was considered statistically significant.
Results
From the 415 infants enroled in the P4 Study, 325 had a recorded 2-year weight (Fig. 1) and are included in this analysis. Those without 2-year weights did not differ from those included in the final sample in terms of gestation at birth, birth weight z-score, 6-month weight z-score, maternal age and BMI at 6 months postpartum and duration of NICU/ SCN stay. Table 1 details the parental demographic and maternal health outcomes of those included in this study. Compared to the NTP group, both GH and PE mothers had significantly higher systolic and diastolic BPs, HOMA-IR scores and BMI at all time points (except first trimester BMI for the PE group), while GH mothers had significantly higher weight at all timepoints. Less than one quarter (n = 14) of PE women experienced early onset PE (≤33 weeks), 16 experienced late onset PE (>33 and <37 weeks), 33 experienced PE at term (≥37 weeks), and 3 experienced PE postpartum.
Birth and infant outcomes are described in Table 2. Compared to the NTP group, GH and PE mothers were less likely to experience spontaneous labour, more likely to deliver via elective or emergency caesarean section, and PE mothers were more likely to have been nulliparous in the index pregnancy. Compared to both the NTP and GH groups, PE-exposed infants were more likely to be born preterm, SGA and be admitted to a NICU/ SCN. Median duration of all NICU/ SCN admissions was 3 days (interquartile range 18), and ranged in length from 1 to 77 days. Infants from both hypertensive groups were breastfed for a shorter duration than those from NTP.
Table 3 details infant growth outcomes after exposure to NTP, GH or PE. Compared to the NTP or GH groups, PE-exposed infants had lower weight, length and corresponding z-scores at birth (all p < 0.001). There were no significant differences in growth outcomes between groups at 6 months or 2 years. From 6 months to 2 years however, GH infants had significantly greater weight gain (p = 0.034), but there were no other significant differences between groups in this period. From birth to 2 years, GH and PE infants experienced significantly greater gains in weight and weight z-scores compared with NTP infants (Fig. 2), and were 2.4 [95% CI: 0.9–6.2] and 2.0 [1.1–3.5] times, respectively, more likely to experience rapid weight gain. In PE infants, weight gain and weight z-score gain from birth to 2 years in those exposed to early onset PE was 10.7 ± 1.3 kg and 0.52 ± 0.86, respectively. In PE infants exposed to late/ term/ postpartum weight gain and weight z-score change from birth to 2 years was 9.8 ± 1.5 kg and 1.04 ± 1.06, respectively. These differences were not statistically significant.
When stratifying our cohort, infants born SGA experienced greater weight z-score gains than those born not SGA (p < 0.001). Similarly, infants born at term experienced greater weight z-score gains than those born preterm (p = 0.002). In infants not born SGA, PE-exposure remained significantly associated with higher weight z-score gain than NTP-exposed infants (p = 0.023) from birth to 2-years, while GH-exposed infants did not significantly differ from NTP-exposed infants. In both term and preterm infants, PE-exposed infants had higher weight z-score gains than NTP-exposed infants (term: p < 0.001, preterm: p = 0.014) (Table 4A, B, Fig. 3A, B).
In univariate linear regression, GH and PE exposure, SGA and prematurity status, 6-month maternal systolic and diastolic BPs, nulliparity, induction of labour and months breastfed to 2 years were all significantly associated with change in weight z-score from birth to 2 years (Supplementary Table 1). Six-month maternal diastolic BP and induction labour onset were highly associated with other variables significant in univariate linear regression and were subsequently excluded from multivariable regression.
Table 5 details multiple regression models that explore the influence of GH and PE exposure on change in weight z-score from birth to 2 years. When adjusting for SGA status (Model 1), both GH and PE exposure remained significantly associated with a greater increase in weight z-score compared to NTP exposure. After adjustments for total months breastfed (Model 2) and other maternal and birth covariates including 6-month maternal systolic BP, nulliparity status and prematurity status (Models 3) and all covariates (Model 4), only PE remained significantly associated with increases in weight z-score.
Discussion
In our population of 325 infants, those exposed to PE were smaller at birth, but caught up to both NTP and GH-exposed infants by 6 months and 2 years. From birth to 2 years, both GH and PE-exposed infants experienced greater gains in absolute weight, weight z-score, and more rapid weight gain, but only PE-exposure remained significantly associated with this change in weight z-score after adjustment for confounding variables.
To our knowledge, we are the first to compare differences in infant growth and trajectories from birth to 2 years between NTP, GH and PE exposure groups. We found no association between GH or PE exposure and infant weight or weight z-scores at 6 months and 2 years compared with NTP. Our findings contrast with earlier studies of preterm and/or VLBW infants,48,50,51 and more recent studies in mixed cohorts,54,65 where those exposed to PE had lower absolute weight or weight z-scores throughout infancy. Martikainen et al.49 also reported this trend in preterm infants, however found no difference in weight in term infants of either exposure. This trend is more likely to reflect the impact of early-onset or severe PE, which are associated with greater uteroplacental dysfunction leading to preterm birth.18,66 Compared to previous studies, PE in our cohort was less severe and less preterm (only 10 (15%) of PE-exposed infants were born before 34 weeks), consistent with the expected rate in an unselected population. Furthermore, earlier studies did not adjust for preterm birth, SGA or VLBW status, common comorbidities experienced by PE-exposed infants, which are all independently associated with infant postnatal growth.14,52,53
Our findings support previous literature finding that PE-exposed infants ‘catch up’ to their normotensive counterparts in respect to growth outcomes.67,68 Correspondingly, we reported that PE-exposed infants experienced greater increases in weight z-score from birth to 2 years, independent of SGA and prematurity status. While FGR and subsequent SGA birth, common complications of PE, are associated with impaired infant growth,14,52,53 many SGA born neonates experience accelerated weight gain, possibly as a response to intrauterine undernutrition or clinical intervention where calories are added to feeds. This is referred to as ‘rapid catch-up growth’, an example of how infants born on weight extremes may experience natural regression to the mean postpartum.44,63 We reported this is our cohort from birth to 6 months,54 as well as in this study from birth to 2 years, irrespective of PE or NTP exposure. However, we also demonstrated that PE exposure is associated with accelerated infant weight gain, independent of confounders including prematurity and SGA status, which supports an intrauterine programming effect of PE as suggested by the DOHaD hypothesis. While adjusted confounders may reflect certain shared genetic predispositions and postnatal environmental factors such as infant feeding practices, we still cannot exclude residual confounding. Although conditional weight gain, which corrects for the regression to the mean that SGA infants may experience, approached significance from birth to 2 years, it was not different between groups, and thus further differentiation of the impact of SGA status is required in similar cohorts.
GH-exposed infants in our cohort did not differ significantly from NTP infants in weight, and while they experienced greater increases in weight z-score in infancy, this was not independent of perinatal confounders. This supports the hypothesis that shared lifestyle or genetic risk factors that predispose to both HDP and childhood morbidity, such as an adverse cardiometabolic profile, are responsible for their increased disease risk rather than an intrauterine programming effect.8,22 This may also explain why GH and late-onset PE, where there is less uteroplacental dysfunction, are associated with increased risks of childhood morbidity.18,66 Whether a result of GH and PE independently or in combination with these external factors, the increased growth trajectories and rapid weight gain experienced by exposed infants have nonetheless been associated with greater risks of obesity and cardiometabolic disease in later childhood.46 Furthermore, by age 20, Davis et al.68 described that those exposed to GH and PE causing preterm birth had a three-fold risk of hypertension, and those exposed to GH resulting in term birth had a two-fold risk of having obesity. Our findings suggest a need in primary care to identify accelerated growth trajectories in infants exposed to HDP and early screening for markers of cardiometabolic disease.69
Interventions to prevent accelerated weight gain trajectories could include strategies to increase the duration of breastfeeding, which is associated with reduced risk of future child and maternal cardiometabolic disease.70 In our study both hypertensive groups breastfed for a shorter duration compared to NTP. Furthermore, the introduction of greater nutrient-rich solid-feeding diets, strategies to combat fussy eating, and the encouragement of greater maternal and infant physical activity may improve future infant health outcomes.54
Strengths of this study include our ethnically diverse cohort, and similar to our previous study,54 our use of the INTERGROWTH-21st Preterm Postnatal Growth Standards,61 which consider the differing postnatal growth patterns of preterm infants.61 Furthermore, our calculation of conditional weight gain was an additional outcome that considered the potential confounding of age, sex, birthweight, and regression to the mean on z-score change that SGA infants in our cohort may have experienced.54,63,64
Limitations of this study include the small sample size of GH infants that reduced statistical power and limited our ability to include further covariates in regression models, and the medium sample of PE infants that prevented sub-analyses based on PE onset or severity. Although SGA and prematurity status are common complications of PE, in some cases they are unrelated to PE, and adjustment for them in regression may have led to the overestimation of the impact of PE. We also lacked data on other confounding factors that may influence infant growth, including paternal factors such as height, weight, cardiometabolic profiles and other genetic or inheritable risk factors, postnatal infant solid feeding practices, activity levels, and the general postnatal environment including infections. Finally, the P4 Study was single-centred, and powered to detect differences in maternal BP outcomes 6 months after NTP, GH or PE exposure rather than paediatric outcomes specifically.
GH and PE are common obstetric complications with known long-term complications for mother and child. We demonstrated that HDP-exposed infants experience accelerated weight gain from birth to 2 years. For PE-exposed infants, this was independent of commonly comorbid intermediates including SGA and prematurity status, suggesting that PE may have an intrauterine programming effect. However, further research in larger, term-born, non-SGA cohorts is needed to further disentangle these covariates to understand the specific pathophysiological implications of GH and PE exposure. Nonetheless, these disorders are commonly comorbid to complications like SGA and prematurity status, and thus our findings provide opportunities for clinical intervention.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Payne, B. A., Hanson, C., Sharma, S., Magee, L. A. & von Dadelszen, P. Epidemiology of the hypertensive disorders of pregnancy. In The FIGO textbook of Pregnancy Hypertension: an evidence-based guide to monitoring, prevention and management (Magee, L. A., von Dadelszen, P., Stones, W. & Matthews, M. eds.) 63-74 (The Global Library of Women’s Medicine, London, 2016).
Roberts, C. L. et al. Population-based trends in pregnancy hypertension and pre-eclampsia: An international comparative study. BMJ Open 1, e000101 (2011).
Gestational Hypertension and Preeclampsia: Acog Practice Bulletin, Number 222. Obstet. Gynecol. 135, e237-e260 (2020).
Umesawa, M. & Kobashi, G. Epidemiology of hypertensive disorders in pregnancy: Prevalence, risk factors, predictors and prognosis. Hypertens. Res. 40, 213–220 (2017).
Magee, L. A. et al. The 2021 International Society for the Study of Hypertension in Pregnancy Classification, Diagnosis & Management Recommendations for International Practice. Pregnancy Hypertens. 27, 148–169 (2022).
Brown, M. A. et al. Recognizing cardiovascular risk after preeclampsia: the P4 study. J. Am. Heart Assoc. 9, e018604 (2020).
Stuart, J. J. et al. Hypertensive disorders of pregnancy and maternal cardiovascular disease risk factor development: an observational cohort study. Ann. Intern. Med. 169, 224–232 (2018).
Wu, P. et al. Preeclampsia and future cardiovascular health: a systematic review and meta-analysis. Circ.: Cardiovasc. Qual. Outcomes 10, e003497 (2017).
Hermes, W. et al. Cardiovascular risk factors in women who had hypertensive disorders late in pregnancy: A cohort study. Am. J. Obstet. Gynecol. 208, 474.e471–478 (2013).
Brown, M. C. et al. Cardiovascular disease risk in women with pre-eclampsia: Systematic review and meta-analysis. Eur. J. Epidemiol. 28, 1–19 (2013).
Habli, M., Levine, R. J., Qian, C. & Sibai, B. Neonatal outcomes in pregnancies with preeclampsia or gestational hypertension and in normotensive pregnancies that delivered at 35, 36, or 37 weeks of gestation. Am. J. Obstet. Gynecol. 197, 401–407 (2007).
Langenveld, J. et al. Neonatal outcome of pregnancies complicated by hypertensive disorders between 34 and 37 weeks of gestation: A 7 year retrospective analysis of a national registry. AJOG 205, 540.e541–547 (2011).
Lowe, S. A. et al. Somanz guidelines for the management of hypertensive disorders of pregnancy 2014. Aust. N. Z. J. Obstet. Gynaecol. 55, e1–e29 (2015).
Gruslin, A. & Lemyre, B. Pre-eclampsia: Fetal assessment and neonatal outcomes. Best. Pract. Res. Clin. Obstet. 25, 491–507 (2011).
Lausman, A. & Kingdom, J. Intrauterine growth restriction: Screening, diagnosis, and management. J. Obstet. Gynaecol. Can. 35, 741–748 (2013).
Bokslag, A., van Weissenbruch, M., Mol, B. W. & de Groot, C. J. M. Preeclampsia; short and long-term consequences for mother and neonate. Early Hum. Dev. 102, 47–50 (2016).
Nomura, Y. et al. Neurodevelopmental consequences in offspring of mothers with preeclampsia during pregnancy: Underlying biological mechanism via imprinting genes. Arch. Gynecol. Obstet. 295, 1319–1329 (2017).
Tranquilli, A. L., Brown, M. A., Zeeman, G. G., Dekker, G. & Sibai, B. M. The definition of severe and early-onset preeclampsia. Statements from the International Society for the Study of Hypertension in Pregnancy. Pregnancy Hypertens. 3, 44–47 (2013).
Pettit, F., Mangos, G., Davis, G., Henry, A. & Brown, M. A. Pre-eclampsia causes adverse maternal outcomes across the gestational spectrum. Pregnancy Hypertens. 5, 198–204 (2015).
Shen, M. et al. Comparison of risk factors and outcomes of gestational hypertension and pre-eclampsia. PLoS One 12, e0175914 (2017).
Brown, M. A. et al. The hypertensive disorders of pregnancy: Isshp classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 13, 291–310 (2018).
Alsnes, I. V. et al. Hypertension in pregnancy and offspring cardiovascular risk in young adulthood: Prospective and sibling studies in the Hunt study (Nord-Trøndelag Health Study) in Norway. Hypertension 69, 591–598 (2017).
Miliku, K. et al. Associations of maternal and paternal blood pressure patterns and hypertensive disorders during pregnancy with childhood blood pressure. J. Am. Heart Assoc. 5, e003884 (2016).
Fraser, A., Nelson, S. M., Macdonald-Wallis, C., Sattar, N. & Lawlor, D. A. Hypertensive disorders of pregnancy and cardiometabolic health in adolescent offspring. Hypertension 62, 614–620 (2013).
Davis, E. F. et al. Cardiovascular risk factors in children and young adults born to preeclamptic pregnancies: A systematic review. Pediatrics 129, e1552–e1561 (2012).
Nahum Sacks, K. et al. In utero exposure to pre-eclampsia as an independent risk factor for long-term respiratory disease. Pediatr. Pulmonol. 55, 723–728 (2020).
Liu, X. et al. Maternal preeclampsia and childhood asthma in the offspring. Pediatr. Allergy Immunol. 26, 181–185 (2015).
Byberg, K. K., Ogland, B., Eide, G. E. & Oymar, K. Birth after preeclamptic pregnancies: Association with allergic sensitization and allergic Rhinoconjunctivitis in late childhood; a historically matched cohort study. BMC Pediatr. 14, 101 (2014).
Keski-Nisula, L., Heinonen, S., Remes, S. & Pekkanen, J. Pre-eclampsia, placental abruption and increased risk of atopic sensitization in male adolescent offspring. Am. J. Reprod. Immunol. 62, 293–300 (2009).
Mann, J. R., McDermott, S., Bao, H., Hardin, J. & Gregg, A. Pre-eclampsia, birth weight, and autism spectrum disorders. J. Autism Dev. Disord. 40, 548–554 (2010).
Griffith, M. I., Mann, J. R. & McDermott, S. The risk of intellectual disability in children born to mothers with preeclampsia or eclampsia with partial mediation by low birth weight. Pregnancy Hypertens. 30, 108–115 (2011).
Grace, T., Bulsara, M., Pennell, C. & Hands, B. Maternal hypertensive diseases negatively affect offspring motor development. Pregnancy Hypertens. 4, 209–214 (2014).
Trønnes, H., Wilcox, A. J., Lie, R. T., Markestad, T. & Moster, D. Risk of cerebral palsy in relation to pregnancy disorders and preterm birth: A national cohort study. Dev. Med. Child Neurol. 56, 779–785 (2014).
Tuovinen, S. et al. Maternal hypertensive disorders during pregnancy: Adaptive functioning and psychiatric and psychological problems of the older offspring. Brit. J. Obstet. Gynaec. 121, 1482–1491 (2014).
Tuovinen, S., Eriksson, J. G., Kajantie, E. & Räikkönen, K. Maternal hypertensive pregnancy disorders and cognitive functioning of the offspring: A systematic review. J. Am. Soc. Hypertens. 8, 832–847.e831 (2014).
Walker, C. K. et al. Preeclampsia, placental insufficiency, and autism spectrum disorder or developmental delay. JAMA Pediatr. 169, 154–162 (2015).
Warshafsky, C., Pudwell, J., Walker, M., Wen, S. W. & Smith, G. N. Prospective assessment of neurodevelopment in children following a pregnancy complicated by severe pre-eclampsia. BMJ Open 6, e010884 (2016).
Safi-Stibler, S. & Gabory, A. Epigenetics and the developmental origins of health and disease: Parental environment signalling to the epigenome, critical time windows and sculpting the adult phenotype. Semin. Cell Dev. Biol. 97, 172–180 (2020).
Lu, H. Q. & Hu, R. Lasting effects of intrauterine exposure to preeclampsia on offspring and the underlying mechanism. Am. J. Perinatol. Rep. 9, e275–e291 (2019).
Barker, D. J. The origins of the developmental origins theory. J. Intern. Med. 261, 412–417 (2007).
Hales, C. N. & Barker, D. J. The thrifty phenotype hypothesis. Br. Med. Bull. 60, 5–20 (2001).
Wang, G. et al. Weight gain in infancy and overweight or obesity in childhood across the gestational spectrum: A prospective birth cohort study. Sci. Rep. 6, 29867 (2016).
Singhal, A. Long-term adverse effects of early growth acceleration or catch-up growth. Ann. Nutr. Metab. 70, 236–240 (2017).
Cho, W. K. & Suh, B.-K. Catch-up growth and catch-up fat in children born small for gestational age. Korean J. Pediatr. 59, 1–7 (2016).
Ounsted, M. & Sleigh, G. The infant’s self-regulation of food intake and weight gain: Difference in metabolic balance after growth constraint or acceleration in utero. Lancet 305, 1393–1397 (1975).
Zheng, M. et al. Rapid weight gain during infancy and subsequent adiposity: A systematic review and meta-analysis of evidence. Obes. Rev. 19, 321–332 (2018).
Vakil, P., Henry, A., Craig, M. E. & Gow, M. L. A review of infant growth and psychomotor developmental outcomes after intrauterine exposure to preeclampsia. BMC Pediatr. 22, 513 (2022).
Szymonowicz, W. & Yu, V. Y. Severe pre-eclampsia and infants of very low birth weight. Arch. Dis. Child. 62, 712 (1987).
Martikainen, A. Growth and development at the age of 1.5 years in children with maternal hypertension. J. Perinat. Med. 17, 259–269 (1989).
Silveira, R. C., Procianoy, R. S., Koch, M. S., Benjamin, A. C. W. & Schlindwein, C. F. Growth and neurodevelopment outcome of very low birth weight infants delivered by preeclamptic mothers. Acta Paediatr. 96, 1738–1742 (2007).
Matić, M., Inati, V., Abdel-Latif, M. E. & Kent, A. L. Maternal hypertensive disorders are associated with increased use of respiratory support but not chronic lung disease or poorer neurodevelopmental outcomes in preterm neonates at <29 weeks of gestation. J. Paediatr. Child Health 53, 391–398 (2017).
Bocca-Tjeertes, I. F. A., Reijneveld, S. A., Kerstjens, J. M., de Winter, A. F. & Bos, A. F. Growth in small-for-gestational-age preterm-born children from 0 to 4 years: The role of both prematurity and Sga status. Neonatology 103, 293–299 (2013).
von Beckerath, A.-K. et al. Perinatal complications and long-term neurodevelopmental outcome of infants with intrauterine growth restriction. Am. J. Obstet. Gynecol. 208, 130.e131–130.e136 (2013).
Gow, M. L. et al. Growth from birth to 6 months of infants with and without intrauterine preeclampsia exposure. J. Dev. Orig. Health Dis. 13, 151–155 (2022).
Davis, G. K. et al. Postpartum physiology, psychology and paediatric follow up study (P4 study) - study protocol. Pregnancy Hypertens. 6, 374–379 (2016).
Physical Status: The Use and Interpretation of Anthropometry. Report of a Who Expert Committee. World Health Organ. Tech. Rep. Ser. 854, 1–452 (1995).
de Onis, M. & Habicht, J. P. Anthropometric reference data for international use: Recommendations from a World Health Organization Expert Committee. Am. J. Clin. Nutr. 64, 650–658 (1996).
Demerath, E. W. & Fields, D. A. Body composition assessment in the infant. Am. J. Hum. Biol. 26, 291–304 (2014).
World Health Organisation: Multicentre Growth Reference Study Group. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age: Methods and Development. (World Health Organisation, Geneva, 2006).
World Health Organisation: WHO Multicentre Growth Reference Study Group. WHO Child Growth Standards: Head Circumference-for-Age, Arm Circumference-for-Age, Triceps Skinfold-for-Age and Subscapular Skinfold-for-Age: Methods and Development. (World Health Organisation, Geneva, 2007).
Villar, J. et al. Postnatal Growth Standards for Preterm Infants: The Preterm Postnatal Follow-up Study of the Intergrowth-21(St) Project. Lancet Glob. Health 3, e681–e691 (2015).
Ong, K. K., Ahmed, M. L., Emmett, P. M., Preece, M. A. & Dunger, D. B. Association between Postnatal Catch-up Growth and Obesity in Childhood: Prospective Cohort Study. BMJ 320, 967–971 (2000).
Cole, T. J. Conditional reference charts to assess weight gain in British infants. Arch. Dis. Child. 73, 8–16 (1995).
Cole, T. J. Presenting information on growth distance and conditional velocity in one chart: Practical issues of chart design. Stat. Med. 17, 2697–2707 (1998).
Byberg, K. K., Øymar, K., Eide, G. E., Forman, M. R. & Júlíusson, P. B. Exposure to preeclampsia in utero affects growth from birth to late childhood dependent on child’s sex and severity of exposure: Follow-up of a nested case-control study. PLoS One 12, e0176627 (2017).
Staff, A. C. & Redman, C. W. G. The Differences between Early- and Late-Onset Pre-Eclampsia. In Preeclampsia. Comprehensive Gynecology and Obstetrics (Saito, S. eds) 157-172 (Springer, Singapore, 2018).
Cheng, S.-W., Chou, H.-C., Tsou, K.-I., Fang, L.-J. & Tsao, P.-N. Delivery before 32 weeks of gestation for maternal pre-eclampsia: Neonatal outcome and 2-year developmental outcome. Early Hum. Dev. 76, 39–46 (2004).
Davis, E. F. et al. Clinical cardiovascular risk during young adulthood in offspring of hypertensive pregnancies: Insights from a 20-year prospective follow-up birth cohort. BMJ Open 5, e008136 (2015).
Bloetzer, C. et al. Screening for cardiovascular disease risk factors beginning in childhood. Public Health Rev. 36, 9 (2015).
Nguyen, B. et al. Breastfeeding and cardiovascular disease hospitalization and mortality in Parous women: Evidence from a large Australian cohort study. J. Am. Heart Assoc. 8, e011056 (2019).
Acknowledgements
We acknowledge the Bidjigal people of the Eora Nation, and Gweagal people who are the traditional custodians of the Land on which our research was conducted. We acknowledge the Indigenous women, infants and families who participated in this study, and thank all women and their families for their ongoing participation in and support for the P4 Study. We also acknowledge the students involved in the collection of data as part of this study as well as paediatricians Prof. Maria Craig, Dr Ana Dosen, Dr Scott Dunlop, Dr Melanie Fentoulis, Dr Joseph Khouri, Dr Lynette Khoury and Dr Darren Shepherd who were all involved in collecting the paediatric growth measures.
Funding
Funding for the P4 Study was provided by the St George and Sutherland Medical Research Foundation and a philanthropic donation from Emeritus Professor Richard Henry. Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Substantial contributions to conception and design: G.D., M.A.B., M.E.C., A.H. Substantial contributions to acquisition of data: L.R., J.M.K., A.D. Substantial contributions to analysis and interpretation of data: M.L.G., PV. Drafting the article or revising it critically for important intellectual content: all authors. Final approval of the version to be published: all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent statement
Written informed consent for mother and infant was obtained at study enrolment, which occurred by 6 months postpartum.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gow, M.L., Vakil, P., Roberts, L. et al. Childhood growth outcomes 2 years after hypertensive versus normotensive pregnancy: a P4 study. Pediatr Res 95, 275–284 (2024). https://doi.org/10.1038/s41390-023-02789-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02789-7
- Springer Nature America, Inc.